Abstract
Migration routes and the depth patterns of anguillid eel larvae migrating long distances from spawning grounds in the ocean remain poorly understood. We used otolith stable isotope analysis to study the oceanic migrations of anguillid eels by reconstructing experienced water temperature histories of larvae. The otolith stable oxygen isotopes (δ18Ootolith) of recruited Anguilla japonica glass eels were analyzed to assess the relationship with the experienced water temperature of the early larval stage in laboratory experiments. A negative linear relationship between rearing water temperature and δ18Ootolith values was observed for eel leptocephali reared at five different temperatures between 19 °C and 27 °C. Subsequently, the obtained equation between water temperature and δ18Ootolith was applied to estimate the water temperature experienced by wild-caught glass eels during their larval oceanic migration according to recruitment latitude, season, and species. The δ18Ootolith values of A. japonica glass eels were significantly higher at higher recruitment latitudes from Taiwan to Japan and later recruitment periods of 6 months, from November to March, indicating that the individuals were exposed to lower water temperatures along the Kuroshio flowing northward as they reached the higher latitudes in the later recruitment seasons. Furthermore, the δ18Ootolith values of the temperate A. japonica were higher than those of the tropical Anguilla marmorata, recruited at the same locations in southern Japan and Taiwan. This suggests that larval migratory behavior in the ocean may differ between the two eel species, although they have sympatric spawning areas in the western North Pacific. Collectively, these results suggest that δ18Ootolith provides a reasonable estimation of the experienced water temperature and may prove useful for reconstructing the early migratory history of anguillid eels in the ocean.
Similar content being viewed by others
Introduction
Pursuing the horizontal migratory routes and vertical depths in the ocean for long-distance migratory marine animals is challenging. Catadromous anguillid eel larvae typically migrate thousands of kilometers from oceanic spawning areas to freshwater or coastal saline growth habitats, and understanding their early life history has been a long-term challenge. Extensive oceanographic investigations using research vessels found the spawning grounds of the European eel Anguilla anguilla and the American eel Anguilla rostratain the Sargasso Sea in the Atlantic Ocean1 and those of the Japanese eel Anguilla japonica near the West Mariana Ridge in the North Pacific2.
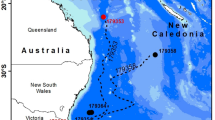
The Japanese eel is the only anguillid species for which wild eggs have been collected3 providing researchers a known starting point of its migration. After the fertilized eggs hatch, the transparent leaf-like eel larvae, called leptocephali, migrate in the ocean for several months and metamorphose into cylindrical glass eel juveniles, and then reach estuaries. Leptocephali of A. japonica are transported from their spawning grounds by the westward-flowing North Equatorial Current and are subsequently transferred to the northward-flowing Kuroshio Current4,5 before being recruited to a latitudinally wide geographic range along the coast of East Asia (Fig. 1). Leptocephalus migration and subsequent glass eel recruitment are thought to be directly affected by oceanographic features such as ocean currents, eddies, and water temperatures6,7. Furthermore, leptocephali exhibit diel vertical migration, alternating between shallow nighttime depths and deeper daytime depths8. However, information on the depths during most of their migration periods remains limited. Consequently, detailed migration routes and vertical depths have not been established. Notably, how glass eel recruitment occurs over a latitudinally wide geographical area and which migration routes are associated with each recruitment area remain unclear.
Map of seven collection locations of Anguilla japonica and A. marmorata glass eels in Japan and Taiwan. Arrow indicates the Kuroshio Current. The map was created using R (version 4.3.2, https://www.r-project.org/) with the rnaturalearth package (using Natural Earth map data, https://www.naturalearthdata.com/), ggplot2 package (https://ggplot2.tidyverse.org/), and Adobe Illustrator (version 25.1, https://adobe.com/).
Anguilla japonica, which mainly uses temperate habitats, spawns sympatrically with the North Pacific population of Anguilla marmorata, which predominantly has tropical habitats9. While some A. marmoratareach tropical regions, including the Philippines and Indonesia, by the southward-flowing Mindanao Current, others reach subtropical Taiwan and southern Japan (mainly the Nansei Islands) via the northward-flowing Kuroshio Current10,11,12,13. However, how A. japonica and A. marmorata glass eels recruit to different locations despite having been spawned in nearly the same area is unclear. Determining the water temperatures experienced during the larval oceanic migration might provide clues to the migratory history of the larval stages of these species. After recruitment to freshwater growth habitats, A. japonica and A. marmorata coexist in rivers in southern Japan and Taiwan, symbolizing the biocultural diversity in river ecosystems14,15. In these sympatric areas, A. japonica, listed as endangered by the International Union for Conservation of Nature (IUCN), is speculated to be in an interspecific competitive relationship with A. marmorata, although they segregate their habitats within rivers16,17. Furthermore, there are concerns regarding the impact on these eel populations due to habitat shifts and oceanographic fluctuations caused by climate change including global warming. Therefore, clarifying the recruitment mechanisms of these two species is important for ecological understanding and resource management.
Stable carbon and oxygen isotopes (δ13C, δ18O) have been used to reconstruct the history of ambient water temperature based on the temperature dependency of isotopic fractionation between water δ18O (δ18Owater) and carbonate δ18O (δ18Ocarbonate)18. The δ18O in biogenic carbonate of aquatic organisms, such as foraminifera, fish otoliths, cephalopod statoliths, and mollusk shells, has been used to reconstruct the experienced temperature or ambient δ18Owater19,20,21. It has been reported that the δ13C of biogenic carbonate reflects the complicated contribution of metabolic rate, δ13C of food sources, and δ13C of dissolved inorganic carbon (DIC)22,23. Although the slopes of the linear relationship between δ18Ocarbonate and temperature are generally consistent, there are certain species-specific offsets between δ18Owater and δ18Ocarbonate24. As biomineralization generally occurs in a compartmentalized space separated from the ambient water, δ18Ocarbonate is controlled not only by thermodynamic processes but also by modifications of physiological and kinetic effects that may be taxon-specific25.
Temperature dependency of δ18O in the otolith (δ18Ootolith) of A. japonica has been evaluated using glass eel samples26. The equation obtained by rearing glass eels under four temperatures (15, 20, 25, and 30 °C) was reported by Shirai et al.26 as follows:
where T is the temperature in degrees Celsius, VPDB is Vienna Pee Dee Belemnite, and VSMOW is Vienna Standard Mean Ocean Water. Ion microprobe analysis was performed only on the otolith growth portion of the glass eel stage, within an approximate 8-µm area on the cross-sectional surface. Due to this analytical method, Eq. (1) may be biased as the study employed an ion microprobe technique specific to microscale solid analysis, which is relatively susceptible to matrix effects26. Hence, the most straightforward strategy for estimating the temperature history from δ18Ocarbonate is to establish a species-specific temperature–δ18Ocarbonate relationship.
In this study, we reared newly hatched A. japonica leptocephali under different temperatures in a laboratory setting and demonstrated the temperature dependency of δ18Ootolith when reconstructing the experienced water temperatures during the oceanic larval stage. Subsequently, we used this relationship to estimate the average temperatures experienced by the recruited of A. japonica and A. marmorata glass eels during their larval migrations from the spawning area to different recruitment locations at widely varying latitudes. This reconstruction facilitated interspecies comparisons of experienced temperatures and niche separation during the larval migration by recruitment location and time. Establishing a new analytical tool to determine the water temperatures and depths that reflect vertical temperature changes experienced by eel leptocephali during oceanic migration can provide useful insights into their oceanic migratory ecology and contribute the management of commercially important fishery resources14,15. Moreover, using the δ18Ootolith analysis method might improve the techniques for the artificial production of glass eels in the laboratory27,28,29.
Materials and methods
Experiment 1: temperature dependency validation of otolith isotopes for artificially reared eel leptocephali
Leptocephali rearing experiment
We conducted the rearing experiments of A. japonica leptocephali at the IRAGO Institute, Aichi Prefecture, Japan. All experiments were performed in accordance with the ARRIVE guidelines as well as the guidelines of the Institutional Animal Care and Use Committee, the University of Tokyo. The fertilized A. japonica eggs were obtained according to the methods of Okamura et al.30. The eggs hatched spontaneously on 11 February 2017. The hatched yolk sac larvae (preleptocephali) were retained without feeding for 5 days in a 180 L polycarbonate cylindrical tank under full darkness at 25 °C. The salinity of the rearing water was maintained at a natural ocean salinity of 35 psu by adding solar salt (Nichien Co. Ltd, Japan) to 32.5 psu groundwater.
A 3-week rearing experiment of A. japonica leptocephali was conducted from 17 February to 10 March 2017. Six days post-hatch (dph), randomly selected larvae were distributed to 20 L planktonkreisel tanks in five water temperature groups (19, 21, 23, 25, and 27 °C), with approximately 2000 individuals each. The water temperature within each tank was altered over 3 h to the target temperatures by using controller systems (E5EC-T, OMRON Corp.). Larvae were fed five times per day during the 3-week rearing experiment. The diet comprised shark Squalus acanthiasegg yolk, Antarctic krill extract, soybean peptide, and vitamins31. The experimental tanks were maintained in darkness throughout the day, with a weak water flow (1.5 L/min), and lights above the tanks were turned on for 10 min only during feeding.
To examine the otolith growth of larvae in each water temperature group, we randomly sampled 14−34 larvae from each tank at the beginning of the experiment and weekly threreafter32. These larvae were anesthetized using tricaine methanesulfonate (MS-222) and fixed in a 5% formalin-seawater solution. Otolith images of 10 larvae in each temperature group were captured using a stereomicroscope (SMZ1500, Nikon) coupled with a camera (DS-Fi1, Nikon); the individual otolith diameters were measured using Image J software (Table S1). Samples collected at the beginning of the experiment and weekly were not used for otolith isotope ratio measurements.
At the end of the 3-week rearing experiment, all leptocephali were anesthetized using MS-222, fixed in 70% ethanol, and used for subsequent otolith analysis (Fig. 2a). The otolith diameters of 10 leptocephali per temperature group were measured at the end of the experiment.
Photographs of Anguilla japonica leptocephali (a) and an enlarged view of the head (b) of 27-day-olds reared at 27 °C, viewed with a stereomicroscope with a polarized filter. (c) One hundred extracted otoliths of A. japonica leptocephali in a distilled-water-filled micro-Petri dish. The arrows in (b) show otoliths. Scale bars = 1 mm, 100 μm, and 5 mm in a, b, and c, respectively.
Groundwater and rearing water were measured daily for salinity with a refractometer (S/Mill-E, ATAGO Co., Ltd.) to monitor the rearing water stability. The rearing water salinity range was 35 ± 0.5 psu throughout the rearing period. The water temperature in the tanks was recorded every 10 min using a thermal recorder (TR-7, T&D Corp.). The mean water temperatures of the 19, 21, 23, 25, and 27 °C tanks were 19.3 ± 0.4, 21.4 ± 0.4, 23.0 ± 0.3, 24.8 ± 0.3, and 26.7 ± 0.1 °C, respectively (Table S2). Rearing water samples for δ18Owater analysis were collected from the tanks in each temperature group on the last day of the experiment.
Preparation of leptocephali otolith samples
Pairs of sagittal otoliths on each side of eel leptocephali (Fig. 2b) reared for 3 weeks were extracted using insect pins and tweezers under a stereomicroscope (SMZ1500, Nikon). The extracted otoliths were cleaned to remove tissues and washed with distilled water. One hundred otoliths were pooled for one isotope measurement batch per water temperature group. Left and right sagittal otoliths of leptocephali were used; hence, 50−63 leptocephali were used to extract 100 otoliths per set. Three sample sets (100 otoliths per set) were prepared for each water temperature. The otoliths were placed in 2-mm diameter micro-Petri dishes filled with distilled water (Fig. 2c) and dried in an oven (Dov-300 A, AS ONE Corp.) at 40 °C for 1 h to evaporate the water. The otoliths were placed in a reaction tube in the micro-Petri dish.
Otolith isotope analysis of reared leptocephali
The δ13Cotolith and δ18Ootolith were measured using an IsoPrime100 isotope ratio mass spectrometer (Isoprime Ltd., Cheadle Hulme, UK) equipped with a customized continuous-flow gas preparation system (MICAL3c) at the National Institute of Technology, Ibaraki College, Hitachinaka, Japan, according to a protocol developed by Ishimura et al.33,34. This system can accurately measure stable isotopes in CaCO3 samples for amounts as low as 0.2 µg. Carbonate samples were reacted with 104% H3PO4 at 25 °C. The evolved CO2 gas was first purified in a stainless-steel vacuum line and then in a helium-purged purification line. The purified CO2 gas was injected into the IsoPrime100 mass spectrometer. The δ13Cotolith and δ18Ootolith values of each carbonate sample were reported in standard δ notation (δ13Cotolith, δ18Ootolith; ‰) relative to the VPDB standard. Detailed analytical conditions have been previously described35. The analytical precision was within 0.1‰ for δ13Cotolith and δ18Ootolith.
The analysis was conducted in three sets from different sample pools for the five water temperature groups; however, one dataset for the 25 °C experimental group could not be analyzed. In cases where a higher sample gas yield volume was obtained in each analysis, the isotope analysis was repeated twice and the average of the two analyses was calculated (Table S3).
As the δ18Ootolith of the entire sagittal otolith was analyzed as a bulk sample, the δ18Ootolith value of otolith growth volume during the temperature experiment was estimated by assuming a mass balance relationship equation for otoliths36. The relationships between the experimental temperature and the estimated δ18Ootolith value of otolith growth volume during the 3-week experiment were mathematically calculated. As the otolith diameter rather than otolith weight was measured, the estimation was conducted by assuming that weight was proportional to length as follows:
where δ18OT−0 is the estimated isotope ratio of otolith growth volume during the temperature experiment; δ18OT is the isotope ratio of the whole otolith measured at the end of the experiment; δ18O0 is the mean isotope ratio from the δ18Ootolith value of the 25 °C experimental group reflecting the rearing temperature of fertilized eggs and preleptocephali until the start of the experiment; L0 and LT are the otolith diameter at the start and end of the experiment, respectively. The otolith volume (LT3) increased 1.4−2.7 times during the experiment: 1.4 times at 19 °C and 2.7 times at 27 °C (Table S1). The same calculation was applied for δ13Cotolith. The calculated otolith isotopes grown during the rearing experiment are referred to hereafter as δ18Ootolith−growth and δ13Cotolith−growth.
Otolith isotope analysis of water samples
The δ18Owater analysis of water samples was performed on nine samples of groundwater collected weekly and rearing waters on the last day of the experiment for the five temperature groups (Table S2), using a Picarro L2130-i Analyzer (Picarro Inc. California, USA) at the National Institute of Technology, Ibaraki College. The long-term analytical precision for seawater was ± 0.05‰. The δ18Owater values were reported relative to VSMOW.
Experiment 2: otolith isotope characterization for wild glass eels
Wild glass eel collection
A total of 1044 glass eels were obtained at four locations in Japan (Tone River, Chiba; Cape Irago, Aichi; Niyodo River, Kochi; and Ihara River, Kagoshima) and three locations in Taiwan (Tansui River, northwestern Taiwan; Yilan River, northeastern Taiwan; and Donggang River, southwestern Taiwan), collected by fishers from November 2016 to March 2017 (Fig. 1, Table S4). The glass eels were preserved in ethanol before measuring total length (TL, mm), body weight (BW, g), and pigmentation stages. These spatiotemporal scales generally cover the timing and location of the main glass eel recruitment of A. japonica on the Pacific coast4,7,37. In addition to the 959 A. japonica glass eels collected from seven locations, 85 A. marmorata glass eels were collected at two southern locations (Ihara River, Kagoshima, Japan, and Yilan River, northeastern Taiwan). For A. japonica, the higher the latitude, the later the recruitment period (r = 0.487, p < 0.001), and the larger the body size (TL: r = 0.172, p < 0.001, BW: r = 0.370, p < 0.001). For A. marmorata, the later the recruitment, the larger the body size (TL: r = 0.326, p = 0.002, BW: r = 0.279, p = 0.010).
Otolith isotope analysis for wild glass eels
In total, 269 A. japonica and 48 A. marmorata glass eels in the early pigmentation stages (VA to VB2) were subjected to otolith stable isotope analysis. A pair of sagittal otoliths was extracted from each individual, washed with distilled water, and dried. The analysis was performed using a stable isotope mass spectrometer (Delta V plus, Thermo Fisher Scientific Inc.) connected to an automated pretreatment device (GasBench II, Thermo Fisher Scientific Inc.) at the Atmosphere and Ocean Research Institute, the University of Tokyo. The detailed analytical settings were previously reported38. The NBS-19 (δ13C = +1.95‰, δ18O = −2.20‰, VPDB) with known isotopic composition distributed by the National Bureau of Standards was used as the standard sample. The reproducibility of NBS-19 for δ13C was +0.06‰ to +0.2‰, and that for δ18O was +0.08‰ to +0.2‰.
The commonly accepted acid fractionation factor of 1.01025 for calcite39 was applied for the MICAL3c and DELTA V analyses to facilitate comparison with previously reported δ18O values40. As the difference in the acid fractionation factor between calcite and aragonite is temperature dependent41, +0.09‰ was subtracted from the δ18Ootolith value determined using DELTA V (reaction temperature: 72 °C) to adjust for the fractionation at a reaction temperature of 25 °C. Experienced water temperatures of eel leptocephali in the ocean were estimated by δ18Ootolith−growth calculated using the relational equation obtained in Experiment 1. Estimating individually experienced salinity, which requires complex conditions for the sample sets and environmental information is difficult42. Thus, the δ18Owater value was set to +0.06‰, which was the average value (mean ± SD: +0.06 ± 0.08‰, min: −0.03‰, max: +0.16‰) collected at different depths (103–324 m) in the North Equatorial Current (16.5°N, 123°E)43, following the method from a previous study26. For comparison, the generalized equation for the temperature–δ18Ootolith relationship calculated for marine fish proposed by Morissette et al.24 was also applied (Eq. (3)) for water temperature estimation to evaluate how the temperature–δ18Ootolith equation affects the ecological interpretation:
Environmental data for wild eel leptocephali in the ocean
To estimate the possible experienced water temperatures at the depths of wild eel leptocephali in the ocean, water temperatures at different depths near the spawning areas and at different latitudes along the Kuroshio Current were examined at the recruitment locations of glass eels. In the waters around the spawning area, CTD observations were conducted by the R/V Hakuho-Maru KH-16-4 research cruise for A. japonica and A. marmorata in the western North Pacific. Sequential water temperature (°C), salinity (psu), dissolved oxygen (mL/L), and chlorophyll a fluorescence (µg/L) were measured for depths from 0 to 1000 m at two stations where A. japonica and A. marmorata leptocephali were collected (16°N, 137°E and 14°N, 137°E, respectively) on 16 and 17 July 2016 (Fig. 3). Based on the vertical water temperature profiles, the distribution depths of leptocephali were estimated in correspondence with the experienced water temperatures estimated from δ18Ootolith values. At the sampling locations of glass eels, water temperature data in the ocean were obtained from the FRA-ROMS website of the Japan Fisheries Research and Education Agency (https://fra-roms.fra.go.jp/) for each month (November 2016 to March 2017) when the glass eels were recruited to the five sampling locations along the Pacific side of the Kuroshio Current (Chiba, Aichi, Kochi, Kagoshima, and Yilan). The monthly average water temperatures were calculated. Water temperature data were obtained from four depths: 0, 50, 100, and 200 m.
CTD profiles in the spawning area region of Anguilla japonica and A. marmorata observed at 16°E, 137°E (St. 2, 16 July 2016) and 14°E, 137°E (St. 4, 17 July 2016) by the KH-16-4 cruise. The thermocline is the area of rapid change in water temperature (orange line) with depth. The maximum salinity (blue line) and chlorophyll concentration (green line) values around 150 m occurred at the top of the thermocline. These two parameters showed subsequent rapid decreases; oxygen (pink line) decreased at depths of ~ 100 m.
Statistical analyses
Variations in the otolith oxygen isotopes of reared A. japonica leptocephali and wild glass eels were examined using linear models. A simple linear regression was used to examine the relationship between the water temperature and the δ13Cotolith−growth and δ18Ootolith−growth − δ18Owater of reared A. japonica leptocephali. To assess whether these otolith isotope values differ between A. japonica and A. marmorata, linear mixed models (LMMs) were constructed for data from two locations where the two species were obtained sympatrically. The dependent variable was δ13Cotolith or δ18Ootolith − δ18Owater, and the explanatory variables were species (fixed effect), location (random intercept), and recruitment date (covariate). LMMs were also constructed to explain the spatiotemporal variation in the δ18Ootolith values of wild glass eels for A. japonica. The dependent variable was δ13Cotolith or δ18Ootolith − δ18Owater, and the explanatory variables were the recruitment date (covariate), recruitment latitude (covariate), and recruitment location (random intercept). For simplicity, the interaction terms among the explanatory variables were not considered. For the recruitment dates of glass eels, the number of days was calculated using 15 November 2016 as the starting point of zero. For glass eels collected at the Ihara River from 1 to 3 January 2017, the collection date was set as 2 January 2017. For glass eels collected in the Yilan River, information was only available for the collection month; thus, the collection date was assumed to be the 15th of each month. To eliminate outliers for the regression between temperature and stable isotope values of reared leptocephali, data with absolute values of studentized residuals exceeding three were excluded from the analysis. Of the measured data points for reared A. japonica leptocephali, one otolith isotope value was omitted as an outlier (Table S3).
Results
Otolith isotopes of reared A. japonica leptocephali.
The δ18Owater values of the weekly collected four groundwater samples ranged from −0.57‰ to −0.56‰ (mean ± SD, −0.56‰ ± 0.01‰; Table S2). The δ18Owater values of the five rearing tanks for each temperature group at the end of the experiment (day 21) ranged from −0.54‰ to −0.51‰ (mean ± SD, −0.52‰ ± 0.015‰; Table S2). Groundwater and rearing water showed small variation ranges, indicating relatively constant δ18Owater values.
The δ13Cotolith and δ18Ootolith values ranged from −3.68‰ to −2.71‰ and −1.57‰ to −0.08‰, and the estimated values for the otolith growth volume of δ13Cotolith−growth and δ18Ootolith−growth ranged from −5.69‰ to −2.63‰ and −1.99‰ to +1.74‰, respectively (Table S3). The δ18Ootolith−growth values of A. japonica leptocephali were measured with slight variations, with a significant negative relationship between the estimated δ18Ootolith−growth and temperature (r2 = 0.603, p = 0.002), excluding one outlier. The relationship between ambient water temperatures and the fractionation of δ18Ootolith−growth from δ18Owater of rearing water was obtained using linear regression (Fig. 4a):
Relationship between water temperature and the δ18Ootolith-growth − δ18Owater (a) and δ13Cotolith-growth (b) of Anguilla japonica leptocephali. Plots are shown as the mean measurement values of each temperature group. Solid line and shading show regression line and 95% confidence intervals, respectively.
where T is the temperature in Celsius; the numbers in parentheses indicate the 95% confidence intervals.
The δ13Cotolith−growth showed an increasing trend with increasing water temperatures (r2 = 0.460, p = 0.008; Fig. 4b). A large variation was observed under the 19 °C condition, with the positive slope becoming marginally significant (p = 0.049) when the bootstrap test for the regression slope was applied using the 75,000 bootstrap samples. However, this study did not analyze the δ13C of DIC, food, or soft tissue, which is necessary to interpret the δ13Cotolith−growth, Thus, these results do not necessarily imply a direct control of water temperature.
Latitudinal, seasonal, and species differences in wild glass eel otoliths
The δ18Ootolith values of A. japonica glass eels collected at seven locations in Japan and Taiwan ranged from −1.27‰ to +0.46‰, and the δ18Ootolith values of A. marmorata glass eels collected at two locations (Ihara River, southern Japan, and Yilan River, northeastern Taiwan) ranged from −2.20‰ to −0.20‰ (Table S5). The dataset from two locations where A. japonica and A. marmorata were sampled revealed that the δ18Ootolith − δ18Owater increased with a later recruitment date and was higher in A. japonica than in A. marmorata on the same recruitment date (Fig. 5, recruitment date: F1,135 = 30.4, p < 0.001; species: F1,122 = 42.9, p < 0.001).
Relationship between sampling day (15 November 2016 = day 0) and the δ18Ootolith − δ18Owater of Anguilla japonica (green symbols) and A. marmorata (red symbols) glass eels at two locations of the Ihara River, Kagoshima, Japan and the Yilan River, northeastern Taiwan.
The δ18Ootolith − δ18Owater values of A. japonica glass eels varied spatiotemporally, tending to increase with later recruitment and at higher latitudes (Fig. 6; recruitment date: F1,263 = 21.56, p < 0.001; latitude: F1,9 = 4.08, p = 0.073). The positive slope for the latitude became highly significant when the bootstrap test for the regression slopes was applied using the 10,000 bootstrap samples (recruitment date: p < 0.001; latitude: p = 0.003). Moreover, the maximum likelihood estimates of the model increased the Akaike information criterion (AIC) when the effect of latitude was removed (∆AIC = 2.32). This suggests that the recruitment date and latitude are useful parameters for explaining spatiotemporal variation in the δ18Ootolith − δ18Owater.
Three-dimensional plots of the sampling location latitude (x), sampling day (y, 15 November 2016 = day 0), and the δ18Ootolith − δ18Owater (z) of Anguilla japonica glass eels. Note: the δ18Owater is set at +0.06‰. The regression plain obtained from the linear mixed model is shown as a green plain (pseudo r2 = 0.185, z = 0.0145x + 0.00254y − 1.30).
Based on Eq. (4) and the assumption of a δ18Owater value of +0.06‰, the mean experienced temperatures for each of the seven locations and each collection month from November to March ranged from 23.8 °C to 26.1 °C for A. japonica and 24.6 °C to 28.3 °C for A. marmorata (Table S5). The lowest water temperature estimated from δ18Ootolith was observed in A. japonica glass eels from Aichi, Japan, collected in the late season of March, while the highest water temperature was observed in A. marmorata glass eels from Yilan, northeastern Taiwan, collected in the early season of November, with a difference of ~ 4.5 °C. The calculation using the generalized Eq. (3) resulted in a temperature 4 °C lower than those calculated using Eq. (4) (Table S5).
The δ13Cotolith values of A. japonica glass eels collected at seven locations in Japan and Taiwan ranged from −8.17‰ to −5.97‰; the δ13Cotolith of A. marmorata glass eels collected at two locations ranged from −8.90‰ to −5.50‰ (Table S5). The dataset from two locations where A. japonica and A. marmorata were sampled suggested that the δ13Cotolith values of glass eels were not related to species or recruitment date (recruitment date: F1,155 = 2.188, p = 0.141; species: F1,155 = 1.931, p = 0.167). The δ13Cotolith values of A. japonica glass eels for all datasets showed no significant spatiotemporal variation (recruitment date: F1,266 = 1.48, p = 0.224; latitude: F1,7 = 0.276, p = 0.616).
Discussion
Otolith isotopes of eels as a temperature indicator
This study is the first evaluation of the relationship between the δ18Ootolith of A. japonica leptocephali and ambient water temperature by using a laboratory-based experiment enabled by recently advanced artificial seedling technology27,28,29. In laboratory experiments for artificial seeding production technology studies, the leptocephali are typically reared at optimum temperatures between 21 and 23 °C, as established by a trial-and-error process45,46,47. However, in this study, we conducted a 3-week water temperature experiment at a wider range of ambient water temperatures (19–27 °C), to obtain further insights into leptocephali thermophysiology. Otolith growth of reared A. japonica leptocephali increased with temperature. Field observations have shown that A. japonica spawns at depths of 150–200 m around the West Mariana Ridge, and eggs and newly hatched larvae are thought to float to the upper part of the thermocline at a 150 m depth3. This depth corresponds to a water temperature of ~ 23 °C (Fig. 3) in the spawning areas, estimated to be the optimal water temperature for hatched larvae of this species. Therefore, in the laboratory experiments conducted in this study, a water temperature of 23 °C served as the median of the five experimental groups. At higher water temperatures of 25 °C and 27 °C, higher otolith growth of leptocephali was observed during the 3-week experiment, but the growth slowed from the second week (Table S1). High water temperatures are associated with an increase in the energy intake and feeding activity of leptocephali, as well as an increase in energy consumption through metabolism. Hence, the increase in energy consumption due to high basal metabolism is a trade-off with nutrient intake32,48. Accordingly, the higher water temperatures were likely not their optimal temperature. In contrast, otolith growth was low in the lower water temperature groups (19 °C and 21 °C), likely due to being below the optimum temperature range for growth. In these 19–27 °C experimental temperature ranges, the influence of otolith deposition during the first five days from hatching was relatively large, accounting for the difficulty in assessing the δ18Ootolith of otolith growth volume during the temperature-controlled experimental period.
The proportion of otolith growth volume during the experiment differed among temperature settings, with a greater proportion at higher water temperatures. Hence, the estimation error might be larger at lower temperatures due to the significant contribution of the otolith growth during initial non-temperature-regulation periods (at 25 °C). Nevertheless, the δ18Ootolith−growth calculation for the growth portion during the rearing experiments demonstrated that the δ18Ootolith of A. japonica leptocephali exhibited a temperature dependence with a negative linear relationship.
The general characteristics of the temperature–δ18Ootolith relationship (Fig. 4a) of eel species in this study and a previous study24 included similar temperature sensitivities (i.e., the slope of the linear equation) and significant offsets (i.e., the intercept when the slope is assumed to be the same). This characteristic is evident in previous studies of other teleost fish species (e.g., Pacific bluefin tuna Thunnus orientalis49, Japanese sardine Sardinops melanostictus50, chub mackerel Scomber japonicus51). The analysis of covariance (ANCOVA) identified significant differences for slope (F1,30 = 11.304, p = 0.002) calculated using Eq. (4) for A. japonica leptocephalus otoliths than those using Eq. (1) for glass eels26; the slope estimate was steeper (−0.348) compared with that for Eq. (1) (−0.153; slope difference = 0.196, 95% CI: 0.077–0.315). The slope also differed from that generally observed in biogenic calcium carbonate (−0.21)24. Such differences in the temperature–δ18Ootolith relationship are likely due to the combined effects of experimental artifacts, such as limited otolith growth during rearing, different analytical methods, and the narrow temperature range of the experimental setting, as well as biological effects, including different developmental stages. However, the most critical issue is that the present study estimated the isotope ratio of otolith growth volume during the temperature-control experiment by subtracting the influence of initial otolith growth using Eq. (2). The proportion of otolith growth volume was smaller in the lower temperature settings, resulting in large uncertainty in the slope estimation. The offset was lower around the initial temperature of 25 °C. Differences in analytical methods also likely contributed to this discrepancy among studies, given that the analysis by Shirai et al.26 used secondary ion mass spectrometry. Hane et al.52 reported a difference of ~ 0.4‰ between secondary ion mass spectrometry and MICAL3c for Pacific bluefin tuna otoliths analysis. Such analytical artifacts likely affect the magnitude of offset but not the slope.
The δ18Owater value for glass eels assumed in this study was a constant +0.06‰, the average value of the possible distribution depths of A. japonica leptocephali in the Pacific Ocean26,43. However, this value should vary according to the diel vertical migration and the migration route of leptocephali over several months in the natural environment. For example, by applying the δ18Owater values (min: −0.03‰, max: +0.16‰) for the distribution depths (103–324 m) in the North Equatorial Current used to calculate the average δ18Owater value43, the difference in water temperature estimated from Eq. (4) ranged from −0.26 °C to +0.29 °C. Similarly, by applying the δ18Owatervalues (mean ± SD: +0.14 ± 0.10‰, min: −0.09‰, max: +0.31‰) near the surface (10–30 m) around the Nansei Islands and the southeast of Kyushu (25–32 °N, 125–133 °E) along the Kuroshio Current53, the estimated temperature difference ranged from −0.43 °C to +0.72 °C. Within the range of these depths and regional variations, the estimated temperature variation was relatively small. Future numerical simulation approaches may allow different δ18Owater values to be applied for different migration routes.
Physiological changes associated with the developmental stages may cause differences in the accumulating mechanism of δ18Ootolith between the leptocephalus and glass eel stages, which undergo drastic physiological and morphological transformations in the metamorphosing process; this effect is included in Eq. (4) obtained in the present study. As Eq. (4) minimizes the physiological and methodological effects, and the degree of offset is small around 25 °C, we propose Eq. (4) as the best tentative indicator of the experienced temperature of wild eel leptocephali.
A linear relationship between δ13Cotolith values and water temperature was also observed in experiments with reared A. japonica leptocephali (Fig. 4b). This trend does not imply a direct control of water temperature, but rather reflect metabolic and/or seawater DIC changes. δ13Cotolith is mainly controlled by relative contribution of DIC and respiratory carbon originated from food source, and the proportion of these two carbon source changes with metabolic activity54,55. In our experiment, the food fed to the larvae remained the same among the five temperature experimental groups, but otolith growth, as well as larval growth and survival rates, was reduced by lower temperatures32. Although the larvae fed on the same food during the experiment, the changes in δ13Cotolith−growth values with increasing temperature might reflect multiple factors such as temperature-induced metabolic differences, changes in the δ13C of dissolved inorganic carbon of water, or a contribution of metabolisms due to the residence time of nutrition from the yolk sac after hatching. Further experimental investigation is necessary to fully establish using δ13Cotolith for ecological reconstruction.
When designing the rearing tank experiment, using replicate tanks would have been ideal. However, the limited facilities available due to technical difficulties with artificially rearing eel larvae made large-scale experiments impractical. To address this limitation, this study standardized the size and shape of the tanks and applied multiple consecutive temperature conditions (i.e., five equally spaced water temperature conditions). Such an approach complements tank replications, as previous studies have suggested the reproducibility of this method30,44. Therefore, although the absence of replications may have widened the confidence interval of the estimated slope, the accuracy of the estimated slope remained consistent. The linear relationship between water temperature and otolith isotopes also indicates that the experiment was successfully performed.
Experienced temperature depends on recruitment latitude and timing
Our fitted model showed that the δ18Ootolith values increased with A. japonica glass eels collected at higher latitudes and during later seasons, indicating that the individuals recruited to the northern areas and in latter seasons may be exposed to lower temperatures for longer during the larval durations in the ocean. The fishing season for recruiting A. japonica glass eel generally begins earlier in the southern regions: October in Taiwan and December in Japan37. In the northernmost island of Hokkaido (42.6°N), glass eels recruit later in the season56. Glass eels recruited during the late season appear to have longer larval durations and delayed arrival to estuaries due to exposure to colder winter temperatures along the Kuroshio Current7,57, which is consistent with the findings of the present study.
The swimming depths during metamorphosis or oceanic glass eel stages remain unclear, but Otake et al.8 reported that when A. japonica leptocephali reach total lengths > 20–30 mm, they perform diel vertical migrations with distribution at 170–220 m during the day and 60–85 m during the night. These depths corresponded to daytime water temperatures of 19–22 °C and nighttime temperatures of 28–30 °C around the spawning areas in the North Equatorial Current (Fig. 3). Estimated temperatures from δ18Ootolith for A. japonica glass eels recruited to Taiwan were approximately 26 °C and those to southern Japan were 24–25 °C (Table S5). These correspond to the temperatures in the middle of the diel vertical migration range. Water temperatures near the recruitment locations were lower than those in the tropical North Equatorial Current, and the temperatures estimated from δ18Ootolith corresponded to near-surface water temperatures in Taiwan and southern Japan in the early season of November and December; water temperatures in later seasons (January to March) and at deeper depths (> 50 m) were even colder and did not correspond to the estimates in the present study (Fig. S1). Therefore, at least the δ18Ootolith values obtained from glass eels in these southern locations may more closely reflect water temperatures during the larval stage in the North Equatorial Current or shallower depths along the Kuroshio Current.
However, the otolith growth rate during the larval period of anguillid eels is not constant, with the highest growth rate at ~ 20–30 mm total length, and otolith daily increment widths are generally larger during metamorphosis from leptocephalus to glass eel58,59. Additionally, the volume of aragonite crystals in the otolith is larger in the later depositional portions, as the oval-shaped fish otoliths are deposited in three dimensions. Therefore, where the entire otolith was analyzed as a bulk sample, the individual δ18Ootolith values may contain bias rather than representing the average experienced temperature from hatching to recruitment. A more detailed analysis of the growth stages is expected to enable more accurate estimation.
Experienced temperatures between eel species
The observed difference in δ18Ootolith between the two anguillid species might indicate niche separation. The δ18Ootolith values of A. marmorata collected in the two locations of southern Japan (Ihara River) and northeastern Taiwan (Yilan River) were significantly lower than those of A. japonica, suggesting that A. marmorata may experience higher water temperatures during the oceanic larval period, at least during the early season in November. The mean difference in the net experienced temperature estimated from δ18Ootolith by Eq. (4) was 2.6 °C higher for A. marmorata than for A. japonica when recruited to northeastern Taiwan in November but was similar in the late season. The reconstructed temperature from δ18O of the whole otoliths represents a cumulative signature from birth till collection of each individual; however, identifying a temperature or depth of a specific timing is difficult due to the signature containing multiple factors associated with temperature and water mass (i.e., salinity) during different life stage. In the western North Pacific, the A. japonica and North Pacific population of A. marmorata spawn sympatrically in the western Mariana region9. These eel leptocephali are transported by the North Equatorial Current, suggesting that although both species generally follow the same migration route by ocean currents, they may experience relatively different water temperatures seasonally. Anguilla marmorata has generally higher otolith growth rates and a younger age of recruitment than A. japonica9,60. This suggests that the oceanic larval period is shorter for A. marmorata and the overall experienced water temperatures from the tropical spawning areas to the recruitment locations remain warm. This interspecies difference could be due to behavioral traits, such as swimming ability and diel vertical migration depths of leptocephali. In flat-shaped leptocephali, buoyancy increases with body depth, and A. marmorata has generally greater body depths than A. japonica61 (Fig. S2). This suggests that A. marmorata may exploit shallower and warmer water masses. Warmer temperatures may allow A. marmorata to grow more rapidly than A. japonica, enabling them to recruit at younger ages. This may allow A. marmorata to attain a higher growth rate than A. japonica, enabling them to recruit at younger ages. These interspecific differences may also reflect variations in the spawning season and optimal water temperatures for the survival and growth of each species11,62.
As artificial production technology for A. marmorata has not yet been established, it was not possible to evaluate potential interspecific differences in δ18Ootolith through rearing experiments.
However, even among individuals collected from the same locations, the difference in δ18Ootolith between the two species varied depending on the collection month (Fig. 5); similar variations were not observed for δ13Cotolith. This cannot be explained solely by interspecific differences. Therefore, it is assumed that these differences in δ18Ootolith reflect the environmental conditions experienced by each individual.
Conclusion
We clarified the water temperature dependency of δ18Ootolith in A. japonica leptocephali, suggesting its possible use as an indicator of ocean temperature experienced by leptocephalus larvae. The δ18Ootolith of eel leptocephali can reveal details of the larval habitat in the wild. The δ18Ootolith of the wild eels was analyzed to determine whether differences exist among recruitment latitude, timing, and species. Results suggest that changes occur in the experienced water temperature by latitude and season and that there are species-specific differences in migratory ecology during the oceanic larval stage. Although previous studies have discussed differences in experienced water temperature based on recruitment latitudes and seasons, this study provides new insights suggesting that experienced water temperature may differ between two eel species recruited to the same locations. The results obtained from otolith isotopic data may reflect both species’ distribution depths and growth rates, which are ecologically relevant.
Biochemical analysis using δ18Ootolith is a valid biological tool for stock assessment and management for of various fish species63,64,65, potentially including anguillid eels. The glass eel recruitment abundance influenced by the spatiotemporal variability of oceanographic conditions7. Investigating the relationship between glass eel recruitment and the water temperatures experienced during migration via the δ18Ootolith indicator may provide useful insights into the potential effects of water temperature on individual recruitment success. Long-term trends in A. japonica recruitment suggest that their distribution areas may have shifted northwards over the past 30 years, possibly influenced by changes in ocean currents66. As the water temperature experienced by eel larvae is thought to influence their migratory behavior, the δ18Ootolith indicator could serve as a valuable tool for assessing biological responses to future climate changes.
Data availability
All fish sample information and data analyzed in the present study are included in the article and its supplementary information files.
References
Schmidt, J. The breeding places of the eel. Philos. Trans. R. Soc. Lond. B. 211, 179–208 (1922).
Tsukamoto, K. Discovery of the spawning area for Japanese eel. Nature 356, 789–791. https://doi.org/10.1038/356789a0 (1992).
Tsukamoto, K. et al. Oceanic spawning ecology of freshwater eels in the western North Pacific. Nat. Commun. 2, 179. https://doi.org/10.1038/ncomms1174 (2011).
Tsukamoto, K. Recruitment mechanism of the eel, Anguilla japonica, to the Japanese coast. J. Fish. Biol. 36, 659–671. https://doi.org/10.1111/j.1095-8649.1990.tb04320.x (1990).
Shinoda, A. et al. Evaluation of the larval distribution and migration of the Japanese eel in the western North Pacific. Rev. Fish Biol. Fish. 21, 591–611. https://doi.org/10.1007/s11160-010-9195-1 (2011).
Zenimoto, K. et al. The effects of seasonal and interannual variability of oceanic structure in the western Pacific North Equatorial Current on larval transport of the Japanese eel Anguilla japonica. J. Fish. Biol. 74, 1878–1890. https://doi.org/10.1111/j.1095-8649.2009.02295.x (2009).
Han, Y. S. Temperature-dependent recruitment delay of the Japanese glass eel Anguilla japonica in East Asia. Mar. Biol. 158, 2349–2358. https://doi.org/10.1007/s00227-011-1739-y (2011).
Otake, T., Inagaki, T., Hasumoto, H., Mochioka, N. & Tsukamoto, K. Diel vertical distribution of Anguilla japonica leptocephali. Ichthyol. Res. 45, 208–211. https://doi.org/10.1007/BF02678565 (1998).
Kuroki, M. et al. Sympatric spawning of Anguilla marmorata and Anguilla japonica in the western North Pacific Ocean. J. Fish. Biol. 74, 1853–1865. https://doi.org/10.1111/j.1095-8649.2009.02299.x (2009).
Shiao, J. C., Iizuka, Y., Chang, C. W. & Tzeng, W. N. Disparities in habitat use and migratory behavior between tropical eel Anguilla marmorata and temperate eel A. japonica in four Taiwanese rivers. Mar. Ecol. Prog. Ser. 261, 233–242. https://doi.org/10.3354/meps261233 (2003).
Wakiya, R., Itakura, H. & Kaifu, K. Age, growth, and sex ratios of the giant mottled eel, Anguilla marmorata, in freshwater habitats near its northern geographic limit: a comparison to tropical regions. Zool. Stud. 58, e34. https://doi.org/10.6620/ZS.2019.58-34 (2019).
Kita, T. & Tachihara, K. Age, growth, and gonadal condition of the giant mottled eel, Anguilla marmorata, in Okinawa-Jima Island, Japan. Environ. Biol. Fish. 103, 927–938. https://doi.org/10.1007/s10641-020-00994-5 (2020).
Kumai, Y., Tsukamoto, K. & Kuroki, M. Growth and habitat use of two anguillid eels, Anguilla marmorata and A. japonica, on Yakushima Island, Japan. Ichthyol. Res. 67, 375–384. https://doi.org/10.1007/s10228-020-00732-y (2020).
Kuroki, M., van Oijen, M. J. & Tsukamoto, K. Eels and the Japanese: an inseparable, long-standing relationship in Eels and Humans (eds Tsukamoto, K. & Kuroki, M.). Springer, Tokyo 91–108. https://doi.org/10.1007/978-4-431-54529-3_6 (2014).
Tzeng, W. N. Freshwater eels and humans in Taiwan in Eels and Humans (eds Tsukamoto, K. & Kuroki, M.). Springer, Tokyo 129–142. https://doi.org/10.1007/978-4-431-54529-3_9 (2014).
Matsushige, K., Yasutake, Y. & Mochioka, N. Contrasting riverine distribution and habitat use of the Japanese eel, Anguilla japonica, and the giant mottled eel, Anguilla marmorata, in a sympatric river. J. Fish. Biol. 101, 1617–1622. https://doi.org/10.1111/jfb.15224 (2022).
Kumai, Y. et al. Ontogenetic habitat shift of Anguilla marmorata and A. japonica in the rivers of southern Japan: implications for habitat use evaluation and conservation. Aquat. Conserv. 33, 1295–1308. https://doi.org/10.1002/aqc.4013 (2023).
Hillaire-Marcel, C. et al. A stable isotope toolbox for water and inorganic carbon cycle studies. Nat. Rev. Earth Environ. 2, 699–719. https://doi.org/10.1038/s43017-021-00209-0 (2021).
Grossman, E. L. & Ku, T. L. Oxygen and carbon isotope fractionation in biogenic aragonite: temperature effects. Chem. Geol. 59, 59–74. https://doi.org/10.1016/0168-9622(86)90057-6 (1986).
Darnaude, A. M. et al. Listening in on the past: what can otolith δ18O values really tell us about the environmental history of fishes? PLoS One. 9, e108539. https://doi.org/10.1371/journal.pone.0108539 (2014).
Chung, M. T., Chen, C. Y., Shiao, J. C., Lin, S. & Wang, C. H. Temperature-dependent fractionation of stable oxygen isotopes differs between cuttlefish statoliths and cuttlebones. Ecol. Indic. 115, 106457. https://doi.org/10.1016/j.ecolind.2020.106457 (2020).
Kalish, J. M. 13C and 18O isotopic disequilibria in fish otoliths: metabolic and kinetic effects. Mar. Ecol. Prog. Ser. 75, 191–203 (1991).
Solomon, C. T. et al. Experimental determination of the sources of otolith carbon and associated isotopic fractionation. Can. J. Fish. Aquat. Sci. 63, 79–89. https://doi.org/10.1139/f05-200 (2006).
Morissette, O., Trueman, C. N., Sturrock, A. M., Geffen, A. J. & Shirai, K. Limited evidence for species-specific sensitivity of temperature‐dependent fractionation of oxygen stable isotope in biominerals: a meta‐analysis. Methods Ecol. Evol. 14, 1719–1731. https://doi.org/10.1111/2041-210X.14122 (2023).
Gilbert, P. U. P. A. et al. Biomineralization: Integrating mechanism and evolutionary history. Sci. Adv. 8, eabl9653. https://doi.org/10.1126/sciadv.abl9653 (2022).
Shirai, K. et al. Temperature and depth distribution of Japanese eel eggs estimated using otolith oxygen stable isotopes. Geochim. Cosmochim. Acta 236, 373–383. https://doi.org/10.1016/j.gca.2018.03.006 (2018).
Okamura, A., Horie, N., Mikawa, N., Yamada, Y. & Tsukamoto, K. Recent advances in artificial production of glass eels for conservation of anguillid eel populations. Ecol. Freshw. Fish. 23, 95–110. https://doi.org/10.1111/eff.12086 (2014).
Tanaka, H. Progression in artificial seedling production of Japanese eel Anguilla japonica. Fish. Sci. 81, 11–19. https://doi.org/10.1007/s12562-014-0821-z (2015).
Okamura, A., Yamada, Y., Horie, N. & Mikawa, N. Larval Rearing in Eel Science, Fisheries Science Series (eds Tsukamoto, K., Kuroki, M. & Watanabe, S.). Springer, Singapore. 201–212. https://doi.org/10.1007/978-981-99-5692-0_15 (2023).
Okamura, A. et al. Growth and survival of eel leptocephali (Anguilla japonica) in low-salinity water. Aquaculture 296, 367–372. https://doi.org/10.1016/j.aquaculture.2009.08.039 (2009).
Okamura, A. et al. Hen egg yolk and skinned krill as possible foods for rearing leptocephalus larvae of Anguilla japonica Temminck & Schlegel. Aquac. Res. 44, 1531–1538. https://doi.org/10.1111/j.1365-2109.2012.03160.x (2013).
Kuroki, M., Okamura, A., Yamada, Y., Hayasaka, S. & Tsukamoto, K. Evaluation of optimum temperature for the early larval growth of Japanese eel in captivity. Fish. Sci. 85, 801–809. https://doi.org/10.1007/s12562-019-01317-z (2019).
Ishimura, T., Tsunogai, U. & Gamo, T. Stable carbon and oxygen isotopic determination of sub-microgram quantities of CaCO3 to analyze individual foraminiferal shells. Rapid Commun. Mass. Spectrom. 18, 2883–2888. https://doi.org/10.1002/rcm.1701 (2004).
Ishimura, T., Tsunogai, U. & Nakagawa, F. Grain-scale heterogeneities in the stable carbon and oxygen isotopic compositions of the international standard calcite materials (NBS 19, NBS 18, IAEA‐CO‐1, and IAEA‐CO‐8). Rapid Commun. Mass. Spectrom. 22, 1925–1932. https://doi.org/10.1002/rcm.3571 (2008).
Nishida, K. & Ishimura, T. Grain-scale stable carbon and oxygen isotopic variations of the international reference calcite, IAEA‐603. Rapid Commun. Mass. Spectrom. 31, 1875–1880. https://doi.org/10.1002/rcm.7966 (2017).
Dorval, E. et al. Temperature record in the oxygen stable isotopes of Pacific sardine otoliths: experimental vs. wild stocks from the Southern California Bight. J. Exp. Mar. Biol. Ecol. 397, 136–143. https://doi.org/10.1016/j.jembe.2010.11.024 (2011).
Yokouchi, K. & Kuroki, M. Fisheries in Eel Science, Fisheries Science Series (eds Tsukamoto, K., Kuroki, M. & Watanabe, S.). Springer, Singapore. 241–252. https://doi.org/10.1007/978-981-99-5692-0_18 (2023).
Shirai, K. et al. Reconstruction of the salinity history associated with movements of mangrove fishes using otolith oxygen isotopic analysis. Mar. Ecol. Prog. Ser. 593, 127–139. https://doi.org/10.3354/MEPS12514 (2018).
Sharma, T. & Clayton, R. N. Measurement of O18O16 ratios of total oxygen of carbonates. Geochim. Cosmochim. Acta 29, 1347–1353. https://doi.org/10.1016/0016-7037(65)90011-6 (1965).
Amano, Y., Shiao, J. C., Ishimura, T., Yokouchi, K. & Shirai, K. Otolith geochemical analysis for stock discrimination and migratory ecology of tunas in Biology and Ecology of Bluefin Tuna (eds Kitagawa, T. & Kimura, S.). CRC Press, Boca Raton. 225–257. https://doi.org/10.1201/b18714 (2015).
Kim, S. T., Mucci, A. & Taylor, B. E. Phosphoric acid fractionation factors for calcite and aragonite between 25 and 75°C: Revisited. Chem. Geol. 246, 135–146. https://doi.org/10.1016/j.chemgeo.2007.08.005 (2007).
Sakamoto, T. et al. Combining microvolume isotope analysis and numerical simulation to reproduce fish migration history. Methods Ecol. Evol. 10, 59–69. https://doi.org/10.1111/2041-210X.13098 (2019).
Östlund, G. H., Craig, H. C., Broecker, W. S. & Spencer, D. W. & GEOSECS. Shorebased measurements during the GEOSECS Pacific expedition. PANGAEA. https://doi.org/10.1594/PANGAEA.743238 (1987).
Okamura, A., Yamada, Y., Mikawa, N., Horie, N. & Tsukamoto, K. Effect of starvation, body size, and temperature on the onset of metamorphosis in Japanese eel (Anguilla japonica). Can. J. Zool. 90, 1378–1385. https://doi.org/10.1139/cjz-2012-0146 (2012).
Tanaka, H., Kagawa, H. & Ohta, H. Production of leptocephali of Japanese eel (Anguilla japonica) in captivity. Aquaculture 201, 51–60. https://doi.org/10.1016/S0044-8486(01)00553-1 (2001).
Tanaka, H., Kagawa, H., Ohta, H., Unuma, T. & Nomura, K. The first production of glass eel in captivity: fish reproductive physiology facilitates great progress in aquaculture. Fish. Physiol. Biochem. 28, 493–497. https://doi.org/10.1023/B:FISH.0000030638.56031.ed (2003).
Okamura, A. et al. Rearing eel leptocephali (Anguilla japonica Temminck & Schlegel) in a planktonkreisel. Aquac. Res. 40, 509–512. https://doi.org/10.1111/j.1365-2109.2008.02127.x (2009).
Okamura, A., Horie, N., Mikawa, N., Yamada, Y. & Tsukamoto, K. Influence of temperature and feeding regimes on growth and notochord deformity in reared Anguilla japonica leptocephali. Fish. Sci. 84, 505–512. https://doi.org/10.1007/s12562-018-1188-3 (2018).
Kitagawa, T. et al. Otolith δ18O of Pacific bluefin tuna Thunnus orientalis as an indicator of ambient water temperature. Mar. Ecol. Prog. Ser. 481, 199–209. https://doi.org/10.3354/meps10202 (2013).
Sakamoto, T. et al. Temperature dependence of δ18O in otolith of juvenile Japanese sardine: laboratory rearing experiment with micro-scale analysis. Fish. Res. 194, 55–59. https://doi.org/10.1016/j.fishres.2017.05.004 (2017).
Nakamura, M. et al. Temperature dependency equation for chub mackerel (Scomber japonicus) identified by a laboratory rearing experiment and microscale analysis. Mar. Freshw. Res. 71, 1384–1389. https://doi.org/10.1071/MF19313 (2020).
Hane, Y. et al. Reconstruction of temperature experienced by Pacific bluefin tuna Thunnus orientalis larvae using SIMS and microvolume CF-IRMS otolith oxygen isotope analyses. Mar. Ecol. Prog. Ser. 649, 175–188. https://doi.org/10.3354/meps13451 (2020).
Kodama, T., Kitajima, S., Takahashi, M. & Ishimura, T. Spatiotemporal variations of seawater δ18O and δD in the Western North Pacific marginal seas near Japan. Geochem. J. 58, 94–108. https://doi.org/10.2343/geochemj.GJ24009 (2024).
Chung, M. T., Trueman, C. N., Godiksen, J. A., Holmstrup, M. E. & Grønkjær, P. Field metabolic rates of teleost fishes are recorded in otolith carbonate. Commun. Biol. 2, 24. https://doi.org/10.1038/s42003-018-0266-5 (2019).
Chung, M. T., Trueman, C. N., Godiksen, J. A. & Grønkjær, P. Otolith δ13C values as a metabolic proxy: approaches and mechanical underpinnings. Mar. Freshw. Res. 70 , 1747–1756. https://doi.org/10.1071/MF18317 (2019).
Morita, K. & Kuroki, M. Japanese eel at the northern edge: glass eel migration into a river on Hokkaido, Japan. Ichthyol. Res. 68, 217–221. https://doi.org/10.1007/s10228-020-00771-5 (2021).
Hsiung, K. M., Kuo, Y. C., Lin, Y. T., Tseng, Y. H. & Han, Y. S. North equatorial current and kuroshio velocity variations affect body length and distribution of the Japanese eel Anguilla japonica in Taiwan and Japan. Sci. Rep. 12, 2888. https://doi.org/10.1038/s41598-022-06669-8 (2022).
Robinet, T. et al. New clues for freshwater eels (Anguilla spp.) migration routes to eastern Madagascar and surrounding islands. Mar. Biol. 154, 453–463. https://doi.org/10.1007/s00227-008-0938-7 (2008).
Kuroki, M., Miller, M. J. & Tsukamoto, K. Diversity of early life-history traits in freshwater eels and the evolution of their oceanic migrations. Can. J. Zool. 92, 749–770. https://doi.org/10.1139/cjz-2013-0303 (2014).
Leander, N. J., Tzeng, W. N., Yeh, N. T., Shen, K. N. & Han, Y. S. Effects of metamorphosis timing and the larval growth rate on the latitudinal distribution of sympatric freshwater eels, Anguilla japonica and A. Marmorata, in the western North Pacific. Zool. Stud. 52, 30. https://doi.org/10.1186/1810-522X-52-30 (2013).
Tsukamoto, K. et al. Positive buoyancy in eel leptocephali: an adaptation for life in the ocean surface layer. Mar. Biol. 156, 835–846. https://doi.org/10.1007/s00227-008-1123-8 (2009).
Han, Y. S., Yambot, A. V., Zhang, H. & Hung, C. L. Sympatric spawning but allopatric distribution of Anguilla japonica and Anguilla marmorata: temperature-and oceanic current-dependent sieving. PLoS One 7, e37484. https://doi.org/10.1371/journal.pone.0037484 (2012).
Von Leesen, G., Ninnemann, U. S. & Campana, S. E. Stable oxygen isotope reconstruction of temperature exposure of the Icelandic cod (Gadus morhua) stock over the last 100 years. ICES J. Mar. Sci. 77, 942–952. https://doi.org/10.1093/icesjms/fsaa011 (2020).
Reis-Santos, P. et al. Reading the biomineralized book of life: expanding otolith biogeochemical research and applications for fisheries and ecosystem-based management. Rev. Fish. Biol. Fish. 33, 411–449. https://doi.org/10.1007/s11160-022-09720-z (2023).
Sakamoto, T., Takahashi, M., Shirai, K., Aono, T. & Ishimura, T. Fisheries shocks provide an opportunity to reveal multiple recruitment sources of sardine in the Sea of Japan. Sci. Rep. 14, 21722. https://doi.org/10.1038/s41598-024-72925-8 (2024).
Chang, Y. L., Morita, K., Muramatsu, K., Kishida, O. & Kuroki, M. Northern shifts in the migration of Japanese glass eels to subarctic Hokkaido Island over the past three decades. Ocean Dyn. 75, 10. https://doi.org/10.1007/s10236-024-01651-6 (2025).
Acknowledgements
We are grateful to Noriyuki Horie and Naomi Mikawa of the IRAGO Institute for their support during rearing experimentation and to Noriko Izumoto of the Atmosphere and Ocean Research Institute at the University of Tokyo for her support for otolith oxygen isotope analysis of glass eels. We also thank Katsumi Tsukamoto, Akira Shinoda, Tsuguo Otake, and Takashi Yamakawa for their suggestions and cooperation.
Author information
Authors and Affiliations
Contributions
MK and KS conceived and designed the experiments and analyzed the data. MK, SH, AO, and YY conducted the rearing experiment of leptocephali. YH provided glass eel samples from Taiwan. SH, KN, TI, and KS conducted the isotopic analyses. All authors contributed to the writing of this paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have declared no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kuroki, M., Hayasaka, S., Nishida, K. et al. Reconstructing the experienced temperature during the larval oceanic migration of anguillid eels from otolith stable oxygen isotopes. Sci Rep 15, 1584 (2025). https://doi.org/10.1038/s41598-025-85523-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-85523-z