Abstract
Reducing the radiation dose and maintaining treatment efficacy during microwave ablation (MWA) of lung tumors is of great importance. The objective of this study is to evaluate the feasibility of using spectral shaping technology (100Sn kV) to reduce radiation dose in CT-guided lung tumor MWA. Here, we conducted a prospective, randomized, double-blind and non-inferiority trial including 114 patients diagnosed with lung malignancy who were scheduled for CT-guided lung tumor MWA. The patients were randomly divided into two groups: the experimental group (n = 57), which received spectral shaping protocol CT-guided lung tumor MWA (with scanning voltage of Sn100 kV and reference current of 96 mAs), and the control group (n = 57), which underwent the standard-dose protocol CT-guided lung tumor MWA (scanning voltage of 120 kV and reference current of 94 mAs). The experimental group exhibited significantly higher image noise compared to the control group, and a lower signal-to-noise ratio and the total radiation dose. No statistical differences in aortic CT values, subjective image quality scores, operation duration and complication rates. Technique efficacy was achieved in more than 98.2% of patients in both two groups. Spectral shaping technology significantly reduces the radiation dose for patients undergoing CT-guided lung tumor MWA compared to the standard-dose protocol, without compromising image quality, operation duration or treatment efficacy.
Trial registration number ChiCTR2000032481.
Highlights
-
λ Reducing the radiation dose and maintaining treatment efficacy during microwave ablation (MWA) of lung tumors is of great importance.
-
λ Spectral shaping technology significantly reduces the radiation dose for patients undergoing lung tumor MWA.
-
λ Spectral shaping technology showed the best performance for safety and treatment efficacy.
Similar content being viewed by others
Introduction
In recent years, microwave ablation (MWA) has been widely used in the treatment of primary and secondary lung tumors. Compared with other thermal ablation techniques, MWA offers advantages such as short ablation times, large ablation zones, and a reduced heat-sink effect1,2. Computed tomography (CT) is the most commonly utilized and the most accurate image-guided method for thermal ablation of lung tumors3,4. However, repeated scans during the ablation procedure can expose patients to excessive radiation5,6. Therefore, effectively reducing the radiation dose during MWA of lung tumors is of great importance for patient protection.
With advancements in CT scanning technology, various low-dose techniques are increasingly being applied to reduce the radiation dose received by patients during CT examinations7,8,9. In recent years, some researchers have explored the use of low-dose CT technology in CT-guided pulmonary interventions. For instance, Fu et al. attempted to reduce the radiation dose during lung biopsies by lowering the scanning tube current. Their results demonstrated that low-dose CT-guided lung biopsies could reduce radiation doses by 90% compared with standard-dose CT-guided lung biopsies, while maintaining comparable diagnostic accuracy10. Similarly, Lee et al. reported that using a 100 kVp protocol for CT-guided percutaneous transthoracic biopsy, as opposed to the conventional 120 kVp scan protocol, could significantly reduce radiation exposure while maintain high diagnostic accuracy and safety11.
In recent years, spectral shaping techniques have been reported to be able to significantly reduce the radiation dose for patients undergoing chest CT scans12,13. Spectral shaping is achieved by using an additional built-in tin filter with a thickness of 0.6 mm14. The tin filter can be positioned in the X-ray beam within the pre-patient tube collimator box. The tin filter removes the lower-energy X-ray photons that are absorbed by the patient without contributing to the measured signal, resulting in substantial hardening of the X-ray spectrum14. For example, the mean photon energy of 100 kVp can be increased from 66.4 to 78.7 keV using the tin filter15. This higher mean energy leads to a higher percentage of X-ray photons reaching the detector and allows for greater dose efficiency15. A previous study showed that chest CT for the detection of pulmonary nodules can be performed with high image quality and sensitivity at a very low effective radiation dose of 0.06 mSv using a tube voltage of 100 kVp with spectral shaping and advanced iterative reconstruction16. However, to our knowledge, there has been little to no research on whether spectral shaping technology could be applied in CT-guided ablation of lung tumors to reduce the radiation dose. Therefore, the present study aimed to explore the feasibility of using spectral shaping technology (100Sn kV) to reduce radiation dose in CT-guided MWA of lung tumors, by comparing it with the conventional dose CT-guided protocol.
Materials and methods
Study design and subjects
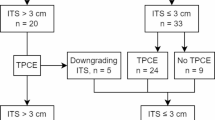
This prospective, randomized, double-blind, non-inferiority trial was approved by the local institutional ethics committee, and informed consent was obtained from all eligible patients. We assessed 131 patients indicated for MWA of lung tumors according to Multi-Disciplinary Treatment guidelines. Ultimately, 114 eligible patients were enrolled in the study (Fig. 1). The inclusion criteria for subjects in the study are: (1) age > 18 years; (2) pathologically verified clinical stage I peripheral non-small cell lung cancer (NSCLC) with the largest tumor diameter ≤ 3 cm, without lymph node or distant metastasis, and who cannot undergo surgery due to other medical conditions (severe dysfunctions of heart, lung, liver, kidney, etc.) or who refused surgery; (3) lung metastases with controlled primary tumors, total number of unilateral lung metastases ≤ 3, total number of bilateral lung metastases ≤ 5, and maximum tumor diameter ≤ 3 cm; (4) progression or recurrence of lung tumors after chemotherapy or molecular targeted therapy for peripheral NSCLC. The exclusion criteria for subjects are: (1) severe bleeding tendency, platelet count < 50 × 10^9/L, or uncorrectable coagulation dysfunction; (2) serious failure of vital organ functions (heart, liver, lung, kidney) or serious uncontrollable infections; (3) pregnant or nursing women; (4) life expectancy < 3 months; 4) eastern Cooperative Oncology Group (ECOG) Performance Status (PS) ≥ 3; (5) known allergy to drugs that may be used in the study. This study was performed in accordance with CONSORT 2010 guidelines and the Declaration of Helsinki. The trial was registered on the Chinese Clinical Trial Registry (www.chictr.org.cn), under the registration number ChiCTR2000032481 on 29/04/2020.
Flowchart of study design. MWA: microwave ablation.
All patients were randomly divided (1:1) into 2 groups: the experimental group, which received the spectral shaping CT-guided lung tumor MWA, and the control group, which underwent the standard-dose CT-guided lung tumor MWA. Randomization in this trial was performed by using a dynamic randomization method (Pocock and Simon minimized randomization method). The randomization process of this experiment was centralized, using central randomization, and the central randomization process will be realized through the Internet. Randomization results are generated immediately and need to be printed for archiving. Once a treatment number is provided by the randomization system, patients are considered to have been randomized. All patients must be registered before starting treatment. After patients are randomized, the investigator will fill in the eligible patients on the Case Report Form (CRF) and record the patient’s assigned number on the CRF. All patients concluded a follow-up at 1 and 3 months after MWA. Demographics and clinical data of patients were collected from the hospital information system.
The primary endpoint of the study was technical success rate. The secondary endpoints included objective and subjective image quality, operation duration, radiation dose, technique efficacy, and complication rates.
Percutaneous microwave ablation procedures
A percutaneous MWA procedure was conducted under the guidance of a dual-source computed tomography (CT) system (SOMATOM Force, Siemens Healthcare, Forchheim, Germany). The MWA system (MTC-3 C, Vison- China Medical Devices R&D Center, Nanjing, China) included a generator operating at 2450 ± 50 MHz, producing a maximum power output of 100 W, and a water-cooled antenna with an outer diameter of either 16G or 18G and effective length of 150–180 mm.
Patients in the experimental group underwent a spectral shaping chest CT protocol, performed at a tube potential of 100 kVp with a tin filter and a quality reference of 96 mAs. In contrast, patients in the control group were scanned using the institutional standard chest CT protocol for MWA, conducted at a fixed voltage of 120 kVp and a quality reference of 94 mAs. Other acquisition parameters were identical for the two groups: automated attenuation-based tube current modulation (CAREDose4D; Siemens Healthcare) was enabled; detector collimation width was 2 × 96 × 0.6 mm; gantry rotation time was 500 ms; and pitch was 1.2. All images were reconstructed using advanced modeled iterative reconstruction (IR) (ADMIRE; Siemens Healthineers) at a strength level of 3. The slice thickness was 2 mm, with an increment of 2 mm. A lung reconstruction kernel (B60f) and a mediastinal reconstruction kernel (B30f) were utilized. All reconstructed images were transferred to a dedicated workstation (3D Workplace; Siemens Healthineers) for data analysis.
The MWA procedure was performed by an experienced interventional radiologist with more than 10 years of experience in CT-guided thermal ablation. The operator reviewed both previously and recently obtained images to evaluate the location, size and adjacent structures of the index lesions, and determined the patient’s scan position. A localized scan of the lesion area was performed to select the optimal puncture point and entry route. Local anesthesia was administrated with 2% lidocaine, both intradermally and deeper to the pleural surface. The microwave antennas were then inserted incrementally under CT guidance (with a longitudinal scan length typically ranging 4–6 cm per scan) until the optimal ablation position was achieved. Following ablation, a localized CT scan of the lesion was conducted to ensure that the ablation zone fully encompassed the tumor and extended 0.5 cm beyond it. Finally, after removing the microwave antennas, an entire pulmonary scan was performed to monitor for potential complications. All patients received a chest CT scan the day after MWA to assess for pneumothorax or hemorrhage. Analgesics and antipyretics were prescribed as necessary.
Image quality (IQ) evaluation
The entire pulmonary scan image obtained after removing the microwave antennas was used for the evaluation of objective image quality. All measurements were performed by a radiologist who was blinded to all clinical information and the scanning protocol. Background noise was defined as the standard deviation of attenuation measured in the air outside the thorax. Attenuation was measured in Hounsfield units (HU) within a circular region of interest (ROI) with an area of approximately 2 cm2 in descending aorta (on the mediastinal window image, the ROI was chosen to exclude parts of the vessel wall, calcification, or plaques). The signal-to-noise ratio (SNR) was calculated as the quotient of the mean CT attenuation of the vessel and corresponding image noise.
Images of localized CT scan obtained after ablation were used for the evaluation of subjective image quality. Two senior observers with 3 and 7 years of diagnostic experience, blinded to the clinical information, independently performed the subjective IQ evaluation. They assessed image noise, sharpness, and artifacts on the mediastinal window images and lung window image. Overall diagnostic IQ was rated using a 4-point Likert scale: (1) 4 points = excellent IQ: minimal image noise, clear lung structures and lesion edge, no artifacts or structural obscurity, and images providing sufficient information for precise ablation localization; (2) 3 points = good IQ: less than average noise, only mild artifacts or structural obscurity, and images providing adequate information for ablation localization; (3) 2 points = moderate IQ: average image noise, moderate artifacts or structural obscurity, and images being basically sufficient for localization; (4) 1 point = fair IQ or not evaluable: above average noise, severe artifacts, significantly reduced sharpness or structural obscurity, and images not providing adequate information for ablation localization; or unacceptable image noise, artifacts, or uncharacterizable structures. Images with scores from 2 to 4 were considered to have diagnostic quality, while those with a score of 1 were deemed non-diagnostic. Discrepancies were resolved through consensus.
Radiation exposure calculation
The tube current, volume CT dose index (CTDIvol) and dose length product (DLP) were recorded for every single scan. Effective radiation dose (ED) was estimated by multiplying the DLP by a conversion factor of 0.014 mSv/(mGy × cm), as shown in the formula below17:
\({\text{ED}}={\text{DLP}} \times k(k=0.0{\text{14 mSv}}/[{\text{mGy}} \times {\text{cm}}]).\)
The total radiation dose received by each patient during the entire ablation process were calculated, encompassing initial localized scan through to the full pulmonary scan after the microwave antennas were removed, excluding the chest CT scan conducted the day following the microwave ablation (MWA). Additionally, the radiation dose for each local scan of the lesion was also calculated.
Therapeutic efficacy assessments
A tumor treated according to the protocol and with a patient who was completely recovered at the time of the procedure was deemed a “technical success”. Follow-up evaluations were conducted 1- and 3-month post-ablation, respectively. These evaluations included physical examination, laboratory tests, and contrast-enhanced CT imaging. Technique efficacy referred to a specific prospective time point when “complete ablation” of the macroscopic tumor was confirmed on follow-up imaging. Complete ablation was defined by lesion disappearance, complete cavitation formation, fibrotic progression or scar formation, solid nodule involution or stability, and/or atelectasis without contrast-enhanced signs on follow-up CT images. Incomplete ablation was indicated by incomplete cavitation with remaining solid or liquid components, partial fibrosis or fibrotic lesions with solid residues, and/or solid nodules that were unchanged or increased in size, displaying irregular peripheral or internal enhancement on follow-up CT images.
Complication rates
Treatment-related complications were noted within 30 days after ablation and were classified in accordance with the Common Terminology Criteria for Adverse Events version 4.018. Pneumothorax and hemoptysis were evaluated by CT scan after operation. It was also recorded if patients needed chest drainage catheter insertion to control pneumothorax. The incidence of air embolism and procedure-related mortality were recorded based on medical records.
Statistical analysis
The sample size estimation for this study was based on the technical success rate of MWA, which is approximately 99% for lung tumors according to current treatment outcomes19. Our goal was to demonstrate that the technical success rate of ultra-low-dose CT-guided MWA is not inferior to that of the conventional dose group by more than 5%. Assuming a type I error of 0.05 (two-sided) and a test power of 80%, the required sample size was calculated to be 52 patients per group. Additionally, considering an estimated 10% rate of non-compliance or loss to follow-up, the minimum required enrolment was determined to be 114 patients, with 57 patients in both the experimental and control groups.
Statistical analysis was performed using SPSS Version 22.0. All continuous data were expressed as mean ± SD, and categorical data were expressed as count (%). The comparisons of continuous data were analyzed with Student’s t test. Statistical differences for the comparison of categorical data were determined by using the chi-square test and Fisher exact test, as appropriate. The Wilcoxon signed-rank test was used to compare subjective image quality scores. p value of < 0 0.05 was considered statistically significant. The consistency of subjective evaluations of image quality between the two physicians was assessed using κ statistics. A κ value of > 0.61–0.80 indicates good correlation.
Results
Baseline data
Between March 2021 and March 2022, 114 patients met the criteria for enrollment in the study and random group assignment. Fifty-seven patients received spectral shaping protocol CT-guided lung tumor MWA, and 57 patients received the standard-dose protocol. Nobody withdrew their informed consent. All patients were analyzed for all outcomes, including safety. Patient demographics (sex, age, and body mass index (BMI)), characteristics of target lesions (nodule size, location, and type and distance from pleura to target lesion), and procedure-related information (patient position, hand position, number of scanning, and procedure time) are summarized in Table 1. There were no statistically significant differences between the two groups in terms of age, sex, BMI, scanning position, pathological type distribution, or basic characteristics of the lesions between the experimental group and control group (Table 1).
MWA procedure
All 114 patients (100%) successfully underwent CT-guided MWA for lung tumors, with no serious ablation complications or deaths occurring. Technique efficacy was achieved in all 57 patients (100%) in the experimental group and in 56 patients (98.2%) in the control group. Additionally, there were no statistically significant differences in the procedure time (31.96 ± 5.94 min vs. 31.12 ± 6.52 min, p = 0.47) or the total number of local lesion scans (4.07 vs. 4.00, p = 0.91)) between the two groups (Table 1).
* Procedure time was defined as the time of first CT scan to the end of CT scan after MWA.
Image analysis results
The consistency of image quality evaluation between the two radiologists was good (κ = 0.733). The image quality analysis results for the two groups of patients are shown in Table 2. The experimental group had significantly higher image noise (14.67 ± 3.66 vs. 10.06 ± 2.27, p < 0.001) and lower SNR (3.08 ± 0.74 vs. 14.59 ± 1.23, p < 0.001) compared to the control group, both with statistical significance. However, there were no statistically significant differences in the CT values and subjective scores of the images between the two groups.
Radiation dose
The actual tube current in the experimental group was 165.50 ± 98.14 mAs, which was significantly higher than that in the control group (103.43 ± 46.76 mAs, p = 0.033). The radiation dose included the radiation dose of each local scan and the total radiation dose. The comparison of radiation doses received by the patients during MWA of lung tumors is shown in Table 3. In terms of local lesion scan dose, the mean DLP (0.60 ± 0.34 vs. 80.73 ± 50.72 mGy cm, p < 0.001) and ED (0.09 ± 0.04 vs. 1.13 ± 0.71 mSv, p < 0.001) in the experimental group were significantly lower in the control group. Consistently, the mean DLP (62.99 ± 32.15 vs. 661.61 ± 362.72 mGy cm, p < 0.001) and ED (0.88 ± 0.45 vs. 9.26 ± 5.08 mSv, p < 0.001) of total radiation dose were also significantly lower in the experimental group. Compared to the control group, the radiation dose received by patients in the experimental group decreased by approximately 90%. Figures 2 and 3 shows the image quality and radiation dose for patients in the control group and experimental group during the ablation process, respectively.
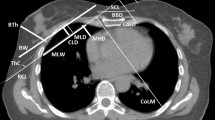
Standard-dose protocol CT-guided microwave ablation of lung tumor. A 73-year-old male patient, with a height of 1.69 m, weight of 73 kg, and a BMI of 25.56. He had a solitary lung adenocarcinoma in the lower lobe of the right lung. He cannot endure surgy because of renal failure, and received standard-dose protocol CT-guided lung tumor MWA. The local scan radiation dose was 0.812 mSv, and the overall radiation dose during ablation was 9.53 mSv for this patient. A: Preoperative local lesion scan clearly shows the solid nodule. B During the puncture process, the ablation needle punctures to a satisfactory ablation site. C: Whole lung scan images after ablation show that the lesion was completely covered by a ground glass halo.
Spectral shaping technology (100Sn kV) CT-guided microwave ablation of lung tumor. A 54-year-old male patient, with a height of 1.70 m, weight of 63 kg, and a BMI of 21.80. He had a solid nodule in the lower lobe of the right lung, and the pathological result was lung metastasis of rectal cancer. He received spectral shaping protocol CT-guided lung tumor MWA. The local scan radiation dose was 0.056 mSv, and the overall radiation dose during ablation was 0.77 mSv for this patient. A Preoperative local lesion scan clearly shows the solid nodule. B During the puncture process, the ablation needle punctures to a satisfactory ablation site. C Immediate whole lung scan images after ablation show that the lesion was completely covered by a ground glass halo and that there was a small amount of bleeding at the puncture site.
Complications
Pneumothorax developed in 22 (38.60%) and 25 (43.86%) patients in the experimental and control groups, respectively (p = 0.445; Table 4). Five patients with severe pneumothorax required chest tube insertion, including three patients (5.26%) in the experimental group and two patients (4.39%) in the control group. Pneumoderm developed in 8 (14.04%) and 7 (12.28%) patients in the experimental and control groups, respectively (p = 0.697; Table 4). Hemoptysis developed in 8 (14.04%) and 7 (12.28%) patients in the experimental and control groups, respectively (p = 0.697; Table 4). The occurrences of pneumoderm and pleural effusion were similar in the two groups. One patient was diagnosed with hemothorax in the control group, and one patient developed lung abscess and one pneumonia during follow-up in the experimental group. No deaths occurred in either group.
Discussion
The present study is a prospective, randomized, double-blind and non-inferiority trial aimed at evaluating the feasibility and clinical value of CT-guided MWA of lung tumor using spectral shaping technology. The results indicate that, compared to the standard-dose scanning protocols, the spectral shaping protocol can reduce the radiation dose by approximately 90% during MWA without compromising image quality or prolonging the procedure duration.
Recent studies have increasingly demonstrated that spectral shaping technology can significantly reduce radiation doses and minimize beam hardening artifacts in patients undergoing chest CT scans14,16,20. Consequently, some researchers have explored the application of spectral shaping technology in CT-guided pulmonary interventions to further reduce the radiation dose received by patients during these procedures, with promising results. For instance, Li et al. performed CT-guided lung lesion biopsy using spectral shaping technology (100Sn kV), which reduced the radiation dose from 1.78 mSv to 0.14 mSv while maintaining high image quality and biopsy success rates21. Similarly, Liu et al. applied spectral shaping technology (100Sn kV) for pre-thoracoscopic hook-wire placement under CT guidance, demonstrating that spectral shaping technology could significantly reduce patient radiation dose without compromising technical success22. The current study is the first to apply spectral shaping technology to CT-guided MWA of lung tumors, and our data confirm that this approach is feasible for such procedures.
It has been reported that the skin radiation dose during ablation can reach 1–2 Gy, approaching or even exceeding the threshold for deterministic skin reactions23. Kloeckner et al. reported that, during CT fluoroscopy-guided lung ablation, the total mean DLPs could reach to 1004 mGy·cm24. Excessive radiation doses can also lead to adverse events such as damage to surrounding tissues and increasing risk of ionizing radiation-related tumors, which have significant impact on patients and society25,26. In our study, the DLPs and ED in control group were 661.61 ± 362.72 mGy·cm and 9.26 ± 5.08 mSv, respectively. While the radiation dose in control group is lower compared to previous studies, further reduction of radiation exposure during the procedure would still benefit patients.
In the present study, CT-guided MWA of lung tumor was performed with acceptable image quality at a very low effective radiation dose of 0.88 ± 0.45 mSv using spectral shaping protocol in experiment group. Typically, reducing the CT radiation dose leads to an increase in image noise or artifacts, which can obscure lesions, especially ground-glass and smaller lesions. This degradation in image quality can complicate procedures such as puncture or the determination of ablation boundaries. To mitigate these issues, we applied ADMIRE (Advanced Modeled Iterative Reconstruction) technology, which reduces image noise and improves the detectability of low-contrast objects. ADMIRE uses a combination of system, statistical, and anatomical models to perform iterative calculations in both the raw data and image domains, thereby creating a non-linear relationship between image noise and radiation dose27. In addition, the actual tube current in the experimental group was significantly higher than that in the control group, partially compensating for the image quality loss associated with lower radiation doses.
During the CT-guided MWA procedure, the image quality and radiation dose were influenced by several factors: Ground-glass and smaller lesions typically require higher image quality and radiation doses; lesions located further from the pleural surface often necessitate more localized scans, increasing radiation exposure; and even patients position or hand placement, can affect the image quality by influencing the automated attenuation-based tube current modulation system. In our study, we noted and compared tumor-related characteristics and patient-related parameters, and no statistically significant differences were found between the two groups in terms of age, sex, BMI, scanning position, pathological type distribution, or the basic characteristics of the lesions.
The spectral shaping protocol reduced radiation exposure by about 90%, which may lower the risk of radiation-induced complications and long-term health problems for patients. This is particularly important in lung cancer treatments, where repeated imaging might be necessary, thus emphasizing the importance of minimizing radiation exposure. Furthermore, the study ensures that the lower dose protocol does not negatively impact critical factors such as technique efficacy, operation duration, or complication rates, making it a viable and safer alternative for clinical practice. This advancement could lead to widespread adoption of spectral shaping technology in CT-guided procedures, improving patient safety and outcomes in oncological management.
The study still has several limitations. First, because of the principles of spectral shaping technology, we only investigated its application in CT-guided MWA of lung tumors. The feasibility of applying this technology to CT-guided interventions in other anatomical locations remains to be further studied. Second, in terms of efficacy evaluation, only technical success rate and technique efficacy were noted and analyzed; survival analysis such as progression-free survival and overall survival should be also included in a further study. Third, the follow-up period is short.
In conclusion, spectral shaping technology significantly reduces the radiation dose for patients undergoing CT-guided lung tumor MWA, without compromising image quality, treatment efficacy, or increasing operation duration.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Moussa, A. M., Ziv, E., Solomon, S. B. & Camacho, J. C. Microwave ablation in primary lung malignancies. Semin. Intervent. Radiol. 36, 326–333 (2019).
Chen, B. et al. The efficacy and complications of computed tomography guided microwave ablation in lung cancer. Ann. Palliat. Med. 9, 2760–2765 (2020).
Tafti, B. A., Genshaft, S., Suh, R. & Abtin, F. Lung ablation: indications and techniques. Semin. Intervent. Radiol. 36, 163–175 (2019).
Hung, W. T. et al. Enhancing precision in lung tumor ablation through innovations in CT-guided technique and angle control. Thorac. Cancer. 15, 867–877 (2024).
Goshima, S. et al. Diffusion kurtosis imaging to assess response to treatment in hypervascular hepatocellular carcinoma. AJR Am. J. Roentgenol. 204, W543–W549 (2015).
Eisenberg, J. D. et al. Radiation exposure from CT-guided ablation of renal masses: effects on life expectancy. AJR Am. J. Roentgenol. 204, 335–342 (2015).
Kjølby, B. F. et al. Fast diffusion kurtosis imaging of fibrotic mouse kidneys. NMR Biomed. 29, 1709–1719 (2016).
Wang, D., Li, Y. H., Fu, J. & Wang, H. Diffusion kurtosis imaging study on temporal lobe after nasopharyngeal carcinoma radiotherapy. Brain Res. 1648, 387–393 (2016).
Liu, X., Zhang, M., Zhang, L., Zhao, Y. & Sun, W. Radiation dose reduction using two orthogonal topograms associated with automatic tube voltage selection for lung CT scanning as compared with a single anteroposterior topogram. Jpn J. Radiol. 37, 292–300 (2019).
Fu, Y. F. et al. Computed tomography-guided lung biopsy: a randomized controlled trial of low-dose versus standard-dose protocol. Eur. Radiol. 30, 1584–1592 (2020).
Lee, H. N. et al. Diagnostic performance of CT-guided percutaneous transthoracic core needle biopsy using low tube voltage (100 kVp): comparison with conventional tube voltage (120 kVp). Acta Radiol. 59, 425–433 (2018).
Weis, M. et al. Radiation dose comparison between 70 kVp and 100 kVp with spectral beam shaping for non-contrast-enhanced pediatric chest computed tomography: a prospective randomized controlled study. Invest. Radiol. 52, 155–162 (2017).
Messerli, M. et al. Ultralow dose CT for pulmonary nodule detection with chest x-ray equivalent dose—a prospective intra-individual comparative study. Eur. Radiol. 27, 3290–3299 (2017).
Primak, A. N., Ramirez Giraldo, J. C., Liu, X., Yu, L. & McCollough, C. H. Improved dual-energy material discrimination for dual-source CT by means of additional spectral filtration. Med. Phys. 36, 1359–1369 (2009).
Agostini, A. et al. Third-generation iterative reconstruction on a dual-source, high-pitch, low-dose chest CT protocol with tin filter for spectral shaping at 100 kV: a study on a small series of COVID-19 patients. Radiol. Med. 126, 388–398 (2021).
Gordic, S. et al. Ultralow-dose chest computed tomography for pulmonary nodule detection: first performance evaluation of single energy scanning with spectral shaping. Invest. Radiol. 49, 465–473 (2014).
Deak, P. D., Smal, Y. & Kalender, W. A. Multisection CT protocols: sex- and age-specific conversion factors used to determine effective dose from dose-length product. Radiology 257, 158–166 (2010).
Chen, A. P. et al. Grading dermatologic adverse events of cancer treatments: the common terminology criteria for adverse events Version 4.0. J. Am. Acad. Dermatol. 67, 1025–1039 (2012).
Healey, T. T., March, B. T., Baird, G. & Dupuy, D. E. Microwave ablation for lung neoplasms: a retrospective analysis of long-term results. J. Vasc Interv Radiol. 28, 206–211 (2017).
Li, J. et al. Chest CT screening in patients with overweight or obesity using spectral shaping at 150 kVp: compared with 120 kVp protocol and spectral shaping at 100 kVp protocol. Jpn J. Radiol. 38, 451–457 (2020).
Li, C., Liu, B., Meng, H., Lv, W. & Jia, H. Efficacy and radiation exposure of ultra-low-dose chest CT at 100 kVp with tin filtration in CT-guided percutaneous core needle biopsy for small pulmonary lesions using a third-generation dual-source CT scanner. J. Vasc Interv Radiol. 30, 95–102 (2019).
Liu, B. et al. Ultralow dose computed tomography protocol for hook-wire localization of solitary pulmonary nodules prior to video-assisted thoracoscopic surgery. Thorac. Cancer. 10, 1348–1354 (2019).
Tsapaki, V. et al. Patient skin dose assessment during CT-guided interventional procedures. Radiat. Prot. Dosimetry. 129, 29–31 (2008).
Kloeckner, R. et al. Radiation exposure in CT-guided interventions. Eur. J. Radiol. 82, 2253–2257 (2013).
Pradella, M. et al. Impact factors for safety, success, duration and radiation exposure in CT-guided interventions. Br. J. Radiol. 92, 20180937 (2019).
Guberina, N. et al. Radiation exposure during CT-guided biopsies: recent CT machines provide markedly lower doses. Eur. Radiol. 28, 3929–3935 (2018).
Hell, M. M. et al. Prospectively ECG-triggered high-pitch coronary angiography with third-generation dual-source CT at 70 kVp tube voltage: feasibility, image quality, radiation dose, and effect of iterative reconstruction. J. Cardiovasc. Comput. Tomogr. 8, 418–425 (2014).
Acknowledgements
This work was supported by the Key Research and Development Plan Project of Heilongjiang Provincial Department of Science and Technology (No.2023ZX06C13).
Author information
Authors and Affiliations
Contributions
D.Z., and P.W. contributed to the conception, design, and supervision of the study; P.W., and D.Z. contributed significantly to data analysis and interpretation; R.S., Y.D., and X.Y. collected the extracted the data, performed the analysis and interpreted the results; X.L. and L.Z., participated in study design, safety review and discussion; D.Z., and P.W. performed the data analyses and wrote the manuscript; All authors discussed the results and read the manuscript. D.Z. have full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
s were obtained from all human subjects in this study, and the protocol was approved by the Ethics Committee of the Renji Hospital, School of Medicine, Shanghai Jiao Tong University.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, P., Sun, R., Deng, Y. et al. Spectral shaping technology reduces the radiation dose of CT-guided lung tumor microwave ablation. Sci Rep 15, 12478 (2025). https://doi.org/10.1038/s41598-025-86945-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86945-5