Abstract
Management of melanoma has changed significantly with the discovery of targeted therapies and immune checkpoint inhibitors (ICI). Our aim in the study is to determine which treatment alternatives, specifically dabrafenib plus trametinib and ICIs, are effective in adjuvant therapy and which treatment is effective as first-line metastatic therapy. This retrospective, multicenter study included 120 patients diagnosed with stage IIIB-IIID melanoma receiving both adjuvant and first-line metastatic treatment between 2007 and 2023. Data on clinicopathologic characteristics, treatment regimens and outcomes were collected. Objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS) and overall survival (OS) were analyzed. Patients treated with dabrafenib plus trametinib as adjuvant therapy had the longest relapse-free survival (RFS) (median: 8.3 months), followed by those treated with interferon (4.1 months) and nivolumab (1.9 months) (p = 0.002). Metastatically, the highest ORR was observed in patients treated with dabrafenib plus trametinib (54.5%), followed by ICI (52.0%) and chemotherapy (33.3%). Similarly, DCR was superior for dabrafenib plus trametinib (86.3%) compared to ICI (70.8%) and chemotherapy (66.6%). Median PFS was 9.7 months (95% CI 7.2–12.2 months) in the whole group. This was 14.3 months (95% CI 9.6–19.0 months) with ICI, 10.3 months (95% CI 4.2–16.4 months) with BRAF/MEK inhibitors and 6.3 months (95% CI 4.7–7.9 months) with chemotherapy, which was statistically significant (p < 0.001). Dabrafenib plus trametinib showed the longest median OS (53.5 months) in metastatic patients and was significantly better than chemotherapy (33.6 months) (p < 0.001). BRAF V600E mutation, RFS > 6 months, ORR are all independent factors for OS prognosis. In our study, dabrafenib plus trametinib combination was more effective in adjuvant treatment of melanoma, while immunotherapy was more effective in metastatic first-line treatment.
Similar content being viewed by others
Introduction
Melanoma is an aggressive type of skin cancer with an increasing incidence over the recent years1. Despite improvements in strategies for early diagnosis, a significant proportion of patients present in the locally advanced or metastatic stage, leading to high mortality2. The main melanoma treatment options available in the past were interferon and chemotherapy, which only partially improved patient survival. However, over the last decade, melanoma management has undergone a significant change with the development of targeted therapies and immunotherapy (ICI)3.
Adjuvant therapies, such as ICIs and BRAF/MEK inhibitors, have made a significant contribution to the management of high-risk melanoma patients after surgical resection. These therapies aim to eliminate microscopic residual disease, reduce recurrence and improve overall survival (OS). Nivolumab, a PD-1 inhibitor, and combination BRAF/MEK inhibitors such as dabrafenib plus trametinib, have shown significant efficacy in prolonging recurrence free survival (RFS) in patients with resected stage III melanoma, making them standard adjuvant therapies4,5. Moreover, there have been recent advances in treatment alternatives for metastatic melanoma ICIs, especially anti-PD-1 drugs such as pembrolizumab and nivolumab, have improved survival rates in patients with metastatic melanoma. Moreover, identification of the BRAF V600E mutation has led to the development of targeted drugs in melanoma6,7,8. While these therapies have significantly improved outcomes, research on combination therapies and optimal treatments in adjuvant and metastatic primary care settings is still ongoing. BRAF-MEK inhibitors and ICI inhibitors are used in the adjuvant treatment of malignant melanoma patients. The same treatment agents are also used in the metastatic period. In this study, the survival and optimal treatment options of patients who received adjuvant treatment and those who received treatment in the metastatic period were investigated. This study aims to evaluate the real-life effectiveness of these therapies in both adjuvant and first-line metastatic management.
Materials and methods
This is a multicenter (20 centers) retrospective study of 120 patients with histologically diagnosed melanoma and stage IIIB-IIID between 2007 and 2023. Patients over 18 years of age who received adjuvant systemic therapy after surgical resection and patients who progressed during follow-up and received first-line treatment in the metastatic period were included in the study. Demographic data, clinicopathologic characteristics, adjuvant and metastatic first-line treatment regimens, response rates and survival outcomes were obtained from the records and files. Data collected also included age, gender, Eastern Cooperative Oncology Group-Performance Status (ECOG-PS), LDH at diagnosis, melanoma subtype, BRAF mutation status, presence of ulceration and metastatic sites.
In this article, staging was based on the Tumor-Node-Metastasis (TNM)-based staging of melanoma according to the 8th editions of the International Union for Cancer Control (UICC) and American Joint Committee on Cancer (AJCC) publications. Patients received one of three standard adjuvant therapies: nivolumab, dabrafenib plus trametinib or high-dose interferon. In metastatic first-line management, patients were treated with chemotherapy, BRAF/MEK inhibitors or ICI according to their BRAF mutation status and clinical indications.
Response Evaluation Criteria In Solid Tumors (RECIST) was used to measure treatment responses. Best response rates with metastatic first-line treatment were evaluated. Objective response rate (ORR) was defined as the sum of complete response (CR) and partial response (PR); disease control rate (DCR) was the sum of CR and PR and stable disease (SD).
RFS: time from the date of surgery to the start of metastatic first-line treatment; Progression-free survival (PFS): time from first-line treatment initiation to first progression, death or last visit; OS is defined as the time from the date of diagnosis (by biopsy or surgery) to the date of last follow-up or death. Age, gender, ECOG-PS, stage, BRAF mutation status, melanoma subtype, localization, ulceration status, RFS and ORR were evaluated as prognostic factors.
Descriptive statistics were used to summarize baseline characteristics. Descriptive statistics were presented as frequency (percent) or median (range, min–max). The χ2 or Fisher’s exact tests were used to compare the proportions in different categorical groups. Continuous variables were analyzed with the Mann–Whitney U test. Kaplan-Meier survival curves were constructed to assess RFS, PFS and OS, and log-rank tests were used to compare survival distributions between treatment groups. We used the Chi-square test to compare independent categorical variables. Multivariate analyses were performed using Cox regression. Statistical analyses were performed using SPSS 23 and a value of p < 0.05 was accepted to indicate statistical significance.
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. Marmara University Ethics Committee approved the study (Approval Number 08.12.2023.1555).
Results
There were 120 patients in total, with a median age of 53 years (IQR: 18–85). Eighty-one patients (67.5%) were male. 89.2% of patients had cutaneous melanoma at diagnosis, and 10.8% had acral melanoma. Adjuvant treatment strategy: nivolumab (32.5%), dabrafenib plus trametinib (30.8%) and interferon (36.7%). (Table 1).
In terms of metastatic stage, 41.7% of patients were classified as M1a, followed by 29.2% as M1b and 14.2% as M1c. Among 120 patients, first-line treatment options included chemotherapy (temozolamid or carboplatin with paclitaxel) (21.7%), dabrafenib plus trametinib (36.7%) and ICI (nivolumab or ipilumumab or pembrolizumab) (41.7%) (Table 2). Metastatic first-line treatment of 37 patients who relapsed after adjuvant dabrafenib plus trametinib was 26 ICI; 5 dabrafenib plus trametinib; 6 chemotherapy. After adjuvant nivolumab, 39 patients who relapsed received 11 ICI; 8 dabrafenib plus trametinib and 20 chemotherapy in metastatic first-line treatment. Meanwhile of the 44 patients who relapsed after adjuvant interferon, 31 received dabrafenib plus trametinib and 13 received ICI in the metastatic first-line setting.
Median follow-up period of all patients was 61 months (95% CI 52.9–70.9 months). RFS was 3.6 months (95% CI 2.1–5.1 months) in the whole group and this duration was significantly different between treatment groups (p = 0.002). Patients treated with dabrafenib plus trametinib had the longest median RFS (8.3 months (95% CI 2.7–14.0 months)), followed by interferon (4.1 months (95% CI 0.0-13.1 months)) and nivolumab (1.9 months (95% CI 0.0-4.1 months)). ORR was highest in the dabrafenib plus trametinib group (54.5%), followed by ICI (52.0%) and chemotherapy (33.3%). DCR was superior in the dabrafenib plus trametinib group (86.3%) compared to ICI (70.8%) and chemotherapy (66.6%) (Table 3).
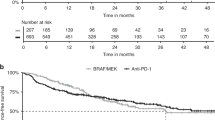
Median PFS was 9.7 months (95% CI 7.2–12.2 months) in the whole group. This was 14.3 months (95% CI 9.6–19.0 months) with ICI, 10.3 months (95% CI 4.2–16.4 months) with BRAF/MEK inhibitors and 6.3 months (95% CI 4.7–7.9 months) with chemotherapy, which was statistically significant (p < 0.001). Median OS was 51.3 months (95% CI 46.3–56.4 months) in the whole group. This was 53.5 months (95% CI 36.6–70.4 months) for first-line BRAF/MEK inhibitors, followed by ICI with a median OS not yet reached and chemotherapy with 33.6 months (95% CI 25.3–41.8 months) (p < 0.001) (Fig. 1).
Progression-free survival and overall survival.
In BRAF mutant melanoma, patients who received adjuvant dabrafenib plus trametinib followed by metastatic first-line immunotherapy and patients who received adjuvant immunotherapy followed by dabrafenib plus trametinib at relapse had similar overall survival that was not stastically significant (p = 0.37).
Univariate and multivariate analyses identified several factors that significantly affected overall survival. In univariate analysis, stage (HR: 0.29, 95% CI: 0.14–0.61, p = 0.001), BRAF V600E mutation (HR: 0.29, 95% CI: 0.10–0.87, p = 0.028), RFS time (HR: 0.51, 95% CI: 0.30–0.87, p = 0.015), ulceration (HR: 0.54, 95% CI: 0.30–0.97, p = 0.039) and ORR (HR: 0.37, 95% CI: 0.21–0.64, p = 0.001) were significantly associated with survival outcomes. Multivariate analysis confirmed the importance of BRAF V600E mutation (HR: 0.17, 95% CI: 0.46–0.66, p = 0.01), RFS time (HR: 0.47, 95% CI: 0.22–0.98, p = 0.04), ulceration (HR: 0.39, 95% CI: 0.17–0.88, p = 0.02) and ORR (HR: 0.23, 95% CI: 0.10–0.49, p = 0.01) as independent predictors (Table 4).
Discussion
In our study, we aimed to evaluate the real life data and survival outcomes of ICI and dabrafenib plus trametinib in both adjuvant and metastatic first-line treatment. We also analysed the factors affecting prognosis in overall survival. The findings emphasise the approach of dabrafenib plus trametinib or ICI, when used both adjuvantly and as first-line treatment for metatsatic cases, in managing this challenging patient population by improving response rates and prolonging survival.
As in many cancers, treatment management of melanoma has improved significantly with the contribution of ICI and BRAF/MEK inhibitors9. Our study aimed to evaluate the real-life data of these therapies in both adjuvant and metastatic periods and to improve survival outcomes.
In the adjuvant data of our study, as in other clinical trials, patients with BRAF mutant melanoma treated with dabrafenib plus trametinib had the longest duration of RFS, followed by interferon and nivolumab10. Similarly, our data regarding the fact that nivolumab showed favorable RFS outcomes is in line with the findings of the study in which nivolumab outperformed ipilimumab as adjuvant therapy11.
Though still widely used, the efficacy of interferon appears to be lower compared to modern ICI’s and targeted therapies12. Up until 6–7 years ago, interferon therapy was given in adjuvant melanoma treatment because ICI or targeted therapies were not available in Turkey. As observed in other studies, our research indicated that patients treated with interferon jad a shorter relapse-free survival (RFS) duration. Nevertheless, interferon remains an option in selected patients and in resource-limited countries, but its use is decreasing as more effective therapies are favored.
In first-line treatment for metastatic melanoma, our study, consistent with other research, demonstrated that targeted therapies and ICIs outperformed traditional chemotherapy. In BRAF mutant metastatic melanoma, patients receiving dabrafenib plus trametinib had significantly higher ORR and DCR compared to those treated with chemotherapy. These findings align with those of other studies13. Notably, ICI had a reduced DCR of 70.8% in contrast to BRAF/MEK inhibitors, which had a DCR of 86.3%; nevertheless, ORR was comparable. ICI, such as nivolumab and pembrolizumab, have demonstrated the ability to induce durable responses, with certain groups of patients achieving prolonged survival despite disease progression. This is one of th primary advantages of ICIs, despite the fact that responses may take longer to manifest compared to the more rapid effects observed with targeted therapy. Like adjuvant interferon, chemotherapy was used in the first-line management of metastatic cases when targeted therapies and ICI could not be achieved in certain periods. Yet similar to other studies, our findings revealed the lowest ORR and DCR, confirming its restricted role in the current management of melanoma12.
In terms of overall survival, multivariate analysis shows that the presence of BRAF V600E mutation and ulceration status are important factors influencing prognosis. Patients who achieved an objective response rate (CR + PR) showed better outcomes. These findings highlight personalized treatment strategies in the management of melanoma for certain high-risk patients with BRAF V600E mutation and ulcer positivity.
PFS and OS results also emphasize the efficacy of treatments in first-line metastatic melanoma. Dabrafenib plus trametinib and ICI provided superior survival benefits compared to chemotherapy; with BRAF/MEK inhibitors, OS was 53.5 months, while this has not yet been reached with ICI. These findings are similar to studies highlighting the durable survival benefits of ICI in metastatic melanoma14,15. Although these therapies are effective, they are not without their challenges. One of the most important problems is resistance to targeted therapies. A group of patients who initially respond rapidly to dabrafenib plus trametinib progress over time due to the development of a resistance mechanism. To solve these problems, combination strategies such as adding ICI to targeted therapies are still under
Limitations
There are many limitations on our search. This study is retrospective in nature, including multicenter, and it is biased in terms of selection and treatment protocols. Longer follow-up durations and prospective studies are needed to verify our results while studying combination therapy approaches in more detail.
Conclusion
In conclusion, dabrafenib plus trametinib was more effective in adjuvant treatment of melanoma, while immunotherapy was more effective in metastatic first-line treatment. Immunotherapy was also more effective in overall survival. RFS > 6 months is an independent factor for prognosis. More comprehensive and prospective studies are needed to optimize melanoma management, investigate mechanisms of drug resistance, and address long-term survival outcomes.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Leonardi, G. et al. Cutaneous melanoma: from pathogenesis to therapy (review). Int. J. Oncol. ; (2018).
Davis, L. E., Shalin, S. C. & Tackett, A. J. Current state of melanoma diagnosis and treatment. Cancer Biol. Ther. 20 (11), 1366–1379 (2019).
Orzan, O. A., Șandru, A. & Jecan, C. R. Controversies in the diagnosis and treatment of early cutaneous melanoma. J. Med. Life. 8 (2), 132–141 (2015).
Long, G. V. et al. Adjuvant dabrafenib plus Trametinib in Stage III BRAF -Mutated Melanoma. N. Engl. J. Med. 377 (19), 1813–1823 (2017).
Weber, J. et al. Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. N. Engl. J. Med. 377 (19), 1824–1835 (2017).
Lazaroff, J. & Bolotin, D. Targeted therapy and immunotherapy in Melanoma. Dermatol. Clin. 41 (1), 65–77 (2023).
Atkins, M. B. et al. Combination Dabrafenib and Trametinib Versus Combination Nivolumab and Ipilimumab for patients with Advanced BRAF -Mutant Melanoma: the DREAMseq Trial—ECOG-ACRIN EA6134. J. Clin. Oncol. 41 (2), 186–197 (2023).
Ascierto, P. A. et al. Sequencing of Ipilimumab Plus Nivolumab and Encorafenib Plus Binimetinib for untreated BRAF -Mutated metastatic melanoma (SECOMBIT): a Randomized, Three-Arm, open-label phase II trial. J. Clin. Oncol. 41 (2), 212–221 (2023).
Seth, R. et al. Systemic therapy for Melanoma: ASCO Guideline Update. J. Clin. Oncol. 41 (30), 4794–4820 (2023).
Dummer, R. et al. Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAFV600-mutant, stage III melanoma (COMBI-AD): exploratory biomarker analyses from a randomised, phase 3 trial. Lancet Oncol. 21 (3), 358–372 (2020).
Larkin, J. et al. Adjuvant Nivolumab versus Ipilimumab in Resected Stage III/IV Melanoma: 5-Year efficacy and Biomarker results from CheckMate 238. Clin. Cancer Res. 29 (17), 3352–3361 (2023).
Shi, Q. et al. Interferon-α1b for the treatment of metastatic melanoma: results of a retrospective study. Anticancer Drugs. 32 (10), 1105–1110 (2021).
Latimer, N. R., Abrams, K. R., Amonkar, M. M., Stapelkamp, C. & Swann, R. S. Adjusting for the Confounding effects of Treatment switching—the BREAK-3 trial: Dabrafenib Versus Dacarbazine. Oncologist 20 (7), 798–805 (2015).
Hodi, F. S. et al. Nivolumab plus Ipilimumab or Nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 19 (11), 1480–1492 (2018).
Robert, C. et al. Pembrolizumab versus Ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 20 (9), 1239–1251 (2019).
Funding
Authors received no specific funding for this work.
Author information
Authors and Affiliations
Contributions
Conceptualization, N.M. and O.S.; methodology, A.O. and N.M.; software, N.M and E.A.; validation, O.D., N.M., T.E., B.Y., I.O.K., C.K. and M.K.; formal analysis, ˙T.B., N.S.D., A.B., M.M.A., F.S. and N.M.; investigation, N.M., H.I.E.,N.S.; resources, N.M. and H.A.; data curation, N.M., A.K.G., Y.C., F.R.O., O.B., G.O.K., B.E.O, Y.S., T.S., S.Y., İ.D., G.A., A.O., M.G., E.S., E.E.D., A.A., O.A., N.I., O.A., and S.A.; writing—original draft preparation, N.M.; writing—review and editing, N.M.; visualization, N.M.; supervision, N.M.; project administration, N.M.; funding acquisition, N.M. All authors have read and agreed to the published version of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. Marmara University Ethics Committee approved the study (Approval Number 08.12.2023.1555)
Informed consent
Informed consent was waived by (Marmara University Ethics Committee approved the study (Approval Number 08.12.2023.1555)) due to retrospective nature of study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Majidova, N., Arak, H., Ozalp, F.R. et al. Prognostic factors and outcomes of adjuvant and first-line metastatic treatments in melanoma a Turkish oncology group study. Sci Rep 15, 3200 (2025). https://doi.org/10.1038/s41598-025-87553-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87553-z