Abstract
The objective of this study was to compare the diagnostic efficacy and safety of ultrasound-guided percutaneous transhepatic gallbladder biopsy (PTGB) with endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) in the assessment of gallbladder tumors. We conducted a retrospective, single-center study involving 101 patients diagnosed with gallbladder cancer who underwent either PTGB or EUS-FNA between January 2019 and December 2022. The study cohort was divided into two groups: 52 patients underwent PTGB, and 49 underwent EUS-FNA. Clinical data, diagnostic outcomes, patient demographics, and complications were systematically documented. The sensitivity, accuracy, and incidence of complications were evaluated for both groups. The sensitivity and accuracy rates were 94.23% and 94.23% for PTGB compared to 97.82% and 97.96% for EUS-FNA, with no significant difference between the two techniques. However, EUS-FNA was associated with significantly lower rate of adverse reactions (2.04%) compared to PTGB (15.38%). Both PTGB and EUS-FNA exhibited high diagnostic efficacy for gallbladder tumors. However, EUS-FNA demonstrated a significantly lower incidence of complications, making it a compelling alternative to PTGB, especially when percutaneous biopsy is unsuccessful or not feasible. High-quality prospective, multicenter trials are recommended to further validate these findings and to refine biopsy guidelines for gallbladder tumors.
Similar content being viewed by others
Introduction
Gallbladder cancer is recognized as the most common and aggressive malignancy of the biliary tract. Its incidence has been steadily increasing in China, currently ranks fifth in incidence and fourth in mortality rate1. Due to its insidious onset and rapid progression, gallbladder cancer is often diagnosed at an advanced stage, resulting in a poor prognosis with an overall five-year survival rate of less than 21%2,3,4,5. Biopsy is generally not required when imaging findings strongly suggest a resectable tumor, in order to avoid tumor seeding and delays in surgery.However, for unresectable or borderline resectable cases, histopathological confirmation is necessary before initiating systemic therapy6. Accurate pathological diagnosis helps determine eligibility for systemic therapy, while also enabling molecular testing for personalized treatment approaches. Additionally, it aids in distinguishing gallbladder cancer from other malignancies, ensuring appropriate management, and preventing unnecessary high-risk surgery in unresectable cases7.
Percutaneous transhepatic gallbladder biopsy (PTGB) is the conventionally preferred method for pathological tissue examination in patients with gallbladder cancer before initiating systemic treatments, including chemotherapy or targeted therapy8.
Concurrently, advancements in endoscopic techniques have enhanced the sensitivity and accuracy of endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) for diagnostic purposes9,10. EUS-FNA demonstrates excellent safety and substantial diagnostic value for pancreatic lesions, subepithelial tumors of the gastrointestinal tract, adjacent organs such as the liver11,12,13,14,15,16. Both PTGB and EUS-FNA are capable of obtaining histological specimens, however, comparative studies have been so far confined to pancreatic and hepatic applications17,18. For biliary system lesions that cannot be adequately sampled via conventional percutaneous methods, or when the sampling results are unsatisfactory, EUS-FNA is recommended when the lesion is accessible, particularly for metastatic lesions or enlarged lymph nodes where biopsy can significantly influence the staging of malignant tumors and subsequently alter clinical treatment strategies.
To date, no study has directly compared the diagnostic efficacy of EUS-FNA and PTGB for gallbladder tumors. This study methodically evaluates the sensitivity, accuracy, and safety of EUS-FNA and PTGB in the context of gallbladder tumors. The study was conducted to provide evidence-based recommendations to facilitate the selection of optimal biopsy strategies.
Methods
Ethics declarations
Ethical approval for this study was granted by the Ethics Committee of The First Affiliated Hospital of Xi’an Jiaotong University (Identifier No. XJTU1AF2023LSK-525). Informed consent was obtained from all study participants. The study strictly adhered to the ethical standards outlined in the Declaration of Helsinki. The methods and procedures of this study were meticulously executed in accordance with the NCCN Guidelines® Insights: Biliary Tract Cancers8.
Patients
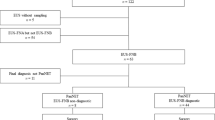
This retrospective, single-center study included 101 patients who underwent gallbladder biopsy at our institution from January 2019 to December 2022 and were clinically diagnosed with gallbladder cancer. Participants were categorized into two groups: those who underwent percutaneous transhepatic gallbladder biopsy (PTGB) and those who underwent endoscopic ultrasound-guided fine-needle aspiration biopsy (EUS-FNA). The selection of either PTGB or EUS-FNA was made by the attending physician, guided by findings from abdominal ultrasound and computed tomography (CT). PTGB was typically employed when lesions were clearly visible on abdominal ultrasound, whereas EUS-FNA was preferred when lesions were less discernible on ultrasound or were located near the intestinal mucosa.
The inclusion criteria were as follows: (1) Age between 18 and 80 years; (2) Patients clinically diagnosed with gallbladder cancer but determined to be unresectable or borderline resectable based on imaging evaluation; (3) Patients are willing to receive systemic therapy or conversion therapy; (4) Availability of complete clinical data and follow-up information.
The exclusion criteria included: (1) Patients who did not undergo puncture procedures or who were punctured using methods other than PTGB or EUS-FNA; (2) Patients with incomplete clinical data or follow-up.
Treatment methods
All biopsies in the EUS group were performed by a single endoscopist, whereas those in the PTGB group were conducted by a single ultrasonologist. Both operators had experience with more than 500 cases. Patients received analgesic premedication; 15 mg of pentazocine was administered for PTGB, while a combination of either 35 mg of piperidine hydrochloride or 7.5–15 mg of pentazocine with 2–5 mg of midazolam was used for EUS-FNA. PTGB was guided by abdominal ultrasound using the Hitachi Preirus system, which involved a transhepatic approach and puncturing the tumor with 17-18G needles. EUS-FNA was performed using a Pentax 3870UTK endoscope and a Hitachi Preirus ultrasound processor. 22G aspiration needles were selected at the operator’s discretion (Fig. 1). Rapid on-site cytological evaluation was performed.
(a) Abdominal contrast-enhanced CT showed solid mass in gallbladder; (b) Abdominal contrast-enhanced CT showed uneven thickening of gallbladder wall; (c) Endoscopic ultrasound showed mass in gallbladder (indicated by left arrow) and gallbladder lumen (indicated by right arrow); (d) Endoscopic ultrasound showed lymph node (indicated by arrow).
Diagnosis and evaluation of complications
In the final pathological diagnosis, the PTGB group was diagnosed using histology, whereas the EUS group was diagnosed using both histology and cytology. All patients underwent a 24-hour clinical observation period following the procedure. The potential for tumor seeding, which might be associated with these procedures, was assessed by evaluating the presence or absence of any apparent tumor involvement in the gastrointestinal wall, along the needle track, and during follow-up via imaging modalities (e.g., CT, MRI). Complications were classified according to the criteria established by the American Society for Gastrointestinal Endoscopy (ASGE)19. Pain was recorded if medication was required within 24 hours, and fever was noted if the patient’s temperature remained above normal for three or more days following the procedure.
Statistical methods
The statistical software SPSS version 26.0 was used for data analysis in this study. Normally distributed measurement data were expressed as mean ± standard deviation. A two-sample t-test was used for comparisons between two groups, whereas one-way analysis of variance (ANOVA) was employed for comparisons among multiple groups. For measurement data with a skewed distribution, the median and range were calculated for descriptive analysis, and non-parametric tests were employed for inter-group comparisons. Categorical data were presented as numbers and percentages. The Chi-square test or Fisher’s exact test was used for group comparisons, as deemed appropriate. A p-value of less than 0.05 was considered indicative of a statistically significant difference.
Results
Patient characteristics
A total of 101 patients were included in the study, with 52 in the PTGB group and 49 in the EUS group (Table 1). The median age of the patients was 62 years (range: 19–80), with 43 males and 58 females. Of the 101 patients, 98 were diagnosed with malignancy, comprising 86 cases of primary gallbladder cancer and 12 cases of metastatic gallbladder cancer. Three patients were ultimately diagnosed with benign tumors: adenoma (1 case), intracystic papillary neoplasm (1 case), and biliary intraepithelial neoplasia (1 case).
Characteristics of the puncture needle and tumor
The puncture needles used in the PTGB group were Tru-Cut type, with 44.23% being 17G and 55.77% being 18G. In the EUS-FNA group, 22G needles were used in all cases. The median lesion diameter was 34 mm in the PTGB group and 29 mm in the EUS-FNA group. In the PTB group, lesions were localized as follows: 67.31% in the fundus, 19.23% in the body, 7.69% in the neck, and 5.77% in the cystic duct of the gallbladder. In the EUS group, lesions were localized as follows: 48.98% in the fundus, 18.37% in the body, 28.57% in the neck, and 4.08% in the cystic duct of the gallbladder. The percutaneous group exhibited a higher prevalence of tumors in the fundus of the gallbladder, whereas the EUS group had a greater proportion of tumors in the neck(Table 2).
Diagnostic ability and complications
Results from PTGB indicated that, out of the 52 cases examined, 49 were diagnosed as malignant, while three were initially classified as benign. Subsequent examinations revealed an increase in size for all three initially benign cases, with adenocarcinoma detected in each, necessitating a reclassification of their diagnoses from benign to malignant. EUS-FNA results identified 45 cases as malignant and four as benign. Of the four cases initially deemed benign, three exhibited no tumor growth following a one-year follow-up, thereby confirming their diagnoses as benign. However, in the remaining case, the tumor exhibited growth, and histopathological examination subsequently detected as a malignant tumor, prompting a reclassification of the diagnosis from benign to malignant.The sensitivity and accuracy of the procedure were 94.23%, 94.23% in the PTGB group.The sensitivity and accuracy 97.82%, 97.96% in the EUS-FNA group, respectively, showing no significant difference between between the two groups.In the PTGB group, complications were reported in 15.38% of patients, with four cases of mild pain, three cases of fever, and one case of abnormal drainage. The causes of wound pain included puncture pain, pain caused by breathing movements tugging on the drainage tube at the puncture site, and individual variations. Analgesic monitoring and treatment proved effective. The causes of fever included infection, cholecystocolonic fistula, and other factors. Anti-infective monitoring and treatment were effective. In the EUS-FNA group, complications were reported in 2.04% of patients, with one case of mild pain. The causes of wound pain were infection-induced fever resulting from biliary obstruction. No instances of needle-tract dissemination were observed (Table 2).
Discussion
The necessity of obtaining a definitive tissue diagnosis in gallbladder cancer is paramount, particularly in unresectable or borderline resectable cases20,21. Although percutaneous methods are traditionally favored our findings indicate that EUS-FNA offers a compelling, patient-centered alternative8.
Endoscopic ultrasound (EUS) is acknowledged for its effectiveness in diagnosing gallbladder lesions and assessing the extent of invasion in gallbladder cancer22. The reported sensitivity and specificity of EUS-FNA for gallbladder tumors are 90.8% and 100%, respectively, with no serious complication reported23. Both percutaneous and EUS-guided biopsies are pivotal in the preoperative diagnosis of gallbladder cancer, providing distinct advantages regarding the amount of tissue sampled and the safety profile. There is a paucity of research comparing EUS-FNA with percutaneous biopsy. Some studies indicate no statistically significant difference in diagnostic efficiency between the two methods24However, EUS-FNA has been reported to exhibit superior diagnostic accuracy for smaller lesions (longest diameter < 3 cm) compared to percutaneous biopsy25. Moreover, the overall complication rate and the incidence of tumor cell seeding along the needle track are reduced with EUS-FNA/B compared to percutaneous biopsy26,27. Our study demonstrated no significant statistical difference in diagnostic efficacy between the two techniques.
Our comparative study has demonstrated that EUS-guided biopsies yield lower complication rates, which can be attributed to the use of finer = needles that cause less tissue trauma28. Complications occur in 0–2% of EUS biopsies, in contrast to 1–5% for the percutaneous approach, which exhibits a higher incidence of bleeding, bile leaks, and post-biopsy pain29. In study cohort, the EUS-FNA group reported less pain compared to the PTGB group, an outcome likely attributable to the minimally invasive nature of EUS-FNA, which involves entry through natural orifices, thereby reducing patient trauma. Conversely, PTGB involves multiple passes through skin and tissue, is associated with an increased risk of bleeding and infection, thereby highlighting the superior safety of EUS-guided biopsy. Compared to the percutaneous approach, which requires penetrating the peritoneum to access the tumor, EUS-FNA avoids traversing the abdominal wall, thereby reducing the risk of tumor seeding along the needle tract to the peritoneal wall or subcutaneous tissue. Additionally, EUS-FNA allows for the assessment of tumor infiltration depth and involvement of adjacent structures, complementing imaging-based staging information.
The minimally invasive nature of EUS-guided biopsy makes it particularly suitable for sampling small, superficial lesions and those located in anatomically challenging sites that are difficult to access percutaneously. In our study, a patient who did not obtain definitive pathological evidence of malignancy via PTGB subsequently presented with mild abdominal pain and distension, along with elevated levels of bilirubin, transaminases, and blood cell counts. After recovery with anti-inflammatory and hepatoprotective treatment, the patient underwent a successful EUS-guided biopsy, which confirmed malignancy without complications. Consequently, EUS-guided biopsy provides advantages when percutaneous biopsy is unsuccessful or impractical, particularly in scenarios where safe access to specific gallbladder segments is unattainable, the lesion is small, there is a risk of damaging critical anatomy, coagulopathy is present, or intolerable pain during percutaneous biopsy occurs30.
For biopsy, the location of the lesion determines the distinct advantages of technique. EUS-FNA is commonly utilized for sampling gallbladder tumors that are deeply located or near the porta hepatis, particularly when the lesion is more accessible via the gastrointestinal tract. When a tumor is situated at the gallbladder neck, the confluence of the bile ducts, or is associated with enlarged lymph nodes in the porta hepatis region, endoscopic ultrasound can closely approach the target, allowing for precise needle aspiration from multiple angles.
The limitations of this study include its single-center, retrospective nature, a relatively small sample size, and a short follow-up period. Prospective, head-to-head comparisons are necessary to develop robust, evidence-based recommendations. Standardization of terminology, procedures, and diagnostic criteria will facilitate meaningful comparisons between institutions. Analyses of cost-effectiveness and their impact on clinical outcomes will further guide the judicious selection of each modality.
In summary, PTGB and EUS-FNA exhibit comparable diagnostic efficacy for gallbladder tumors. Moreover, EUS-FNA showed lower rates of complications compared to PTGB, to some extent. Additionally, PTGB and EUS-FNA might provide distinct advantages for specific patient groups, contingent upon clinical practice. Future high-quality clinical trials investigating PTGB and EUS-FNA are needed.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Roa, J. C. et al. Gallbladder cancer. Nat Rev Dis Primers. 8(1), 69 (2022). https://doi.org/10.1038/s41572-022-00398-y. Erratum in: Nat Rev Dis Primers. 2022, 8(1):75. doi: 10.1038/s41572-022-00408-z.
Siegel, R. L., Miller, K.D., Wagle, N.S. & Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 73 (1), 17–48. https://doi.org/10.3322/caac.21763 (2023).
John, S., Moyana, T., Shabana, W., Walsh, C. & McInnes, M. D. F. Gallbladder cancer: imaging appearance and pitfalls in diagnosis [formula: see text]. Can. Assoc. Radiol. J. 71 (4), 448–458. https://doi.org/10.1177/0846537120923273 (2020).
Hundal, R. & Shaffer, E. A. Gallbladder cancer: epidemiology and outcome. Clin. Epidemiol. 6, 99–109. https://doi.org/10.2147/CLEP.S37357 (2014).
Ganeshan, D. et al. Current update on gallbladder carcinoma. Abdom. Radiol. (NY). 46 (6), 2474–2489. https://doi.org/10.1007/s00261-020-02871-2 (2021).
Zemour, J., Marty, M., Lapuyade, B., Collet, D. & Chiche, L. Gallbladder tumor and pseudotumor: diagnosis and management. J. Visc. Surg. 151 (4), 289–300. https://doi.org/10.1016/j.jviscsurg.2014.05.003 (2014).
Chaudhari, V. A. et al. Outcome of neoadjuvant chemotherapy in locally advanced/borderline resectable gallbladder cancer: the need to define indications. HPB (Oxford) 20 (9), 841–847. https://doi.org/10.1016/j.hpb.2018.03.008 (2018).
Benson, A. B. et al. NCCN Guidelines® insights: biliary tract cancers, version 2.2023. J. Natl. Compr. Canc Netw.21 (7), 694–704. https://doi.org/10.6004/jnccn.2023.0035 (2023).
Faias, S., Cravo, M., Pereira da Silva, J., Chaves, P. & Dias Pereira, A. Endoscopic ultrasound with fine needle aspiration is useful in pancreatic cysts smaller than 3 cm. BMC Gastroenterol. 20 (1), 413. https://doi.org/10.1186/s12876-020-01565-9 (2020).
Chen, Y. Z. et al. Diagnostic performance of serum macrophage inhibitory cytokine-1 in pancreatic cancer: a meta-analysis and meta-regression analysis. DNA Cell Biol. 33(6), 370-7. doi: 10.1089/dna.2013.2237. Epub 2014 Mar 4. Retraction in: DNA Cell Biol. 2015;34(12):742. (2014).
Arruda do Espirito et al. Endoscopic ultrasound-guided versus percutaneous liver biopsy: a systematic review and meta-analysis of randomized controlled trials. Endoscopy. https://doi.org/10.1055/a-2368-4608 (2024).
Kuraishi, Y. et al. Diagnostic performance and safety of endoscopic ultrasound-guided fine-needle aspiration/biopsy for gallbladder lesions. Dig. Endosc. 36 (2), 206–214. https://doi.org/10.1111/den.14576 (2024).
Baran, B. et al. Endoscopic ultrasound-guided parenchymal liver biopsy: a systematic review and meta-analysis. Surg. Endosc. 35 (10), 5546–5557. https://doi.org/10.1007/s00464-020-08053-x (2021).
Mohan, B. P., Shakhatreh, M., Garg, R., Ponnada, S. & Adler, D. G. Efficacy and safety of EUS-guided liver biopsy: a systematic review and meta-analysis. Gastrointest. Endosc. 89 (2), 238–246e3. https://doi.org/10.1016/j.gie.2018.10.018 (2019).
Weilert, F. et al. EUS-FNA is superior to ERCP-based tissue sampling in suspected malignant biliary obstruction: results of a prospective, single-blind, comparative study. Gastrointest. Endosc. 80 (1), 97–104. https://doi.org/10.1016/j.gie.2013.12.031 (2014).
Bossuyt, P. M. et al. STARD 2015: an updated list of essential items for reporting diagnostic accuracy studies. Radiology 277 (3), 826–832. https://doi.org/10.1148/radiol.2015151516 (2015).
Gheorghiu, M. et al. Endoscopic ultrasound-guided fine-needle biopsy versus fine-needle aspiration in the diagnosis of focal liver lesions: prospective head-to-head comparison. Diagnostics (Basel) 12 (9), 2214. https://doi.org/10.3390/diagnostics12092214 (2022).
Maruta, A. et al. Evaluation of preoperative diagnostic methods for resectable pancreatic cancer: a diagnostic capability and impact on the prognosis of endoscopic ultrasound-guided fine needle aspiration. BMC Gastroenterol. 21 (1), 382. https://doi.org/10.1186/s12876-021-01955-7 (2021).
Sheth, S. G. et al. American Society for Gastrointestinal Endoscopy guideline on the role of endoscopy in the management of chronic pancreatitis: methodology and review of evidence. Gastrointest. Endosc. https://doi.org/10.1016/j.gie.2024.05.017 (2024).
Fujiwara, K., Masatsugu, T., Abe, A., Hirano, T. & Sada, M. Preoperative diagnoses and identification rates of unexpected gallbladder cancer. PLoS One 15 (9), e0239178. https://doi.org/10.1371/journal.pone.0239178 (2020).
.Pavlidis, E. T., Galanis, I. N. & Pavlidis, T. E. New trends in diagnosis and management of gallbladder carcinoma. World J. Gastrointest. Oncol. 16 (1), 13–29. https://doi.org/10.4251/wjgo.v16.i1.13 (2024).
Hashimoto, S. et al. The role of endoscopic ultrasound in the diagnosis of Gallbladder lesions. Diagnostics (Basel). 11 (10), 1789. https://doi.org/10.3390/diagnostics11101789 (2021).
Singla, V. et al. Role of EUS-FNA for gallbladder mass lesions with biliary obstruction: a large single-center experience. Endosc Int. Open. 7 (11), E1403–E1409. https://doi.org/10.1055/a-0982-2862 (2019).
Kandel, P. & Wallace, M. B. Recent advancement in EUS-guided fine needle sampling. J. Gastroenterol. 54 (5), 377–387. https://doi.org/10.1007/s00535-019-01552-2 (2019).
Micames, C. et al. Lower frequency of peritoneal carcinomatosis in patients with pancreatic cancer diagnosed by EUS-guided FNA vs. percutaneous FNA. Gastrointest. Endosc. 58 (5), 690–695. https://doi.org/10.1016/s0016-5107(03)02009-1 (2003).
Okasha, H. H. et al. Endoscopic ultrasound-guided fine needle aspiration versus percutaneous ultrasound-guided fine needle aspiration in diagnosis of focal pancreatic masses. Endosc Ultrasound. 2 (4), 190–193. https://doi.org/10.4103/2303-9027.121239 (2013).
Volmar, K. E., Vollmer, R. T., Jowell, P. S., Nelson, R. C. & Xie, H. B. Pancreatic FNA in 1000 cases: a comparison of imaging modalities. Gastrointest. Endosc. 61 (7), 854–861. https://doi.org/10.1016/s0016-5107(05)00364-0 (2005).
Jenssen, C., Alvarez-Sánchez, M. V., Napoléon, B. & Faiss, S. Diagnostic endoscopic ultrasonography: assessment of safety and prevention of complications. World J. Gastroenterol. 18 (34), 4659–4676. https://doi.org/10.3748/wjg.v18.i34.4659 (2012).
DeWitt, J. M. et al. Interventional endoscopic ultrasound: current status and future directions. Clin. Gastroenterol. Hepatol. 19 (1), 24–40. https://doi.org/10.1016/j.cgh.2020.09.029 (2021).
Turan, A. S. et al. Complications of percutaneous transhepatic cholangiography and biliary drainage, a multicenter observational study. Abdom. Radiol. (NY). 47 (9), 3338–3344. https://doi.org/10.1007/s00261-021-03207-4 (2022).
Author information
Authors and Affiliations
Contributions
Fangzhou Wang (FW) conducted the primary analysis and drafted the manuscript. Zheng Wu (ZW) supervised the study and co-authored the manuscript. Jie Hao (JH), Kongyuan Wei (KW), Cancan Zhou (CZ), Zhimin Geng (ZG), and Qingyong Ma (QM) contributed to the manuscript as co-authors. FW, JH, ZW, CZ, ZG, ZD, SH, ZW, QM, and ZW contributed to the design of the study. FW, JH, ZW, and QM calculated the sample size. FW and JH coordinated the study during the inclusion phase.All authors critically assessed the study design and contributed to patient inclusion in the study. They all edited, read, and approved the final manuscript.Fangzhou Wangand Jie Hao contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, F., Hao, J., Wei, K. et al. Comparative diagnostic efficacy and safety of ultrasound-guided percutaneous transhepatic biopsy and endoscopic ultrasound-guided fine-needle aspiration biopsy for gallbladder tumors. Sci Rep 15, 12155 (2025). https://doi.org/10.1038/s41598-025-87847-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87847-2