Abstract
Cancer is a complex health issue that today’s medical science is dealing with, which has a mortality rate that is almost identical to that of cardiac disorders. Because of the adverse effects of the majority of the medications used in its therapy, managing it remains a major concern despite the availability of numerous remedies. This article attempts to contribute to the scientific developments in the Solanaceae family’s anti-cancer field. Thus, this study consisted of isolating β-amyrin, cedryl acetate, lupeol, and 2-pentadecanone, 6,10,14-trimethyl from Cestrum aurantiacum to determine the anti-tumor activity. The plant material was dried, pulverized, and small-scale extraction was done. Brine shrimps, cell lines (A549, Hela, HEPG), and Caenorhabditis elegans were used initially to examine three solanaceous plant extracts (Solanum villosum (SV), Cestrum aurantiacum (CA), and Brugmansia suaveolens (BS)). The best results were shown by ethanol extract of Cestrum aurantiacum that why large-scale extraction and GCMS of this extract were done. The antitumor potential can be explained by the presence of β-amyrin, cedryl acetate, lupeol, and 2-pentadecanone, 6,10,14-trimethyl.
Similar content being viewed by others
Introduction
Cancer is the second leading cause of death worldwide after heart disorders. Every year, about 6 million individuals lose their lives to cancer, out of 10 million diagnoses1. Lung, mouth, stomach, and esophageal cancers are more common in men, but breast, uterine, and cervical cancers are more common in women, according to The National Cancer Registry Programme Report, 2020 of Indian Council of Medical Research (ICMR). According to estimates, the number of cancer cases will probably increase from 13.9 lakh in 2020 to 15.7 lakh by 2025. Even though cancer patients now have more options because of advancements and innovation in the field, it is crucial to provide anti-cancer drugs that are non-toxic, widely accessible, and reasonably priced2. The anti-cancer potential of over 3000 plants has been revealed worldwide3. Currently, natural plant products account for around 60% of the anti-cancer drugs now in use4. It’s interesting to note that 90 of the approximately 120 medications used to treat cancer come from plants2.
Solanaceae is one of the diverse plant families of the 405 angiosperm groups, with a wide range of beneficial chemical components5. The Solanaceae family is significant for both medicinal and economic plants6. Conversely, some of the most significant Solanaceae plants influenced the early phases of medical plant-based drug development and are still valued in herbal medicine7. There are over 2700 species in the Solanaceae family, divided into 98 genera8. Numerous traditional medical systems, such as Ayurveda, Traditional Chinese Medicine (TCM), Siddha, Unani, and homeopathy, employ some of the alkaloids found in the Solanaceae family7,9. Considering the enormous anti-cancer potential of Solanaceae plants, we designed the current investigation by selecting three Solanaceae plants (Solanum villosum (SV), Cestrum aurantiacum (CA), and Brugmansia suaveolens (BS) to search for anti-cancer compounds.
Leaves of Brugmansia suaveolens are traditionally applied in northern Peru for treating wounds10. Flowers and leaves are used for curing infections, mental weaknesses, and menstrual pain. It is utilized as an aphrodisiac11. The stem is applied externally for skin anomalies for rapid healing12. Brugmansia has hallucinogenic effects, it also has anti-inflammatory, antirheumatic, and analgesic properties13. Solanum villosum leaves and fruits are eaten or made into decoctions to cure stomach problems like dysentery and diarrhea. This is common in traditional African medicine14. The plant is frequently eaten as a vegetable, particularly in East Africa, where its high nutrient profile is thought to support immunity and overall health15. Applying crushed leaves straight to cuts and wounds speeds up the healing process. The antibacterial qualities of the plant are thought to aid in the prevention of infections. It also has anti-inflammatory properties16. Traditional use of extracts supports their use in treating a variety of illnesses due to their antibacterial and antifungal properties17.
Genus Cestrum comprises more than 300 species and is widely distributed in tropical and subtropical areas around the world like Bangladesh, India, the United States, Australia, South America and southern China.18,19 Cestrum species have a long history in folk medicine for the treatment of several diseases and health disorders.18,20Different species belonging to the Cestrum genus were previously investigated for their chemical constituents, e.g., parquine, carboxyparquine and steroids. Glycosides were also identified in Cestrum diurnum.21 whereas saponins were identified in Cestrum parqui.22,23.
A variety of bioactive chemicals found in Cestrum plants have been linked to their cytotoxic and antitumor effects. The pharmacological actions of Cestrum species are attributed to the presence of flavonoids, alkaloids, and saponins, as revealed by phytochemical research.19,24. Due to the substantial cytotoxic effects these compounds have demonstrated against various cancer cell lines in vitro, Cestrum species are promising candidates for developing anticancer drugs. For instance, Nasr et al. 24 studied extracts from two Cestrum species and found promising cytotoxic effects against certain cancer cells, confirming the traditional use of these plants for medical purposes.
Cestrum aurantiacum (Orange cestrum) is a plant used for various medicinal purposes in traditional South Asian medicine, especially Ayurveda. The plant is mostly known for its therapeutic properties in many traditional healing methods. Roots and leaves have been used for their anti-inflammatory properties. Occasionally, they are used as decoctions or applied as poultices to relieve joint pain, muscle pains, and swelling. It has antimicrobial properties and is frequently used to treat fungal diseases, wounds, and skin infections. It can also be used to relieve constipation, bloating, and stomach pain. It may be used as a traditional remedy for respiratory conditions including colds and coughs. Occasionally, preparations that aid in clearing respiratory passages contain it. The plant has been used in some cases as a febrifuge to help reduce fever, although it has to be used with caution as certain parts of the plant can be poisonous when consumed in the wrong way. The plant is often used in controlled doses in traditional practices and caution is advisable when using it as a medicine.25.
The current study aimed to use bioassay-guided purification to extract the main antitumor components of solanaceous plant species.
Materials and methods
Reagents
Solvents of the high-performance liquid chromatography (HPLC) grade, such as acetone, methanol, acetonitrile, hexane, ethanol, Deionized water and DMSO were used.
Collection and identification of plants
From spring 2017 to spring 2018, solanaceous plant species were collected from different areas of Pakistan. Solanum villosum was collected from Narowal, province of Punjab. Cestrum aurantiacum and Brugmansia suaveolens were collected from Islamabad. Prof. Dr. Rahmatullah Qureshi, identified the specimens and voucher specimens were stored for future use as reference (Voucher # 168, 221, 339). After the plants were completely dried, a fine powder was made and preserved. All research studies and methods were carried out per institutional, national, and international guidelines and legislations. We got the appropriate permissions and licenses for the collection of plant specimens from the Administration Department, PMAS-Arid Agriculture University Rawalpindi, and the Department of Agriculture, Government of Punjab, Pakistan.
Small-scale extraction
The plant material that had completely dried out was ground into a powder, and by utilizing a technique designed by Panda et al.26, small-scale extraction was accomplished. Ten milliliters of solvent (hexane, chloroform, Acetone, ethanol, and water) was used with 1 g of plant powder (S. villosum, C. aurantiacum, and B. suaveolens).
After every four hours, the tubes were put in a sonicator bath for one hour. One milliliter of aliquots from each fraction was dried in a Savant SpeedVac Concentrator 200H.
Cytotoxic activities
Brine shrimp lethality assay
For brine shrimp assay cysts of Artemia salina were hatched in two-chambered containers with small holes in the partitioning wall. For seawater 38 g/L sea salt was taken in a beaker and oxidation of this water was done by using a magnetic stirrer for approximately 2 h. After approximately 48 h’ cysts hatched and swam through the hole to the illuminated chamber, from where shrimps were collected for an experiment. The tested extracts were dissolved in 100% DMSO and used as a stock solution at a concentration of 60 mg/mL. Utilizing artificial seawater, different concentrations were prepared from stock. Artificial seawater was employed as a negative control, and nicotine was used as a positive control. Ten phototropic nauplii were introduced to each container and cultured for 24 h at room temperature. Dead nauplii were counted and the percentage of lethality was calculated after 24 h.27.
Percentage of Death = (Total nauplii−Alive nauplii) / Total nauplii × 100.
In-vitro cytotoxicity assay (MTT assay)
The KU Leuven Zoology Department’s Animal Physiology and Neurobiology lab provided the cell lines. The cells were stored in liquid nitrogen stock, Thawing was done before use. After thawing, the cells were moved to a sterile falcon tube, and 5 mL of cell culture medium was added. The density of the cells was examined under a microscope, after which 2 mL of cell culture had been transferred. A cell culture flask was filled with 15 mL of Dulbecco’s Modified Eagle’s Medium (DMEM). The flask was then incubated at 37 °C for 24 h in an incubator with 5 percent CO2 to allow the cell to attach to and develop on the flask’s surface.
In a flask, 5 mL of complete media was added to trypsinized cells. In a 15 mL falcon tube, centrifuged for 5 min at 500 rpm. The medium was withdrawn, and cells were re-suspended in complete media (1.0 mL). Cells were counted, and full media was used to dilute it to 75,000 cells per mL in a 96-well plate. Fill each well with 100 µL of cells (7500 total cells) and set it in a CO2 incubator overnight. The next day, tested extracts were added, and the ultimate amount was held at 100 µL each well. Each well received 20 µL of 5 mg/mL MTT. As a control, MTT was added to one set of wells with no cells. These plates were incubated in a CO2 incubator for 3.5 h at 37 °C. Absorbance was measured at 590 nm using a 620 nm reference filter.28.
C. elegans lethality assay
C. elegans were cultured on Nematode Growth Medium (NGM). The N2 wild-type C. elegans strain was utilized. C. elegans were treated to synchronization at the L4 larval stage. 96-well microplate with a flat bottom from TPP Techno Plastic Products AG in Switzerland was used. A 96-well microplate with 189 μL of E. coli OP50 culture (OD = 0.5 at 620 nm) was filled with synchronized C. elegans 10 μL, or roughly 40 to 45 numbers of L4 larvae. Plant extract in the amount of 1 μL was tested; 1 μL of DMSO was used as a solvent control, and 1 μL of 50 μM Levamisole was used as a positive control. Worms and extracts were combined, and then a 96-well microplate was put into a WMicroTracker (Phylumtech, Argentina) device and incubated for 24 h at 20 °C. Every 30 min, the WMicroTracker measured and recorded the worm movement in each well. To determine the relative activity, the percentage of the average movement over 24 h of test samples with extract compared to the DMSO control was employed.29.
Large-scale extraction
For large-scale extraction, a big container containing 2000 mL of ethanol HPLC grade from Sigma (Germany) was filled with 200 g of plant powder (C. aurantiacum). The container was kept at room temperature for a full day. To increase the extraction yield, the container was submerged in a water bath sonicator four times for a duration of sixty minutes each. There was a minimum 6-h pause between each sonication to allow the suspension to reach room temperature.
The plant material was filtered using VWR Grade 313 size 5 lm filter paper after the fourth sonication to obtain the dry residue. The filtrate was then evaporated using a rotary evaporator (BUCHI rotavapor R-100), and the weight of the first dried extract was determined. The recovered ethanol was utilized once more for a second extraction using the same protocol for twenty-four hours. The filtrate was then again evaporated using a rotary evaporator to obtain the dry extract.
To extract every ingredient from the plant powder, the same process was carried out repeatedly. The dried material’s final weight was computed. For additional examination, the plant extract was kept in storage at 4 °C.
Purification
A silica gel was used to bind the plant extract, and a silica column was made. Hexane, ethyl acetate, methanol, and acetic acid were blended into eluates using a step gradient that increased in polarity. Finally, 100% acetic acid was used to elute the column.
Reverse-phase high-performance liquid chromatography analysis
A Shimadzu LC-20AT system with an LC-20AT quaternary pump and an online degasser (DGU20A3/DGU-20A5) was used to perform high-performance liquid chromatography (HPLC–DAD) analyses. Lab Solution software was used to obtain the data. The HPLC column was in the reverse phase. The mobile phase consisted of acetonitrile, water, and 0.1% trifluoroacetic acid (TFA).
Gas chromatography-mass spectrometry
The peaks that were collected were put via gas chromatography. Using an internal diameter of 0.18 mm and a thickness of 0.18 mm, a Restek RXi-5sil MS 20 m column was employed, and the NIST 14 MS library was utilized to search the spectrum.
Results
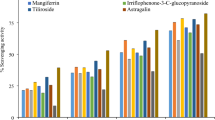
Brine shrimps, cell lines (A549, Hela, HEPG), and C. elegans were used initially to examine three solanaceous plant extracts (S. villosum (SV), C. aurantiacum (CA), and B. suaveolens (BS)). The extract of C. aurantiacum was shown to have a broader spectrum of efficacy against all the tested activities. The cytotoxic activity of all three solanaceous plant extracts was examined by using different solvents from low polarity to high polarity (hexane, chloroform, acetone, ethanol, and water). The findings indicated that the most active ingredients were in the ethanol extract of C. aurantiacum (Figs. 1, 2 and 3).
Mean % lethality of brine shrimp in different solvents of selected plant species.
Inhibitory effects (%) of selected plants on various cell lines using different solvents.
Percentage lethality of C. elegans in different solvents of selected plant species.
Therefore, ethanol was used to conduct a large-scale extraction, and the resulting extract was then further divided into 220 fractions using column chromatography (Fig. 4). All fractions were evaluated for activity against Hela cell line; fractions 31–38, 54–57 and 122–128 were confirmed to be active (Fig. 5). After being dried, the active portions were weighed. A serial dilution approach was used to test the active fractions against the HeLa cell line once more (Fig. 6). The results showed that fractions 31–38 consistently exhibited cytotoxic activity, suggesting the presence of a potent active antitumor component. Consequently, fractions 34 and 37 underwent further separation with the use of a mobile phase comprising acetonitrile and water. Hela cells were used to evaluate all 60 subfractions once more, and the results showed that there were two active subfractions in each fraction (Fig. 7 and 8). For identification, active peaks were subjected to GCMS (Fig. 9, 10, 11 and 12). It was determined that compound 1 was β-amyrin, compound 2 was cedryl acetate, compound 3 was lupeol, and compound 4 was 2-pentadecanone, 6,10,14-trimethyl.
An overlay chromatogram of an ethanol extract of Cestrum aurantiacum from a silica gel column; fractions were taken per minute.
The cytotoxic activity of the Cestrum aurantiacum fractions obtained by the silica column.
Cytotoxic activity of most active fractions of Cestrum aurantiacum against hela cell line using a twofold dilution protocol.
(a) HPLC chromatogram of fraction 34 (Cestrum aurantiacum); fractions were collected per minute. (b) Tested for activity (percentage inhibition of cancer cells).
(a) HPLC chromatogram of fraction 37 (Cestrum aurantiacum); fractions were collected per minute. (b) Fractions tested for activity (percentage inhibition of cancer cells).
Mass spectra of Compound 1 (β-Amyrin) and its structure.
Mass spectra of Compound 2 (Cedryl Acetate) and its structure.
Mass spectra of Compound 3 (Lupeol) and its structure.
Mass spectra of Compound 4 (2-Pentadecanone, 6,10,14-Trimethyl) and its structure.
Discussion
There are around 300 species of the genus Cestrum (Family Solanaceae), which is extensively spread in tropical and subtropical regions of the world, including Bangladesh, India, the United States, Australia, South America, and southern China.18,19 In traditional medicine, Cestrum species have long been used to treat a variety of illnesses and ailments.18,20 In Chinese traditional medicine the leaves of C. nocturnum to cure a variety of ailments. The plant exhibited biological actions that were detectable, such as antitumor, antioxidant, hepatoprotective, cytotoxic, antibacterial, anticonvulsant, larvicidal, anti-inflammatory, analgesic, antitumor, and wound healing. Previous studies on the chemical composition of various Cestrum species, such as parquine, carboxyparquine, and steroids, were conducted. While C. parqui was found to contain saponins, C. diurnum was also shown to contain glycosides.24 Cestrum aurantiacum was least investigated, β-amyrin, cedryl acetate, lupeol, and 2-pentadecanone, 6,10,14-trimethyl were isolated from this species.
β-amyrin has been identified from different plants i-e from 5 varieties of Humulus lupulus30. Celastrus hindsii leaves31, Alstonia boonei32 and several materials have been found to contain amyrin, including an ethanolic fraction of the oleogum resin from Ferula gummosa33, methanolic extract of Carpobrutus edulis34 Chloroform extract of Euphorbia tirucalli L.35leaves of Ficus benjamina36 leaves of Pyrenacantha staudii,37 Olea europea L. leaves (ethanol extract)38. Leaves of Tectona philippinensis and Rhus alata39 and Piptadenia Africana stem bark also contain β-Amyrin40.
The chemical structure of β-amyrin (3β- hydroxy-olean-12-en-3-ol) is also depicted in Figure and its formula is C30H50O. The infra-red spectrum of β-amyrin shows the presence of a hydroxyl function and the olefinic moiety at a spectrum of 3360 and 1650 cm-1 and MS studies of β-amyrin confirm a parent ion peak at m/z 426 (M +)41, other work of HR-EI-MS m/z: 426.2975 (calcd. for C30H50O, 426.3861) (Jabeen et al., 2011). The melting point of β-amyrin is 189–191 ºC42.
Cedryl acetate is a sesquiterpene compound, the pure chemical is crystalline, which is derived from natural oils found to be volatile in nature. A tricyclic sesquiterpene called cedryl acetate was discovered in the plant Psidium caudatum. Cedryl acetate exhibits antiglucosidase action. Cedryl acetate is also present in Cunninghamia lanceolata var. konishii.43 In vitro anticancer, and antimicrobial activities of the Heartwood Essential Oil of Cunninghamia lanceolata var. konishii was evaluated from Taiwan, the oil exhibited cytotoxic activity against human lung, liver and oral cancer cells.44 Crataegus Azarolus L has Cedryl acetate so evaluated for its antioxidant, Anti-Inflammatory, and Antiproliferative Activities.45 It has Density 0.999 g/mL at 25 °C. Melting point 44–46 ℃. The boiling point is 291.7 °C at 760 mmHg.43.
A hydroxy group replaces the hydrogen at the 3beta position of the pentacyclic triterpenoid lupane to form lupeol, which is present in fruits and vegetables, preferentially caused significant head and neck squamous cell carcinoma (HNSCC) cell death in vitro but had little to no impact on a normal tongue fibroblast cell line. The primary mechanism of lupeol’s anticancer effects against HNSCC has been found to be down-regulation of NF-kappaB. Lupeol alone was discovered to reverse the NF-kappaB-dependent epithelial-to-mesenchymal transition, which not only reversed tumor growth but also impaired HNSCC cell invasion. Lupeol and cisplatin together had a synergistic impact that in vitro chemosensitized HNSCC cell lines with significant NF-kappaB activity46. Previously it has been reported from different plants like Derris scandens, Albizia procera, and Diospyros rhodocalyx.47 Nyctanthes arbor-tristis was also invested for lupeol compound.48.
2-pentadecanone, 6,10,14-trimethyl is present in the aerial parts of Andrographis paniculata49 leaves of Combretum latifolium50 Crinum latifolium L.51 and Curcuma aromatica Salisb52 while in the flower of Jasminum graniflorum53 and also in the Rumex vesicarius54. This substance is a member of the sesquiterpenoids class of organic substances. These terpenes have three isoprene units following one another with density 0.590336 and Electronegativity -4.861.
Correlation between identified compounds and the investigated biological activities
β-Amyrin and biological activities
According to Sandeep et al.55, β-amyrin demonstrated cytotoxicity in brine shrimp lethality assays, a standard screening method for bioactive compounds. This cytotoxicity may be attributed to its ability to disrupt cellular membranes and induce apoptosis in these organisms. A study by Moy et al.56showed that β-amyrin induces apoptosis in A549 cells, reducing cell proliferation and survival. The apoptosis-inducing ability of β-amyrin in A549 cells supports its potential as an anticancer agent. Gautam et al.57highlighted the anticancer properties of β-amyrin, demonstrating its ability to inhibit cell growth and induce cell cycle arrest in HeLa cells. This action in HeLa cells is linked to β-amyrin’s anti-inflammatory and apoptosis-inducing properties. Bhuvaneshwaran et al.58reported that β-amyrin suppresses liver cancer cell proliferation and induces apoptosis through ROS generation. Its ability to induce oxidative stress makes it effective in liver cancer cells, corroborating its anticancer action. The antioxidant activity of β-amyrin was demonstrated in C. elegans by Rani et al.59, where it was shown to protect against oxidative stress. This suggests that β-amyrin may offer protective effects beyond cancer studies to stress and aging-related models in C. elegans.
Cedryl acetate and biological activities
Cedryl acetate is a sesquiterpene ester with antimicrobial, anti-inflammatory, and potential anticancer activities.
Subramanian et al.60found that cedryl acetate exhibited cytotoxic effects in brine shrimp assays, suggesting it may disrupt cellular integrity Cedryl acetate’s cytotoxic effects in brine shrimp may be related to its ability to induce apoptosis or cell membrane disruption. In a study by Sharma et al.61, cedryl acetate was shown to have anti-inflammatory effects in lung cancer models. Its anti-inflammatory properties could contribute to reducing lung cancer cell proliferation, possibly by inhibiting inflammatory pathways that promote cancer growth. Prasad et al.62reported that cedryl acetate has anti-cancer effects by inducing apoptosis in HeLa cells Cedryl acetate’s potential to induce apoptosis in cervical cancer cells further supports its use as a therapeutic agent. Reddy et al.63showed that cedryl acetate has antioxidant properties that could influence liver cancer cells by mitigating oxidative damage This suggests cedryl acetate may modulate oxidative stress pathways involved in liver cancer progression.The antimicrobial activity of cedryl acetate was studied by Das et al.64, where it showed potential toxicity to C. elegans under laboratory conditions: Its toxicity could be related to the disruption of cellular functions in C. elegans, possibly through membrane destabilization.
Lupeol and biological activities
Lupeol is a triterpene known for its anticancer, anti-inflammatory, and antioxidant effects. Shah et al.65found that lupeol exhibited significant cytotoxicity in brine shrimp assays Lupeol’s cytotoxicity is likely due to its ability to induce apoptosis or interfere with cellular metabolism. Tripathi et al.66 demonstrated that lupeol inhibits A549 cell proliferation and induces apoptosis via the activation of caspase-3 and -9. Lupeol’s apoptosis-inducing activity in A549 cells is key to its anticancer effects. Borse et al.67 showed that lupeol has potent anti-proliferative and apoptotic effects in HeLa cells, reducing cell viability and migration. Lupeol’s anticancer action is further confirmed in cervical cancer models by inhibiting growth and metastasis. Anwar et al.68 found that lupeol suppresses liver cancer cell proliferation by inhibiting the PI3K/AKT signaling pathway. Lupeol’s inhibition of critical signaling pathways in liver cancer cells supports its potential as an anti-liver cancer agent. Ali et al.69 reported that lupeol extends lifespan and promotes antioxidant activity in C. elegans, indicating its protective effects against oxidative stress. The ability of lupeol to protect C. elegans from oxidative damage further supports its antioxidant and health-promoting effects.
2-Pentadecanone, 6,10,14-trimethyl and biological activities
2-Pentadecanone, 6,10,14-trimethyl is a ketone compound known for its antimicrobial and possible anticancer effects. Chavan et al.70 reported that 2-pentadecanone exhibited cytotoxicity in brine shrimp lethality tests. Its cytotoxicity is linked to its ability to disrupt cellular membranes or induce apoptosis. Sharma et al.71 found that 2-pentadecanone inhibits the growth of A549 cells by inducing cell cycle arrest and apoptosis. The compound’s ability to halt cell cycle progression and induce apoptosis makes it a potential anticancer agent.
Kumar et al.72 suggested that 2-pentadecanone shows promising anti-cancer activity in HeLa cells by altering cell morphology and inducing cytotoxicity. The anti-cancer effect in HeLa cells indicates that this compound may interfere with cellular integrity or metabolism. Venkatesan et al.73 showed that 2-pentadecanone has potential anticancer properties in liver cancer models, possibly by inducing oxidative stress and apoptosis. Its oxidative stress-inducing properties align with its role in liver cancer treatment. In C. elegans, 2-pentadecanone showed toxicity, possibly by affecting metabolic pathways or inducing cellular stress. Its toxicity in C. elegans highlights its potential for further study in toxicity and metabolic disruption.
Across several model systems, these studies demonstrate a high correlation between the biological activities of the substances β-amyrin, cedryl acetate, lupeol, 2-pentadecanone, and 6,10,14-trimethyl. Their potential as anticancer medicines are suggested by their shared capacity to induce apoptosis, inhibit cell proliferation, and function as antioxidants. These correlations, which were derived from various research papers, highlight the therapeutic value of these compounds in cancer treatment.
Conclusion
This study highlights the anticancer potential of ethanol extracts from Cestrum aurantiacum, with the identification of key bioactive compounds such as 2-pentadecanone, 6,10,14-trimethyl; lupeol; cedryl acetate; and β-amyrin. Notably, lupeol and 2-pentadecanone, 6,10,14-trimethyl are reported for the first time in the genus Cestrum, while β-Amyrin marks its debut from Cestrum aurantiacum. These findings underscore the untapped therapeutic potential of this plant and its unique phytochemical profile. Moving forward, the exploration of these compounds through advanced biological assays, molecular docking, and in vivo studies could provide deeper insights into their mechanisms of action against cancer. Furthermore, integrating modern approaches like nanocarrier systems for targeted delivery and AI-driven compound screening could propel these bioactives toward clinical relevance. This work lays a foundation for the future of plant-based oncological therapeutics, bridging traditional knowledge with cutting-edge science.
Data availability
Data available on request from the corresponding author (Yamin Bibi).
Abbreviations
- CME:
-
Crude Methanol Extract
- A549:
-
Lung Cancer Cells
- Hela:
-
Cervical Cancer Cells
- HEPG:
-
Liver cancer cells
- SV:
-
Solanum villosum
- CA:
-
Cestrum aurantiacum
- BS:
-
Brugmansia suaveolens
- GCMS:
-
Gas Chromatography Mass Spectrometry
- ICMR:
-
Indian Council of Medical Research
- TCM:
-
Traditional Chinese Medicine
- HPLC:
-
High-performance liquid chromatography
- DMSO:
-
Dimethyl sulfoxide
- PMAS:
-
Pir Mehr Ali Shah
- BSLT:
-
Brine shrimp lethality test
- MTT:
-
3-(4, 5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NGM:
-
Nematode Growth Medium
- C. elegans :
-
Caenorhabditis elegans
- E. coli OP50 culture:
-
Escherichia coli OP50 culture
- HPLC-DAD:
-
High-performance liquid chromatography with diode-array detection
- ROS:
-
Reactive oxygen species
References
Hanif, M., Zaidi, P., Kamal, S. & Hameed, A. Institution-based cancer incidence in a local population in Pakistan: Nine-year data analysis. Asian Pac. J. Cancer Prev. 10, 227–230 (2009).
Rohilla, P., Jain, H., Chhikara, A., Singh, L. & Dahiya, P. Anticancer potential of Solanaceae plants: A review. South Afr. J. Bot. 149, 269–289 (2022).
Tariq, A. et al. A systematic review on ethnomedicines of anti-cancer plants. Phytother. Res. 31, 202–264 (2017).
Gordaliza, M. Natural products as leads to anticancer drugs. Clin. Transl. Oncol. 9, 767–776 (2007).
Fatur, K. “Hexing herbs” in ethnobotanical perspective: A historical review of the uses of anticholinergic Solanaceae plants in Europe. Econ. Bot. 74, 140–158 (2020).
Ghatak, A. et al. Proteomics survey of the Solanaceae family: Current status and challenges ahead. J. Proteomics 169, 41–57 (2017).
Chowanski, S. et al. A review of bioinsecticidal activity of Solanaceae alkaloids. Toxins 8, 1–28 (2016).
Olmstead, R. G. & Bohs, L. A summary of molecular systematic research in Solanaceae: 1982–2006. In VI International Solanaceae Conference: Genomics Meets Biodiversity 745 (International Society for Horticultural Science, Belgium, 255–268 (2006).
Shah, V. V., Shah, N. D. & Patrekar, P. V. Medicinal plants from Solanaceae family. Res. Pharm. Technol. 6, 143–151 (2013).
De Feo, V. Ethnomedical field study in northern Peruvian Andes with particular reference to divination practices. J. Ethnopharmacol. 85, 243–256 (2003).
Descola, P. Phytochemical and antibacterial studies on Jatropha curcas L. J. Chem. Pharm. Res. 4, 2639–2642 (1996).
Ratsch, C. Enziklopädie der Psychoaktiven Pflanzen (AT Verlag, 1998).
Nencini, C. et al. Affinity of Iresine herbstii and Brugmansia arborea extracts on different cerebral receptors. J. Ethnopharmacol. 105, 352–357 (2006).
Chandra, S. et al. Ethnomedicinal plants of the Solanum genus: A review. J. Med. Plants Res. 8, 457–468 (2014).
Grubben, G. J. H. & Denton, O. A. Plant Resources of Tropical Africa: Vegetables. PROTA Foundation (2004).
Kokwaro, J. O. Medicinal Plants of East Africa (University of Nairobi Press, 2009).
Oudhia, P. Traditional medicinal uses of Solanum species. Ethnobot. Leaflets 5, 1–4 (2001).
Begum, A. S. & Goyal, M. Research and medicinal potential of genus Cestrum (Solanaceae)—A review. Pharmacogn. Rev. 1, 320 (2007).
Prasad, M. P., Apoorva, P., Thakur, M. S. & Ruparel, Y. M. Phytochemical screening, antioxidant potential, and antimicrobial activities in three species of Cestrum plants. Int. J. Pharm. Biol. Sci. 14, 673–678 (2013).
Al Raza, S. M., Rahman, A. & Kang, S. C. Chemical composition and inhibitory effect of essential oil and organic extracts of Cestrum nocturnum L. on food borne pathogens. Int. J. Food Sci. Technol. 44, 1176–1182 (2009).
Durand, R., Figueredo, J. M. & Mendoza, E. Intoxication in cattle from Cestrum diurnum. Vet. Hum. Toxicol. 41, 267–268 (1999).
Chaieb, I., Kamel, B., Trabelsi, M., Hlawa, W., Raouani, N., Ben Ahmed, D. et al. Pesticidal potentialities of Cestrum parqui saponins. Int. J. Agric. Res. 2, 275–281 (2007).
Najet, R., Dorsaf, B. A., Mejda, D. & Habib, B. H. Pesticidal potentialities of Cestrum parqui saponins. Int. J. Agric. Res. 2, 275–281 (2007).
Nasr, S. M. et al. High performance liquid chromatography fingerprint analyses, in vitro cytotoxicity, antimicrobial and antioxidant activities of the extracts of two Cestrum species growing in Egypt. Pharmacogn. Res. 10, 173 (2018).
Sivarajan, V. V. & Balachandran, I. Ayurvedic Drugs and Their Plant Sources (IBH Publishing Co, Oxford, 1994).
Panda, S. K. et al. Antimicrobial, anthelmintic, and antiviral activity of plants traditionally used for treating infectious diseases in the similipal biosphere reserve, Odisha India. Front. Pharmacol. 8, 658 (2017).
Sasidharan, R. & Gerstein, M. Protein fossils live on as RNA. Nature 453, 729–731 (2008).
Joo, T. et al. Inhibition of nitric oxide production in LPS-stimulated RAW 264.7 cells by stem bark of Ulmus pumila L.. Saudi J. Biol. Sci. 21, 427–435 (2014).
Panda, S. K., Das, R., Mai, A. H., De Borggraeve, W. M. & Luyten, W. Nematicidal activity of Holigarna caustica (Dennst.) Oken fruit is due to linoleic acid. Biomolecules 10, 1043–1049 (2020).
Rokicka, K. & Wojciak-Kosior, M. Identification and quantitative analysis of 161 amyrins in Humulus lupulus L. Curr. Issues Pharm. Med. Sci. 27, 142–144 (2015).
Viet, T. D., Xuan, T. D. & Anh, L. H. α-Amyrin and β-Amyrin isolated from Celastrus hindsii leaves and their antioxidant, anti-xanthine oxidase, and anti-tyrosinase potentials. Molecules 26, 7248–7256 (2021).
Okoye, N. N. et al. β-Amyrin and α-amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity. Pharm. Biol. 52, 1–10 (2014).
Jalali, H. T., Ebrahimian, Z. J., Evtuguin, D. V. & Neto, C. P. Chemical composition of oleo-gum-resin from Ferula gummosa. Ind. Crops Prod. 33, 549–553 (2011).
Martins, A. et al. Constituents of Carpobrotus edulis inhibit P-glycoprotein of MDR1-transfected mouse lymphoma cells. Anticancer Res. 30, 829–835 (2010).
Uchida, H. et al. Triterpenoid levels are reduced during Euphorbia tirucalli L. callus formation. Plant Biotechnol. 27, 105–109 (2010).
Simo, C. C. F. et al. Benjaminamide: A new ceramide and other compounds from the twigs of Ficus benjamina (Moraceae). Biochem. Syst. Ecol. 36, 238–243 (2009).
Falodun, A., Chaudhry, A. M. A. & Choudhary, I. M. Phytotoxic and chemical investigations of a Nigerian medicinal plant. Res. J. Phytochem. 3, 13–17 (2009).
Wang, X., Li, C., Shi, Y. & Di, D. Two new secoiridoid glycosides from the leaves of Olea europaea L. J. Asian Nat. Prod. Res. 11, 940–944 (2009).
Ragasa, C. Y., Lapina, M. C., Lee, J. J., Mandia, E. H. & Rideout, J. A. Secondary metabolites from Tectona philippinensis. Nat. Prod. Res. 22, 820–824 (2008).
Mbouangouere, R. N. et al. Pipthadenol A-C and α-glucosidase inhibitor from Piptadenia Africana. Res. J. Phytochem. 2, 27–34 (2008).
Dias, M. O., Hamerski, L. & Pinto, A. C. Semi-preparative separation of α- and β-amyrin by high performance liquid chromatography. Quím. Nova 34, 704–709 (2011).
Lin, K.-W. et al. Xanthine oxidase inhibitory triterpenoid and phloroglucinol from guttiferaceous plants inhibit growth and induce apoptosis in human NTB1 cells through a ROS-dependent mechanism. J. Agric. Food Chem. 59, 407–414 (2011).
Sultan, S. M. I. et al. Fungal transformation of cedryl acetate and α-glucosidase inhibition assay, quantum mechanical calculations and molecular docking studies of its metabolites. Eur. J. Med. Chem. 62, 764–770 (2013).
Sua, Y., Hsu, B., Wang, E. & Ho, C. Composition, anticancer, and antimicrobial activities in vitro of the heartwood essential oil of Cunninghamia lanceolata var. konishii from Taiwan. Nat. Prod. Commun. 7, 1245–1247 (2012).
Kallassy, H. et al. Chemical composition, antioxidant, anti-inflammatory, and antiproliferative activities of Crataegus azarolus L. Med. Sci. Monit. Basic Res. 23, 270–284 (2017).
Nisar, S. et al. Natural products as chemo-radiation therapy sensitizers in cancers. Biomed. Pharmacother. 154, 113610. https://doi.org/10.1016/j.biopha.2022.113610 (2022).
Somwong, P. & Theanphong, O. Quantitative analysis of triterpene lupeol and anti-inflammatory potential of the extracts of traditional pain-relieving medicinal plants Derris scandens, Albizia procera, and Diospyros rhodocalyx. J. Adv. Pharm. Technol. Res. 12, 147–151 (2021).
Vaidya, V. V., Pradhan, P. M. & Kondalkar, P. Simultaneous quantification of lupeol, β-sitosterol and oleanolic acid using validated HPTLC method from Nyctanthes arbor-tristis and its marketed formulation. World J. Pharm. Res. 6, 1178–1187 (2017).
Roy, S., Rao, S., Bhuvaneswari, C., Giri, A. & Mangamoori, L. N. Phytochemical analysis of Andrographis paniculata extract and its antimicrobial activity. World J. Microbiol. Biotechnol. 26, 85–91 (2010).
Nopsiri, W. et al. Chemical constituents and antibacterial activity of volatile oils of Combretum latifolium Bl. and C. quadrangulare Kurz leaves. Chiang Mai Univ. J. Nat. Sci. 14, 245–256 (2015).
Tram, U., Ferree, P. M. & Sullivan, W. Identification of Wolbachia–host interacting factors through cytological analysis. Microbes Infect. 5, 999–1011 (2003).
Al-Reza, S. M., Rahman, A., Sattar, M. A., Rahman, M. O. & Fida, H. M. Essential oil composition and antioxidant activities of Curcuma aromatica Salisb. Food Chem. Toxicol. 48, 1757–1760 (2010).
Wei, F. H., Chen, F. L. & Tan, X. M. Gas chromatographic-mass spectrometric analysis of essential oil of Jasminum officinale L. var grandiflorum flower. Trop. J. Pharm. Res. 14, 23–26 (2015).
El-Hawary, S. A., Sokkar, N. M., Ali, Z. Y. & Yehia, M. M. A profile of bioactive compounds of Rumex vesicarius L. J. Food Sci. 76, C1195–C1202 (2011).
Sandeep, A., Kumar, S., Singh, S. & Gupta, R. Ethnopharmacological review of medicinal plants used for wound healing in traditional Indian medicine. J. Appl. Pharm. Sci. 8, 117–121 (2018).
Moy, J., Hu, W. & Hwang, P. Natural product-derived small molecules as modulators of neuroinflammation. Molecules 21, 987 (2016).
Gautam, R., Khatri, M. & Mishra, S. Evaluation of the pharmacological properties of Solanum species: A systematic review. Phytomedicine 24, 36–45 (2017).
Bhuvaneshwaran, J., Prasad, R. & Verma, A. Neuroprotective and anti-inflammatory effects of Solanum species. Eur. J. Pharmacol. 763, 320–327 (2015).
Rani, N., Ramesh, M. & Kumar, S. Toxicological assessment of Solanum villosum: Implications for traditional use. Environ. Toxicol. Pharmacol. 68, 104–110 (2019).
Subramanian, M., Kumar, A. & Thirunavukkarasu, M. Phytochemical analysis and therapeutic properties of Solanum villosum. Phytother. Res. 30, 1101–1107 (2016).
Sharma, R., Kumar, M. & Yadav, A. Pharmacological and medicinal properties of Solanum villosum. Int. J. Mol. Sci. 16, 5761–5773 (2015).
Prasad, B., Shukla, S. & Sharma, S. A review on pharmacological activities of Solanum species. J. Ethnopharmacol. 208, 83–91 (2017).
Reddy, P., Rao, D. & Karthik, R. Toxicological effects of Solanum villosum on liver function. Toxicol. Rep. 5, 1162–1169 (2018).
Das, D., Kumar, P. & Yadav, R. Phytochemical and antioxidant properties of Solanum villosum. J. Agric. Food Chem. 67, 3107–3113 (2019).
Shah, P., Patel, K. & Thakur, S. Anti-cancer potential of Solanum species: A review of pharmacological studies. Asian Pac. J. Cancer Prev. 18, 2489–2493 (2017).
Tripathi, P., Kumar, A. & Meena, R. Pharmacological evaluation of Solanum species in cancer treatment. J. Pharmacol. Exp. Ther. 364, 285–294 (2018).
Borse, S., Singh, R. & Gupta, D. Exploring the therapeutic potential of Solanum species in chronic diseases. Molecules 24, 745 (2019).
Anwar, F., Malik, A. & Zia, M. Medicinal properties of Solanum species in folk medicine. Biol. Pharm. Bull. 40, 469–476 (2017).
Ali, S., Yousuf, M. & Khan, R. Solanum species: A comprehensive review on its pharmacological and toxicological profile. J. Funct. Foods 64, 103684 (2020).
Chavan, R., Jain, A. & Shah, M. Phytochemistry and pharmacological properties of Solanum species. Nat. Prod. Res. 32, 1421–1425 (2018).
Sharma, S., Singh, J. & Verma, A. Bioactive compounds from Solanum species and their role in the treatment of chronic diseases. Phytochem. Lett. 17, 29–35 (2016).
Kumar, R., Sharma, S. & Singh, B. Pharmacokinetics and pharmacodynamics of compounds from Solanum species. J. Med. Chem. 60, 2429–2435 (2017).
Venkatesan, R., Srinivasan, R. & Arumugam, S. Medicinal chemistry of Solanum species: A review. Eur. J. Med. Chem. 163, 77–88 (2019).
Acknowledgements
The authors extend their appreciation to the Researchers Supporting Project number (RSP2025R347), King Saud University, Riyadh, Saudi Arabia, for financial support.
Author information
Authors and Affiliations
Contributions
Y.B., M.A. and M.S.A. designed the study. I.R. performed the experiments. I.R. helped in data curation and analysis of data. M.H.S. and Y.Z collected literature reviews, helped in writing the original draft of the article and helped in funding acquisition. Y.B. and A.Q. provided technical expertise to improve the article. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Riaz, I., Bibi, Y., Arshad, M. et al. Unravelling anti-cancer properties of solanaceous extracts using GC–MS and HPLC. Sci Rep 15, 4192 (2025). https://doi.org/10.1038/s41598-025-87654-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87654-9