Abstract
Infertility is increasingly linked to obesity, especially visceral fat accumulation. The Body Roundness Index (BRI) provides a precise quantification of body and visceral fat but its implications for infertility risks and diagnostic value are yet to be fully determined. This study analyzed data from 1,305 married women aged 20–45 years, drawn from the National Health and Nutrition Examination Survey (NHANES). Logistic regression was utilized to examine the relationship between BRI and infertility, incorporating univariate and multivariate regression analyses, smooth curve fittings, and subgroup analyses. Additionally, Receiver Operating Characteristic (ROC) analysis assessed the predictive capability of BRI for infertility. An increase in one unit of BRI correlated with a 13% higher prevalence of infertility after adjusting for covariates (OR = 1.13; 95% CI: 1.05, 1.21). Women with higher BRI exhibited greater infertility odds compared to the reference group (OR: 2.20, 95% CI: 1.42, 3.42). Smooth curve fitting analysis revealed a positive, nonlinear relationship, with an inflection point observed at a BRI value of 7.95. Subgroup analyses upheld the robustness of this association across varied demographic segments. ROC curves indicated significant diagnostic potential of BRI in younger women, whereas its predictive relevance diminished in those over 35, akin to other obesity markers. This study is the first to rigorously affirm a strong link between increased BRI and the higher prevalence of infertility among married women aged 20–45. To optimize fertility, maintaining a healthy weight and waist circumference is recommended. The integration of BRI into clinical practice could assist in early intervention for women at risk of infertility. However, further research is required to identify effective predictive markers for infertility in women aged 36–45.
Similar content being viewed by others
Introduction
Infertility is medically defined as the inability to conceive after 12 months of regular, unprotected sexual intercourse1. This condition is a major global reproductive health issue, affecting an estimated 186 million people worldwide2. In the United States, infertility impacts approximately 6.7–15.5% of women of reproductive age3. Given its significant impact on human development, the US Centers for Disease Control and Prevention (CDC) emphasizes the importance of prioritizing the diagnosis and treatment of infertility4. Despite increasing international attention to infertility as a public health concern, the underlying factors contributing to it require further exploration5,6,7.
Obesity has become a global epidemic, with reliable estimates indicating that over 1.1 billion people are affected worldwide8. The negative effects of obesity on reproductive health are well-documented, as obese women often experience irregular menstruation, impaired ovulation, and endometriosis9. However, much of the existing research on the relationship between obesity and infertility has relied primarily on body mass index (BMI) as an indicator. While BMI is a common measure of obesity, it is limited in its ability to differentiate between lean and fat tissues10,11. Recently, more precise measures have been developed to assess visceral fat accumulation, which is closely linked to reproductive dysfunction12. For instance, the weight-adjusted waist circumference index (WWI) shows a positive association with infertility among U.S. women13. Similarly, the Visceral Adiposity Index (VAI), which reflects dysfunction and accumulation of visceral adipose tissue, has been positively correlated with increased infertility incidence14. Introduced in 2013, the Body Roundness Index (BRI) offers a superior estimation of body fat distribution and visceral adiposity than other traditional anthropometric measures15,16. Unlike the BMI, which is based solely on height and weight, the BRI incorporates waist circumference, offering a more comprehensive perspective on body shape17.This feature enhances the BRI’s ability to identify abdominal obesity, particularly in individuals with excess visceral fat, providing a more accurate assessment of this condition18. Furthermore, the BRI is less influenced by muscle mass, making it a more reliable indicator of fat-related health risks in individuals with higher muscle mass19. Additionally, the inclusion of height in the BRI formula distinguishes it from other obesity indices such as WWI or VAI, allowing it to more thoroughly reflect an individual’s body composition and fat distribution14,20,21. Despite its promising advantages, the significance of the BRI in relation to infertility among married women has yet to be explored. This study aims to fill this gap by examining the association between BRI and infertility. Furthermore, this research will evaluate and compare four obesity indices—BRI, BMI, WWI, and the VAI—as screening tools for identifying American married women at heightened odds of infertility. By pinpointing individuals at increased risk, targeted preventive interventions can be devised to mitigate the incidence of infertility.
Methods
Study design and population
This study utilizes data from the ongoing National Health and Nutrition Examination Survey (NHANES), managed by the National Center for Health Statistics under the Centers for Disease Control and Prevention. Further details on NHANES are accessible via the CDC’s dedicated website (http://www.cdc.gov/nchs/nhanes/). NHANES employs a multistage probability sampling technique to collect detailed data from a representative sample of the non-institutionalized, civilian U.S. population. The original collection of NHANES data received ethical approval from the Institutional Review Board of the National Center for Health Statistics (NCHS), and informed consent was obtained from all participants. Following ethical guidelines, de-identified data from the NHANES program are publicly available. For this study, researchers not involved with the initial NHANES program accessed and analyzed these data; therefore, no further ethics approval was required for this secondary analysis.
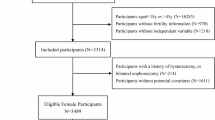
The dataset from 2013 to 2018 was chosen for analysis to include responses from a detailed questionnaire on infertility. The initial dataset consisted of 29,400 participants who had completed assessments regarding demographics, health conditions, and laboratory tests. We systematically excluded participants for several reasons: male participants (n = 14,452), individuals with marital status other than married (n = 10,875), those outside the 20–45 age range (n = 2,368), and individuals lacking data on BMI, WWI, VAI, BRI, or fertility status (n = 384). Additionally, extreme values of BRI were identified and excluded based on a threshold of mean ± 3 standard deviations (n = 16). After these exclusions, the final sample consisted of 1,305 participants. Figure 1 illustrates the sample selection flowchart.
Sample selection flowchart from NHANES 2013–2018.
Definition of infertility and BRI, BMI, WWI, VAI
Infertility was assessed using self-reported data from the Reproductive Health Questionnaire, specifically question RHQ074, which asked if the participant had attempted to conceive for over a year. Answers affirming this were categorized as ‘infertile’, and all others as ‘fertile’. Anthropometric measurements, including body height, body weight, and waist circumference (WC), were collected by trained examiners at a mobile examination center equipped with standardized instruments. Body weight was measured using calibrated platform scales with a precision of 0.1 kg, and height was assessed with stadiometers to the nearest 0.1 cm, with participants standing upright. These measurements were taken while participants wore light clothing and no shoes. Triglycerides (TG) were measured using the Wahlefeld method, while high-density lipoprotein cholesterol (HDL) was quantified using the magnesium sulfate/glucan method. Both TG and HDL measurements were performed on serum samples, and participants were instructed to fast for at least 9 h prior to blood collection. BMI was calculated by dividing weight in kilograms by the square of height in meters22. BRI was computed by 364.2-365.5*(1-[WC(m)/2π]2/[0.5*height(m)]2) [½ 23. The WWI, indicating obesity levels by normalizing WC to body weight, is determined by the square root of the WC divided by body weight24. For females, the VAI is calculated as WC/(36.58 + (1.89*BMI))*(TG/0.81)*(1.52/HDL-C), where TG and HDL-C are measured in mmol/L and WC in cm12.
Covariates
Our analyses adjusted for a range of covariates identified from prior research12,24,25: age, race/ethnicity (Mexican American, Other Hispanic, Non-Hispanic White, Non-Hispanic Black, and Other), poverty-to-income ratio (PIR), educational level (below high school, at least high school), diabetes status, hyperlipidemia, hypertension, smoking status (defined as having smoked 100 or more cigarettes lifetime), ,menstrual regularity over the past 12 months, history of pelvic infection or pelvic inflammatory disease treatment, hormone usage, physical activity level, and alcohol consumption (categorized as drinking at least once a month or not). Diagnoses of diabetes, hyperlipidemia, and hypertension were verified through medical records, current medication use, or if fasting plasma glucose levels exceeded 7.0 mmol/L. Information on these variables was obtained from the NHANES database (https://www.cdc.gov/nchs/nhanes/).
Statistical analysis
In adherence to guidelines set by the NCHS, we applied sample weights in our statistical estimates to accurately represent the noninstitutionalized civilian U.S. population. Participant demographics and baseline characteristics were summarized using means ± standard deviations for continuous variables and percentages for categorical variables. To assess differences between participants with and without infertility, we compared means and proportions.
Initially, relationships were explored univariately. The link between BRI and infertility was further analyzed through multivariate logistic regression. We utilized four models: Model 1 was unadjusted; Model 2 adjusted for age and race; Model 3 further adjusted for health behaviors and comorbidities; and Model 4 adjusted for a comprehensive set of variables including age, race, educational level, PIR, smoking status, alcohol consumption, diabetes, hyperlipidemia, hypertension, physical activity level, menstrual regularity in the past 12 months, history of pelvic infection treatment, and hormone use.
To investigate potential nonlinear relationships, we employed generalized additive models (GAM) and smooth curve fitting techniques. Nonlinear associations prompted the use of a recursive method to pinpoint the inflection point, followed by a two-piecewise linear regression to analyze segments on either side of this point. Subgroup analyses were performed using stratified multivariate regression, incorporating interaction terms to explore variability across different subgroups. The predictive capacity of obesity indices for identifying infertility was evaluated using receiver operating characteristic (ROC) analysis and area under the curve (AUC) values. A p-value of 0.05 or less was considered statistically significant. All analyses were conducted using R (http://www.Rproject.org) and EmpowerStats (http://www.empowerstats.com).
Results
Baseline characteristics of participants
Our analysis included 1,305 participants. Table 1 outlines the clinical characteristics stratified by the presence of infertility. Compared to controls, individuals with infertility were generally older, exhibited higher BMI values, a greater prevalence of diabetes, and a higher BRI level. No significant differences were observed between the groups in other baseline characteristics, including race, PIR, education level, physical activity, smoking status, alcohol consumption, menstrual regularity in the past 12 months, history of pelvic infection treatment, and hormone use, hypertension, and hyperlipidemia (p > 0.05).
Associations between BRI and the prevalence of infertility
Table 2 details the results from our multivariate logistic regression analyses. In Model 1, which was unadjusted, BRI demonstrated a positive association with the prevalence of infertility [Odds Ratio (OR) = 1.13; 95% Confidence Interval (CI): 1.06, 1.20; p < 0.0001]. This association persisted in Model 2, which adjusted for age and race (OR = 1.13; 95% CI: 1.06, 1.20; p < 0.0001). When further adjusted for health behaviors and comorbidities in Model 3, the positive association was still evident (OR = 1.10; 95% CI: 1.03, 1.18; p = 0.0038). The fully adjusted Model 4 confirmed a significant positive association (OR = 1.13; 95% CI: 1.05, 1.21; p = 0.0009), indicating a 13% increase in the prevalence of infertility per unit increase in BRI. Furthermore, the highest tertile of BRI was associated with an 120% increased prevalence of infertility compared to the lowest tertile in Model 4, underscoring a significant trend (p for trend = 0.0004).
The generalized additive models and smooth curve fits employed to delineate the nonlinear relationship between BRI and the prevalence of infertility. Notably, an inflection point was identified, as shown in Fig. 2. A subsequent threshold effect analysis employing a two-piecewise linear regression model pinpointed the inflection point at 7.95 (Table 3). The likelihood ratio tests comparing the piecewise models to standard linear models revealed a significant improvement (p = 0.038), suggesting that the piecewise model more accurately represented the data.
Association between BRI and the prevalence of infertility.
The area between the upper and lower dashed lines is represented as 95% CI. Each point shows the magnitude of the BRI and is connected to form a continuous line. age, race, educational level, poverty-to-income ratio, smoking status, alcohol consumption, diabetes, hyperlipidemia, hypertension, physical activity level, menstrual regularity in the past 12 months, history of pelvic infection treatment, and hormone use were adjusted.
Subgroup analysis
We conducted subgroup analyses to evaluate the robustness and consistency of the BRI-infertility relationship, as detailed in Table 4. These analyses considered potential modifiers such as age, race, alcohol consumption, smoking status, diabetes, hyperlipidemia, menstrual regularity in the past 12 months, hormone use. No significant interactions were observed (all p for interaction > 0.05), indicating that these factors did not significantly modify the observed relationship.
Diagnostic efficacy of obesity indices for infertility
The efficacy of various obesity indices in distinguishing individuals with infertility was assessed through ROC curves and AUC values (Fig. 3). In the subgroup of participants aged ≤ 35 years, the AUC values for BRI, BMI, WWI, and VAI were 0.6546 (95% CI: 0.5882,0.7210), 0.6412 (95% CI: 0.5749,0.7076), 0.6449 (95% CI: 0.5845,0.7052), and 0.5808 (95% CI: 0.5186,0.6430), respectively. Notably, BRI demonstrated the highest AUC among these indices (Table 5). Conversely, in participants older than 35 years, the diagnostic utility of these indices was limited, with AUC values for BRI, BMI, WWI, and VAI were 0.5347 (95% CI: 0.4782, 0.5912), 0.5542 (95% CI: 0.4974, 0.6111), 0.5157 (95% CI: 0.4583, 0.5731), and 0.5309 (95% CI: 0.4748, 0.5870), respectively (Table 6).
ROC curves of different obesity indices for the prediction of infertility risk.
Discussion
This pioneering study explores the association between the BRI and infertility within a nationally representative sample of American women aged 20 to 45 who are married. We found a consistent positive nonlinear association between BRI and infertility, maintained across both unadjusted and adjusted models. Notably, we identified a critical inflection point: below this threshold, each additional unit increase in BRI was associated with 23% higher odds of infertility (OR = 1.23; 95% CI: 1.10–1.36; p = 0.0002). Subgroup analyses confirmed the robustness of this association across diverse demographic groups. While indices such as BRI, BMI, WWI, and VAI showed significant diagnostic value in younger women, their relevance diminished in the 36 to 45 age group.
The burgeoning prevalence of obesity is now a major public health concern, affecting approximately 20% of American women of reproductive age1.Traditionally, BMI has been the standard for assessing overweight conditions in studies investigating obesity’s impact on fertility. However, the diverse presentations of obesity—generalized versus central—suggest that BMI alone may not capture the nuances of body fat distribution and muscle composition effectively26,27. This realization has prompted the introduction of novel obesity indices over recent decades, designed specifically to measure central obesity12,13,14,21,22,24,25,28,29,30,31 .The BRI, by providing a more accurate estimation of visceral fat, offers significant insights into the role of abdominal obesity in reproductive challenges15. The BRI is well-documented as a robust predictor of diabetes, including prediabetes, cardiometabolic disorders, and metabolic syndrome32,33,34.In this investigation, we expanded the scope of BRI to assess its relevance as a potential determinant of infertility. Our results affirm the association between increased visceral fat, as measured by BRI, and a higher prevalence of infertility, with a notable 13% increase in infertility odds for each unit increment in BRI among women with marital status. It effectively bridges a critical gap in the current literature by linking specific body composition metrics with reproductive health outcomes.
Despite these promising associations, the underlying mechanisms through which higher BRI levels influence infertility remain unclear, and there exist several possible explanations. Excess fat, particularly in the abdominal region, can elevate estrogen levels in obese women, disrupting hormonal balance and causing irregularities in ovulation and menstrual cycles35,36. Additionally, abdominal obesity is often associated with conditions such as polycystic ovary syndrome (PCOS), which exacerbates ovulatory dysfunction and hormonal imbalances37. Moreover, the inflammatory cytokines released by visceral fat may negatively impact fertility by impairing the endometrial receptivity necessary for successful implantation20,38.These findings collectively highlight the complex interconnections between abdominal obesity, reproductive health, and cellular mechanisms, emphasizing the need for further research to elucidate these intricate biological processes and their implications for fertility.
Magnetic resonance imaging (MRI) and computed tomography (CT) are universally recognized as the gold standards for assessing visceral fat. Nevertheless, the widespread adoption of these imaging modalities is curtailed by their significant costs and the complex nature of the procedures involved39. Consequently, exploring alternative obesity metrics for evaluating infertility presents a pragmatic approach. Our ROC analysis established that four obesity indices, including BRI, BMI, WWI, and VAI, demonstrate good diagnostic prowess in women under the age of 35, Notably, BRI exhibited the highest AUC among these indices. However, the efficacy of these indices in women aged 36 to 45 remains uncertain, underscoring an urgent need for targeted research to develop more effective diagnostic tools for this age group. This insight directs future investigations aimed at enhancing fertility evaluations and interventions.
Our study’s conclusions are substantiated by data obtained from the NHANES, with a strategic focus on married women to enhance the accuracy of responses regarding infertility issues. By excluding divorced, separated, or unmarried individuals from our analysis, we minimized potential biases, thereby improving the reliability of our findings. However, we acknowledge that this approach resulted in a smaller sample size for each cycle. To address this limitation, we included data from three cycles of the NHANES, spanning from 2013 to 2018. This adjustment enhances the robustness of our findings. Furthermore, the incorporation of appropriate sample weights ensures not only the reliability but also the broader applicability of our results. We advocate for the adoption of these obesity metrics in clinical practice to facilitate timely interventions for women under 35 who are at risk of or currently experiencing infertility. However, the study’s reliance on self-reported infertility via questionnaires could introduce bias due to potential recall inaccuracies. Furthermore, as the study leverages data exclusively from the United States, its generalizability to other populations might be limited. Additionally, the cross-sectional design of this study precludes definitive causal inferences between BRI and infertility, underscoring the need for future longitudinal research to substantiate and expand upon these findings.
Conclusions
Our analysis confirms a positive nonlinear association between BRI and infertility. To improve fertility outcomes, it is recommended that women prioritize maintaining a healthy weight and waist circumference. Considering the cost-effectiveness and accessibility of these obesity indices, we strongly recommend their integration into clinical protocols. However, it is vital to note that these conclusions are primarily relevant to the cohort of U.S. women studied. Therefore, further research is essential to validate whether these findings are applicable to female populations in other geographical areas.
Data availability
Publicly available datasets were analyzed in this study. Those data can be found here: www.cdc.gov/nchs/nhanes/.
References
Broughton, D. E. & Moley, K. H. Obesity and female infertility: Potential mediators of obesity’s impact. Fertil. Steril. 107, 840–847. https://doi.org/10.1016/j.fertnstert.2017.01.017 (2017).
Inhorn, M. C. & Patrizio, P. Infertility around the globe: New thinking on gender, reproductive technologies and global movements in the 21st century. Hum. Reprod. Update. 21, 411–426. https://doi.org/10.1093/humupd/dmv016 (2015).
Thoma, M. E. et al. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil. Steril. 99, 1324–1331 e1321, (2013). https://doi.org/10.1016/j.fertnstert.2012.11.037
Macaluso, M. et al. A public health focus on infertility prevention, detection, and management. Fertil. Steril. 93 (16 e11-10). https://doi.org/10.1016/j.fertnstert.2008.09.046 (2010).
Zhu, F. et al. Elevated blood mercury level has a non-linear association with infertility in U.S. women: data from the NHANES 2013–2016. Reprod. Toxicol. 91, 53–58. https://doi.org/10.1016/j.reprotox.2019.11.005 (2020).
Lee, S., Min, J. Y. & Min, K. B. Female infertility associated with blood lead and cadmium levels. Int. J. Environ. Res. Public. Health. 17 https://doi.org/10.3390/ijerph17051794 (2020).
Sharma, R., Biedenharn, K. R., Fedor, J. M. & Agarwal, A. Lifestyle factors and reproductive health: Taking control of your fertility. Reprod. Biol. Endocrinol. 11, 66. https://doi.org/10.1186/1477-7827-11-66 (2013).
Haslam, D. W., James, W. P. & Obesity Lancet 366, 1197–1209, doi:https://doi.org/10.1016/s0140-6736(05)67483-1 (2005).
Ennab, F. & Atiomo, W. Obesity and female infertility. Best Pract. Res. Clin. Obstet. Gynaecol. 89, 102336. https://doi.org/10.1016/j.bpobgyn.2023.102336 (2023).
Cai, S. et al. Association of the weight-adjusted-waist index with risk of all-cause mortality: A 10-year follow-up study. Front. Nutr. 9, 894686. https://doi.org/10.3389/fnut.2022.894686 (2022).
Park, Y., Kim, N. H., Kwon, T. Y. & Kim, S. G. A novel adiposity index as an integrated predictor of cardiometabolic disease morbidity and mortality. Sci. Rep. 8, 16753. https://doi.org/10.1038/s41598-018-35073-4 (2018).
Deng, C., Ke, X., Lin, L., Fan, Y. & Li, C. Association between indicators of visceral lipid accumulation and infertility: A cross-sectional study based on U.S. women. Lipids Health Dis. 23, 186. https://doi.org/10.1186/s12944-024-02178-x (2024).
Zhong, H. et al. Associations between weight-adjusted-waist index and infertility: Results from NHANES 2013 to 2020. Medicine 102, e36388. https://doi.org/10.1097/MD.0000000000036388 (2023).
Zhuang, J., Wang, Y., Wang, S., Hu, R. & Wu, Y. Association between visceral adiposity index and infertility in reproductive-aged women in the United States. Sci. Rep. 14, 14230. https://doi.org/10.1038/s41598-024-64849-0 (2024).
Thomas, D. M. et al. Relationships between body roundness with body fat and visceral adipose tissue emerging from a new geometrical model. Obesity 21, 2264–2271. https://doi.org/10.1002/oby.20408 (2013).
Zhang, X. et al. Body roundness index and all-cause mortality among US adults. JAMA Netw. Open. 7, e2415051. https://doi.org/10.1001/jamanetworkopen.2024.15051 (2024).
Zhou, D., Liu, X., Huang, Y. & Feng, Y. A nonlinear association between body roundness index and all-cause mortality and cardiovascular mortality in general population. Publ Health Nutr. 25, 3008–3015. https://doi.org/10.1017/S1368980022001768 (2022).
Li, M. et al. The pathophysiological associations between obesity, NAFLD, and atherosclerotic cardiovascular diseases. Horm. Metab. Res. 56, 683–696. https://doi.org/10.1055/a-2266-1503 (2024).
Tian, S., Zhang, X., Xu, Y. & Dong, H. Feasibility of body roundness index for identifying a clustering of cardiometabolic abnormalities compared to BMI, waist circumference and other anthropometric indices: the China health and nutrition survey, 2008 to 2009. Med. (Baltim). 95, e4642. https://doi.org/10.1097/MD.0000000000004642 (2016).
Wang, W., Hou, S., Wang, K., Ling, B. & Yu, H. Association of body roundness index with female infertility: 2013–2018 NHANES. Front. Nutr. 11, 1416637. https://doi.org/10.3389/fnut.2024.1416637 (2024).
Sun, F. et al. Associations of weight-adjusted-waist index and depression with secondary infertility. Front. Endocrinol. 15, 1330206. https://doi.org/10.3389/fendo.2024.1330206 (2024).
Zhu, L. et al. Association between body mass index and female infertility in the United States: data from national health and nutrition examination survey 2013–2018. Int. J. Gen. Med. 15, 1821–1831. https://doi.org/10.2147/IJGM.S349874 (2022).
Wei, C. & Zhang, G. Association between body roundness index (BRI) and gallstones: results of the 2017–2020 national health and nutrition examination survey (NHANES). BMC Gastroenterol. 24, 192. https://doi.org/10.1186/s12876-024-03280-1 (2024).
Wen, Z. & Li, X. Association between weight-adjusted-waist index and female infertility: a population-based study. Front. Endocrinol. 14, 1175394. https://doi.org/10.3389/fendo.2023.1175394 (2023).
Ke, J., Feng, Y. & Chen, Z. Association between waist circumference and female infertility in the United States. PLoS One. 18, e0295360. https://doi.org/10.1371/journal.pone.0295360 (2023).
Carmienke, S. et al. General and abdominal obesity parameters and their combination in relation to mortality: a systematic review and meta-regression analysis. Eur. J. Clin. Nutr. 67, 573–585. https://doi.org/10.1038/ejcn.2013.61 (2013).
Moltrer, M. et al. Body mass index (BMI), waist circumference (WC), waist-to-height ratio (WHtR) e waist body mass index (wBMI): which is better? Endocrine 76, 578–583. https://doi.org/10.1007/s12020-022-03030-x (2022).
Jiang, N., Zhang, S., Chu, J., Yang, N. & Lu, M. Association between body roundness index and non-alcoholic fatty liver disease detected by Fibroscan in America. J. Clin. Lab. Anal. 37, e24973. https://doi.org/10.1002/jcla.24973 (2023).
Zhang, J. et al. Associations between novel anthropometric indices and the prevalence of gallstones among 6,848 adults: A cross-sectional study. Front. Nutr. 11, 1428488. https://doi.org/10.3389/fnut.2024.1428488 (2024).
Gao, W. et al. The association between the body roundness index and the risk of colorectal cancer: A cross-sectional study. Lipids Health Dis. 22 https://doi.org/10.1186/s12944-023-01814-2 (2023).
Yin, Y. H. et al. Higher waist circumference is associated with increased likelihood of female infertility: NHANES 2017–2020 results. Front. Endocrinol. 14, 1216413. https://doi.org/10.3389/fendo.2023.1216413 (2023).
Wang, J., Wu, M., Wu, S. & Tian, Y. Relationship between body roundness index and the risk of heart failure in Chinese adults: The Kailuan cohort study. ESC Heart Fail. 9, 1328–1337. https://doi.org/10.1002/ehf2.13820 (2022).
Li, Y. et al. Body roundness index and waist-hip ratio result in better cardiovascular disease risk stratification: Results from a large Chinese cross-sectional study. Front. Nutr. 9, 801582. https://doi.org/10.3389/fnut.2022.801582 (2022).
Zhao, Q. et al. Capacity of a body shape index and body roundness index to identify diabetes mellitus in Han Chinese people in Northeast China: A cross-sectional study. Diabet. Med. 35, 1580–1587. https://doi.org/10.1111/dme.13787 (2018).
Jurczewska, J. et al. Abdominal obesity in women with polycystic ovary syndrome and its relationship with diet, physical activity and insulin resistance: A pilot study. Nutrients 15 https://doi.org/10.3390/nu15163652 (2023).
Incedal Irgat, S. & Bakirhan, H. The effect of obesity on human reproductive health and foetal life. Hum. Fertil. (Camb). 25, 860–871. https://doi.org/10.1080/14647273.2021.1928774 (2022).
Tatarchuk, T. et al. The effect of gastric sleeve resection on menstrual pattern and ovulation in premenopausal women with classes III–IV obesity. Obes. Surg. 32, 599–606. https://doi.org/10.1007/s11695-021-05820-0 (2022).
Vannuccini, S. et al. Infertility and reproductive disorders: impact of hormonal and inflammatory mechanisms on pregnancy outcome. Hum. Reprod. Update. 22, 104–115. https://doi.org/10.1093/humupd/dmv044 (2016).
Cornier, M. A. et al. Assessing adiposity: A scientific statement from the American heart association. Circulation 124, 1996–2019. https://doi.org/10.1161/CIR.0b013e318233bc6a (2011).
Acknowledgements
We thank all the contributors and participants in the National Health and Nutrition Examination Survey.
Author information
Authors and Affiliations
Contributions
FY and XQY designed the research. FY, RDF, XQY, and PJL collected and analyzed the data. FY, XQY, and RDF drafted the manuscript. FY and XQY revised the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fang, Y., Ren, D., Pan, J. et al. The nonlinear association between body roundness index and infertility in married women. Sci Rep 15, 2431 (2025). https://doi.org/10.1038/s41598-025-86948-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86948-2
This article is cited by
-
Anthropometric indices and mortality in postmenopausal women: NHANES 1999–2018 evidence
BMC Women's Health (2025)
-
Association between relative fat mass and female infertility among reproductive-aged women from NHANES 2013–2020
Scientific Reports (2025)