Abstract
Yutangba, situated in Enshi City, Hubei Province, is globally noted for its high selenium (Se) content. Soil invertebrates are essential to the functionality and services of terrestrial ecosystems, yet their community composition in this region remains under-explored. This study utilized environmental DNA metabarcoding to investigate the interrelations among environmental factors, soil invertebrate diversity, and community characteristics concerning soil Se content, pH, and moisture content in the region. Environmental factors such as Se concentration, water content, and pH were strongly associated with species beta diversity of soil invertebrates in Se-rich areas, reshaping their distribution and abundance, while alpha diversity remained relatively stable. Among these, Se notably emerges as the primary regulatory factor increasing soil invertebrate diversity. The acidic soil pH, along with moisture, plays a fine-tuning role in regulating the species richness and community composition. Unexpectedly, certain species, such as the Formicidae (ants, e.g., Odontomachus troglodytes), the Noctuidae (e.g., Diarsia rosaria), and the annelid Haplotaxida Perionyx excavates, exhibit a strong positive association with Se, indicating a high level of Se tolerance among the native species. This novel perspective reveals the complex role of Se in soil ecosystems, emphasizing the necessity of understanding its ecological functions and potential implications for ecosystem health and stability.
Similar content being viewed by others
Introduction
The diversity of soil invertebrates is crucial for ecosystem health, significantly impacting nutrient cycling, organismal growth, and climate regulation through greenhouse gas emissions1. Soil invertebrates, including a wide range of species such as nematodes, springtails, mites, and earthworms, play a pivotal role in maintaining soil structure and fertility. Beyond their well-known role in the decomposition of organic matter, these invertebrates actively break down complex organic materials through enzymatic processes, thereby influencing both soil and plant dynamics2,3. Research has shown that invertebrates can independently degrade organic materials without microbial assistance, making significant contributions to the global recycling of plant matter2,3. Furthermore, the activities of soil invertebrates are essential for soil formation and development. They modify soil properties, facilitate the translocation of materials, and convert energy, all of which are critical for maintaining the integrity and functionality of soil ecosystems4,5,6.
Studies examining the relationship between soil invertebrates and environmental conditions have identified parent rock type, soil pH, nutrient availability, organic matter content, and climate significantly influence the distribution of soil animal communities7,8,9. These communities are key indicators of forest health and disturbances. Selenium (Se), an essential trace element for numerous organisms10, exhibits beneficial or toxic effects based on its concentration11,12,13. Soil pH significantly affects animal diversity and the composition of functional communities, with distinct decomposer species thriving in either acidic or alkaline environments14,15,16. This indicates that regional abiotic factors, such as soil pH, exert a greater influence on decomposer communities than local biotic factors. Soil pH, determined by parent rock and stand type, is fundamental to the availability and structure of resources within soil food webs17,18,19. Consequently, soil Se content and pH are primary determinants of soil organism distribution.
Se distribution exhibits significant variability across the globe, with concentrations reaching up to 1000 mg/kg in certain Se-rich regions, juxtaposed with areas where it is nearly nonexistent20. Outside these enriched zones, Se is also found in uranium and phosphate ore deposits. Yutangba, recognized worldwide for its high Se content, is notable for containing the only known independent Se deposit, located within black rock series21,22,23. Despite its small area of merely 0.01 km², Yutangba demonstrates considerable heterogeneity in soil Se distribution, predominantly shaped by topographical variations and soil particle size24. Soil organisms significantly influence soil formation, yet the effect of the region’s uneven Se distribution on their distribution is not well understood. While soil invertebrates near ancient Se mines may have evolved adaptive mechanisms due to extended exposure25, the current research lacks evidence regarding the direct or indirect effects of soil Se on soil invertebrate biodiversity. Therefore, the hypothesis that Se-rich environments support greater soil invertebrate diversity cannot be substantiated at this time. Further research is needed to explore the relationships between Se content, soil pH, and soil invertebrate diversity.
Environmental DNA (eDNA) technology, utilizing molecular sequence analysis of genetic material in environmental samples, allows for efficient identification and monitoring of biological species through comparison with reference species databases. This technology marks a substantial advancement in biodiversity science since the early 21st century26,27. This study employed eDNA technology to compile foundational data on the soil animal community in the region. By analyzing the relationships between soil invertebrate diversity indicators and soil properties such as Se content, the research established a framework for understanding the association between soil Se levels and soil fauna and monitoring soil ecological health. The results offer valuable insights for future research into the characteristics and evolutionary dynamics of soil animal diversity in the area.
Materials and methods
Sample collection and the separation of soil organisms
The designated soil sampling route at Yutangba, Enshi City, Hubei Province, as illustrated in Fig. 1, targeted the central zone of a distinct Se reserve, beginning at sites K and A. The route traced two watercourses, merging at site G and then extending downstream. Eleven sampling locations were established, with three subplots at each site arranged in S-shaped or daisy-shaped patterns to ensure randomness and equitable volume, representing a composite of multiple points. Soil sampling was conducted thrice at each of the 11 sites in July, September, and November 2015, yielding 99 soil specimens. Post-sampling, the soil specimens were transported to the laboratory, separated using modified Tullgren funnels, and subjected to a 48-hour drying period. The dried samples were then categorized and preserved in an 85% ethanol solution. For environmental DNA analysis, 33 composite samples were prepared from the corresponding sample points.
Determination of moisture content
Upon returning to the laboratory, labeled soil samples were collected. Each sample was weighed in a clean culture dish, recording the mass as Mdish. After adding the soil to the dish, the combined mass was documented as Mdish+soil sample. Samples were then dried to a constant weight in a temperature-controlled drying oven, and the mass was noted as Mdish+dried. Moisture content of the soil sample was calculated using the formula: Moisture content (%) = [(Mdish + soil sample - Mdish + dried) / (Mdish + dried - Mdish)] × 100%.
Determination of selenium content
Determining soil Se concentration adhered to the Chinese National Standard GB 5009.93—201728. Dried soil samples were ground and sieved through a 200-mesh screen before being stored in sealed plastic bags. A precise 0.05 g portion of each soil sample was weighed and placed in a digestion tube. To enable digestion, 8 mL of concentrated nitric acid, 2 mL of concentrated hydrochloric acid, and 2 mL of hydrogen peroxide were added to each tube, with the samples soaking for 24 h. The digestion process entailed a gradual temperature increase in a digestion furnace until the solution achieved clarity and transparency. The solution volume was then reduced, and hydrochloric acid was added to maintain clarity. The digested solutions containing Se were transferred to volumetric flasks and diluted to a known volume with ultrapure water. Analysis was performed using an AFS-922 dual-channel atomic fluorescence spectrometer, as detailed in Appendix A. The Se content, expressed as micrograms per gram (µg/g), was calculated by comparing the initial weight of the soil samples with the final volume of the digested solutions for subsequent analysis.
Determination of pH
Post moisture content assessment, the remaining soil samples were exposed to natural air drying and subsequently sieved through a 1 mm mesh. A 20-gram portion of the sieved, air-dried soil was weighed and transferred into a 120 ml wide-mouth bottle with a cover. Then, 100 ml of CO2-free distilled water was added, maintaining a soil-to-water ratio of 1:5. The mixture was blended for 2 min and allowed to settle for 30 min. The pH of the resulting solution was measured with a pH meter, and the data were recorded.
DNA extraction and PCR amplification
DNA from soil eukaryotic invertebrates was extracted from 33 samples using the E.Z.N.A.® Soil DNA Kit (Omega Biotek, Norcross, GA, U.S.), adhering to the manufacturer’s protocols. The COI gene underwent PCR amplification under these conditions: initial denaturation at 95°C for 2 minutes, followed by 25 cycles of 95°C for 30 seconds, 55°C for 30 seconds, and 72°C for 30 seconds, with a final extension at 72°C for 5 minutes. Two oligonucleotide primers, mlCOlintF (5’-GGWACWGGWTGAACWGTWTAYCCYCC-3’) and jgHCO2198 (5’-TAIACYTCIGGRTGICCRAARAAYCA-3’), were utilized. Each PCR was performed in triplicate in a 20 µL volume containing 4 µL of 5 × FastPfu Buffer, 2 µL of 2.5 mM dNTP mix, 0.8 µL of each primer at 5 µM, 0.4 µL of FastPfu Polymerase, and 10 ng of DNA template. DNA fragments were isolated from 2% agarose gels and purified using the AxyPrep DNA Gel Extraction Kit (Axygen Biosciences, Union City, CA, U.S.), following the manufacturer’s guidelines precisely.
DNA sequencing and data processing
Concentrations of the PCR products were measured using the Qubit® 3.0 Fluorometer (Life Invitrogen). Equal molar amounts of the twenty-four amplicons, tagged with a specific barcode, were combined to create a composite sample. This composite DNA was utilized to construct a paired-end library suitable for Illumina sequencing platforms, following the provided protocol for genomic DNA library preparation. The library underwent paired-end sequencing (2 × 250 bp) on an Illumina MiSeq system (provided by Shanghai BIOZERON Co., Ltd.), adhering to the standard sequencing protocols.
Raw fastq files were processed with custom Perl scripts to demultiplex data using unique barcode sequences for each sample. The demultiplexing adhered to stringent criteria: (i) Reads of 250 base pairs were truncated if a sliding window of 10 bases had an average quality score below 20, and truncated reads shorter than 50 base pairs were discarded. (ii) Reads with exact barcode matches, more than 2 nucleotide mismatches in primer sequences, or ambiguous nucleotide codes were excluded. (iii) Overlapping sequences were assembled only if the overlap exceeded 10 base pairs, and reads failing to meet this overlap requirement were rejected.
Statistical analysis
Statistical analyses and visualizations were conducted using R (version 4.3.0) and the ggplot2 package29, except where noted. Data from eleven sample site groups were assessed for normality. Normally distributed data were analyzed using ANOVA and the least significant difference (LSD) multiple comparison test. Non-normally distributed data were evaluated using the Kruskal-Wallis (KW) test and the Wilcoxon test. Statistical significance was set at P < 0.05.
Species richness estimation and diversity comparison among samples employed rarefaction and extrapolation methods. Invertebrate alpha diversity and abundance were assessed using the Chao1 estimator, Abundance-based Coverage Estimator (ACE), and observed species index, calculated via R scripts. Beta diversity of invertebrates was analyzed using PCoA based on Bray-Curtis distances and PERMANOVA, utilizing the vegan package in R30. Soil community structure differences were investigated with a nonparametric multivariate analysis of variance (Adonis). Additionally, Mantel correlograms with 9,999 permutations tested the significance of correlation trends in concentration changes. The Mantel test further examined correlations between species and environmental variables, including Se, H2O, and pH, using Spearman’s rank correlation, with significance determined by 9,999 permutations. A Spearman’s rank correlation coefficient (Spearman’s rho) greater than 0.6 and a significance level (P) less than 0.05 were considered significant.
Results
Analysis of environmental factors
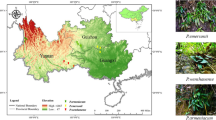
Samples were collected from 11 locations adjacent to the stream, where two streams converge downstream of site G and upstream of site F. Site A was positioned at the selenium cave, while site K was located near selenium mining waste (Fig. 1).
Sampling sites near selenium mines. (a) Locations of 11 sampling sites in Enshi City, Hubei Province. The stream was illustrated based on relative positioning. (b) Selenium cave at site A. (c) Sampling site at site G. Scale bar: 50 m.
To assess environmental factor variations, month and site relationships were analyzed. The results showed no significant differences among the months (Fig. 2a–c, all P > 0.05), but significant site-specific differences were observed (Fig. 2d–f, all P < 0.05, Appendix B). All sites exhibited acidic pH values below 7.0, with the highest at 6.4, suggesting a generally weakly acidic soil environment. Sites with higher Se concentrations had more acidic pH levels (Fig. 2d,e). Soil water content ranged from 27.6 to 48.7%, peaking at site C with 62.56% and the lowest at site A. Despite site A’s low water content, it had a high Se concentration of 254.37 µg/g.
Environmental factor Analysis. Relationships between months (a–c) and sampling points (d–f) with environmental factors, including Se (µg/g dry soil), pH, and H2O (%), were examined. Variations in environmental factors across sites were assessed using the Kruskal-Wallis test with subsequent pairwise comparisons, as detailed in Appendix B.
Taxa composition and abundance analysis
In July, September, and November 2015, 33 samples were collected from 11 sites. COI amplification sequencing produced 1,478,635 circular consensus sequencing (CCS) sequences with an average length of 312.23 bp. Optimized sequences totaling 1,468,280, ranging from 301 to 350 bp, represented 99.30% of the total, ensuring annotation accuracy (Appendix C).
Analysis of taxonomic composition and differential relative abundance of soil invertebrates revealed distinct patterns across various sites (Fig. 3). The phyla Arthropoda and Annelida consistently exhibited high abundances. Within these groups, Diptera (including Culicidae and Tachinidae) in the Insecta class and Haplotaxida (including Henicopidae and Megascolecidae) in the Chilopoda class were particularly prominent. Additionally, sites C, H, and I, which recorded the highest humidity levels, showed a greater abundance of Anopheles mosquitoes, indicating their preference for aquatic environments. Variations in environmental factors, such as Se concentration and water content, are linked to the distribution and abundance patterns of soil invertebrates.
Taxonomic composition and differential relative abundance of soil invertebrates. The top 10 relative abundance groups for phyla (a), classes (b), orders (c), and families (d) were visualized, with each taxonomic group represented by a distinct color.
Alpha diversity signatures
A comparative analysis was conducted to investigate the impact of various environmental factors on alpha diversity. Despite variations in selenium concentrations, pH levels, and water content across different sites, there were no significant differences in alpha diversity among the sites (Fig. 4a-c) even in areas with high selenium concentrations. This suggests that the species present in these areas may have developed tolerance mechanisms or that selenium does not play a primary role in shaping alpha diversity at the studied scales.
The alpha diversity analysis of OTUs across various sample sites. The differences in alpha diversity indices among different sites, including observed species (a), ACE (b), and Chao1, were analyzed using the Kruskal-Wallis test.
Beta diversity
PCoA of OTU data on species diversity indicated that three axes accounted for 21.5% of the variance (Fig. 5). The composition of invertebrates in the Yutangbae area was significantly different among the various sites (PERMANOVA R2 = 0.37, P = 0.001), while no significant differences were observed among different months (PERMANOVA R2 = 0.03, P = 0.23). It suggests that spatial variations have a greater impact on soil invertebrate diversity than temporal variations. These results highlight the essential roles of Se, H2O, and pH in determining species composition. The strong association between environmental factors and community structure highlights the necessity of considering these variables in ecological studies and management.
PCoA and CCA of soil organisms. PCoA based on Bray-Curtis distances and PERMANOVA of the archaeal community were performed. Differences between PCoA1 and PCoA2 across various sample sites were compared.
Analysis of environmental variables pH, H2O, and Se revealed no significant correlation between Se and the other two factors (Fig. 6a). However, a significant positive correlation was identified between H2O and pH (rho = 0.47, P < 0.01) (Fig. 6a). The stability of pH across different sample sites may be due to their proximity to water sources, where pH remains relatively constant. A Mantel test was performed to evaluate the relationship between species beta diversity and specific environmental factors, revealing a significant positive correlation between species beta diversity and sample sites (P < 0.05) (Fig. 6a). Additionally, H2O significantly influenced site A (rho = 0.23, P = 0.046). Sites A, J, and K, characterized by high species diversity and elevated Se concentrations (Figs. 2d and 6a), suggest that Se significantly enhances species diversity at Se-enriched areas.
To analyze the relationship between species diversity and changes in environmental factors such as alpha diversity, Se content, H2O content, or pH, we used Mantel correlograms with 9999 permutations for significance tests (Fig. 6b). Results indicate a significant negative correlation between species diversity and Se content in Se-rich areas with low soil Se (r = − 0.52, P = 0.008), implying a necessary Se threshold for soil invertebrate survival. Unexpectedly, in regions with high Se concentrations, a significant positive correlation emerges between species diversity and Se concentration (r = 0.68, P = 8E−4), indicating species tolerance to elevated Se levels. Furthermore, significant positive correlations were observed with high alpha diversity (r = 0.39, P = 0.036), high water content (r = 0.78, P = 5E−04), and high pH (r = 0.42, P = 7E−4), suggesting that these environmental factors were also associated with species beta diversity.
The relationships between species diversity and environmental factors based on the Mantel test. (a) The heatmap displays correlations between various environmental factors, with each row and column representing a specific factor. The color intensity indicated the strength and direction of the correlation coefficient, where darker shades signified stronger positive or negative correlations, and lighter shades denoted weaker or nonsignificant correlations. Connecting lines between the sites and environmental factors highlighted significant Mantel test correlations. Asterixis indicate significant differences: **P value < 0.01. (b) The Mantel correlogram for beta diversity and alpha diversity, Se, H2O, and pH. The value of the Mantel correlation coefficient is shown on the y-axis for each condition, while the x-axis displays the different levels of the factors. The red dots indicating statistical significance at P < 0.05.
Relationships between environmental variables and soil invertebrates
Correlation analysis between soil invertebrates and environmental factors (Fig. 7, Appendix D) identified significant associations for 83 distinct species with Se concentration, soil moisture content, and pH. Insects demonstrated notable correlations with environmental factors, with 74% of species belonging to the order Insecta. The majority of soil organisms exhibited a strong and statistically significant relationship with Se concentration, as 67% of the correlation coefficients (rho) exceeded 0.5. The Formicidae family (ants), including species such as Odontomachus troglodytes, Lasius emarginatus, and Lasius niger, along with the Noctuidae family, such as Diarsia rosaria, and Haplotaxida, specifically Perionyx excavates, showed a high correlation with Se. In contrast, the aquatic larvae of Diptera, such as Drosophila littoralis, were strongly associated with moisture content. In addition, Coleoptera (beetles) exhibited a notable correlation with soil pH. The number of species associated with Se significantly exceeded those linked to water and pH, indicating that Se distribution and concentration had a more substantial impact on insect diversity and distribution. Furthermore, the correlation coefficient (rho) between species related to Se and water is typically higher than that between species related to pH. This suggests that Se and water may have a more direct and significant impact on insects, particularly during the larval stage when they are more sensitive to environmental changes. Although five species exhibited significant correlations with both Se concentration and pH, the correlation coefficients (rho) with Se were consistently higher than those with pH. This indicates that the abundance of these soil invertebrates is more significantly influenced by Se concentration.
Heatmap analysis of soil organisms in Se-enriched areas based on environmental factors, using a Mantel test with permutation, shows a Spearman’s correlation coefficient. Asterixis indicate significant differences: *P value < 0.05.
Discussion
Robinson et al.31 investigated the impact of soil temperature on the structure and diversity of invertebrate communities. In our study, despite fluctuations in local moisture content and temperature regimes across different months, species diversity remained unaffected, suggesting that environmental conditions in Se-rich areas are relatively stable over time. Therefore, future research should focus on how spatial variations in these factors influence soil invertebrate diversity, rather than temporal changes.
The soil pH exerts a decisive influence on the selenium speciation: in acidic soils, selenium is predominantly reduced to tetravalent selenite32,33,34, which readily complexes with oxides of iron and manganese to form stable complexes or composite precipitates35,36. As the soil environment shifts towards a more alkaline condition, tetravalent selenite can be easily oxidized to hexavalent selenate, which is more soluble in water and consequently more readily dissolved in the soil for plant uptake and utilization32,33. However, Wan et al.37found that all tested plants are capable of accumulating selenium to concentrations that could be potentially harmful to animals when plant root zones are exposed to selenate, suggesting that selenite has a lower toxicity. In this study, we found that the content of selenium showed no significant correlation with soil pH, which may be due, in part, to the similar pH values near water sources. Furthermore, this result indirectly suggests that the effect of selenium on soil invertebrates may play a predominant role. In the soil pH sampling survey conducted in the Yutangba region by Guo38, it was observed that all soil samples were acidic. Consistent with this, we also found that the pH values in the sampling areas were predominantly acidic, suggesting that the concentration of selenate, which is more readily utilized by plants, may be lower compared to selenite in the soil. Due to the lower absorption rate of selenite compared to selenate during animal metabolic processes39,40, this may indirectly reduce the amount of selenium effectively utilized by insects in the soil. However, pH showed a significant correlation with water content (Fig. 6a), implying that moisture content and pH may act as fine-tuning factors in the dynamics of selenium availability41,42,43.
It is well established that selenium is an essential element for animal survival, and its deficiency can negatively impact species viability44. This study not only confirms the importance of Se but also unexpectedly reveals that Se enhances species diversity at high concentrations (Fig. 6b). This finding challenges the conventional understanding that high selenium concentrations typically inhibit growth and potentially affect survival45,46. The pivotal role of earthworms in soil ecosystems47,48 is further underscored by the significant positive correlation observed between Se concentration and the abundance of earthworm species, such as P. excavates (Fig. 7and Appendix D). Additionally, earthworms can reduce local heavy metal content48, which may indirectly contribute to Se tolerance. Previous research has highlighted the importance of moisture and pH for earthworm distribution49. This study, however, found no significant correlation between the beta diversity of P. excavatus and moisture or pH levels, suggesting that selenium concentration is the predominant factor influencing earthworm distribution in Se-enriched areas.
Besides, insects demonstrate a certain level of tolerance to Se and have evolved strategy to regulate its absorption into their tissues50. In Yutangba’s soil, Se was widely enriched, providing insects with additional nutrients. This nutrient benefits plant absorption and utilization38,51,52, as evidenced in Cardamine hupingshanesis(Brassicaceae), a plant highly tolerant to Se53. It also influences the distribution of soil microbial populations54,55,56,57. Consequently, these soil nutrients may directly and indirectly impact soil invertebrate biodiversity58. Additionally, parasitic insects expose larvae directly to organic Se rather than selenate, thereby circumventing selenium toxicity40. This mechanism may contribute to the higher Se tolerance observed in native species25. Due to the relatively lower concentration of water-soluble Se in Enshi’s aquatic environments59, a Se gradient forms in small streams, enabling species to select optimal Se levels for growth and reproduction within the high Se soil conditions. Furthermore, certain native species have developed unique adaptabilities to selenium. For example, the abundance of ants like O. troglodytes, L. emarginatus, and L. nigeris positively correlated with Se concentrations in this study, which is consistent with research indicating high Se tolerance in ants relative to other native species25. This correlation implies that extended exposure to Se-rich environments, such as mine caves, may have led to greater Se tolerance in these invertebrates compared to those in low Se areas. Long-term exposure to Se appears to enhance Se tolerance in soil invertebrates in such areas, supporting our hypothesis.
In summary, environmental factors intricately impact soil health and biodiversity. In Se-enriched habitats, Se significantly shapes species diversity, with its bioavailability being further modulated by moisture and pH levels, thus finely tuning the ecological dynamics in these regions. Notably, the sampled site, a natural Se mine, indicates that certain native species have likely adapted to high-Se environments. Balanced Se concentrations in soil enhance species diversity. This study elucidates the extensive implications of Se on invertebrate biodiversity and provides valuable insights into the intricate interplay between Se and soil ecosystem health.
Conclusions
This study conducted at Yutangba, within the Se-rich Enshi region, examined the correlation between soil invertebrate diversity and environmental variables, with a specific emphasis on Se, moisture, and pH. The findings revealed a substantial impact of these factors on the alpha diversity of soil invertebrates. High alpha diversity areas demonstrated a close association between beta diversity and soil moisture as well as Se content. In contrast, regions with fluctuating pH and moisture levels exhibited beta diversity that was independent of Se concentrations. These data suggest that in Se-rich areas, Se primarily influences beta diversity, whereas in low-Se conditions, moisture content also significantly regulates beta diversity.
Data availability
The data obtained in this study are available from the NCBI under the GenBank number PRJNA1055709. All other data are available in this text and in the Appendixes.
References
Griffiths, H. M., Ashton, L. A., Parr, C. L. & Eggleton, P. The impact of invertebrate decomposers on plants and soil. New Phytol. 231 (6), 2142–2149 (2021).
Joly, F. X. et al. Detritivore conversion of litter into faeces accelerates organic matter turnover. Commun. Biology. 3 (1), 660 (2020).
Kane, J. L., Kotcon, J. B., Freedman, Z. B. & Morrissey, E. M. Fungivorous nematodes drive microbial diversity and carbon cycling in soil. Ecology 104 (1), e3844 (2023).
Wall, D. et al. Global decomposition experiment shows soil animal impacts on decomposition are climate. Glob. Change Biol. 14 (11), 2661–2677 (2008).
García-Palacios, P., Maestre, F. T., Kattge, J. & Wall, D. H. Climate and litter quality differently modulate the effects of soil fauna on litter decomposition across biomes. Ecol. Lett. 16 (8), 1045–1053 (2013).
Briones, M. J. I. The Serendipitous Value of Soil Fauna in Ecosystem Functioning: the unexplained explained. Front. Environ. Sci., 6. (2018).
Bird, S., Coulson, R. N. & Crossley, D. A. Impacts of silvicultural practices on soil and litter arthropod diversity in a Texas pine plantation. For. Ecol. Manag. 131, 65–80 (2000).
Ponge, J. F. et al. Collembolan communities as bioindicators of land use intensification. Soil Biol. Biochem. 35 (6), 813–826 (2003).
Cassagne, N., Gauquelin, T., Bal-Serin, M. C. & Gers, C. Endemic Collembola, privileged bioindicators of forest management. Pedobiologia 50, 127–134 (2006).
National Research Council (US) Subcommittee on Selenium. Selenium in Nutrition: Revised Edition (National Academies Press (US), 1983).
See, K. A., Lavercombe, P. S., Dillon, J. & Ginsberg, R. Accidental death from acute selenium poisoning. Med. J. Aust. 185 (7), 388–389 (2006).
MacFarquhar, J. K. et al. Acute selenium toxicity associated with a dietary supplement. Arch. Intern. Med. 170 (3), 256–261 (2010).
Zhang, H. Y. et al. Association between fatality rate of COVID-19 and selenium deficiency in China. BMC Infect. Dis. 21 (1), 452 (2021).
Schaefer, M. The soil fauna of a beech forest on limestone: trophic structure and energy budget. Oecologia 82 (1), 128–136 (1990).
Salmon, S., Mantel, J., Frizzera, L. & Zanella, A. Changes in humus forms and soil animal communities in two developmental phases of Norway spruce on an acidic substrate. For. Ecol. Manag. 237 (1–3), 47–56 (2006).
Pollierer, M. M. et al. Diversity and functional structure of soil animal communities suggest soil animal food webs to be buffered against changes in forest land use. Oecologia 196 (1), 195–209 (2021).
Ruess, L., Sandbach, P., Cudlin, P., Dighton, J. & Crossley, A. Acid deposition in a spruce forest soil: effects on nematodes, mycorrhizas and fungal biomass. Pedobiologia 40 (1), 51–66 (1996).
Lauber, C. L., Strickland, M. S., Bradford, M. A. & Fierer, N. The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biol. Biochem. 40 (9), 2407–2415 (2008).
Rousk, J. et al. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 4 (10), 1340–1351 (2010).
Hartikainen, H. Biogeochemistry of selenium and its impact on food chain quality and human health. J. Trace Elem. Med Biol. 18 (4), 309–318 (2005).
Yao, L., Gao, Z., Yang, Z. & Long, H. The formation of selenium-rich siliceous rocks in the Yutangba selenium deposit. Sci. China Ser. D-Earth Sci. (in Chinese). 32 (1), 54–63 (2002).
Zhu, J., Zheng, B. & Liu, S. R. Some new forms of native selenium and their genetic investigation. Acta Mineral. Sinica. 20 (4), 337–341 (2000).
Wen, H. et al. Discovery and indicative significance of the largest selenium isotopic fractionation in Yutangba selenium deposit in Hubei Province. Chin. Sci. Bulletin(in Chinese). 52 (17), 1845–1848 (2007).
Zhu, J. M. et al. Distribution pattern of soil selenium and its influencing factors in the micro-local high-selenium environment of Yutangba. Environ. Sci. (in Chinese). 19 (6), 33–36 (1998).
De La Riva, D. G. & Trumble, J. T. Selenium exposure results in reduced reproduction in an invasive ant species and altered competitive behavior for a native ant species. Environ. Pollut. 213, 888–894 (2016).
Wang, P. et al. Environmental DNA: an Emerging Tool in Ecological Assessment. Bull. Environ Contam. Toxicol. 103 (5), 651–656 (2019).
Zhang, X. W. Environmental DNA shaping a new era of ecotoxicological research. Environ. Sci. Technol. 53 (10), 5605–5612 (2019).
The Ministry of Health of the People’s Republic of China. Determination of selenium in foods (GB 5009.93—2017). (in Chinese). (2017).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016).
Oksanen, J. et al. vegan: Community Ecology Package: R package version 2.5–3. 2018. Available online at (2018). https://CRAN.R-project.org/package=vegan.
Robinson, S. I., McLaughlin Ó, B., Marteinsdóttir, B. & O’Gorman, E. J. Soil temperature effects on the structure and diversity of plant and invertebrate communities in a natural warming experiment. J. Anim. Ecol. 87 (3), 634–646 (2018).
Zhang, P., Ganje, T. J., Page, A. L. & Chang, A. C. Growth and uptake of Selenium by Swiss Chard in Acid and Neutral soils. J. Environ. Qual. 17, 314–316 (1988).
Elrashidi, M. A., Adriano, D. C. & Lindsay, W. L. Solubility, Speciation, and Transformations of Selenium in Soils. In Selenium in Agriculture and the Environment, pp. 51–63. (1989).
Qin, H. et al. Selenium speciation in seleniferous agricultural soils under different cropping systems using sequential extraction and X-ray absorption spectroscopy. Environ. Pollut. 225, 361–369 (2017).
Balistrieri, L. S. & Chao, T. T. Adsorption of selenium by amorphous iron oxyhydroxide and manganese dioxide. Geochim. Cosmochim. Acta. 54, 739–751 (1990).
Balboni, E., Han, S. C. & Zavarin, M. Selenium interaction with iron minerals: quantitative comparison of sorption and coprecipitation impacts on mobility. Appl. Geochem. 175, 106194 (2024).
Wan, H. F., Mikkelsen, R. L. & Page, A. L. Selenium Uptake by some agricultural crops from Central California Soils. J. Environ. Qual. 17, 269–272 (1988).
Guo, Y. Geochemistry of selenium in Enshi area and experimental study of selenium-enriched crop cultivation. Dissertation for the Doctoral Degree. Wuhan: China University of Geosciences. (in Chinese). (2012).
Burk, R. F. & Hill, K. E. Regulation of Selenium Metabolism and Transport. Annu. Rev. Nutr. 35, 109–134 (2015).
Vickerman, D. B., Trumble, J. T., George, G. N., Pickering, I. I. & Nichol, H. Selenium biotransformations in an insect ecosystem: effects of insects on phytoremediation. Environ. Sci. Technol. 38 (13), 3581–3586 (2004).
Sylvain, Z. A. et al. Soil animal responses to moisture availability are largely scale, not ecosystem dependent: insight from a cross-site study. Glob. Change Biol. 20 (8), 2631–2643 (2014).
Kökdener, M. & Şahin Yurtgan, M. The Effect of Soil Type and Moisture Level on the development of Lucilia Sericata (Diptera: Calliphoridae). J. Med. Entomol. 59 (2), 508–513 (2022).
Wu, B. et al. Precipitation seasonality and soil pH drive the large-scale distribution of soil invertebrate communities in agricultural ecosystems. FEMS Microbiol. Ecol. 99 (11), fiad131 (2023).
So, J., Choe, D. H., Rust, M. K., Trumble, J. T. & Lee, C. Y. The impact of selenium on insects. J. Econ. Entomol. 116 (4), 1041–1062 (2023).
Song, K., Liu, M., Chen, J., Li, H., Wang, K. & D, and Differences in the bioaccumulation of selenium by two earthworm species (Pheretima Guillemi and Eisenia fetida). Chemosphere 202, 560–566 (2018).
Yue, S., Huang, C., Wang, R. & Qiao, Y. Selenium toxicity, bioaccumulation, and distribution in earthworms (Eisenia fetida) exposed to different substrates. Ecotoxicol. Environ. Saf. 217, 112250 (2021).
Butenschoen, O., Marhan, S., Langel, R. & Scheu, S. Carbon and nitrogen mobilisation by earthworms of different functional groups as affected by soil sand content. Pedobiologia 52, 263–272 (2009).
Singh, S., Singh, J. & Vig, A. P. Earthworm as ecological engineers to change the physico-chemical properties of soil: soil vs vermicast. Ecol. Eng. 90, 1–5 (2016).
Singh, S., Sharma, A., Khajuria, K., Singh, J. & Vig, A. P. Soil properties changes earthworm diversity indices in different agro-ecosystem. BMC Ecol. 20 (1), 27 (2020).
Lalitha, K., Rani, P. & Narayanaswami, V. Metabolic relevance of selenium in the insect Corcyra Cephalonica. Uptake of 75 Se and subcellular distribution. Biol. Trace Elem. Res. 41 (3), 217–233 (1994).
Wang, Y. et al. The combined impacts of selenium and phosphorus on the fate of arsenic in rice seedlings (Oryza sativa L). Chemosphere 08 (Pt 3), 136590 (2022).
Chen, Y. et al. Role of soil nutrient elements transport on Camellia oleifera yield under different soil types. BMC Plant Biol. 23 (1), 378 (2023).
Yuan, L. et al. A novel selenocystine-accumulating plant in selenium-mine drainage area in Enshi, China. PLoS One. 8 (6), e65615 (2013).
Wang, Z., Huang, W. & Pang, F. Selenium in Soil-Plant-Microbe: a review. Bull. Environ Contam. Toxicol. 108 (2), 167–181 (2022).
Wang, M. et al. Seasonal changes of soil microbiota and its association with environmental factors in coal mining subsidence area. AMB Express. 13 (1), 147 (2023).
Yuan, L., Xia, Z. & He, C. A novel selenite-tolerant rhizosphere bacterium Wautersiella enshiensis sp. nov., isolated from Chinese selenium hyperaccumulator, Cardamine Hupingshanensis. J. Basic Microbiol. 63 (11), 1305–1315 (2023).
Zang, H. et al. Life-cycle selenium accumulation and its correlations with the rhizobacteria and endophytes in the hyperaccumulating plant Cardamine hupingshanensis. 264:115450. (2023).
Yang, H. et al. The beneficial and hazardous effects of selenium on the health of the soil-plant-human system: an overview. J. Hazard. Mater. 422, 126876 (2022).
Shao, P. W. Methods for Analyzing Selenium Speciation and Their Application in the Enshi Selenium-Rich Area. Dissertation for the Master’s Degree. Shandong: Qingdao University. (in Chinese). (2020).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 32070483, 31460572), Natural Science Foundation of Hubei Province (Grant No. 2020CFB757), Scientific Research Starting Foundation for Ph.D. of Huanggang Normal University (Grant No. 2020010, 2042024378), Scientific Research Project of Education Department of Hubei Province (Grant No. Q20232901), Huanggang Normal University Major Project Incubation Plan-Funded Program (Grant No. 204202315204).
Author information
Authors and Affiliations
Contributions
Conceptualization, Y.F., B.M; methodology, B.M., X.-L.F.; software, B.M., X.-L.F.; formal analysis, B.M., X.-L.F.; investigation, Y.F.; resources, Y.F.; writing—original draft preparation, Y.F., B.M., H.-L.L.; writing—review and editing, Y.F., Y.-L.X., B.M.; visualization, B.M.; supervision, Y.F.; project administration, Y.F.; funding acquisition, Y.F., B.M.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, B., Fang, X., Lei, H. et al. Unexpected species diversity in the understanding of selenium-containing soil invertebrates. Sci Rep 15, 3647 (2025). https://doi.org/10.1038/s41598-025-87917-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87917-5