Abstract
The context of rapid global environmental change underscores the pressing necessity to investigate the environmental factors and high-risk areas that contribute to the occurrence of brucellosis. In this study, a maximum entropy (MaxEnt) model was employed to analyze the factors influencing brucellosis in the Aksu Prefecture from 2014 to 2023. A distributed lag nonlinear model (DLNM) was employed to investigate the lagged effect of meteorological factors on the occurrence of brucellosis. A total of 17 environmental factors were identified as affecting the distribution of brucellosis to varying degrees. The largest contributing was the normalized difference vegetation index (NDVI), followed by gross domestic product (GDP), and then meteorological factors such as average temperature, average relative humidity, and average wind speed. The receiver operating characteristic (ROC) curve demonstrated that the MaxEnt model exhibited a high degree of predictive efficacy, with an area under the curve (AUC) value of 0.921. The impact of high temperature (25℃ with a 2-month lag, RR = 3.130, 95% CI 1.642 ~ 5.965), low relative humidity (28% with a 2.5-month lag, RR = 1.795, 95% CI 1.298 ~ 2.483), and low wind speed (1.9 m/s with a 0-month lag, RR = 2.408, 95% CI 1.360 ~ 4.264) are the most significant meteorological factors associated with the incidence of brucellosis. The trends in the impact of extreme meteorological conditions on the spread of brucellosis were found to be generally consistent. Stratified analyses indicated that males were more affected by meteorological factors than females. The prevalence of brucellosis is influenced by a range of socio-economic and meteorological factors, and a multifaceted approach is necessary to prevent and control brucellosis.
Similar content being viewed by others
Introduction
Brucellosis is a zoonotic bacterial infectious disease caused by infection with the bacterium Brucella, which affects approximately 500,000 individuals annually1. It is almost always transmitted to humans from infected livestock (e.g., animals such as cattle, sheep, pigs, and dogs) as well as from contaminated livestock products, and its high-risk groups include those who live or travel in areas where the disease is endemic (e.g., pastoral areas)2. The epidemic has now reached more than 170 countries and territories globally, with annual economic losses of billions of dollars worldwide. It poses a significant threat to global public health3,4. Since the 1990s, brucellosis in China has undergone a transition from a novel outbreak to a re-emergence5. Brucellosis outbreaks have occurred in Shaanxi Province, Xinjiang Uygur Autonomous Region, and Gansu Province, which are the main epidemiologic regions for brucellosis6,7. Northwest China is primarily an area of animal husbandry, with a significant livestock breeding base in China8. The herders of Northwest China are predominantly located in rural areas, where conditions are relatively poor due to a lack of awareness regarding protection and a high risk of exposure to brucellosis in the population, particularly in light of large-scale reproduction9.
With the increasingly drastic environmental changes, the number of brucellosis cases in China has been on the rise, with the disease spreading to all provinces in the country. This trend has been observed to increase year by year, with a significant spatial correlation10. Many researchers have begun studying to the potential impact of climate change on the prevalence of brucellosis. Several studies have found possible associations between the incidence of human brucellosis in China and various environmental factors11,12,13, including temperature, precipitation, sunshine duration, relative humidity, and average wind speed6. A study conducted in Inner Mongolia, China, employed ecological niche modeling to investigate the spatial distribution of environmental influences on brucellosis14. However, the Aksu Prefecture, situated at the eastern part of the Eurasian continent, is geographically isolated from the oceans and exhibits a warm-temperate arid climate. Its economy is primarily based on animal husbandry15. Due to its distinctive geographical location and behavioural patterns, the potential for brucellosis to be a significant issue may be greater. Therefore, identifying the spatiotemporal development changes in the high-incidence areas of brucellosis will help understand the spatiotemporal distribution pattern of brucellosis in Aksu Prefecture.
In this study, Aksu Prefecture, located in northwestern China, was selected as the study area. Data from the region were collected over a 10-year period to estimate the lag effects and non-linearity of meteorological factors on the risk of brucellosis. This was accomplished by constructing a MaxEnt model and a Distributed Lag Non-Linear Model (DLNM), which facilitated the identification of environmental factors associated with a heightened risk of brucellosis in the region. This information provides a valuable foundation for the prevention and control of brucellosis in Aksu Prefecture.
Materials and methods
Study area
The Aksu Prefecture is situated in northwestern China, between 78°03′ and 84°07′ east longitude and 39°30′ and 42°41′ north latitude (Fig. S1). It is characterised by a diverse topography, including numerous mountain peaks in the north and vast plains in the south. The Aksu Prefecture is situated between the vast Taklamakan Desert in the south and the central part of the foothills of the gravelly fan-shaped land, alluvial plains area, and the Gobi. The region is characterized by medium and low hills, which are distributed in large areas and are home to numerous basins. The area is rich in natural grassland, which is a valuable resource. The region’s climate is arid throughout the year, with minimal precipitation and long sunshine hours. Climate change has had a significant impact, with notable shifts in temperature, particularly in winter and summer. The unique geographic location and well-developed animal husbandry in the Aksu region have made the region a hotspot for brucellosis. Despite the region’s status as one of the most endemic areas for brucellosis in China, no study has yet investigated the effect of environmental factors on brucellosis in Aksu Prefecture.
Data collection
The monthly average of reported brucellosis cases was obtained from the information system of the Center for Disease Control and Prevention and brucellosis surveillance data covering the period from January 1, 2014, to December 31, 2023, in Aksu prefecture, Xinjiang. The map of the administrative boundaries of Aksu prefecture was obtained from the Resource and Environmental Science Data Center of the Chinese Academy of Sciences (http://www.resdc.cn/). Utilize the Amap Application Programming Interface (API) to acquire the latitude and longitude coordinates of reported case residences, and subsequently establish a geospatial database using ArcGIS 10.7. The data and sources of environmental variables utilized in this study are presented in Table 1, which enables the reflection of environmental constraints and their impact on brucellosis16. In order to meet the data format requirements of MaxEnt software, ArcGIS 10.7 was employed to perform the following pre-processing operations: the generation of 1 km×1 km resolution raster layers for each environmental variable through the application of Kriging interpolation17, and the reclassification of land use and vegetation type classification data. All raster data were resampled to a 1 km×1 km pixel resolution and extracted from the map of Aksu prefecture as a mask, thus ensuring consistency in resolution and geographic range.
Statistical analysis
The initial analysis of the data, which included variables related to brucellosis and other factors, was conducted using both SPSS and R statistical analysis software. This study employed the MaxEnt 3.4.3 software to construct an ecological niche model for the Aksu Prefecture. The dependent variable in this model was the distribution of cases, while the independent variables were the environmental variables. The MaxEnt method, which is based on the maximum entropy principle, has been shown to have high predictive power even when used with small sample sizes18,19. In recent years, ecological niche modeling has also been employed in epidemiological studies20,21. In order to avoid multicollinearity between variables, the covariance and correlation matrix between raster data were calculated. If the correlation between two variables was greater than |0.85| 22,23, only one variable was selected to be included in the model. The risk areas were predicted using case and environmental data from 2014 to 2023. A random selection of 75% of the distribution points was designated as the training set, while the remaining 25% were utilized as the test set. To ensure the robustness of the model, a ten-fold cross-validation method was employed, whereby the modelling process was repeated 10 times. The optimal regularization multiplier and the maximum number of background points are filtered. The former is set to 0.5-2 in 0.5 increments, and the latter is set to 5,000–20,000 in 5000 increments. The final output ASCII result file is the average of the 10 repetitions. The MaxEnt modeling program evaluated the individual and collective significance of each variable in the distribution of human brucellosis cases through the Jackknife test, the average area under the curve (AUC) value over 10 model iterations, the mean percentage contribution of each variable to the model, and the elucidation of the relationship between the variables and the occurrence of brucellosis through environmental variable response curves.
The DLNM is based on the definition of a cross basis, a bi-dimensional function represented by the combination of two basis functions, describing the effects of predictors and lags24,25. This study constructed a DLNM model to explore the effect of meteorological factors on the incidence of brucellosis. The main model was as follows:
\(~Log[E(Y_{t} )] = \alpha + cb(A_{j} ,\beta _{j} ) + ns(times,df_{1} ) + ns(B,df_{2} ) + month\)
Log[E(Yt)] represents the model linked by the “log” function; Yt represents the dependent variable, which is the number of brucellosis cases observed on the t-th month. The value of t represents the observation date. The variable j denotes the lag month, while A represents the independent variable, which is temperature, relative humidity or wind speed. The corresponding coefficients are denoted by βj. The variable B indicates factors other than those included in the model (except for A); cb represents the cross-basis function, selecting the natural spline function (ns) as the basis function; According to the Quasi-Akaike information criterion (QAIC) and previous research9, the maximum lag month is defined as 4 months. Other works on the same subject set the lag to 1 and 6 months, respectively26,27. The sensitivity analysis results are shown in the Fig. S9. In particular, we changed the maximum lag months of meteorological factors (1–10 months). Based on the results and previous research28,29, the df1 of a smoothed time function in the regression model was set to 3. The degrees of freedom (df2) for confounding factors was set at 3.
Finally, we conducted an attribution analysis and examined the interactions of meteorological factors based on DLNM. Attributable Fractions (AF), also known as attributable risks, represent the proportion or percentage of all cases of a specific outcome in a population that can be attributed to a particular risk factor. We assessed additive interactions by incorporating pairs of meteorological factors into the DLNM to evaluate their combined effects on brucellosis incidence. The primary evaluation indicators were Relative Excess Risk due to Interaction (RERI), Attributable Proportion (AP), and Synergy Index (SI). These three indicators were calculated using the methodology described in the previous study30.
In addition, we conducted stratification analyses by potential individual-level association modifiers, including sex (male and female), and age (≤ 14, 15–64 and ≥ 65y). We defined the 2.5th percentile (P2.5) and the 75th percentile (P97.5) for each meteorological factor as extreme weather, with the objective of studying the effect of extreme conditions on the risk of brucellosis and population of high risk. To achieve this, we used the relative risk (RR) and cumulative relative risk (CRR) to estimate the above effects along with corresponding 95% confidence intervals (CI). The average value of each variable is used as the basic standard for calculating the RR value. All statistical analyses were conducted using the R software (version 4.2.2), with the packages dlnm and splines employed primarily. All statistical tests were two-sided, and a P-value of less than 0.05 was considered statistically significant.
Results
Data analysis
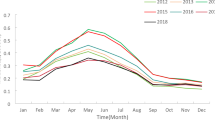
A total of 6, 027 cases were reported in Aksu Prefecture during the period from 2014 to 2023. The minimum of 4 cases, while the maximum of 208 cases, with an average of 50.23 cases per month. The summary statistics of each meteorological factor are presented in Table 2. The mean (standard deviation) of the average temperature, relative humidity, wind speed and precipitation were 9.37 (11.39) ℃, 49.04 (10.98) %, 4.81 (1.08) m/s, and 0.34 (0.35) mm, respectively. Figure 1 shows the time series curves of monthly meteorological data. A periodic pattern is evident in the mean temperature, relative humidity, wind speed, and precipitation.
The time series curves of monthly meteorology in Aksu, China, 2014–2023.
Analysis of environmental factors and spatial analysis based on the MaxEnt model
Table S1 shows the correlation analysis of the environmental factors for the period 2014–2023. When the correlation coefficient is greater than 0.85 (|r|>0.85), it is deemed that multicollinearity exists. After the final elimination of multicollinearity, the variables NDVI, GDP, TEM, RHU, WIN, SDD, NPP, and VEG were included in the MaxEnt model. The analysis of all brucellosis data from 2014 to 2023 in Aksu Prefecture showed that the cumulative contribution of NDVI, GDP, TEM, RHU, and WIN, which is an important influence on the occurrence of brucellosis. The results of the jackknife test (Fig. 2) were similar to the results in Table 3. If NDVI is omitted, it has the most information that the other variables do not. With the omission of any variable, the model is the least weakened due to the relative influence of the other variables. Fig. S2 depicts the training outcomes of the MaxEnt model. The average training area under the curve (AUC) for the replicate runs was 0.921, with a standard deviation (SD) of 0.002 (Fig. 2a). Furthermore, the model demonstrated a superior predictive fit in the test set, with an AUC of 0.920 and an SD of 0.005.
Figure 3 illustrates that the high-risk areas for brucellosis in Aksu Prefecture are primarily concentrated in the south of Baicheng County, the south of Wensu County, the northeast of Awati County, and the northwest of Kuqa City. A comparison of the reported cases with the risk zone predicted by the MaxEnt model revealed that the vast majority of cases fell within this zone. The incorporation of the aforementioned screened variables into the model for prediction resulted in a high degree of agreement when compared to real reported cases.
The exposure-response curves of the top five high-impact factors, as determined by the established niche model, are presented in Fig. S3. The curves for each variable demonstrate nonlinearity. A positive correlation was observed between the probability of human brucellosis in Aksu Prefecture and the NDVI, with an increase in the probability observed as the NDVI increased above 0.3. NDVI is the most commonly used vegetation index for detecting vegetation growth status, vegetation cover, and removing some of the radiometric errors, etc. It reflects background influences on the plant canopy, such as soil, wet ground, snow, dead leaves, roughness, etc., and is related to vegetation cover. The probability of human brucellosis exceeded 50% when the average temperature was above 0℃, the relative humidity was low, or the wind speed was low.
The results of the jackknife test of variable importance. The values presented are the mean values obtained from replicate runs. (a) The jackknife of regularized training gain test. (b) The jackknife of test gain. (c) The jackknife of AUC.
Area of potential risk for brucellosis in Aksu Prefecture. The map show the point-wise mean and standard deviation of the 10 output grids. The map was drawn from Maximum Entropy Species Distribution Modeling, Version 3.4.4 (https://biodiversityinformatics.amnh.org/open_source/maxent/).
The associations between meteorological factors and the transmission of human brucellosis
Non-linear and lagged effects of meteorological factors on the transmission of human brucellosis
Figure 4 illustrates the correlation between climate variables in varying time lags and the risk of brucellosis. Referring to the average monthly temperature (9.37 ºC), relative humidity (49.04%), and wind speed (4.81 m/s) in Aksu Prefecture from 2014 to 2023, each meteorological factor demonstrated a non-linear trend with the risk of brucellosis in the different lag months. Figure 5 presents a curve plot of the meteorological factors and the risk of brucellosis at lag 1–4 months. The results indicated that low temperatures exhibited a protective effect, whereas high temperatures were identified as a risk factor for brucellosis. The protective or risk effects exhibited a gradual decrease with the extension of the lag time. When the lag period was two months and the temperature was 25 ℃, the relative risk reached its maximum (RR = 3.130, 95% CI 1.642 ~ 5.965). With regard to relative humidity, the impact of low or high humidity on the incidence of brucellosis was more pronounced in a shorter lag period. Low relative humidity is a risk factor for brucellosis, with the effect decreasing as humidity increases. The relative humidity at 28% at lag 2.5 months exhibited the greatest effect on brucellosis, with an RR of 1.795 (95% CI 1.298 ~ 2.483). The risk of brucellosis will increase when the relative humidity is above 68%. With regard to wind speed, the risk impact is greater at wind speeds of 1.9 m/s (lag 0 month, RR = 2.408, 95% CI 1.360 ~ 4.264). Low wind speeds facilitate the spread of human brucellosis in Aksu prefecture, with a lag effect on its spread of more than one month when wind speeds reach 3–4.5 m/s.
Contour graphs and Three-dimension of exposure-lag-response for meteorological factors and brucellosis. (A) temperature. (B) relative humidity. (C) wind speed.
Plot of meteorological factors and brucellosis risk with a lag of 1 to 4 months. (A) temperature. (B) relative humidity. (C) wind speed.
Single-lag effects of extreme meteorological factors
The distribution of the lagged effects of P2.5, P25, P75, and P97.5 on the risk of brucellosis for each meteorological factor was plotted using the mean value of each meteorological factor as a reference value (Fig. S4). The results demonstrated that low temperatures (P2.5 and P25), extreme high relative humidity (P75 and P97.5), and high wind speeds (P75 and P97.5) all exhibited a modest and essentially equal effect on the risk of brucellosis transmission. Conversely, the effect of extremely hot weather on the risk of brucellosis was more significant. The effect of temperature on brucellosis risk increases and then decreases with lag time. Similarly, the effect of low relative humidity on the risk of brucellosis exhibited a slight increase with increasing lag time.
Furthermore, the total population and the at-risk population were examined in conjunction with various meteorological variables under P2.5 and P97.5 conditions. The lagged effects of meteorological factors on the total population are illustrated in Fig. S5, and their specific values are shown in Table S2. The results indicated that the RR values of extreme monthly high temperature (P97.5: 24.29 °C) were all greater than 1 at lags of 1–4 months, and the results were statistically significant. Notably, the results showed that the RR value of extreme monthly low temperature (P2.5: −9.63 °C) exceeded 1 at lags of 0 and 0.5 months, yet was not found to be statistically significant. For extreme relative humidity, the results showed that the RR values were all greater than 1 and statistically significant at extreme low relative humidity (P2.5: 31.85%). Extreme low monthly wind speed (P2.5: 3.01 m/s) had RR values greater than 1 and statistically significant at a lag of 0–1 month. Fig. S6 shows the results after stratifying the analysis by gender. The trend of effects across gender and age over the lag period of the study was consistent with the overall. Among these, high temperature (P97.5: 24.29 °C) had the largest effect effect value on males at a lag of 2 months. Low relative humidity (P2.5: 31.85%) presented as a risk factor throughout the lag period, but there was no major difference in the effect on men and women. Low wind speed (P2.5: 3.01 m/s) showed an immediate effect, such as the risk was greatest in the lag 0 month, with a consistent trend between genders, and no significant differences were observed. The analysis of brucellosis risk associated with extreme meteorological factors, stratified by age groups (≤ 14 years, 15–64 years, and ≥ 65 years), is presented in Fig. S7. The findings indicate that individuals aged 65 years and older are more susceptible to brucellosis compared to those aged 15 to 64 years. Additionally, extreme low relative humidity was associated with a high risk of morbidity across all age groups. The impact of extreme low wind speed at a lag of 0 months was found to be more pronounced in individuals under the age of 14. However, no significant variations in the observed trends were noted across the different age groups.
Cumulative lag effect of meteorological factors
Figure S8 illustrates the cumulative effects of meteorological factors on brucellosis. The relationships between temperature, relative humidity, and wind speed and the risk of brucellosis are characterized by ‘S-shaped,’ ‘W-shaped,’ and ‘N-shaped’ curves, respectively. These results align with the exposure-response curves derived from the MaxEnt model. Temperatures below the mean reference of 9.37 °C exert a protective effect against brucellosis. However, once temperatures exceed 9.37 °C, the risk of brucellosis increases with rising temperature. Specifically, when the temperature was less than or equal to 9 °C, the temperature was the cumulative relative risk (CRR) of brucellosis was 0.927 (95% CI 0.871 ~ 0.988). For relative humidity, CRR values exceed 1 when humidity levels are less than or equal to 50%, with the relative risk of brucellosis increasing as relative humidity declines. Conversely, when relative humidity is greater than or equal to 69% (CRR = 1.054, 95% CI 0.710 ~ 1.565), the relative risk gradually increases, although no statistically significant difference was observed. Regarding wind speed, the cumulative lag effect on brucellosis peaks at wind speeds of 3.9 m/s, with a CRR of 4.972 (95% CI 3.325 ~ 7.435).
Disease burden analysis of meteorological factors on the transmission of human brucellosis
The results of the analysis of attributable fractions and numbers of brucellosis caused by meteorological factors are presented in Table 4. The results from the attributable analysis are consistent with the ranking of the percentage contribution of meteorological factors in Max-Ent model. The burden of brucellosis influenced by temperature was found to be higher among males and populations aged 64 years and older. Specifically, 12.098% (95% CI: 12.016%~12.277%) of brucellosis were attributable to temperature, with a significant difference observed between the sexes. Brucellosis was attributable to temperature in 13.465% (95% CI: 12.821%~15.184%) of populations aged 64 years and older. The burden of relative humidity (AF = 12.440%, 95% CI: 11.308%~12.421%) and wind speed (AF = 7.404%, 95% CI: 6.152%~8.354%) was higher in populations under 15 years of age.
Additive interaction effects of different meteorological factors on human brucellosis
The additive interactions of meteorological factors on human brucellosis are presented in Table 5. The results indicate that the additive interaction effect of temperature and relative humidity was greatest for males (RERI = 0.974, 95% CI 0.237 ~ 1.710) and for individuals aged 64 years and older (RERI = 0.902, 95% CI 0.273 ~ 1.532). Additionally, the interaction between temperature and wind speed was found to increase the risk of human brucellosis, with a more pronounced impact on males (RERI = 0.979, 95% CI 0.030 ~ 1.929) and individuals over 64 years of age (RERI = 0.697, 95% CI 0.543 ~ 0.852). Conversely, the interaction between relative humidity and wind speed did not demonstrate a significant additional effect on human brucellosis.
Discussion
In this study, we first estimated the probability distribution of environmental factors and brucellosis from 2014 to 2023 by the MaxEnt model. This was done in order to explore the environmental factors that affect the occurrence of brucellosis in Aksu Prefecture and to map the potential high-risk areas. Secondly, among the environmental factors with high contribution based on the MaxEnt model, meteorological factors were selected to construct the DLNM model to explore the correlation analysis and lag effect of meteorological factors that have a large impact on brucellosis in Aksu Prefecture. The model effectively evaluated the relationship between meteorological factors and climate-sensitive infectious diseases, including hand, foot and mouth disease, dengue fever and chickenpox31,32,33,34. The results of our study showed that the risk of brucellosis in Aksu Prefecture was nonlinearly correlated with environmental factors such as NDVI, GDP, temperature, relative humidity and wind speed. A positive correlation was observed between NDVI, GDP, and temperature, while a negative correlation was evident between relative humidity and wind speed with a certain lag effect.
The largest contribution was NDVI, which is similar to another Chinese study35. It found that the effect of NDVI was more significant in predicting brucellosis based on environmental factors. Another study also showed that the incidence of human brucellosis was significantly associated with increases in mean temperature and NDVI36. Dense vegetation can protect bacteria from UV radiation and desiccation. The second highest contribution of environmental factors to brucellosis was GDP. GDP was positively correlated with the occurrence of brucellosis in Aksu Prefecture. A study of spatial and temporal trends and drivers of human brucellosis in China, based on interpretable analyses, found that the expansion of human brucellosis was positively correlated with GDP and negatively correlated with humidity37. Overcrowded or densely populated herds or flocks increase the chances of direct animal-to-animal transmission. It indicated that higher GDP contributes to improved sanitation and increased access to health care, which in turn increases the likelihood of detecting disease. The dependency plot between it and SHapley Additive exPlanations (SHAP) values is consistent with the corresponding plot of exposure in this study. Another analysis of the spatial and temporal distribution of human brucellosis and socioeconomic factors in mainland China also showed a positive correlation between GDP and high temperature38. Through the above discussion and in conjunction with the previous39,40, we can conclude that socioeconomic drivers may play a crucial role in the expansion of human brucellosis. As people’s standard of living rises, the demand for animal protein increases, leading to an increase in livestock production, slaughter and transportation for meat consumption41, and more people are thus exposed to animal hosts and their vectors, thereby increasing the risk of disease transmission to humans5.
The meteorological factors that contributed more to the occurrence of brucellosis in Aksu Prefecture were temperature, relative humidity, and wind speed, in that order. Research has shown that average temperature, wind speed, and relative humidity have a significant impact on brucellosis from May to July every year (warm season)15. This is consistent with the meteorological factors (average temperature, relative humidity, and wind speed) included in the DLNM in this study. The results of this study showed that high temperature, low humidity and low wind speed are the main risk factors for brucellosis transmission with certain lag and cumulative effects. It may be related to the natural environment suitable for the survival of Brucella. Previous studies have found that arid areas are more prone to brucellosis42. Our study found that the lagged effect of temperature at 25 ℃ reached its maximum at 2 months lag, and its maximum cumulative effect reached its maximum at 4 months. This may be due to the particular geographic location of the Aksu region, which leads to extensive feeding activities in late spring, when temperatures provide a favorable environment for Brucella and good conditions for host animals to reproduce, thus increasing the risk of infection from exposure to these infected animals43. Warmer temperatures increase the chances of bacterial survival, similar to other infectious diseases, in a relationship that reflects complex interactions at the pathogen-host-human interface. This is more conducive to bacterial production, adaptation and transmission, thus increasing the risk of illness and infection44,45. However a study from Inner Mongolia, China, suggests that cooler regions are more likely to be highly endemic for human brucellosis, which is less consistent with the results of this study46. This may be due to the geographical location and environment of different regions47. Meanwhile we are in agreement with the results of more studies. For example the results of a study in Datong showed the highest delayed effect on brucellosis with a lag of 4 months at low monthly temperatures7. Another result from Yongding City, Gansu Province, showed that temperature and hours of sunshine increased the risk of brucellosis at different lag times27.The warm season favors rutting in animals (e.g., cattle, sheep, dogs, etc.), which not only increases sheep densities but also promotes the spread of brucellosis among animals. This also increases livestock production and thus the likelihood of contact between humans and infected animals26.
For relative humidity, our findings are consistent with those of a study in Shaanxi, where the risk and lag effect of human brucellosis were greater in environments with low relative humidity12. In addition, the results of a study on the spatial and temporal distribution of human brucellosis and the factors influencing it have shown that low relative humidity plays a crucial role in the survival and activity of pathogens in the environment46 and there is a negative correlation, which directly or indirectly affects the enzymatic activity of Brucella and its survival and spread48. For wind speed, our study found that extreme low wind speeds increased the risk of brucellosis transmission after 0-1.5 months. A study from Jilin, China, indicated that low wind speed (1.4 m/s) was a risk factor for brucellosis at a two-month lag, with a maximum RR of 1.68 (95% CI = 1.25 ~ 2.26) and a cumulative lag effect9. Another study found that low wind speed lagging 1 month predicted the highest incidence of brucellosis49. High wind speeds can cause bacteria to have a shorter lifespan in the air48, low wind speeds can cause air to stagnate, especially in enclosed or semi-enclosed environments (e.g., domesticated animals), promoting the retention and spread of brucellosis in the local environment50.
In this study, stratified analyses revealed that the risk of brucellosis was higher in males and ≥ 65 years of age at extreme high temperatures, whereas both males and females were at greatest risk of infection in the month of exposure at low wind speeds. The interaction of temperature and wind speed puts people over the age of 65 at increased risk of infection, possibly due to climatic conditions of high temperatures and low wind speeds that favor the growth and reproduction of Brucella. When temperatures rise, Brucella bacteria have an increased rate of metabolism and an increased ability to reproduce, while low wind speeds reduce air currents, making it easier for the bacteria to become trapped and spread through the air. As we age, the body’s immune system weakens and becomes less resistant to pathogens. As a result, people over the age of 65 are more susceptible to Brucella infections. A study in the Kingdom of Saudi Arabia found a significantly higher risk of brucellosis in male than in female during the warm season51. This is consistent with the results of the present study, which may be due to the fact that male prime age is more likely to go out for grazing in late spring and summer, thus increasing the risk of infection by exposure to high temperatures and low wind speed52. When temperatures are relatively warm, people over 65 are more likely to sit on doorsteps in the sun or be out and about, and they are in close contact with sheep, goats and dogs as they play and feed the animals, increasing their risk of contracting brucellosis. Our study shows that extreme high relative humidity and extreme high wind speeds do not differ in trend by gender and are not statistically significantly different in increasing the transmission of brucellosis. The increased likelihood of couples going out to graze together in low wind speed also increases the risk of exposure to animals such as cattle and sheep, which increases the risk of bacterial infections.
Humans tend to keep animals in captivity during the winter and spring. However, this is also the period when small ruminants are usually in the cycle of pregnancy, abortion and birth. Their secretions containing high levels of Brucella spp. are released into the environment, accelerating animal-to-animal transmission during such periods of susceptibility. In addition, climatic factors are critical in increasing or decreasing human exposure to infected animals. Our study significantly enhances the understanding of the environmental factors influencing the high-risk areas of brucellosis, particularly in Aksu Prefecture, China. By analyzing the interplay between ambient temperature, relative humidity, and wind speed, we have identified critical conditions that exacerbate the incidence of brucellosis. Specifically, we found that elevated ambient temperatures, coupled with low relative humidity and reduced wind speed, are correlated with an increased activity of brucellosis in regions such as Baicheng and Wensu counties. These findings are particularly pertinent given the ongoing challenges posed by climate change, which may further alter these environmental conditions and potentially escalate the risk of brucellosis outbreaks. Understanding these dynamics is crucial for local health departments, as it provides a scientific basis for developing targeted and effective prevention strategies. Moreover, our research underscores the importance of continuous environmental monitoring and the need for integrated approaches that consider both ecological and epidemiological factors. By establishing predictive models based on environmental indicators, health authorities can better anticipate and mitigate the risks associated with brucellosis.
There are some limitations in our study. The study area of this study is only a part of Northwest China, and there is a need to conduct relevant studies on larger areas in the future. In addition, other meteorological factors (e.g., dusty weather) that may have a greater impact could not be taken into account. Finally, more possible potential individual factors (e.g., level of exposure to animals, etc.) could not be taken into account due to the lack of individual data.
In summary, this study not only contributes to the existing body of knowledge regarding brucellosis transmission but also serves as a vital resource for policymakers and public health officials in Aksu Prefecture. The insights gained from this research can guide the formulation of proactive health interventions, ultimately aiming to reduce the burden of brucellosis and protect the health of local communities.
Conclusion
This study provides the first evidence for the close relationship between brucellosis transmission and climatic factors in Aksu Prefecture, Xinjiang. The results showed that Baicheng County and Wensu County were the high incidence areas of brucellosis in Aksu Prefecture. High temperature, low relative humidity, and low wind speed were found to be more favorable for brucellosis activity in Aksu Prefecture, and the lag effect was observed to be significant over a four-month period. The findings of this study provide a valuable reference for local public health personnel in the formulation of preventive and control measures.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to governance restrictions or the identifiable nature of the data, but are available from the corresponding author (send requests to 357935099@qq.com) on reasonable request.
References
Shakir, R. & Brucellosis. J. Neurol. Sci. 420, 117280. https://doi.org/10.1016/j.jns.2020.117280 (2021).
Zhai, M. et al. Research on the predictive effect of a combined model of ARIMA and neural networks on human brucellosis in Shanxi Province, China: a time series predictive analysis. BMC Infect. Dis. 21, 280. https://doi.org/10.1186/s12879-021-05973-4 (2021).
Ran, X. et al. Brucellosis seroprevalence in dairy cattle in China during 2008–2018: a systematic review and meta-analysis. Acta Trop. 189, 117–123. https://doi.org/10.1016/j.actatropica.2018.10.002 (2019).
Wang, Y. et al. Temporal trends analysis of human brucellosis incidence in mainland China from 2004 to 2018. Sci. Rep. 8, 15901. https://doi.org/10.1038/s41598-018-33165-9 (2018).
Ran, X. et al. Brucellosis seroprevalence in ovine and caprine flocks in China during 2000–2018: a systematic review and meta-analysis. BMC Vet. Res. 14, 393. https://doi.org/10.1186/s12917-018-1715-6 (2018).
Li, Y. J., Li, X. L., Liang, S., Fang, L. Q. & Cao, W. C. Epidemiological features and risk factors associated with the spatial and temporal distribution of human brucellosis in China. BMC Infect. Dis. 13, 547. https://doi.org/10.1186/1471-2334-13-547 (2013).
Guan, P., Wu, W. & Huang, D. Trends of reported human brucellosis cases in mainland China from 2007 to 2017: an exponential smoothing time series analysis. Environ. Health Prev. Med. 23, 23. https://doi.org/10.1186/s12199-018-0712-5 (2018).
Sun, M. J. et al. Genotyping of Brucella melitensis and Brucella abortus strains currently circulating in Xinjiang, China. Infect. Genet. Evol. 44, 522–529. https://doi.org/10.1016/j.meegid.2016.07.025 (2016).
Huang, S. et al. Risk effects of meteorological factors on human brucellosis in Jilin province, China, 2005–2019. Heliyon 10, e29611. https://doi.org/10.1016/j.heliyon.2024.e29611 (2024).
Yang, Z. R. et al. Characteristics on spatial and temporal distribution as well as the driving effect of meteorological factors on brucellosis in Datong City, Shanxi Province, 2005–2015]. Zhonghua Liu Xing Bing Xue Za Zhi 39, 1165–1171. https://doi.org/10.3760/cma.j.issn.0254-6450.2018.09.005 (2018).
Sun, Z. X. et al. Socioeconomic, meteorological factors and spatiotemporal distribution of human brucellosis in China between 2004 and 2019-A study based on spatial panel model. PLoS Negl. Trop. Dis. 17, e0011765. https://doi.org/10.1371/journal.pntd.0011765 (2023).
Yang, Z. et al. Spatiotemporal expansion of human brucellosis in Shaanxi Province, Northwestern China and model for risk prediction. PeerJ 8, e10113. https://doi.org/10.7717/peerj.10113 (2020).
Chen, H. et al. Driving role of climatic and socioenvironmental factors on human brucellosis in China: machine-learning-based predictive analyses. Infect. Dis. Poverty 12, 36. https://doi.org/10.1186/s40249-023-01087-y (2023).
Jia, P. & Joyner, A. Human brucellosis occurrences in inner mongolia, China: a spatio-temporal distribution and ecological niche modeling approach. BMC Infect. Dis. 15, 36. https://doi.org/10.1186/s12879-015-0763-9 (2015).
Cao, B. et al. Tracing the future of epidemics: Coincident niche distribution of host animals and disease incidence revealed climate-correlated risk shifts of main zoonotic diseases in China. Glob. Chang. Biol. 29, 3723–3746. https://doi.org/10.1111/gcb.16708 (2023).
Alemu, W. G. & Wimberly, M. C. Evaluation of remotely sensed and interpolated environmental datasets for vector-borne disease monitoring using in situ observations over the Amhara Region, Ethiopia. Sens. (Basel) 20 https://doi.org/10.3390/s20051316 (2020).
Sun, J. M. et al. Factors associated with spatial distribution of severe fever with thrombocytopenia syndrome. Sci. Total Environ. 750, 141522. https://doi.org/10.1016/j.scitotenv.2020.141522 (2021).
Cao, Y. T. et al. Maximum entropy modeling the distribution area of morchella dill. Ex pers. Species in China under changing climate. Biology (Basel) 11 https://doi.org/10.3390/biology11071027 (2022).
Tang, X., Yuan, Y., Li, X. & Zhang, J. Maximum entropy modeling to predict the impact of climate change on pine wilt disease in China. Front. Plant. Sci. 12, 652500. https://doi.org/10.3389/fpls.2021.652500 (2021).
Pigott, D. M. et al. Mapping the zoonotic niche of ebola virus disease in Africa. Elife 3, e04395. https://doi.org/10.7554/eLife.04395 (2014).
Johnson, E. E., Escobar, L. E. & Zambrana-Torrelio, C. An ecological framework for modeling the geography of disease transmission. Trends Ecol. Evol. 34, 655–668. https://doi.org/10.1016/j.tree.2019.03.004 (2019).
Song, S. et al. Impacts of environmental heterogeneity on moss diversity and distribution of didymodon (Pottiaceae) in Tibet, China. PLoS ONE 10, e0132346. https://doi.org/10.1371/journal.pone.0132346 (2015).
Dai, X. et al. MaxEnt model-based prediction of potential distributions of parnassiawightiana (Celastraceae) in China. Biodivers. Data J. 10, e81073. https://doi.org/10.3897/BDJ.10.e81073 (2022).
Gasparrini, A. Distributed lag linear and non-linear models in R: the package dlnm. J. Stat. Softw. 43, 1–20 (2011).
Gasparrini, A., Armstrong, B. & Kenward, M. G. Distributed lag non-linear models. Stat. Med. 29, 2224–2234. https://doi.org/10.1002/sim.3940 (2010).
Liu, K. et al. Effect of climatic factors on the seasonal fluctuation of human brucellosis in Yulin, northern China. BMC Public Health 20, 506. https://doi.org/10.1186/s12889-020-08599-4 (2020).
Zheng, H. et al. Influence and prediction of meteorological factors on brucellosis in a northwest region of China. Environ. Sci. Pollut. Res. Int. 30, 9962–9973. https://doi.org/10.1007/s11356-022-22831-1 (2023).
Di, Q. et al. Association of short-term exposure to air pollution with mortality in older adults. Jama 318, 2446–2456. https://doi.org/10.1001/jama.2017.17923 (2017).
Xu, M. M. et al. Analysis on influence and lag effects of meteorological factors on incidence of hand, foot and mouth disease in Shijiazhuang, 2017–2019. Zhonghua Liu Xing Bing Xue Za Zhi 42, 827–832. https://doi.org/10.3760/cma.j.cn112338-20200930-01213 (2021).
Wu, D. et al. Investigating the impact of extreme weather events and related indicators on cardiometabolic multimorbidity. Arch. Public Health 82, 128. https://doi.org/10.1186/s13690-024-01361-x (2024).
Hao, J. et al. Impact of ambient temperature and relative humidity on the incidence of hand-foot-mouth disease in Wuhan, China. Int. J. Environ. Res. Public Health 17 https://doi.org/10.3390/ijerph17020428 (2020).
Xiao, X. et al. The exposure-response relationship between temperature and childhood hand, foot and mouth disease: a multicity study from mainland China. Environ. Int. 100, 102–109. https://doi.org/10.1016/j.envint.2016.11.021 (2017).
Zhang, T., Qin, W., Nie, T., Zhang, D. & Wu, X. Effects of meteorological factors on the incidence of varicella in Lu’an, Eastern China, 2015–2020. Environ. Sci. Pollut. Res. Int. 30, 10052–10062. https://doi.org/10.1007/s11356-022-22878-0 (2023).
Xiang, J. et al. Association between dengue fever incidence and meteorological factors in Guangzhou, China, 2005–2014. Environ. Res. 153, 17–26. https://doi.org/10.1016/j.envres.2016.11.009 (2017).
Zhao, Y. et al. Prediction of human brucellosis in China based on temperature and NDVI. Int. J. Environ. Res. Public Health 16 https://doi.org/10.3390/ijerph16214289 (2019).
Peng, R. et al. Driving effect of multiplex factors on human brucellosis in high incidence region, implication for brucellosis based on one health concept. One Health 15, 100449. https://doi.org/10.1016/j.onehlt.2022.100449 (2022).
Wen, X., Wang, Y. & Shao, Z. The spatiotemporal trend of human brucellosis in China and driving factors using interpretability analysis. Sci. Rep. 14, 4880. https://doi.org/10.1038/s41598-024-55034-4 (2024).
Peng, C., Li, Y. J., Huang, D. S. & Guan, P. Spatial-temporal distribution of human brucellosis in mainland China from 2004 to 2017 and an analysis of social and environmental factors. Environ. Health Prev. Med. 25, 1. https://doi.org/10.1186/s12199-019-0839-z (2020).
Chen, S. et al. Increasing threat of brucellosis to low-risk persons in urban settings, China. Emerg. Infect. Dis. 20, 126–130. https://doi.org/10.3201/eid2001.130324 (2014).
Tan, Z. et al. A familial cluster of human brucellosis attributable to contact with imported infected goats in Shuyang, Jiangsu Province, China, 2013. Am. J. Trop. Med. Hyg. 93, 757–760. https://doi.org/10.4269/ajtmh.15-0149 (2015).
Zhao, C. et al. Epidemic characteristics and transmission risk prediction of brucellosis in Xi’an city, Northwest China. Front. Public Health 10, 926812. https://doi.org/10.3389/fpubh.2022.926812 (2022).
Silva, I., Dangolla, A. & Kulachelvy, K. Seroepidemiology of Brucella abortus infection in bovids in Sri Lanka. Prev. Vet. Med. 46, 51–59. https://doi.org/10.1016/s0167-5877(00)00136-7 (2000).
Lee, H. S., Her, M., Levine, M. & Moore, G. E. Time series analysis of human and bovine brucellosis in South Korea from 2005 to 2010. Prev. Vet. Med. 110, 190–197. https://doi.org/10.1016/j.prevetmed.2012.12.003 (2013).
Xu, L. & Deng, Y. Spatiotemporal pattern evolution and driving factors of brucellosis in China, 2003–2019. Int. J. Environ. Res. Public Health 19 https://doi.org/10.3390/ijerph191610082 (2022).
Revich, B., Tokarevich, N. & Parkinson, A. J. Climate change and zoonotic infections in the Russian Arctic. Int. J. Circumpolar. Health 71, 18792. https://doi.org/10.3402/ijch.v71i0.18792 (2012).
Liang, D. et al. Spatiotemporal distribution of human brucellosis in Inner Mongolia, China, in 2010–2015, and influencing factors. Sci. Rep. 11, 24213. https://doi.org/10.1038/s41598-021-03723-9 (2021).
Imai, C., Armstrong, B., Chalabi, Z., Mangtani, P. & Hashizume, M. Time series regression model for infectious disease and weather. Environ. Res. 142, 319–327. https://doi.org/10.1016/j.envres.2015.06.040 (2015).
Cao, L. T. et al. Relationship of meteorological factors and human brucellosis in Hebei Province, China. Sci. Total Environ. 703, 135491. https://doi.org/10.1016/j.scitotenv.2019.135491 (2020).
Mohammadian-Khoshnoud, M., Sadeghifar, M., Cheraghi, Z. & Hosseinkhani, Z. Predicting the incidence of brucellosis in Western Iran using Markov switching model. BMC Res. Notes 14, 79. https://doi.org/10.1186/s13104-020-05415-5 (2021).
Poli, C. et al. Management strategies for lamb production on pasture-based systems in subtropical regions: a review. Front. Vet. Sci. 7, 543. https://doi.org/10.3389/fvets.2020.00543 (2020).
Aloufi, A. D., Memish, Z. A., Assiri, A. M. & McNabb, S. J. Trends of reported human cases of brucellosis, Kingdom of Saudi Arabia, 2004–2012. J. Epidemiol. Glob. Health 6, 11–18. https://doi.org/10.1016/j.jegh.2015.09.001 (2016).
Bagheri, H., Tapak, L., Karami, M., Amiri, B. & Cheraghi, Z. Epidemiological features of human brucellosis in Iran (2011–2018) and prediction of brucellosis with data-mining models. J. Res. Health Sci. 19, e00462 (2019).
Acknowledgements
This study was supported and funded by grants from the National Natural Science Foundation of China and the "Tianshan Talents" medical and health high-level talent cultivation program. We thank the Center for Disease Control and Prevention of Xinjiang Uygur Autonomous Region and Aksu for its invaluable assistance with administration, and data collection.
Funding
This work was supported by “Tianshan Talents” Program for Training High-Level Talents in Medicine and Healthcare (Grant Nos. TSYC202301B130 and 2022TSYCJC0008) and the National Natural Science Foundation of China (Grant Nos. 82260648).
Author information
Authors and Affiliations
Contributions
Conception and design of study:D. Wu ; acquisition of data: X.X. Shen, Q. Zhou and C.C. Wang; analysis and/or interpretation of data: D. Wu, R.N. Fu and J. Zhou; Drafting the manuscript: D. Wu, J. Zhou and C. Wang; Revising the manuscript critically for important intellectual content: X.X. Shen, Q. Zhou, Y.H. Ma and C.C. Wang; Approval of the version of the manuscript to be published: D. Wu, X.X. Shen, Q. Zhou, J. Zhou, R.N. Fu, C. Wang, Y.H. Ma, C.C. Wang.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors have approved the final manuscript and have given consent for publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, D., Shen, X., Zhou, Q. et al. Risk effects of environmental factors on human brucellosis in Aksu Prefecture, Xinjiang, China, 2014–2023. Sci Rep 15, 2908 (2025). https://doi.org/10.1038/s41598-025-86889-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86889-w
Keywords
This article is cited by
-
Assessing the spatiotemporal dynamics and driving factors of human brucellosis in Northern Xinjiang, China (2015–2023)
Tropical Medicine and Health (2026)
-
Spatiotemporal trends and ecological determinants of human brucellosis among 31 provinces in mainland China, 2004–2021: a Bayesian spatiotemporal modeling study
BMC Public Health (2025)
-
Interactions between brucellosis and environmental factors: spatiotemporal epidemiology from Xinjiang, China
BMC Public Health (2025)
-
Ecological study of human brucellosis in Iran from 2000 to 2023 and the limited role of climatic factors
Scientific Reports (2025)