Abstract
This study evaluated the impact of aspirin on the biochemical recurrence (BCR) rate following robot-assisted radical prostatectomy (RARP) in patients. A database search identified patients who underwent RARP for pT2-3N0M0 disease at any of 25 centers between 2011 and 2022, categorized into aspirin (n = 350) and control groups (n = 5857). Adjustment by 1:1 propensity score matching (PSM) and Mahalanobis distance matching (MDM) created 350 matched pairs. The effect of aspirin on the BCR rate was evaluated by analysis of BCR-free survival. After PSM and MDM, the 3-year BCR-free rate was significantly better in the aspirin group (85.0%, 95% confidence interval [CI] 80.8–89.4) than in the control group (PSM, 74.5%, 95% CI 66.5–83.5, p = 0.021; MDM, 74.7%, 95% CI 66.3–84.3, p = 0.037). In the analysis of high-risk subgroups, patients in the aspirin group with an ISUP (International Society of Urological Pathology) grade ≥ 4 had a significantly lower recurrence rate in both matched groups (PSM, hazard ratio 0.44, 95% CI 0.22–0.88; MDM, hazard ratio 0.45, 95% CI 0.23–0.90). In conclusion, this study suggests that aspirin could enhance BCR-free survival post-RARP, especially in patients with higher ISUP grades.
Similar content being viewed by others
Introduction
Aspirin is an antithrombotic agent that inhibits the function of cyclooxygenase (COX)-1 by acetylating serine residues, thereby reducing the synthesis of thromboxane A2, which promotes thrombosis1. While aspirin is often administered in patients at high risk of cardiovascular disease2, its tumor-suppressive effects in various types of cancer are also attracting attention3,4,5. These antitumor effects are attributed to inhibition of proliferation of cancer cells and angiogenesis through the putative acetylation of COX-2, a COX-dependent mechanism, and inhibition of inhibitory kappa B kinase beta (IKK-β), a COX-independent mechanism6. The double-blind CAPP2 study3 highlighted the antitumor benefits of aspirin in patients with mismatch repair gene mutations (i.e., Lynch syndrome), and the ongoing CAPP3 trial is investigating the effects of aspirin in doses as low as 100 mg/day.
The development of prostate cancer (PCa) has also been reported to be associated with chronic inflammation7. On the other hand, PCa is also known to be an immunologically cold tumor that is resistant to immune checkpoint inhibitors8. In addition, the effects of aspirin on PCa also remain controversial. Aspirin has been also reported to suppress metalloproteinase (MMP)-9 activity and urokinase-type plasminogen activator (uPA) expression by inhibiting IKK-β-mediated nuclear factor-kappa B (NF-κB) activation, thereby suppressing PCa cell invasion9. However, the anti-tumor effect of aspirin on PCa is controversial in clinical research.
While several studies have reported potential benefits, showing antitumor effects of aspirin in patients with PCa10,11,12, other studies have presented conflicting results with negative outcomes13,14. In a meta-analysis of randomized controlled trials and population based real-world studies, Ma et al.15 demonstrated that aspirin reduces the incidence (relative rate [RR] 0.96, 95% CI 0.95–0.98, p < 0.001) and mortality (RR 0.88, 95% CI 0.82–0.95, p < 0.001) of PCa.
However, there is limited evidence specifically evaluating the effectiveness of aspirin after radical prostatectomy for PCa. Choe et al.10 investigated the prognostic value of anticoagulants including aspirin after radiotherapy or radical prostatectomy in patients with PCa and demonstrated that 10-year PCa-specific mortality was significantly lower in the anticoagulants group (3% vs. 8%, p < 0.01). However, this study did not include postoperative data, such as Gleason score (GS) based on surgical specimens, pathological T stage (pT stage), and resection margin status, and was conducted before the introduction of robotic surgery. Since the advent of robotic surgery, robot-assisted radical prostatectomy (RARP) has played an important role in the treatment of patients with non-metastatic PCa who are candidates for surgery, such as those with a life expectancy of more than 10 years16,17. While advancements in radiation therapy have improved its efficacy, RARP remains a key surgical option achieving postoperative biochemical recurrence (BCR) rate comparable to those of open and laparoscopic surgery18.
Therefore, it is essential to accumulate evidence concerning the antitumor effects of aspirin post-RARP, particularly in patients with PCa who are at high risk of recurrence and expected to benefit from aspirin. The aim of this study was to assess the impact of aspirin on the risk of BCR of PCa after RARP.
Patients and methods
Data sources and patient eligibility
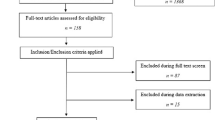
The study had a retrospective observational design and analyzed the clinical data for 8194 patients identified in the Daimonji Clinical Application Database (Dai-CAD)19 to have undergone RARP at any of 25 centers between January 2011 and January 2022. We defined patient eligibility using the following inclusion criteria: pathological diagnosis of PCa based on surgical specimens; no lymph node or distant metastasis; and pT2 or pT3 stage disease, excluding pT4 stage because of the small number involved (n = 5) (Fig. 1). Using these criteria, we extracted patients with pT2 or pT3, cN0 or pN0, and cM0 disease. Patients were excluded if they had received preoperative hormone therapy because of its impact on the prognosis. Patients taking either dutasteride or chlormadinone acetate orally before surgery were also excluded because of the effect of these agents on the prostate-specific antigen (PSA) level.
Flow diagram showing the patient selection process.
Definition of aspirin group
Patients taking aspirin at the time of surgery, including those who temporarily discontinued it perioperatively, were defined as the aspirin group, regardless of whether they were taking anticoagulant medication or concomitant antiplatelet therapy other than aspirin. Duration of aspirin therapy was not considered owing to lack of relevant information in the database.
Statistical analysis
Patient characteristics were compared between the control group and the aspirin group using Pearson’s chi-squared test. Multivariable Cox regression analysis was performed to assess the prognostic impact of each variable in all eligible patients. There were conflicting reports20,21 on the effect of obesity on BCR after radical prostatectomy, so we included obesity as a factor in multivariable Cox analysis. Propensity scores were calculated using logistic regression analysis to adjust for differences in patient characteristics between the two groups. Propensity score matching (PSM) was performed by the optimal matching method in a ratio of 1:1 based on propensity scores. The variables matched included PSA (< 10 vs. ≥ 10 ng/mL), ISUP (International Society of Urological Pathology) grade (≤ 3 vs. ≥ 4), pT stage (pT2 vs. pT3), and resection margin status (negative vs. positive). The ISUP grade was based on the GS diagnosed at the time of histopathological examination of the surgical specimens. Although PSM is one of the most popular methods for between-group adjustment in non-randomized studies, there is also a report showing that PSM alone is insufficient for balancing the groups and recommending the use of other matching methods22. Thus, Mahalanobis distance matching (MDM) was performed as an additional matching method to improve the robustness of our results. Both PSM and MDM were performed using the “MatchIt” R package23. In the subgroup analysis, these two matching methods were used for each high-risk group, namely, PSA ≥ 10 ng/mL, ISUP grade ≥ 4, pT3 stage, and positive resection margin. The effect of aspirin on the risk of BCR after RARP was evaluated by assessing the time to BCR and tested by the log-rank test and Cox regression analysis. The date of BCR was defined as (i) the date of surgery when the PSA did not fall below 0.2 ng/mL after surgery, or (ii) the first date of PSA > 0.2ng/mL when the PSA was below 0.2 ng/mL one month after surgery and the PSA measured at an interval of two to four weeks was > 0.2 ng/mL on two consecutive occasions24,25. All analyses were performed using R (version 4.4.0 for Windows®; The R Foundation for Statistical Computing, Vienna, Austria)26. All statistical analyses were two-sided, and a p-value < 0.05 was considered statistically significant.
Ethics statement
The study was approved by the Institutional Review Board of Kyoto University Hospital (R3168) and performed in accordance with the 1964 Declaration of Helsinki and its subsequent amendments or comparable ethical standards. Due to the retrospective nature of the study, informed consent was not required. An opt-out mechanism with clear instructions was provided for individuals who did not wish to participate. This procedure was approved by the Institutional Review Board of Kyoto University Hospital to ensure compliance with ethical standards, participant protection, anonymity, and secure data handling.
Results
Patient characteristics before matching
Patients who met the study eligibility criteria (Fig. 1) were divided into a control group (n = 5857) and an aspirin group (n = 350). Table 1 shows the patient characteristics in each study group. The proportion of patients aged > 70 years was significantly higher in the aspirin group than in the control group (56.9% vs. 41.5%, p < 0.001), as were the proportions with PSA ≥ 10 ng/mL (35.7% vs. 28.5%, p = 0.005) and pT3 disease (30.9% vs. 25.2%, p = 0.021). The proportion of overweight or obese patients with a BMI of 25 or more was not significantly different between the two groups. As shown in Figs. 2 and 84% of the reasons for taking aspirin in the aspirin group were due to vascular disease (coronary artery disease, stroke, and carotid artery stenosis) or preventive administration for high risk of vascular disease.
Reasons for taking aspirin.
Risk factors for BCR after RARP identified before matching
Before adjustment, aspirin was not associated with BCR in multivariable analysis (hazard ratio [HR] 0.91, 95% confidence interval [CI] 0.68–1.21, p = 0.52) (Fig. S1). However, PSA ≥ 10 ng/mL (HR 1.42, CI 1.24–1.64, p < 0.001), ISUP grade ≥ 4 (HR 2.44, 95% CI 2.12–2.82, p < 0.001), pT3 stage (HR 2.43, 95% CI 2.10–2.81, p < 0.001), and a positive resection margin (HR 2.09, 95% CI 1.81–2.40) were identified to be significant predictors of a poor prognosis. Being overweight or obese with a BMI of 25 or more did not significantly affect prognosis.
After adjustment by PSM and MDM
After PSM, there were 350 patients in each group, with no significant between-group differences in patient characteristics. Similarly, there were no differences in patient characteristics after MDM (Table 2). As shown in Fig. S2, the absolute standardized mean differences for each variable were < 0.1 after both matching methods, and the balance of covariates was better than before adjustment. Therefore, both matching methods were considered adequately conducted.
Effect of aspirin on BCR rate after RARP
Figure 3 showed the results of Kaplan–Meier analysis of the time to BCR after RARP before and after matching. Before matching, there was no significant difference in the 3-year BCR-free survival rate between the aspirin group and the control group (85.0% [95% CI 80.8–89.4] vs. 85.6% [95% CI 84.5–86.6]; p = 0.926, log-rank test). However, after adjustment by PSM, the 3-year BCR-free survival rate was significantly higher in the aspirin group than in the control group (85.0% [95% CI 80.8–89.4] vs. 74.5% [95% CI 66.5–83.5]; p = 0.021, log-rank test). Furthermore, after adjustment by MDM, the 3-year BCR-free rate was significantly better in the aspirin group (85.0% [95% CI 80.8–89.4] vs. 74.7% [95% CI 66.3–84.3]; p = 0.037, log-rank test).
Results of Kaplan–Meier analysis of time to BCR before and after adjustment. (a) Before matching. (b) After propensity score matching. (c) After Mahalanobis distance matching. BCR, biochemical recurrence; HR, hazard ratio; CI, confidence interval.
Effect of aspirin in subgroups at high risk of recurrence
We evaluated the effect of aspirin on the BCR rate in the subgroups identified to have a poor prognosis in the multivariable analysis before adjustment, namely, those with a PSA level ≥ 10 ng/mL, those with an ISUP grade of ≥ 4, those with pT3 stage disease, and those with a positive resection margin. After adjustment of patient characteristics by PSM and MDM, there was no significant between-group difference in any patient characteristic in any of the high-risk subgroups (Tables S1 and S2). In patients with ISUP grade ≥ 4, aspirin had a significant beneficial effect on time to BCR after both PSM (HR 0.44, 95% CI 0.22–0.86, p = 0.017) and MDM (HR 0.45, 95% CI 0.23–0.90, p = 0.024) (Fig. 4). In contrast, patients with pT3 stage disease or a PSA ≥ 10 ng/mL did not derive significant benefit from aspirin. Moreover, in patients with a positive resection margin, the impact of aspirin was beneficial only after PSM (HR 0.51, 95% CI 0.29–0.90, p = 0.019) (Fig. 4a). These findings suggest that the benefit of aspirin depends on tumor grade and that its beneficial effect in patients with a positive margin status after resection seen after PSM warrants further investigation.
Impact of aspirin on time to biochemical recurrence in high-risk subgroups. (a) After propensity score matching. (b) After Mahalanobis distance matching. HR, hazard ratio; CI, confidence interval; PSA, prostate-specific antigen; ISUP, International Society of Urological Pathology.
Discussion
The BCR rate after RARP varies according to risk group. A study in which risk was classified using the D’Amico system found that the 10-year postoperative BCR-free rate was significantly higher in the low-risk group than in the high-risk group (85.7% vs. 43.2%)27. Although preventing BCR after RARP remains a controversial issue, especially in high-risk patients, COX inhibitors, including aspirin, may be candidate agents for prevention of recurrence. The present study demonstrated the efficacy of aspirin in prevention of recurrence in patients after RARP in a retrospective multicenter analysis using two types of matching adjustment. We did not detect a prognostic benefit of aspirin before adjustment (HR 0.91, 95% CI 0.68–1.21, p = 0.515), which may reflect between-group differences in the proportion of patients with a PSA ≥ 10 ng/mL (35.7% vs. 28.5%, p = 0.005) and those with pT3 stage disease (30.9% vs. 25.2%, p = 0.021) in the pre-adjusted patient population. After adjustment for this imbalance in patient characteristics, the prognosis was better in the aspirin group. Our findings suggest that COX inhibitors, such as aspirin, may be effective as adjunctive therapy for PCa after radical treatment. The impact of aspirin on the risk of developing PCa is controversial. Lapi et al.12 investigated the correlation between aspirin and the risk of PCa in Italy and found that the incidence of PCa was lower in patients who received aspirin < 100 mg/day (HR 0.64, 95% CI 0.48–0.86), particularly when used for more than 5 years (HR 0.43, 95% CI 0.21–0.91), than in those who received aspirin ≥ 100 mg/day (HR 0.87, 95% CI 0.51–1.51). Conversely, Elwood et al.28 analyzed the effects of aspirin on various types of cancer using a meta-analysis and reported that the cancer mortality rate for PCa was HR 0.89 (0.78–1.02). Furthermore, Nordstrom et al.14 showed that the initial PSA level was lower in men in the Stockholm cohort who received low-dose aspirin (75 mg) than in their counterparts who did not receive aspirin (–3.92%, 95% CI − 5.76, − 2.05, p < 0.001); however, aspirin did not reduce the occurrence risk of PCa at any dose (odds ratio 1.115, 95% CI 0.995–1.250, p = 0.061), including high-grade PCa (GS > 7; odds ratio 1.025, 95% CI 0.894–1.174, p = 0.723). As a concern regarding the decrease in PSA caused by aspirin, Mantica et al.29 reported that in patients with PCa diagnosed by prostate biopsy, aspirin administration was predictive of a higher Gleason grade group (OR 2.24, 95% CI 1.01–4.87, p = 0.04). Thus, they suggested that aspirin may interfere with the detection of PCa, and the cutoff value for PSA should be considered lower in patients taking aspirin. On the other hand, Choe et al.10 demonstrated that the effect of aspirin on recurrence rate after radical treatment was even more pronounced in patients with high-risk PCa (4% vs. 19%, p < 0.01). Therefore, the effect of aspirin on the prognosis of PCa may vary according to duration of administration and dosage and tumor characteristics. Wang et al.30 evaluated dose-response relationship between aspirin and cancer risk in a meta-analysis and reported that the RR of PCa varied with the dose of aspirin administered. These differences in efficacy may reflect the COX-2 expression level in PCa, which has been correlated with the prognosis of PCa and is highly expressed in metastatic PCa31. Our finding after adjustment using the two matching methods that the prognosis was significantly better in patients with an ISUP of ≥ 4 who took aspirin may be explained by the efficacy of COX inhibitors in patients with high-risk PCa and high COX-2 expression. In this study, we defined a positive margin as a high risk of recurrence, which was identified as a significant risk factor for BCR in multivariable analysis before matching group, but there are conflicting reports regarding the impact of positive surgical margins after radical prostatectomy on BCR. Some studies suggest it is a risk factor32, others report it is not33, and some indicate it only poses a risk in cases of high-grade tumors at the resection margins34,35, making the overall impact unclear. Unfortunately, the database used in this study did not include information on the Gleason status of the positive resection margins, so we could not evaluate it. However, by analyzing the effects of aspirin in different tumor statuses of positive resection margins, it may be possible to examine the effects of aspirin in more detail.
Aspirin-associated adverse events are not a negligible concern. Although continuing low-dose aspirin or anticoagulant therapy in the perioperative period has been reported not to increase bleeding during RARP19,36, long-term aspirin therapy is known to increase the risks of major bleeding (RR 1.66, 95% CI 1.41–1.95) and gastrointestinal bleeding (RR 1.37, 95% CI 1.15–1.62)37. Given this bleeding risk, COX-2 selective inhibitors are also candidates for adjunctive therapy, as well as aspirin. COX-2 selective inhibitors such as celecoxib are thought to be associated with fewer adverse events than non-selective non-steroidal anti-inflammatory drugs38 and can be expected to have a suppressive effect on PCa similar to that of aspirin39. Pruthi et al.40 reported that celecoxib delayed elevation of PSA after BCR in patients with PCa who have been treated with radiotherapy or radical prostatectomy. However, in the present study, we could not explore the effects of COX-2 selective inhibitors on PCa owing to the lack of relevant information in the database. Therefore, prospective studies are needed to evaluate the efficacy of aspirin and COX-2 selective inhibitors in patients with high-risk PCa, such as those with a high ISUP grade.
This study had several limitations. First, no information on the duration and dosage of aspirin therapy was available in the database similar to the study previously reported by Choe et al.10 Therefore, the correlation between the duration or dosage of aspirin and the recurrence rate could not be analyzed. However, previous studies have suggested that prolonged daily use of low-dose aspirin (≥ 75 mg) may reduce cancer-related mortality, including PCa, with significant effects observed after 5 years and further benefits seen with over 7.5 years of use41. Similarly, long-term aspirin use may also play a role in reducing the risk of BCR. Unlike mortality, which requires a longer observation period to assess, BCR could have been detected much earlier due to the sensitivity of biochemical tests, which may have allowed an earlier assessment of the effect of aspirin. Since the patients in this study were matched based on pathological findings and most were taking aspirin continuously postoperatively, the continuation of aspirin therapy after surgery might be particularly important for influencing BCR outcomes. Furthermore, as shown by the reasons for taking aspirin among the participants in this study (Fig. 2), approximately 80% of the patients had vascular disease requiring continuous low-dose aspirin (usually 100 mg in Japan), so it was speculated that few patients used higher doses of aspirin or discontinued aspirin in the early postoperative period. Second, no information on mortality was available, making it difficult to determine whether aspirin improves overall survival or cancer-specific survival. In addition, we could not evaluate EAU-defined high-risk BCR after radical prostatectomy16,42 (ISUP ≥ 4 or PSA doubling time ≤ 1 year) due to insufficient postoperative PSA trend data. Third, the information on oral medications in this database was limited. For example, there have been reports that proton pump inhibitors, often used to prevent gastrointestinal disorders caused by aspirin, may affect PCa progression43. Therefore, it cannot be ruled out that other oral medications may have influenced the risk of BCR. Finally, because this is a retrospective study, it might contain potential bias, limiting its generalizability and causal interpretation. For reasons that are unclear, the aspirin group included a proportion of patients with advanced age or comorbidities who underwent RARP rather than radiotherapy even though they would be typically considered high risk for surgery. Therefore, the possibility of selection bias cannot be excluded. However, use of two matching methods helped to reduce the risk of bias, making the present study valuable in terms of demonstrating the tumor-suppressive effect of aspirin on PCa even in the era of robotic surgery.
In conclusion, aspirin may have beneficial effects in terms of BCR-free survival after RARP, even in groups with high recurrence rates, such as those with ISUP grade ≥ 4. The findings of this study provide a rationale for conducting prospective studies of the prognostic impact of low-dose aspirin in patients with PCa, particularly those at high risk of poor prognosis.
Data availability
This research database is not publicly available due to privacy concerns but can be provided by the corresponding author upon reasonable request.
References
Gelbenegger, G. et al. Aspirin for primary prevention of cardiovascular disease: a meta-analysis with a particular focus on subgroups. BMC Med. 17. https://doi.org/10.1186/s12916-019-1428-0 (2019).
Visseren, F. L. J. et al. ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 42, 3227–3337. https://doi.org/10.1093/eurheartj/ehab484 (2021).
Burn, J. et al. Cancer prevention with aspirin in hereditary colorectal cancer (Lynch syndrome), 10-year follow-up and registry-based 20-year data in the CAPP2 study: a double-blind, randomised, placebo-controlled trial. Lancet 395, 1855–1863. https://doi.org/10.1016/S0140-6736(20)30366-4 (2020).
Reimers, M. S. et al. Aspirin use after diagnosis improves survival in older adults with colon cancer: a retrospective cohort study. J. Am. Geriatr. Soc. 60, 2232–2236. https://doi.org/10.1111/jgs.12033 (2012).
Ricciotti, E., Wangensteen, K. J. & FitzGerald, G. A. Aspirin in Hepatocellular Carcinoma. Cancer Res. 81, 3751–3761. https://doi.org/10.1158/0008-5472.CAN-21-0758 (2021).
Alfonso, L., Ai, G., Spitale, R. C. & Bhat, G. J. Molecular targets of aspirin and cancer prevention. Br. J. Cancer 111, 61–67. https://doi.org/10.1038/bjc.2014.271 (2014).
Hatano, K., Fujita, K. & Nonomura, N. Application of anti-inflammatory agents in prostate Cancer. J. Clin. Med. 9. https://doi.org/10.3390/jcm9082680 (2020).
Maleki Vareki, S. High and low mutational burden tumors versus immunologically hot and cold tumors and response to immune checkpoint inhibitors. J. Immunother Cancer 6, 157. https://doi.org/10.1186/s40425-018-0479-7 (2018).
Shi, C. et al. Aspirin inhibits IKK-beta-mediated prostate Cancer Cell Invasion by Targeting Matrix Metalloproteinase-9 and urokinase-type plasminogen activator. Cell. Physiol. Biochem. 41, 1313–1324. https://doi.org/10.1159/000464434 (2017).
Choe, K. S. et al. Aspirin use and the risk of prostate cancer mortality in men treated with prostatectomy or radiotherapy. J. Clin. Oncol. 30, 3540–3544. https://doi.org/10.1200/JCO.2011.41.0308 (2012).
Downer, M. K. et al. Regular aspirin use and the risk of Lethal prostate Cancer in the Physicians’ Health Study. Eur. Urol. 72, 821–827. https://doi.org/10.1016/j.eururo.2017.01.044 (2017).
Lapi, F. et al. Risk of prostate cancer in low-dose aspirin users: a retrospective cohort study. Int. J. Cancer. 139, 205–211. https://doi.org/10.1002/ijc.30061 (2016).
Assayag, J., Pollak, M. N. & Azoulay, L. The use of aspirin and the risk of mortality in patients with prostate cancer. J. Urol. 193, 1220–1225. https://doi.org/10.1016/j.juro.2014.11.018 (2015).
Nordstrom, T., Clements, M., Karlsson, R., Adolfsson, J. & Gronberg, H. The risk of prostate cancer for men on aspirin, statin or antidiabetic medications. Eur. J. Cancer 51, 725–733. https://doi.org/10.1016/j.ejca.2015.02.003 (2015).
Ma, S. et al. Effect of aspirin on incidence, recurrence, and mortality in prostate cancer patients: integrating evidence from randomized controlled trials and real-world studies. Eur. J. Clin. Pharmacol. 79, 1475–1503. https://doi.org/10.1007/s00228-023-03556-7 (2023).
Conford, P. & Guidelines on Prostate Cancer. EAU-EANM-ESTRO-ESUR-ISUP-SIOG. https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-EANM-ESTRO-ESUR-ISUP-SIOG-Guidelines-on-Prostate-Cancer-2024_2024-04-09-132035_ypmy_2024-04-16-122605_lqpk.pdf (2024).
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Prostate Cancer. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf (2024).
Novara, G. et al. Systematic review and meta-analysis of studies reporting oncologic outcome after robot-assisted radical prostatectomy. Eur. Urol. 62, 382–404. https://doi.org/10.1016/j.eururo.2012.05.047 (2012).
Kubota, M. et al. Effect of continued perioperative anticoagulant therapy on bleeding outcomes following Robot-assisted radical prostatectomy. Urology 148, 151–158. https://doi.org/10.1016/j.urology.2020.08.095 (2021).
Suh, J. et al. Differences in risk factors for biochemical recurrence after radical prostatectomy stratified by the degree of obesity: focused on surgical methods. Sci. Rep. 10, 10157. https://doi.org/10.1038/s41598-020-67237-6 (2020).
Luo, R., Chen, Y., Ran, K. & Jiang, Q. Effect of obesity on the prognosis and recurrence of prostate cancer after radical prostatectomy: a meta-analysis. Transl Androl. Urol. 9, 2713–2722. https://doi.org/10.21037/tau-20-1352 (2020).
King, G. & Nielsen, R. Why propensity scores should not be used for matching. Political Anal. 27, 435–454. https://doi.org/10.1017/pan.2019.11 (2019).
Ho, D., Imai, K., King, G. & Stuart, E. MatchIt: nonparametric preprocessing for Parametric Causal Inference. J. Stat. Softw. 42(8), 1–28. https://doi.org/10.18637/jss.v042.i08 (2011).
The Japanese Urological Association. The Japanese Society of Pathology, Japan Radiological Society. General Rule for Clinical and Pathological Studies on Prostate Cancer 5th edn (Medical Review Co., Ltd., 2022).
Pound, C. R. et al. Natural history of progression after PSA elevation following radical prostatectomy. JAMA 281, 1591–1597. https://doi.org/10.1001/jama.281.17.1591 (1999).
R Core Team. R A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, (2024).
Diaz, M. et al. Oncologic outcomes at 10 years following robotic radical prostatectomy. Eur. Urol. 67, 1168–1176. https://doi.org/10.1016/j.eururo.2014.06.025 (2015).
Elwood, P. C. et al. Aspirin and cancer survival: a systematic review and meta-analyses of 118 observational studies of aspirin and 18 cancers. Ecancermedicalscience 15, 1258. https://doi.org/10.3332/ecancer.2021.1258 (2021).
Mantica, G. et al. Correlation between long-term acetylsalicylic acid use and prostate Cancer screening with PSA. Should we reduce the PSA Cut-off for patients in chronic therapy? A Multicenter Study. Res. Rep. Urol. 14, 369–377. https://doi.org/10.2147/RRU.S377510 (2022).
Wang, L. et al. Aspirin Use and Common Cancer risk: a Meta-analysis of Cohort studies and Randomized Controlled trials. Front. Oncol. 11, 690219. https://doi.org/10.3389/fonc.2021.690219 (2021).
Richardsen, E., Uglehus, R. D., Due, J., Busch, C. & Busund, L. T. COX-2 is overexpressed in primary prostate cancer with metastatic potential and may predict survival. A comparison study between COX-2, TGF-beta, IL-10 and Ki67. Cancer Epidemiol. 34, 316–322. https://doi.org/10.1016/j.canep.2010.03.019 (2010).
Zhang, L. et al. Positive surgical margin is associated with biochemical recurrence risk following radical prostatectomy: a meta-analysis from high-quality retrospective cohort studies. World J. Surg. Oncol. 16, 124. https://doi.org/10.1186/s12957-018-1433-3 (2018).
Fairey, A. S. et al. Long-term cancer control after radical prostatectomy and bilateral pelvic lymph node dissection for pT3bN0M0 prostate cancer in the prostate-specific antigen era. Urol. Oncol. 32, 85–91. https://doi.org/10.1016/j.urolonc.2013.03.005 (2014).
Cao, D., Kibel, A. S., Gao, F., Tao, Y. & Humphrey, P. A. The Gleason score of tumor at the margin in radical prostatectomy is predictive of biochemical recurrence. Am. J. Surg. Pathol. 34, 994–1001. https://doi.org/10.1097/PAS.0b013e3181e103bf (2010).
Savdie, R. et al. High Gleason grade carcinoma at a positive surgical margin predicts biochemical failure after radical prostatectomy and may guide adjuvant radiotherapy. BJU Int. 109, 1794–1800. https://doi.org/10.1111/j.1464-410X.2011.10572.x (2012).
Tamhankar, A. S., Patil, S. R., Ahluwalia, P. & Gautam, G. Does Continuation of Low-Dose Aspirin during Robot-assisted radical Prostatectomy Compromise Surgical outcomes? J. Endourol. 32, 852–858. https://doi.org/10.1089/end.2018.0390 (2018).
Raju, N., Sobieraj-Teague, M., Hirsh, J., O’Donnell, M. & Eikelboom, J. Effect of aspirin on mortality in the primary prevention of cardiovascular disease. Am. J. Med. 124, 621–629. https://doi.org/10.1016/j.amjmed.2011.01.018 (2011).
Rahme, E. & Nedjar, H. Risks and benefits of COX-2 inhibitors vs non-selective NSAIDs: does their cardiovascular risk exceed their gastrointestinal benefit? A retrospective cohort study. Rheumatol. (Oxford) 46, 435–438. https://doi.org/10.1093/rheumatology/kel428 (2007).
Patel, M. I. et al. Celecoxib inhibits prostate cancer growth: evidence of a cyclooxygenase-2-independent mechanism. Clin. Cancer Res. 11, 1999–2007. https://doi.org/10.1158/1078-0432.CCR-04-1877 (2005).
Pruthi, R. S. et al. Phase II trial of celecoxib in prostate-specific antigen recurrent prostate cancer after definitive radiation therapy or radical prostatectomy. Clin. Cancer Res. 12, 2172–2177. https://doi.org/10.1158/1078-0432.CCR-05-2067 (2006).
Rothwell, P. M. et al. Effect of daily aspirin on long-term risk of death due to cancer: analysis of individual patient data from randomised trials. Lancet 377, 31–41. https://doi.org/10.1016/S0140-6736(10)62110-1 (2011).
Van den Broeck, T. et al. Prognostic value of biochemical recurrence following treatment with curative intent for prostate Cancer: a systematic review. Eur. Urol. 75, 967–987. https://doi.org/10.1016/j.eururo.2018.10.011 (2019).
Gesmundo, I. et al. Proton pump inhibitors promote the growth of androgen-sensitive prostate cancer cells through ErbB2, ERK1/2, PI3K/Akt, GSK-3beta signaling and inhibition of cellular prostatic acid phosphatase. Cancer Lett. 449, 252–262. https://doi.org/10.1016/j.canlet.2019.02.028 (2019).
Acknowledgements
We would like to express our gratitude to the following people for their cooperation: Toshihide Hosomi, Masafumi Tsuruta, Naoki Hayata, Mizuho Akahane, Takashi Matsuoka, Jin Kono, and Yuki Kita, Department of Urology, Kyoto University Graduate School of Medicine; Yuto Hattori, Yohei Abe, Takanari Kambe, Naofumi Tsutsumi, Tasuku Fujiwara, Hiroki Hagimoto, Yuta Mine, Akihiko Nagoshi, Shiori Murata, Hidetoshi Kokubun, Toshinari Yamasaki, and Mutsushi Kawakita, Department of Urology, Kobe City Medical Center General Hospital; Naoto Takaoka, Hiroki Watanabe, Yurina Funahashi, Maki Fujiwara, Hiroaki Kawanishi, and Kazuhiro Okumura, Department of Urology, Tenri Yorozu Hospital; Masatoshi Kumagai, Masakatsu Ueda, Masaaki Imamura, Koji Yoshimura, Department of Urology Shizuoka General Hospital; Takahiro Kirisawa, and Ayumu Matsuda, Department of Urology, National Cancer Center Hospital; Kyohei Sugiyama, and Koji Inoue, Department of Urology, Kurashiki Central Hospital; Yu Miyazaki, Atsushi Igarashi, Hiromasa Araki, Takayoshi Miura, Mutsuki Mishina, Hiroshi Okuno, and Toru Kanno, Department of Urology, National Hospital Organization Kyoto Medical Center; Noriyuki Makita, Shinya Abe, Kazuma Hiramatsu, and Yuki Kamiyama, Department of Urology, Kyoto City Hospital; Yosuke Shimizu, Sojun Kanamaru, Department of Urology, Kobe City Nishi-Kobe Medical Center; Hideto Ota, Tatsuya Hazama, Yuta Yamada, Masahiro Tamaki, Toshifumi Takahashi, Masakazu Nakashima, and Noriyuki Ito, Department of Urology, Japanese Red Cross Wakayama Medical Center; Takehiro Takahashi, Masakatsu Hirano, and Ken Maekawa, Department of Urology, Japanese Red Cross Osaka Hospital; Kazunari Tsuchihashi, Satoshi Ishitoya, Department of Urology, Japanese Red Cross Otsu Hospital; Takeshi Soda, Ryosuke Ikeuchi, Shuhei Koike, Kosuke Nishizaki, and Takuya Okada, Department of Urology, Kitano Hospital;, Koji Nishizawa and Toru Yoshida, Department of Urology, Shiga General Hospital; Takaaki Takuma, Yuichi Uemura, and Jun Watanabe, Department of Urology, Toyooka Hospital; Daisuke Takahashi, Kana Kohashiguchi, and Takeru Fujimoto, Department of Urology, Himeji Medical Center; Kosuke Ogawa, Suguru Ito, and Kazutoshi Okubo, Department of Urology, Kyoto Katsura Hospital; Naoki Kohei, Department of Urology, Numazu City Hospital; Shotaro Hatano, Shuhei Goto, Department of Urology, Shimada General Medical Center; Toshiya Akao, Department of Urology, Rakuwakai Otowa Hospital; Tomomi Kamba, Department of Urology, Faculty of Life Sciences, Kumamoto University; Motohiro Taguchi, and Shingo Yamamoto, Department of Urology, Hyogo Medical University; Atsushi Koizumi, and Tomonori Habuchi, Department of Urology, Akita University Graduate School of Medicine; Koji Yamasaki, Department of Urology, Miyazaki University; We would also like to acknowledge the members of the Daimonji Clinical Application Database (Dai-CAD). In addition, we thank Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
Conception and design: HNe; Acquisition of data: TO, TS, SF, HO, KH, KN, RK, KS, MK, HNe; Analysis and interpretation of data: SS, HNe; Drafting of the manuscript: SS; Critical revision of the manuscript for important intellectual content: HNe, MK, TG; Statistical analysis: SS; Supervision: HNi, TG, TK; Other: DaiCAD project administration: MK, TS, RS, YS, HNe, AS, SA.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Suzuki, S., Negoro, H., Kubota, M. et al. Impact of aspirin on biochemical recurrence of prostate cancer after robot assisted radical prostatectomy in a multicenter retrospective cohort study. Sci Rep 15, 2025 (2025). https://doi.org/10.1038/s41598-025-86521-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86521-x