Abstract
Alexithymia, characterized by difficulty in expressing and recognizing emotions, is prevalent among young and middle-aged stroke survivors and can significantly impact rehabilitation outcomes. This study aims to develop and validate a dynamic nomogram to predict the risk of alexithymia in this population. This cross-sectional study was conducted from November 2022 to August 2023 at two tertiary hospitals in Jinzhou City and Cangzhou City, enrolling 319 patients. Predictive factors for alexithymia, such as Activities of Daily Living (ADL) scores, social support levels, lesion location, educational background, and National Institutes of Health Stroke Scale (NIHSS) scores, were identified through univariate and multivariate analyses. These factors were integrated into a web-based dynamic nomogram. The model’s accuracy was evaluated using Receiver Operating Characteristic (ROC) curves and 1000 bootstrap resamples. In the training cohort, 47.8% of patients were diagnosed with alexithymia. The nomogram demonstrated excellent fit and reliability, with an Area Under the Curve (AUC) of 0.837 (95% CI: 0.787–0.889) in the training cohort and 0.847 (95% CI: 0.767–0.928) in the validation cohort, enabling reliable early detection of alexithymia. The dynamic nomogram provides healthcare professionals with an important tool for early detection and management of alexithymia in young and middle-aged stroke survivors. While the model shows high predictive accuracy, its applicability may be limited to similar clinical settings. Future studies should evaluate its utility across diverse healthcare systems. This tool has the potential to significantly improve rehabilitation outcomes by supporting personalized therapeutic strategies and interventions.
Similar content being viewed by others
Introduction
Globally, stroke stands as a leading cause of death and disability, a fact underscored by Feigin et al.1. Recent decades have witnessed a significant surge in the incidence of stroke among young and middle-aged adults, a trend particularly pronounced in China where over two-thirds of stroke cases occur within this demographic2. The consequences of stroke for these younger individuals are profound, often resulting in disabilities during their most productive years. This not only affects individual health but also poses broader socio-economic challenges, as these younger patients face greater difficulties across various life domains compared to older stroke survivors3.
Alexithymia, characterized by a compromised capacity to recognize and articulate emotions, is frequently observed in individuals with neurological disorders. This condition is distinguished by notable difficulties in identifying personal feelings, conveying emotions to others, and a notable lack of externalized thinking and imagination, as delineated by Guillén et al.4. It encompasses deficits not only in emotional awareness and expression but also in distinguishing between emotions and physical sensations, a distinction further explored by Ricciardi et al.5.
Young and middle-aged individuals recovering from stroke frequently confront significant psychological distress. This distress is further heightened by an elevated risk of suicide6, depression7, and anxiety8. Key factors contributing to these psychological challenges include societal stigma9, physical disabilities, and diminished social engagement10. Alexithymia, characterized by difficulties in identifying and describing emotions, poses significant challenges for stroke rehabilitation. It adversely affects emotional and psychological well-being, influencing recovery outcomes. Research indicates that alexithymia is prevalent among patients with various neurological conditions, including stroke. Additionally, alexithymia has been associated with cognitive impairments in stroke patients, contributing to worse functional outcomes. A systematic review revealed that neuropsychological disorders, including alexithymia, are significant barriers to effective rehabilitation11. Patients with post-stroke apathy, often co-occurring with alexithymia, face challenges in engaging with therapy, further complicating recovery efforts12. The impact of alexithymia extends beyond emotional and cognitive domains. It influences social interactions, as patients often struggle to articulate their needs and emotions, limiting their ability to form supportive relationships during recovery13,14. This isolation exacerbates feelings of depression and anxiety, creating a feedback loop that impedes rehabilitation progress. Moreover, alexithymia affects moral decision-making, which is particularly relevant in rehabilitation contexts where patients must navigate complex emotional and ethical dilemmas related to their care15. The presence of alexithymia exacerbates vulnerability to mental health conditions, complicating emotional processing and intensifying the overall burden of the disease11,16. Additionally, alexithymia influences the perception of symptoms17 and can pose significant obstacles to effective treatment and rehabilitation efforts18,19.
Existing psychological assessment tools, such as the Toronto Alexithymia Scale (TAS-20) and the Perth Alexithymia Questionnaire (PAQ), are insufficient for evaluating alexithymia in young and middle-aged stroke patients20,21,22. These tools lack the adaptability to capture the fluctuating nature of alexithymia, which is influenced by situational and neurological factors, especially in stroke patients. Tools like TAS-20 and PAQ fail to effectively address these dynamic changes23. This variability highlights the necessity of developing tailored assessments that consider the dynamic emotional needs of stroke survivors. Studies demonstrate that stroke patients frequently experience significant emotional distress, which exacerbates alexithymic traits24,25. For example, the relationship between alexithymia and psychological distress is particularly pronounced in patients with chronic conditions, such as fibromyalgia, where emotional regulation plays a pivotal role in quality of life24,26. These findings suggest that stroke patients could similarly benefit from interventions targeting both alexithymia and its associated psychological distress to enhance rehabilitation outcomes27.
Cultural factors further influence the manifestation and recognition of alexithymia. For instance, emotional suppression, collectivist values, and stigma surrounding mental health in Chinese society may impact the self-reporting and diagnosis of alexithymia, underscoring the need for culturally adapted diagnostic tools. Research demonstrates that norms around emotional expression vary significantly across cultures, necessitating the development of assessment instruments tailored to specific sociocultural contexts.
In the realm of medicine, nomograms are extensively employed for the prediction and visualization of risk events, particularly in the context of disease diagnosis and prognosis. The primary aim of this study is to harness the potential of nomograms for estimating the risk of alexithymia in young and middle-aged stroke patients. We endeavor to develop a dynamic, web-based tool designed to offer an efficient, effective, and objective approach to diagnosis, thereby facilitating personalized clinical management. Advancing beyond conventional alexithymia diagnostics, our model adopts a probabilistic method to evaluate the likelihood of alexithymia’s occurrence. This approach is particularly relevant given the current scarcity of robust tools for alexithymia risk evaluation in this specific patient demographic, rendering our model a critical addition to clinical practice. Its dynamic nature, accessible via a web link, enables continuous monitoring and adaptation of intervention strategies and treatment plans. The overarching objective of this research is to introduce a precise and practical tool for the assessment of alexithymia risk, aiming to enhance the efficacy of treatment and improve the prognosis for young and middle-aged stroke survivors.
Methods
Elaborate overview of study design
This cross-sectional study, meticulously planned and executed from November 2022 to August 2023, was intricately structured in two pivotal phases, each designed to maximize the robustness and validity of the research findings. The first phase, spanning November 2022 to May 2023, focused on the assembly of the modeling group. Employing a convenience sampling strategy, our team selected 228 stroke patients from a highly-regarded tertiary hospital in Jinzhou City, Liaoning Province. This phase was instrumental in creating a foundational dataset that would inform and shape our subsequent analysis.
The second phase of the study, running from June 2023 to August 2023, shifted our focus to validating the initial findings. In this phase, 91 young and middle-aged stroke patients were selected from a tertiary hospital in Cangzhou City, Hebei Province, to form the validation group. This group played a critical role in assessing the reproducibility and external validity of the models developed from the initial cohort, thereby enhancing the overall credibility of the study.
Detailed rationale for sample size determination
The determination of sample size was guided by a thorough review of existing literature, which identified 15 potential factors influencing the incidence of alexithymia in stroke patients, with an estimated prevalence of 48.2%. Key variables included lesion location, as right hemisphere damage has been widely linked to emotional dysregulation and difficulties in emotional awareness and processing; NIHSS (National Institutes of Health Stroke Scale), a measure of stroke severity that correlates with an increased risk of psychological complications such as alexithymia; ADL (Activities of Daily Living) scores, which reflect functional limitations associated with emotional distress, with greater dependency leading to higher alexithymia risk; social support, a protective factor, as lower levels of social support are strongly associated with higher prevalence of alexithymia; and educational background, which influences cognitive and emotional processing abilities, with lower education levels often correlating with a higher likelihood of alexithymia. Employing a detailed sample size calculation formula that recommended 5–10 patients per variable and factoring in a 10% attrition rate, we arrived at a minimum requirement of approximately 173 patients for the modeling group, calculated as (15 × 5/0.482)/0.9. To maintain a balanced and statistically robust approach, we adhered to a 7:3 ratio between the modeling and validation groups, setting the minimum number for the validation group at 74 patients. The total sample size eventually included 319 patients, with 228 in the modeling group and 91 in the validation group, thus exceeding the minimum thresholds and enhancing the study’s statistical power and potential for generalizable findings.
Comprehensive inclusion and exclusion criteria
Inclusion Criteria:
-
1.
1. Confirmed stroke diagnosis through cranial CT or MRI, in accordance with the “Diagnostic Points for Various Major Cerebrovascular Diseases in China 2019”.
-
2.
2. Age range of 18–59 years, capturing a broad spectrum of the younger and middle-aged demographic.
-
3.
3. Voluntary and informed consent, along with a willingness to participate, ensuring ethical adherence and participant autonomy.
Exclusion Criteria:
-
4.
1. Presence of organic mental disorders or a history of psychiatric illness, to maintain a clear focus on stroke-related outcomes.
-
5.
2. Intellectual disability, impaired consciousness, or significant cognitive impairment, to ensure the reliability and validity of self-reported data.
-
6.
3. Communication barriers, like aphasia, to ensure accurate and complete data collection.
-
7.
4. Refusal to participate in the questionnaire survey, vital for ensuring comprehensive data collection.
-
8.
5. Experiencing other significant stressful life events during hospitalization, which could potentially skew the study results.
Ethical compliance and rigorous participant stratification
Adhering to the highest ethical standards, the study received approval from the Ethics Committee of Jinzhou Medical University and was conducted in strict alignment with the principles of the Declaration of Helsinki. The participant group was meticulously categorized based on their scores on the Toronto Alexithymia Scale (TAS-20), leading to the formation of two distinct groups: those exhibiting symptoms of alexithymia and those without. This careful stratification allowed for a more nuanced analysis of the prevalence and impact of alexithymia among the stroke patient population, shedding light on both clinical and psychological aspects of stroke recovery.
Data collection section
Methodological foundation and team training
The data collection process was underpinned by a comprehensive and systematic approach. To ensure consistency and accuracy, the research team underwent a rigorous one-day training session. This training was designed to equip the team members with a thorough understanding of the research methodology and the nuances of data collection. Such a preparatory step was critical in standardizing the data collection process across the team.
Data acquisition and patient interaction
Eligible patients, identified based on predefined criteria, were approached for data collection on the day of their admission to the hospital. The primary source of data was the hospital’s medical record system, which provided a wealth of detailed disease-related and personal information. Complementing this, additional data was gathered through direct, face-to-face interviews with the patients. These interviews not only allowed for the collection of qualitative data but also facilitated a deeper understanding of the patients’ experiences and perspectives.
During these interactions, the research team took great care to explain the study’s purpose, content, and process in a comprehensive manner. Emphasis was placed on the academic nature of the research, ensuring patients that their data would be treated with the utmost confidentiality and would not be used beyond the scope of this study without their explicit consent.
Data management and validation
In total, our study surveyed 319 patient cases. The process of data compilation and entry was meticulously managed using a dual-entry method. This involved two separate team members independently entering the data, a practice that significantly reduces the likelihood of errors and enhances the accuracy of the dataset.
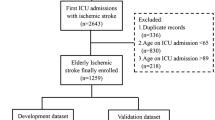
Initially, 450 young and middle-aged stroke patients were evaluated for their eligibility to participate in the study (refer to Fig. 1 for details). Of these candidates, 46 opted not to participate, while 73 did not meet the necessary inclusion criteria. This led to an initial inclusion of 331 patients. However, post-data collection, 12 questionnaires were discarded due to incompleteness, often resulting from patient non-cooperation or fatigue. Consequently, the analysis was conducted with data from 319 patients.
Nomogram of the risk prediction model for alexithymia in young and middle-aged stroke patients.
Demographic and clinical profile of the training cohort
The training cohort, comprising 228 stroke patients, presented a diverse range of demographic and clinical characteristics. Notably, 28.5% of the patients were aged between 18 and 44 years, indicating a significant representation of younger individuals. The gender distribution was predominantly male, with 66.7% of the cohort being male. In terms of daily functional abilities, 15.4% of the patients exhibited normal levels of activity. Clinically, a majority (61.0%) had right-sided lesions. The marital status of the cohort showed that a large majority (88.2%) were married. In terms of neurological impact, 54.4% of the patients displayed mild neurological impairment. Additionally, a notable proportion (47.8%) of the patients were diagnosed with alexithymia, underscoring the psychological impact of stroke in this population.
Research tool
The study used the General Demographic Characteristics Survey, the Toronto Alexithymia Scale (TAS-20), the Social Support Rating Scale (SSRS), the National Institutes of Health Stroke Scale (NIHSS), and the Activities of Daily Living Scale (ADL) as research tools.
General population characteristics questionnaire
Lesion location, NIHSS score, ADL score, place of residence, educational level, marital status, sex, age, family per capita income, first stroke onset, medical expenses, number of comorbidities and alcohol consumption.
Toronto Alexithymia Scale (TAS-20)
In this study, the Chinese version of the Toronto Alexithymia Scale (TAS-20) was used to assess patients’ alexithymia. The scale was compiled by Taylor in 198528. Yi Jie et al. translated TAS-20 into Chinese with high reliability and validity. Cronbach’s α = 0.840, retest reliability is 0.791. The scale consists of 3 subscales and 20 items, of which 7 items measure the difficulty in identifying emotions (DIF subscale), 5 items measure the difficulty in describing emotions (DDF subscale), and 8 items measure extraverted thinking (EOT subscale). Each item was rated using a 5-point Likert scale, ranging from 1 (strongly disagree) to 5 (strongly agree). Therefore, the possible range of TAS total score is 20 to 100 points. A higher score indicates a higher level of alexithymia, and a total score of > 60 indicates the presence of alexithymia.
Perceived Social Support Scale (PSSS)
The perceptive Social Support Scale (PSSS) was used to assess patients’ social support. Compiled by Zimet in 1987 and revised by Jiang Qianjin in 2001, the questionnaire is now one of the commonly used tools to measure social support in China. The questionnaire consists of 12 items, including support within the family (4 items), support from friends (4 items), and other support (4 items), 3 dimensions and 12 items. Likert 7-level scoring method was adopted for all students, with 1 to 7 points from strongly agree to strongly agree. The total score is 12–84, the higher the score, the better the level of understanding social support. Cronbach a coefficient was 0.896. The Cronbach a coefficient of this scale in this study was 0849.
National Institutes of Health Stroke Scale (NIHSS)
The NIHSS Injury Scale to measure stroke severity was used in this study (Goldstein and Samsa, 1997). Originally developed in 1989, it is now widely used in clinics and is recommended as an effective tool for assessing stroke severity in the emergency department. NIHSS includes the following areas: level of consciousness, sensation, neglect, visual field, gaze, facial palsy, motor arms, motor legs, limb ataxia, and speech and articulation disorders. Each area is scored on a scale of 0 to 2, 0 to 3, or 0 to 4. Item scores are added on a scale from 0 to 42 (the higher the score, the more severe the stroke).
Activities of Daily Living Scale (ADL)
In this study, the Activities of daily Living Scale (ADL) was used to assess the patients’ activities of daily living (Morris et al., 1999). The scale includes three assessment levels: diet, bathing, makeup and washing, dressing, stool control, urine control, toilet use, bed chair transfer, flat walking and stair climbing, each item has a score of 0, 5 and 10. The scale has a total score of 100, with higher scores indicating better ability to perform activities of daily living.
Statistical analysis
Our stroke research employed advanced statistical tools, IBM SPSS Statistics 22.0 and R, to analyze continuous and categorical variables, focusing on identifying predictors of alexithymia. We developed both static and dynamic nomograms, with the dynamic version created using R-shiny’s “DynNom” package for real-time risk prediction. This tool was built by integrating independent variables identified through logistic regression, calculating risk probabilities with weights based on their influence on alexithymia risk. Clinicians can input patient characteristics to receive instant risk assessments with 95% confidence intervals. The nomogram underwent rigorous validation, including bootstrap methods and ROC curve analysis, ensuring accuracy and generalizability.
Statistical power analysis and effect sizes
The sample size calculation for the study was based on a power level of 80% (β = 0.80) and a significance level of 0.05 (alpha), assuming a medium effect size (f2 = 0.15) based on prior stroke population studies. With 15 predictors, the required minimum sample size was calculated to be 173 participants for the modeling group, adjusted for a 10% attrition rate. The final sample size was 319 patients, with 228 in the modeling group and 91 in the validation group, which provided sufficient power for the analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated for each predictor using multivariable logistic regression. For example, right hemisphere lesions had an OR of 2.38 (95% CI: 1.38–4.16), social support had an OR of 0.97 (95% CI: 0.95–0.99), and NIHSS scores showed an OR of 4.67 for moderate stroke (95% CI: 2.57–8.66) and 25.7 for severe stroke (95% CI: 7.00–180). Statistical power was validated using Receiver Operating Characteristic (ROC) curves and bootstrap resampling (1000 resamples), with an Area Under the Curve (AUC) of 0.837 (95% CI: 0.787–0.889), indicating strong predictive accuracy.
Results
Baseline characteristics of the study population
This study included 228 stroke patients, categorized into non-alexithymic (N = 119) and alexithymic (N = 109) groups based on their scores on the Toronto Alexithymia Scale. The baseline characteristics of each group are summarized below (Table 1):
Lesion Location: A significant difference in lesion location was observed between the groups (p = 0.003). Participants with right hemisphere lesions were more likely to exhibit alexithymia (71.6% in alexithymic vs. 51.3% in non-alexithymic; OR = 2.38, 95% CI [1.38, 4.16], p = 0.002).Social Support: Lower average social support scores were associated with alexithymia (59.2 ± 12.1 in alexithymic vs. 63.8 ± 14.3 in non-alexithymic; OR = 0.97 per unit increase, 95% CI [0.95, 0.99], p = 0.011).NIH Stroke Scale (NIHSS): Severity of stroke differed significantly across groups (p < 0.001), with moderate (OR = 4.67, 95% CI [2.57, 8.66], p < 0.001) and severe strokes (OR = 25.7, 95% CI [7.00, 180], p < 0.001) being more prevalent in the alexithymic group compared to mild strokes. Activities of Daily Living (ADL): Functional status was significantly associated with alexithymia (p < 0.001). Moderate (OR = 6.77, 95% CI [2.70, 19.8], p < 0.001) and severe impairments (OR = 18.5, 95% CI [5.65, 72.3], p < 0.001) in ADL were more common among alexithymic participants. Residence: Urban residency was less commonly associated with alexithymia compared to rural (OR = 0.54, 95% CI [0.31, 0.91], p = 0.022).Education: Higher educational levels were linked with lower risk of alexithymia. Participants with college or higher education showed a significantly higher prevalence of alexithymia compared to those with junior middle school or lower (24.8% vs. 8.40%, OR = 4.92, 95% CI [2.14, 12.1], p < 0.001).Marital Status, Sex, Age, and Other Socio-demographic Factors: No significant differences were found in marital status (p = 0.229) and sex (p = 0.146) between the groups. Age and family per-capita monthly income also showed no significant differences in distribution between the alexithymic and non-alexithymic groups. First Seizure After Stroke and Number of Comorbidities: These factors did not significantly differ between groups, suggesting that the presence of alexithymia is influenced more by stroke characteristics and social support than by these variables.
Multifactorial analysis of alexithymia in young and middle-aged stroke patients
Multivariate logistic regression analysis identified key predictors of alexithymia, including lesion location, NIHSS score, ADL impairment, social support, and educational background. For instance, patients with right-sided lesions exhibited significantly higher odds of alexithymia compared to those with left-sided lesions (OR = 2.38, 95% CI: 1.38–4.16, p = 0.002). This underscores the importance of tailoring rehabilitation efforts to address emotional dysregulation associated with right hemisphere damage. Similarly, lower social support levels were strongly associated with alexithymia (OR = 0.97 per unit increase, 95% CI: 0.95–0.99, p = 0.011). This finding highlights the critical need for psychosocial interventions aimed at strengthening support systems for stroke patients, which could mitigate emotional difficulties and improve recovery outcomes. The results are presented in Table 2.
Construction of a dynamic nomogram model
Utilizing these independently correlated risk factors, we constructed a risk prediction model for alexithymia (Fig. 1). The nomogram is used to locate the position of each variable on its respective axis, and a line is drawn on the point axis to obtain the corresponding score for that risk factor. The total score is calculated by summing the scores of each factor. Based on this, an online dynamic nomogram is plotted, as shown in Fig. 2. The web page is accessible. The internal validation of the dynamic nomogram model showed an Area Under the Curve (AUC) of 0.837 (Fig. 3), with a 95% Confidence Interval (CI) of [0.787–0.889]. The calibration curves were closely aligned with the ideal curve. The external validation demonstrated an AUC of 0.847 (Fig. 4), with a 95% CI of [0.7667–0.928]. Similarly, the calibration curves in this case were also closely aligned with the ideal curve. Both closely align with the ideal diagonal line, reinforcing the model’s accuracy in predicting alexithymia risk (Figs. 5 and 6). These findings highlight the robustness of the nomogram, establishing it as a valuable tool for the early identification and intervention of individuals with alexithymia, potentially improving clinical outcomes through timely treatment.
Dynamic nomogram of the risk prediction model for alexithymia in young and middle-aged stroke patients.
Receiver operating characteristic (ROC) curve for the training cohort.
Receiver operating characteristic (ROC) curve for the validation cohort.
Effectiveness of the nomogram in predicting the risk of alexithymia in the training cohort.
Effectiveness of the nomogram in predicting the risk of alexithymia in the validation cohort.
Nomogram development and application
For example, consider a stroke patient with the following characteristics: a mild ADL score, a right-sided lesion location, a social support score of 62, a moderate NIHSS score, and an education level of junior middle school or below. Based on our model, the predicted probability of this patient achieving good functional recovery is 53.3% (95% CI: 32.1–73.3%). Each of the variables in the nomogram is assigned a specific weight, reflecting its contribution to the recovery prediction. The ADL score, representing functional impairment, decreases the likelihood of recovery, while a higher social support score positively influences the prognosis. The right-sided lesion and moderate NIHSS score account for the severity of the stroke, with right-sided lesions and higher NIHSS scores being associated with a lower recovery probability. Additionally, the education level, which is an indicator of socio-economic factors, influences recovery, with lower educational attainment associated with a less favorable outcome. By summing the points from each of these factors, clinicians can calculate the patient’s predicted probability of recovery, offering a personalized approach to treatment planning and intervention.
Explanation of figures (ROC curves and nomograms)
The ROC curves for the training (AUC = 0.8368) and validation (AUC = 0.8472) cohorts demonstrate the model’s strong predictive performance in distinguishing between patients at risk of alexithymia and those not at risk. The x-axis represents the false positive rate (FPR), while the y-axis represents the true positive rate (TPR), with the diagonal line indicating random chance. The AUC values, closer to 1, reflect the model’s ability to separate positive and negative cases effectively. The nomogram, meanwhile, provides a visual representation of the prediction model, where variables like social support, lesion location, education level, and ADL score contribute to a total score that determines the probability of alexithymia. For instance, a patient with moderate NIHSS and a right-sided lesion would accumulate a higher risk score, reflecting a greater likelihood of alexithymia.
Discussion
Alexithymia in young and middle-aged stroke patients: a research gap and our contribution
The research focus on young and middle-aged stroke patients has traditionally concentrated on physical rehabilitation and outcomes, often overlooking the prevalence and impact of alexithymia in this demographic. This oversight has resulted in a scarcity of studies constructing dynamic nomogram prediction models for alexithymia in these patients. Our study addresses this gap by identifying independent risk factors associated with alexithymia in young and middle-aged stroke survivors and developing corresponding dynamic nomogram prediction models. These models are vital for facilitating early and targeted treatment interventions. A notable finding of our study is that 47.8% of the patients exhibited alexithymia, highlighting a significant incidence rate that warrants enhanced clinical attention. Through rigorous univariate and multivariate analyses, we have identified several independent risk factors for alexithymia in this patient group. These factors include the Activities of Daily Living (ADL) scores, levels of social support, lesion location, educational attainment, and National Institutes of Health Stroke Scale (NIHSS) scores. The identification of these risk factors is crucial in formulating more effective treatment strategies. It underscores the need for healthcare providers to not only focus on the physical aspects of stroke recovery but also to proactively address the emotional and psychological challenges, particularly alexithymia, that young and middle-aged stroke patients face. This comprehensive approach can potentially enhance the overall rehabilitation process and improve the quality of life for these patients.
Impact of daily living activities on alexithymia
Our study reveals a direct correlation between the severity of impairment in daily living activities and the heightened occurrence of alexithymia. Specifically, we observed a positive correlation between higher Activities of Daily Living (ADL) scores and the incidence of alexithymia among young and middle-aged stroke patients which suggests that stroke-induced limitations in performing daily activities can substantially reduce the quality of life for these patients. The diminished ability for self-care, often necessitating assistance or being completely unachievable, can have profound emotional consequences. This is particularly true for younger patients who may face the added burden of worrying about increasing the caregiving load on their families, potentially leading to a range of adverse emotional states29,30. Furthermore, a reduced capacity for activity can limit opportunities for social interaction, thereby impeding the timely expression and processing of negative emotions and exacerbating alexithymia31. Given these insights, it is critical for medical professionals to thoroughly assess the daily living activities of stroke patients. Selecting appropriate and practical training methods tailored to the patients’ level of impairment is essential. Encouraging patients to engage in activities within their capabilities can be beneficial. Additionally, the integration of psychological and rehabilitative care is vital in addressing the mental health issues stemming from disability. Proper guidance in managing emotions and enhancing overall quality of life is essential in the comprehensive care of young and middle-aged stroke patients.
Influence of social support on alexithymia
Our study delineates a clear negative correlation between social support and the incidence of alexithymia among young and middle-aged stroke patients. We found that patients with lower levels of social support are more likely to experience higher rates of alexithymia. This finding aligns with the research conducted by Selda Celik in patients with diabetes32, and it is33. A robust social support system plays a pivotal role in the well-being of stroke patients. It provides a nurturing environment where patients feel cared for, respected, and supported, which in turn enhances their sense of existence and self-worth. This supportive atmosphere significantly increases opportunities for patients to express their emotions34. Moreover, a strong social network can effectively diminish feelings of alienation and loneliness, which are known contributing factors to alexithymia35. Given these findings, it is essential for medical staff and family members, who often represent the primary points of contact for stroke patients, to take proactive steps in strengthening their communication with patients. Healthcare professionals should be adept at patiently understanding and responding to patients’ emotional needs. Assessing the level of social support available to patients and encouraging them to actively seek and utilize effective social support is crucial. Furthermore, it is advisable for relevant departments to establish a rehabilitation communication platform specifically designed for stroke patients. Such platforms can foster a supportive social environment and enhance public awareness, understanding, and support for stroke patients through targeted public relations campaigns. By creating these supportive structures and increasing societal empathy towards stroke patients, there is a significant opportunity to reduce the prevalence and severity of alexithymia in this patient population.
Lesion location and its relation to alexithymia
Our study uncovers a significant association between the location of cerebral lesions and the occurrence of alexithymia in stroke patients. We found that patients with lesions in the right hemisphere displayed a notably higher rate of alexithymia compared to those with lesions in the left hemisphere. This result corroborates the findings of Spalletta36, which investigated the lateralization of alexithymia in stroke patients. A potential explanation for this phenomenon can be traced to the deficits typically associated with right hemisphere cortical damage, particularly in the area of facial emotion perception. Patients with right hemisphere lesions are often more challenged in recognizing negative emotions and generally exhibit more severe disorders in emotional recognition than those with left hemisphere lesions, as elucidated by Cotovio and Oliveira-Maia37. Additionally, the right hemisphere plays a crucial role in cognitive and emotional integration, involving key areas such as the corpus callosum, frontal lobe, and cingulate gyrus. These regions are responsible for regulating emotion-related autonomic functions and responses to pain. Damage to these areas is closely linked to the emergence of alexithymia, as highlighted in the research by Yount38. However, the current body of research on the structural neural abnormalities associated with alexithymia presents a complex and somewhat conflicting picture. Studies by Xiaosi Gu, Geoffrey Bird, and Björn Enzi have pointed to a relationship between alexithymia and abnormalities in the left hemisphere of the brain39,40,41. This divergence in findings underscores the necessity for continued research in this area. A more comprehensive understanding of the neural mechanisms underlying alexithymia is essential to deepen our knowledge of its pathogenesis and risk factors, which could have significant implications for the diagnosis, treatment, and management of alexithymia in stroke patients.
Educational level and its relationship with alexithymia in stroke patients
Our research has established a significant negative correlation between educational level and the severity of alexithymia in young and middle-aged stroke patients. We observed that lower educational attainment is linked with higher alexithymia scores and more pronounced symptoms. This correlation resonates with the findings from studies conducted by J.K. Salminen, P. Kokkonen, and Matthias Franz, which investigated similar trends in Finnish and German populations42,43,44.
The reasons underlying this correlation may be multifaceted. Lower educational levels often correspond with reduced cognitive capabilities, including diminished learning capacities, cognitive processing skills, critical thinking abilities, and vocabulary acquisition. These cognitive limitations can lead to poorer emotional recognition, analysis, and expression skills, thereby increasing the propensity for alexithymia. Furthermore, individuals with lower educational backgrounds may have limited access to pertinent information and resources, potentially compromising their understanding of their medical condition and their ability to effectively manage the emotional consequences associated with it45. This gap in understanding and limited emotional expression are contributing factors to heightened alexithymia scores.
Given these insights, it becomes imperative for healthcare professionals to pay closer attention to stroke patients with lower educational levels. Proactively identifying and addressing their emotional needs, and enhancing the screening for alexithymia, are vital steps. Implementing initiatives such as regular patient group meetings can offer these individuals a supportive platform to articulate their feelings and emotions. Additionally, incorporating psychological education into patient care, fostering a barrier-free communication environment, and utilizing visually engaging health education methods can significantly aid patients in understanding their emotions and the treatment process. Such initiatives not only facilitate emotional expression but are also instrumental in mitigating the occurrence of alexithymia among this patient demographic.
NIHSS score and its association with alexithymia risk
Our study highlights a critical observation: an NIHSS (National Institutes of Health Stroke Scale) score of 5 or higher is a significant risk factor for the development of alexithymia. This finding suggests that patients experiencing moderate to severe neurological impairment following a stroke are more susceptible to alexithymia. The onset of alexithymia in these patients can be attributed to the necrosis of a large number of nerve cells, leading to various neurological dysfunctions such as sensory and motor impairments. This neurologic compromise can precipitate mental symptoms, including alexithymia. Furthermore, the prevalence of alexithymia is notably higher in patients with serious physical ailments. These conclusions are in line with the findings of N A Antonova et al.46, who observed a similar trend in patients undergoing recovery from late-stage cerebral infarction. The correlation between higher NIHSS scores and increased alexithymia risk underscores the need for medical professionals to closely monitor patients with an NIHSS score of 5 or above, considering their clinical conditions. In response to this risk factor, it is essential for healthcare providers to initiate early exercise training for these patients. Such intervention is crucial not only in enhancing neuroplasticity but also in facilitating the recovery of neural function. Early and focused rehabilitation can play a significant role in mitigating the risk of alexithymia and improving the overall recovery trajectory of stroke patients, especially those with moderate to severe neurological impairments.
The advantages and clinical impact of the r-shiny-based predictive model
The construction of our dynamic predictive model using R-shiny, and its subsequent deployment on a web page through shiny apps, marks a significant step forward in the field of medical diagnostics. This model transcends the capabilities of traditional nomogram models by offering a direct and rapid assessment of the probability of occurrence and 95% confidence interval for alexithymia in young and middle-aged stroke patients. The ease of access and user-friendly interface of the model significantly enhances its utility in early screening processes. In this study, the dynamic nomogram model for alexithymia, tailored specifically for young and middle-aged stroke patients, has been meticulously developed using R-shiny. The model’s robustness is evident from its internal verification Area Under the Curve (AUC) of 0.884 and external Calibration AUC of 0.879. Moreover, both internal and external calibration curves closely align with the ideal curve, underscoring the model’s high degree of accuracy in distinguishing and calibrating. The model’s scientific reliability in predicting alexithymia among this patient group is thus firmly established. In practical clinical settings, this translates into a significant advantage for medical professionals. They can quickly ascertain the alexithymia risk probability for patients without the need for complex formulaic calculations. This immediate access to risk assessment facilitates the development of personalized intervention plans, tailored to the specific needs and risk profiles of patients. Overall, the introduction of this dynamic, web-based model represents a notable innovation in clinical practice. It not only streamlines the diagnostic process but also empowers healthcare providers to make more informed, efficient, and patient-centric decisions, ultimately enhancing the standard of care for young and middle-aged stroke patients.
Clinical relevance
To facilitate clinical application, we developed a dynamic nomogram tool, now available on shinyapps.io (https://dzzhuang.shinyapps.io/outcome/). Users can input five key patient characteristics (e.g., ADL score, NIHSS score) via computer or smartphone to calculate the real-time risk of language disorders, along with the 95% confidence interval and model parameters. The tool simplifies the process by requiring only basic demographic and medical history data, making it accessible for use in outpatient, inpatient, or telemedicine settings. It enables primary healthcare providers to screen for high-risk patients and refer them for intervention, while rehabilitation teams can identify patients needing psychological support early in recovery. The tool’s flexibility optimizes clinical decision-making and improves screening and rehabilitation efficiency. The strong association between social support and alexithymia suggests that enhancing social networks through interventions such as peer support groups, family counseling, and community outreach could be beneficial in stroke rehabilitation centers. Additionally, patients with high NIHSS scores or right hemisphere lesions could benefit from early psychological interventions aimed at improving emotional regulation and recognition, such as cognitive-behavioral therapy or emotion-focused therapy. Tailoring rehabilitation programs to address these predictors, including strategies for improving ADL functioning, could improve both psychological and functional recovery, leading to more comprehensive post-stroke rehabilitation. Future research could further evaluate the effectiveness of these targeted interventions to optimize recovery outcomes and improve the quality of life for stroke survivors.
Cultural considerations in alexithymia
Alexithymia is influenced by cultural norms around emotional expression and psychological well-being. In China, factors such as collectivism, emotional restraint, and the stigma surrounding mental health may affect the perception and self-reporting of alexithymia. Emotional suppression in such cultures could lead to underreporting difficulties in identifying or expressing emotions. These cultural factors may limit the generalizability of our findings, despite the validation of the TAS-20 scale in Chinese populations. Future studies should use culturally adapted tools or conduct cross-cultural validations and explore how cultural norms interact with alexithymia over time to deepen understanding.
Predictors of psychological complications in stroke patients
High NIHSS scores, right hemisphere lesions, and low social support are significant predictors of post-stroke psychological complications, including alexithymia, depression, and anxiety47,48,49. Elevated NIHSS scores correlate with increased risks of these conditions, indicating that greater stroke severity heightens the likelihood of psychological issues. Right hemisphere lesions are linked to deficits in emotional processing, such as impaired facial recognition and altered emotional experiences, which contribute to these complications. Additionally, low social support is associated with higher rates of post-stroke depression and anxiety, underscoring the importance of robust social networks in patient recovery. Recognizing these shared predictors suggests that interventions targeting stroke severity, right hemisphere rehabilitation, and social support enhancement could mitigate multiple psychological outcomes, thereby improving overall patient well-being and recovery.
Global perspectives and implications for stroke-alexithymia prediction models
Our study develops a dynamic nomogram for predicting alexithymia in young and middle-aged stroke patients, incorporating both clinical and psychosocial predictors. Globally, similar nomograms have been developed, but with varying emphases. For instance, a Chinese nomogram integrates demographic characteristics, lifestyle factors, and emotional aspects to predict stroke risk, highlighting the importance of emotional health in stroke outcomes50. In contrast, a UK-based model focuses on clinical variables, such as comorbidities and stroke severity, to predict mortality rates among ischemic stroke patients51. While both models share common predictors like age and comorbid conditions, our study uniquely emphasizes the role of psychosocial factors, such as social support and emotional expression.
Cultural factors play a crucial role in the expression and assessment of alexithymia, as emotional regulation varies across societies. In Western societies, where individualism and emotional introspection are emphasized, alexithymia is more readily recognized and assessed. However, in collectivist cultures, such as those in many Asian countries, emotional suppression is more common, which can affect the prevalence and self-reporting of alexithymia52. As a result, the nomogram developed in our study may need to be adjusted to account for these cultural differences in emotional expression and reporting. Understanding how cultural norms influence emotional processing is crucial for refining the model’s global applicability.
Socioeconomic status (SES) is another important factor influencing stroke outcomes and emotional health. Studies have shown that lower SES is associated with higher stroke prevalence and worse recovery outcomes. In China, higher income levels paradoxically correlate with increased stroke risk, likely due to lifestyle factors such as diet and stress53. In contrast, research from the US and Europe links lower SES with worse stroke outcomes, suggesting that access to resources, including healthcare and rehabilitation, significantly affects recovery54. Our nomogram’s emphasis on social support levels as a predictor of alexithymia is particularly relevant in this context, as socioeconomically disadvantaged individuals may have limited access to emotional support networks, which exacerbates the risk of alexithymia.
Healthcare systems also play a critical role in the effectiveness of stroke rehabilitation and emotional health interventions. Variations in healthcare access, the availability of post-stroke psychological support, and the quality of rehabilitation services can influence both the prevalence of alexithymia and the success of interventions. In regions with limited access to mental health services, such as parts of low-income countries, alexithymia may go undiagnosed, and patients may not receive the necessary support. In contrast, in high-income countries with robust healthcare systems, such as the US or UK, more comprehensive approaches to stroke rehabilitation that include psychological assessments are more likely to address alexithymia early in the recovery process55. This disparity highlights the importance of adapting the nomogram to local healthcare contexts and ensuring that it is accessible and actionable within varying resource environments.
Overcoming barriers to the adoption of the nomogram for stroke patients
A key challenge for implementing the nomogram is the training required for healthcare providers to accurately interpret it and integrate it into their decision-making processes. Many clinicians may lack familiarity with predictive models, emphasizing the need for accessible training materials such as user guides, instructional videos, or tailored workshops. Another significant barrier is resource availability, particularly in under-resourced settings where access to computers, software, or technical support may be limited. Feedback from pilot implementations in a tertiary care setting highlighted that while clinicians found the nomogram user-friendly and valuable for personalized risk assessment, seamless integration into existing workflows, such as linking it with electronic health record (EHR) systems, is crucial to automate data entry, reduce errors, and enhance efficiency. Additionally, web-based and mobile-friendly versions could ensure broader accessibility in outpatient and community healthcare settings. Targeted solutions include interactive training programs, developing lightweight and user-friendly platforms, and expanding pilot studies to diverse settings, such as primary care and rehabilitation centers, to refine usability and address user feedback. By addressing these barriers, the nomogram can become a valuable tool for improving the prediction and management of alexithymia in stroke patients across varied healthcare environments.
Interdisciplinary perspectives
We propose incorporating advanced neuroimaging techniques, such as functional MRI, in future studies to map structural and functional brain changes associated with alexithymia in stroke patients. This approach could provide direct evidence for the neural correlates of identified predictors, including lesion location and stroke severity. Psychological models, such as the emotional regulation model and the interoception framework, offer valuable theoretical perspectives for understanding alexithymia in stroke patients. These models highlight the disruption of sensory and emotional processing pathways, particularly in the context of right hemisphere lesions. Additionally, sociological insights suggest that cultural norms, such as collectivism and emotional suppression, may influence how alexithymia is expressed and reported. Integrating sociological frameworks can enhance the cultural applicability of the nomogram. By combining these interdisciplinary perspectives, we aim to deepen our understanding of the mechanisms linking identified predictors with alexithymia, thereby informing personalized treatment strategies for stroke patients.
Limitations
The cross-sectional design of this study helps identify risk factors for alexithymia but cannot establish the temporal sequence or causality between them. For example, it is unclear whether insufficient social support causes alexithymia or vice versa. Additionally, this design cannot capture the dynamic changes in risk factors and alexithymia over time. Longitudinal studies are needed to confirm temporal relationships and causal effects. Additionally, geographic and cultural limitations arise from the study’s conduct in two tertiary hospitals in Jinzhou City and Cangzhou City, potentially restricting the generalizability of findings to other regions or cultural contexts. Variations in healthcare systems, patient demographics, and cultural attitudes toward emotional expression could influence resultst. The use of convenience sampling may introduce selection bias, as the sample may not be fully representative of the broader population of young and middle-aged stroke survivors. We note that this could limit the generalizability of our findings. Future studies should consider using a more randomized sampling approach to enhance the external validity and minimize selection bias. Our study utilized self-report questionnaires, which are susceptible to response biases, including social desirability and acquiescence biases. Participants may have underreported symptoms or provided socially acceptable responses, potentially leading to an underestimation of the prevalence of alexithymia. Future research employing objective measures or clinician-administered assessments could mitigate this limitation. While we controlled for several known confounders, there may be other unmeasured variables influencing the relationship between stroke and alexithymia. Factors such as pre-stroke psychological conditions, genetic predispositions, or environmental stressors were not assessed in our study. The absence of data on these potential confounders limits our ability to fully account for all variables affecting the outcomes. Incorporating comprehensive assessments in future studies could provide a more nuanced understanding of these relationships.
Future
Given the limitations of the cross-sectional design, future research should focus on longitudinal studies to establish the temporal sequence and causal relationships between risk factors and alexithymia. Such studies could track changes in social support, cognitive function, and alexithymia over time, helping to clarify whether factors like social support lead to alexithymia, or whether alexithymia affects the perception of social support. Additionally, intervention-based studies could assess how targeted interventions, such as enhancing social support or emotional processing skills, can reduce the risk of alexithymia and improve recovery outcomes in stroke patients. Furthermore, future research should explore how these findings apply across different cultural contexts, as cultural factors may influence the expression and reporting of alexithymia. Multi-center studies involving diverse populations would enhance the generalizability and robustness of the model.
Conclusion
This study developed and validated a dynamic nomogram for predicting alexithymia in young and middle-aged stroke patients, demonstrating high predictive accuracy with AUC values of 0.837 and 0.847 in the training and validation cohorts. Key predictors include ADL scores, social support levels, lesion location, educational background, and NIHSS scores, all crucial for identifying high-risk patients and facilitating early intervention. We believe the nomogram can play a vital role in stroke rehabilitation by integrating it into routine assessments, allowing clinicians to identify high-risk patients and provide timely psychological support, potentially improving both emotional and physical recovery outcomes.
In addition to these findings, future research should explore several key areas to further enhance the understanding and application of the nomogram. First, longitudinal studies are needed to track the stability of alexithymia post-stroke and understand its evolution over time. This will provide valuable insights into whether alexithymia persists, fluctuates, or improves with recovery. Additionally, validation of the nomogram in diverse populations, including different cultural, geographic, and socioeconomic groups, will help confirm its generalizability and robustness across broader settings. Finally, clinical trials should investigate whether the use of the nomogram in clinical practice can guide targeted interventions, improving emotional and psychological support for stroke survivors and ultimately enhancing their overall rehabilitation outcomes.
Data availability
The datasets generated and/or analyzed during the current study are available upon reasonable request from the corresponding author.
References
Global National burden of stroke and its risk factors, 1990–2019: a systematic analysis for the global burden of Disease Study 2019. Lancet Neurol. 20 (10), 795–820 (2021).
Béjot, Y., Delpont, B. & Giroud, M. Rising stroke incidence in young adults: more epidemiological evidence, more questions to be answered. J. Am. Heart Assoc. 5(5) (2016).
Edwards, J. D., Kapral, M. K., Lindsay, M. P., Fang, J. & Swartz, R. H. Young stroke survivors with no early recurrence at high long-term risk of adverse outcomes. J. Am. Heart Assoc. 8(1), e010370 (2019).
Guillén, V. et al. Toronto alexithymia scale for patients with eating disorder: [corrected] performance using the non-parametric item response theory. Compr. Psychiatry 55(5), 1285–1291 (2014).
Ricciardi, L., Demartini, B., Fotopoulou, A. & Edwards, M. J. Alexithymia in neurological disease: a review. J. Neuropsychiatry Clin. Neurosci. 27(3), 179–187 (2015).
Kye, S. Y. & Park, K. Suicidal ideation and suicidal attempts among adults with chronic diseases: a cross-sectional study. Compr. Psychiatry. 73, 160–167 (2017).
Maaijwee, N. A. et al. Long-term depressive symptoms and anxiety after transient ischaemic attack or ischaemic stroke in young adults. Eur. J. Neurol. 23(8), 1262–1268 (2016).
Broomfield, N. M., Scoular, A., Welsh, P., Walters, M. & Evans, J. J. Poststroke anxiety is prevalent at the population level, especially among socially deprived and younger age community stroke survivors. Int. J. Stroke. 10(6), 897–902 (2015).
Deng, C. et al. Factors associated with stigma in community-dwelling stroke survivors in China: a cross-sectional study. J. Neurol. Sci. 407, 116459 (2019).
Woodman, P., Riazi, A., Pereira, C. & Jones, F. Social participation post stroke: a meta-ethnographic review of the experiences and views of community-dwelling stroke survivors. Disabil. Rehab. 36(24), 2031–2043 (2014).
Sampogna, F. et al. Prevalence of alexithymia in patients with psoriasis and its association with disease burden: a multicentre observational study. Br. J. Dermatol. 176(5), 1195–1203 (2017).
van Dalen, J. W., van Moll, E. P., Nederkoorn, P. J., van Gool, W. A. & Richard, E. Poststroke apathy. Stroke 44(3), 851–860 (2013).
Peng, P. et al. Alexithymia in Chinese patients with chronic schizophrenia: prevalence, clinical correlates, and relationship with neurocognition and empathy. Asia-Pac. Psychiatry 15(4), e12547 (2023).
Neumann, D., Malec, J. F. & Hammond, F. M. Reductions in alexithymia and emotion dysregulation after training emotional self-awareness following traumatic brain injury: a phase I trial. J. Head Trauma Rehab. 32(5), 286–295 (2017).
Chen, X., Wu, Z., Zhan, B., Ding, D. & Zhang, X. Effects of alexithymia on moral decision-making in sacrificial dilemmas: high alexithymia is associated with weaker sensitivity to moral norms. Psychol. Res. Behav. Manag. 16, 2315–2325 (2023).
Cherrez-Ojeda, I. et al. Alexithymia in patients with psoriasis: a cross-sectional study from Ecuador. Psychol. Res. Behav. Manag. 12, 1121–1126 (2019).
Brown, E. L., Fukuhara, J. T. & Feiguine, R. J. Alexithymic asthmatics: the miscommunication of affective and somatic states. Psychother. Psychosom. 36(2), 116–121 (1981).
Martino, G. et al. The relationship between alexithymia and type 2 diabetes: a systematic review. Front. Psychol. 11, 2026 (2020).
Vanegas, E. et al. Influence of alexithymia on the management of latin American patients with asthma: a cross-sectional study. SAGE Open Med. 8, 2050312120930913 (2020).
Larionow, P., Preece, D. A. & Mudło-Głagolska, K. Assessing alexithymia across negative and positive emotions: psychometric properties of the Polish version of the Perth Alexithymia Questionnaire. Front. Psychiatry 13, 1047191 (2022).
Preece, D. A., Petrova, K., Mehta, A., Sikka, P. & Gross, J. J. Alexithymia or general psychological distress? Discriminant validity of the Toronto Alexithymia Scale and the Perth Alexithymia Questionnaire. J. Affect. Disord. 352, 140–145 (2024).
Fynn, D. M. et al. Assessing alexithymia in adults with acquired brain injury: psychometric properties of the Perth Alexithymia Questionnaire. J. Affect. Disord. 302, 224–233 (2022).
de Haan, H. et al. Alexithymia is not a stable personality trait in patients with substance use disorders. Psychiatry Res. 198(1), 123–129 (2012).
Castelli, L. et al. Alexithymia and psychological distress in fibromyalgia: prevalence and relation with quality of life. Clin. Exp. Rheumatol. 30(6 Suppl 74), 70–77 (2012).
Castelli, L. et al. Alexithymia, anger and psychological distress in patients with myofascial pain: a case-control study. Front. Psychol. 4, 490 (2013).
Gutiérrez Hermoso, L., Velasco Furlong, L., Sánchez-Román, S. & Salas Costumero, L. The importance of alexithymia in post-surgery. Differences on body image and psychological adjustment in breast cancer patients. Front. Psychol. 11, 604004 (2020).
Liu, L. et al. Impact of alexithymia on suicidal ideation among patients with ovarian cancer: a moderated mediation model of self-perceived burden and general self-efficacy. Support. Care Cancer 31(3), 177 (2023).
Taylor, G. J., Ryan, D. & Bagby, R. M. Toward the development of a new self-report alexithymia scale. Psychother. Psychosom. 44(4), 191–199 (1985).
Rathore, S. S., Hinn, A. R., Cooper, L. S., Tyroler, H. A. & Rosamond, W. D. Characterization of incident stroke signs and symptoms: findings from the atherosclerosis risk in communities study. Stroke 33(11), 2718–2721 (2002).
McCurley, J. L. et al. Preventing chronic emotional distress in stroke survivors and their informal caregivers. Neurocrit. Care 30(3), 581–589 (2019).
Obeid, S. et al. Factors associated with alexithymia among the Lebanese population: results of a cross-sectional study. BMC Psychol. 7(1), 80 (2019).
Celik, S., Taskin Yilmaz, F., Yurtsever Celik, S., Anataca, G. & Bulbul, E. Alexithymia in diabetes patients: its relationship with perceived social support and glycaemic control. J. Clin. Nurs. 31(17–18), 2612–2620 (2022).
Du, C., Wu, S., Liu, H., Hu, Y. & Li, J. Correlation of long-term medication behaviour self-efficacy with social support and medication knowledge of kidney transplant recipients. Int. J. Nurs. Sci. 5(4), 352–356 (2018).
Ji, K. et al. Relationship between social capital and quality of life among adult stroke patients: a cross-sectional study in Anhui Province, China. Health Qual. Life Outcomes. 20(1), 19 (2022).
Lay-Yee, R. et al. Prevalence of loneliness and its association with general and health-related measures of subjective well-being in a longitudinal bicultural cohort of older adults in advanced age living in New Zealand: LiLACS NZ. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 77(10), 1904–1915 (2022).
Spalletta, G. et al. Alexithymic features in stroke: effects of laterality and gender. Psychosom. Med. 63(6), 944–950 (2001).
Cotovio, G. & Oliveira-Maia, A. J. Functional neuroanatomy of mania. Transl. Psychiatry 12(1), 29 (2022).
Yount, R. et al. Traumatic brain injury and atrophy of the cingulate gyrus. J. Neuropsychiatry Clin. Neurosci. 14(4), 416–423 (2002).
Bird, G. et al. Empathic brain responses in insula are modulated by levels of alexithymia but not autism. Brain 133(Pt 5), 1515–1525 (2010).
Enzi, B., Amirie, S. & Brüne, M. Empathy for pain-related dorsolateral prefrontal activity is modulated by angry face perception. Exp. Brain Res. 234(11), 3335–3345 (2016).
Gu, X., Hof, P. R., Friston, K. J. & Fan, J. Anterior insular cortex and emotional awareness. J. Comp. Neurol. 521(15), 3371–3388 (2013).
Franz, M. et al. Alexithymia in the German general population. Soc. Psychiatry Psychiatr. Epidemiol. 43(1), 54–62 (2008).
Kokkonen, P. et al. Prevalence and sociodemographic correlates of alexithymia in a population sample of young adults. Compr. Psychiatry 42(6), 471–476 (2001).
Salminen, J. K., Saarijärvi, S., Aärelä, E., Toikka, T. & Kauhanen, J. Prevalence of alexithymia and its association with sociodemographic variables in the general population of Finland. J. Psychosom. Res. 46(1), 75–82 (1999).
Stephenson, R. Introducing alexithymia: a concept within the psychosomatic process. Disabil. Rehab. 18(4), 209–214 (1996).
Antonova, N. A., Kuznetsova, E. B. & Sholomov, I. I. Alexithymia in patients with left-hemispheric cerebral infarction in the late recovery period. Zhurnal Nevrologii i psikhiatrii imeni SS Korsakova 115(10 Pt 2), 3–6 (2015).
Guo, J., Wang, J., Sun, W. & Liu, X. The advances of post-stroke depression: 2021 update. J. Neurol. 269(3), 1236–1249 (2022).
Álvarez-Fernández, S. et al. Emotional processing in patients with single brain damage in the right hemisphere. BMC Psychol. 11(1), 8 (2023).
Butsing, N., Zauszniewski, J. A., Ruksakulpiwat, S., Griffin, M. T. Q. & Niyomyart, A. Association between post-stroke depression and functional outcomes: a systematic review. PLoS One 19(8), e0309158 (2024).
Wu, C. et al. Development, validation, and visualization of a novel nomogram to predict stroke risk in patients. Front. Aging Neurosci. 15, 1200810 (2023).
Szlachetka, W. A. et al. Predicting 10-year stroke mortality: development and validation of a nomogram. Acta Neurol. Belg. 122(3), 685–693 (2022).
Dere, J. et al. The cultural shaping of alexithymia: values and externally oriented thinking in a Chinese clinical sample. Compr. Psychiatry. 54(4), 362–368 (2013).
Xu, F., Ah Tse, L., Yin, X., Yu, I. T. & Griffiths, S. Impact of socio-economic factors on stroke prevalence among urban and rural residents in Mainland China. BMC Public. Health 8, 170 (2008).
Yuan, Y. et al. The prevalence and characteristics of alexithymia in stroke patients: a systematic review and meta-analysis. J. Stroke Cerebrovasc. Dis. 33(7), 107712 (2024).
Naftchi, A. et al. Abstract 1122‐000134: Social and cultural barriers to global stroke care. Stroke Vasc. Interv. Neurol. 1 (S1), e12067 (2021).
Author information
Authors and Affiliations
Contributions
T.Z. and F.S. made equal contributions to this study. T.Z. was responsible for the study design, data collection and analysis, and drafting of the manuscript. F.S. was responsible for the study design, data collection and analysis and participated in drafting the manuscript. X.M. conducted the data analysis and literature research. Y.L. and F.L. contributed equally to the data collection and analysis. L.Z. handled correspondence, provided guidance, and reviewed the manuscript. All authors are responsible for the content of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
Before starting the study, all participants were briefed on its purpose and significance and provided their written consent. Data confidentiality was maintained throughout the study. The procedures adhered to the guidelines of the 1964 Helsinki Declaration and its later amendments. The study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Jinzhou Medical University (Approval Number: JZMULL2023076).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, T., Sun, F., Ma, X. et al. Development and validation of a dynamic nomogram to predict alexithymia in young and middle aged stroke patients. Sci Rep 15, 2637 (2025). https://doi.org/10.1038/s41598-025-86835-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86835-w