Abstract
Aiming at the problem that it is difficult to realize low-cost, high-performance and large-scale utilization of cementitious materials prepared from bulk solid wastes, this paper constructs a set of composite cementitious system based on alkaline activation of slag and fly ash (FA) by calcium carbide slag (CCS) and synergistic activation of sodium sulfate (Na2SO4) as a chemical dopant. The influence of factors such as solid waste type, mixing ratio, and Na2SO4 content on the mechanical properties of composite cementitious systems was investigated by assessing compressive strength and analyzing microstructure using XRD, SEM-EDS, and FTIR. The test results indicate that CCS and Na2SO4 exert significant influences on the strength of the composite cementitious system. CCS acts as an alkali activator, enhancing the system’s hydration with an optimal dosage of 25%. Low Na2SO4 content also promotes hydration, but higher concentrations disrupt the internal structure of the cementitious system post-coagulation, with an optimal content of 6%. The Projection Pursuit Regression (PPR) strength prediction model can fit the actual experimental data very well, which provides a feasible method for the proportion design and mechanical strength prediction of all-solid-waste cementitious systems.
Similar content being viewed by others
Introduction
Traditional Portland cement has been widely used in the field of building materials, but its long-term use inevitably leads to a large amount of carbon dioxide emissions and energy consumption, thus exacerbating the greenhouse effect1. In recent years, with the introduction of the concepts of “Carbon Peaking and Carbon Neutrality2” and “Green Building Materials3”, the use of solid waste has attracted the attention of researchers. Alkali-activated materials are a new type of cementitious system with pozzolanic activity that is based on silicon and aluminum as the main elements. These methods rely mainly on the introduction of alkalis (such as NaOH and sodium silicate) to cause the silicon and aluminum in the raw materials to form a polyaluminate material with a three-dimensional network structure. Because the good mechanical properties of alkali-activated materials are primarily due to silicon and aluminum rather than calcium, these materials are also referred to as geopolymers4. According to their chemical composition, precursors can be divided into high-calcium materials (slag) and low-calcium or calcium-free materials (fly ash, silica fume, etc.). Geopolymers have attracted the attention of many researchers because of their unique microstructural properties and excellent mechanical properties. FA has played a role in the manufacturing of geopolymers and promoted the development of environmentally friendly building materials5. Slag contains chemical components such as CaO and SiO2. The silicon-rich phase C-S-H formed under the action of an activator has stronger atomic bonds and greater strength than the calcium-rich phase cementitious products formed from traditional Portland cement6.
Strongly corrosive saline soils are widely distributed in Northwest China, and severe erosion from sulfate and other types of salt crystallization has occurred. In particular, salt lakes are widely distributed in Northwest China, and underground engineering structures have been in erosive solutions for a long time or in dry and wet cycle areas, which significantly damages and deteriorates the structure7. Could the idea of “turning danger into safety” have been used to convert the hazard of sulfate erosion into a way to improve the strength of geopolymers? Sulfate is a basic metal sulfate. As an activator, sodium sulfate is more economical and environmentally friendly than sodium hydroxide and sodium silicate are8. Adding sodium sulfate to the gelation system could promote the hydration reaction9. Some scholars have reported that the addition of sodium sulfate to the gelation material significantly affects the hydration reaction rate in the gelation system and plays a positive role10,11,12,13,14. Sodium sulfate participates in the hydration reaction, promotes the formation of the AFt phase, increases the alkalinity of the sample, and stimulates gel formation, and the hydration products fill the pores of the material, thereby optimizing the microstructure of the gelation material15. However, a high sodium sulfate content accelerates the production of more expansive products, destroys the microstructure of the cementitious material, and leads to a sharp decrease in strength16,17. Since the pH of a sulfate solution is generally between 9 and 10, the hydration of cementitious material is promoted more slowly than that of other strongly alkaline materials, and early strength often does not meet the requirements. If an alkaline material was added to a sulfate-activated cementitious material system, the early strength of the paste could be significantly improved, but the rate of increase in the late strength would be reduced18.
As a strongly alkaline waste residue product generated in industrial production, CCS can cause serious environmental damage if it is landfilled or dumped. How to achieve the reuse of CCS has become an important issue that needs to be addressed today19. The comprehensive utilization and environmental management of CCS face many challenges, and further research and technological improvements are needed to increase the efficiency of its application and reduce its environmental impact.
CCS is strongly alkaline and can be used as an alkali activator to establish an alkali-activated solid waste gel system. CCS had a good activating effect on cementitious materials such as slag. The alkaline material CCS can form a stable alkaline environment, promote the initial hydration reaction20, and provide many Ca2+ ions, which provide raw materials for the hydration products21,22,23. An analysis of the evolution of the mechanical strength and the development of the microstructure of the CCS alkali-activated gelling material revealed that the NaCl, CaCl2 and Ca(OH)2 components in the alkali residue participated in the hydration process, resulting in a denser microstructure and lower porosity, which increased the compressive strength and density24,25.
To improve the early strength of sodium sulfate-activated geopolymer materials and ensure the late strength of the gelation system, CCS was added to the geopolymer material. Many previous studies have focused on enhancing the properties of FA/Slag-based alkali-activated materials via sulfate or CCS26,27,28. However, there has been very limited research on the combined effects of these two factors on the mechanical properties and microstructures of hydrated products in multifunctional solid waste gelation systems.
In this study, composite cementitious systems were prepared using common industrial byproducts, including CCS, FA and slag, as the primary materials. Additionally, Na2SO4 was added as an activator to further stimulate the gelling material. The impacts of the solid waste type, mixing ratio and Na2SO4 content on the mechanical properties of composite cementitious systems were examined by determining the compressive strength of each system. The microstructure was analyzed via XRD, SEM‒EDS and FTIR to elucidate the mechanism of action of each component. A compressive strength prediction model of the composite cementitious system was established via PPR, thereby providing a theoretical basis for the safe and efficient utilization of industrial waste materials such as CCS, FA and slag.
Materials and methods
Raw materials
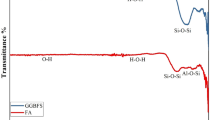
The raw materials for this study include Class I C FA from thermal power plants in Chang ji City, CCS from acetylene production at Xinjiang Hua tai Heavy Chemical Co., and S75 Slag from Xinjiang Bao Xin Sheng yuan Co. Anhydrous Na2SO4 (99% analytically pure) was obtained from Tianjin Zhiyuan Chemical Reagent Co., and tap water was used for testing. Table 1 presents the main chemical compositions and apparent densities of the materials used in the cementitious system. The raw material XRD spectrum was shown in Fig. 1.
XRD patterns of FA, Slag and CCS.
Mix proportions and preparation of sample
Mix proportions
Three cementitious systems were prepared, comprising CCS-Slag (C-S), CCS-FA (C-F), and CCS-Slag-FA (C-S + F). The proportion of mixture is shown in Table 2.
Preparation of sample specimen preparation
The sample preparation process was conducted `in accordance with the JGJ/T 70-200929 standard, with material proportions determined based on the experimental program table. Initially, the ingredients should be weighed and subsequently blended for a duration of 30 s. Subsequently, a precise quantity of Na2SO4 is weighed and dissolved in distilled water to produce a salt solution. Subsequently, the prepared salt solution is introduced to the mixed ingredients, and the mixture is subjected to a 60-second period of agitation. Once mixing was complete, the slurry was poured into molds with dimensions of 40 × 40 × 160 mm. The molded specimens were subsequently placed in a standard curing box under controlled conditions (temperature of 20 ± 3 °C and humidity of 90%) for curing. After 24 h, the specimens were demolded and returned to the curing box to continue the curing process. After 28 days of curing, the specimens underwent unconfined compressive strength testing and microstructure analysis. The detailed procedure is illustrated in Fig. 2.
Production of specimens and testing procedures flow chart.
Test methods
Compressive strength test
Compressive strength is tested using TYA − 300B automatic pressure tester. The compressive strength tests were carried out on samples with curing time of 28 days. The loading rate of the apparatus was fixed at 2400N/s ± 200N/s during the tests and each set of tests was repeated three times and the strength values used in the study were the average values calculated from the measured data.
XRD test
XRD is a technique used to identify the mineralogical composition of materials and is widely used in many studies. First, the crushed specimens after compressive strength testing were immersed in an anhydrous ethanol solution to stop their hydration reaction, and then dried in a vacuum oven at 45 °C until constant weight. Subsequently, the dried specimens were ground to a powder, sieved through a 75 µm sieve, and then the powder was filled and pressed into the grooves of the test slides for XRD testing. The XRD tests were performed using a Bruker D8 Advance X-ray Diffractometer, with the scanning range of the tests set to 10–80° 2θ and the scanning speed of the instrument set to 10°/min.
FTIR test
The test was carried out with an infrared spectrometer produced by Bruker, model Vertex 70, with a test range of 400 cm− 1~4000 cm− 1, and the samples were prepared by the KBr pressing method.
The preparation method of the samples to be tested for FTIR test was the same as that of the XRD test specimens. First, weigh 1.5 mg of a dried solid sample and mix it with 150 mg of dried KBr powder (sample mass: KBr mass = 1:100), pour it into an agate mortar, and grind it thoroughly. After grinding, press the tablets. Test a pure KBr sample as a calibration before starting the formal test.
SEM-EDS test
SEM was utilized to examine and analyze the differences in micro-morphology of the specimens at different ratios. For this test, a Thermo Fisher (formerly FEI) Quattro s field emission ambient SEM was used to observe the micromorphology of the test specimens and the hydration products were analyzed by EDS energy spectroscopy.
The preparation method of the samples to be tested for SEM and EDS test was the same as that of the XRD test specimens.
Results
The primary factors influencing the mechanical properties of various solid waste gelling systems included the amount of CCS mixed, the Na2SO4 content, and the type of gelling system. The experimental program employed a fixed-variable method to systematically investigate the impact of these different factors on the mechanical properties of the gelling systems.
Compressive strength
Effect of CCS on compressive strength
CCS acted as an alkali activator in the gelling system, exerting a considerable influence on its mechanical properties. Figure 3 illustrates the compressive strength test results for specimens with varying CCS dosages and Na₂SO₄ contents, evaluated after 28 days of curing.
As illustrated in Fig. 3, the compressive strength of the three gelling systems demonstrates a pattern of initial increase followed by a decline with the elevation of the CCS dosage. The majority of the samples reached their maximum strength when the CCS dosage was 25%. The C-S and C-F exhibited a pronounced influence from calcium carbide slag, with a discernible trajectory of enhanced strength. The compressive strength of the C-S increased by approximately 45%. In the C-F, when the Na₂SO₄ content is 6%, the compressive strength can be increased from 4.5 MPa to nearly 24.2 MPa with an increase in the CCS dosage, representing a 447.78% increase. The compressive strength of the C-S + F changes more slowly, with an increase of 15–20%.
The highest compressive strength was exhibited by C-S, reaching a peak of 49.2 MPa, followed by C-S + F at 35.8 MPa. The compressive strength of C-F was found to be the lowest, reaching a peak of only 24.2 MPa.
The strength of the three cementitious systems is observed to increase with the addition of CCS, reaching a maximum at 25% CCS, when the Na2SO4 content is held constant. For the C-S, when the CCS dosage was low (CCS dosage % < 25%), the strength of the system increased with the Na2SO4 content, reaching a maximum at 8% Na2SO4 content. When the CCS dosage was ≥ 25%, the optimal Na2SO4 content was 6%.The law of the influence of Na2SO4 and CCS on C-S + F was the same as that of C-S, but as the content of Na2SO4 increased, the strength of the system increased more, indicating that the influence of Na2SO4 on C-S + F was more significant than that of C-S. For the C-F, when the CCS content was low (CCS dosage % < 10%), the strength of the system increased with increasing Na2SO4 content, and at that time, the Na2SO4 excitation effect dominated. As the CCS content increased, the two influencing factors jointly affected the cementitious system.
Effect of CCS dosage on the mechanical strength of multi-component solid waste cementitious system. (CCS dosage of 5%, 10%, 15%, 20%, 25%, 30%). (a) CCS-Slag system. (b) CCS-Slag + FA system. (c) CCS-FA system.
The low early strength observed in the C-F could be attributed to the poor early hydration activity of FA, which resulted in a slow hydration reaction30,31. However, as the Ca2+ and OH− generated from CCS hydration increase, FA begin to hydrate and form cementitious compounds. The incorporation of CCS provides both Ca2+ and an alkaline environment, enhancing the early hydration of FA.
Slag can be rapidly decomposed in an alkaline environment, and Ca2+ ions released from dissolved CCS particles also promote the formation of C-(A)-S-H type gels. Therefore, the compressive strength of the C-S and C-S + F are higher than that of the C-F32.As the amount of CCS increases, the strength of the cementitious system improves. This suggests that using CCS as an alkali activator positively impacts the strength of the cementitious system. Based on the compressive strength data for each system, a 25% CCS dosage is identified as optimal for achieving higher strength in the cementitious systems.
Effect of Na2SO4 on compressive strength
The test results indicated that the cementitious system achieved its optimal mechanical properties with a CCS dosage of 25%. In order to further investigate the effect of Na2SO4 content on these properties, experiments were carried out to vary the Na2SO4 content with the aim of exploring how different Na2SO4 contents affect the macroscopic mechanical properties of the cementitious system. Experimental results illustrating these effects are shown in Fig. 4.
Effect of Na2SO4 dosage on the mechanical strength of multi-component solid waste cementitious system. (Na2SO4 dosage of 2%, 4%, 6%, 8%, 10%). (a) CCS-Slag system. (b) CCS-Slag + FA system. (c) CCS-FA system.
In the C-S, the optimal Na2SO4 content decreases from 8 to 4% as the dosage of CCS increases. For the C-F, the optimal Na2SO4 content decreases from 10 to 6% as the dosage of CCS increases. In the C-S + F, the optimal Na2SO4 content decreases from 8 to 6% with an increase in CCS dosage. The CaO content of the Slag is 39.0%, which allows for the production of Ca(OH)2 during hydration. FA contains 12.7% CaO, while the Slag-FA mixture has a CaO content of approximately 25.9%. At lower CCS dosages (≤ 15%), a higher Na2SO4 content is necessary to achieve effective activation. Conversely, at higher CCS dosages (> 15%), effective activation can be achieved with a lower Na2SO4 content. The optimal Na2SO4 content also depends on the CaO content in the geopolymer. Sulfate is most doped at 6% when the CCS dosage is 25%.
When the CCS content is 25%, with an increase in the Na2SO4 content, the compressive strength of C-S increases from 42.9 MPa to 49.2 MPa, an increase of 15.3%; the compressive strength of C-S + F increased from 24.3 MPa to 35.4 MPa, an increase of 45.7%; the compressive strength of C-F increased from 12.2 MPa to 24.2 MPa, an increase of 98.4%. The effect of sodium sulfate on the compressive strength of C-F is the most significant, followed by C-S + F, and the effect on C-S is the least. This indicated that sodium sulfate could effectively activate volcanic ash materials. The effect of activation was more pronounced for low-activity volcanic ash materials, which could significantly increase the volcanic ash activity of the material and improve utilization.
SO₄²⁻ ions react with the hydration products of the gelling system to form the AFt. Simultaneously, SO42− ions can react with Ca²⁺ to form CaSO₄33,34. CaSO4 and AFt crystals can fill the pores in the network structure of the hydration products, and this filling increases the overall compactness of the hydrated product, leading to an enhancement in its compressive strength. However, as the sulfate concentration continued to increase, the compressive strength of the material started to decrease, especially during the later stages of hydration. This decline can be attributed to two factors: first, as the reaction progresses, some Na2SO4 crystals are consumed, which creates pores and reduces compressive strength; second, excessive SO42− ions can cause sulfate attack on the hydration products within the gel system.
Microscopic analysis
XRD
Figure 5 displays XRD patterns of three gelling systems with CCS dosages of 5%, 25%, and 30%, and 6% Na2SO4 content after 28 days of curing. With increasing CCS dosage, the diffraction peaks of hydration products became notably more pronounced. This suggested that incorporating CCS creates an alkaline environment favorable for cementitious system hydration, thus accelerating the hydration reaction. Significant hydration products such as AFt, C-(A)-S-H gel, and mullite are observed in increased amounts.
XRD comparison of different cementitious systems with 5%, 25%, and 30% CCS dosage. (a) CCS-Slag system.
(b) CCS-Slag + FA system. (c) CCS-FA system.
As can be seen in Fig. 5, as the CCS content increased, the hydration products continued to increase, and diffraction peaks of Ca(OH)₂ and CaCO₃ appeared at the same time, indicating that CCS hydration produced excess Ca(OH)₂, which reacted with CO₂ to form CaCO₃. A comparison of the three gelling systems showed that as the CCS dosage increased, the Ca(OH)₂ diffraction peak in C-S was relatively stronger, and the Ca(OH)₂ diffraction peak in C-F was relatively weaker, indicating that a larger amount of Ca(OH)₂ was consumed during the hydration of FA, and the effect of CCS dosage was more significant in C-F. This could also be seen from the change in the diffraction peak of Na2SO4. As the CCS content increased, the diffraction peak of Na2SO4 decreased, indicating that Na2SO4 was consumed and the hydration product AFt was formed.
Figure 6 showed the XRD patterns of samples with different gelling systems with a CCS content of 25%, which were kept unchanged for 28 days and had different Na₂SO₄ contents. As could be seen in Fig. 6, when the Na2SO4 content was 2%, the products in the three systems were C-(A)-S-H, Ca(OH)2, and a small amount of AFt. As the Na2SO4 content increased, the AFt diffraction peak increased and the amount of AFt generated increased. When the Na2SO4 content was 10%, the hydration product contained more obvious CaSO4, and a large amount of CaSO4 was produced. Figure 6a showed that when the Na2SO4 content was increased from 2–6%, the diffraction peaks corresponding to Ca(OH)235 decreased significantly, while the peaks associated with the C-(A)-S-H gel36 increased correspondingly, and the diffraction peaks corresponding to AFt also increased slightly. When the Na2SO4 content was increased to 10%, a distinct diffraction peak of Na2SO4 was observed, indicating that excess Na2SO4 crystals precipitated and the CaSO4 product also increased. Figure 6b showed that as the Na2SO4 content increased, the Ca(OH)2 diffraction peak weakened, while the diffraction peaks of the hydration products increased. This indicated that sulfate had a positive effect on the hydration of both monocomponent and composite cementitious systems. Figure 6c showed a similar trend, where an increase in Na2SO4 content led to a decrease in the Ca(OH)2 diffraction peak and an increase in the C-(A)-S-H gel peak and the AFt diffraction peak, and a new product, mullite, appeared. This showed that sulfate promoted the hydration reaction. When the Na2SO4 content reached 10%, diffraction peaks of both crystalline Na2SO4 and gypsum appeared, reflecting changes in the hydration products. Overall, the right amount of sulfate promoted the hydration reaction, but excess sulfate not only precipitated crystals that hindered the hydration process, it also formed gypsum and other low-strength products, which adversely affected the strength of the cementitious system.
XRD images of different cementitious systems at 2%, 6% and 10% Na2SO4 contents. (a) CCS-Slag system (b) CCS-Slag + FA system. (c) CCS-FA system.
FTIR
Figure 7 shows the FTIR plots of specimens cured for 28 days at 5% CCS and 25% CCS for the three cementitious systems. Where the low wave number region (450 cm− 1-820 cm− 1) was the bending vibration of Si-O-Si (451 cm− 1) and Si-O-Al (619 cm− 1)chemical bonds in Slag and FA37,38, it could be seen that along with the increase of the dosage of CCS, the low-band transmittance in the C-S and C-F increases significantly. It shown that increasing the dosage of CCS could accelerate the hydration rate of Slag and FA, which was consistent with the compressive strength results. The strong absorption peak in the middle wave number region (820 cm− 1-1200 cm− 1) belonged to the hydration product C-(A)-S-H (Si-O-Si(Al) (962 cm− 1))39 of the gelling system, and the appearance of this peak characterized the production and transformation of C-(A)-S-H, and the C-F showed a significant difference, indicating that the CCS can effectively stimulate the FA for hydration reaction. In addition, analysis of Fig. 7 shown that the Si-O-Si(Al) (962 cm− 1) of C-S absorption peak was narrow and sharp, indicating that the hydration product had a high degree of polymerization of silica-aluminate ions, and generates more and denser C-(A)-S-H, which was the reason for the highest compressive strength of the C-S. At the same time, the absorption peak of SO42− appeared around the wave number of 1117 cm− 1 40, indicating that sulfate existed in the gelling system at this time. The absorption peak near 1420 cm− 1 was the characteristic peak of O-C-O41 chemical bonding of CO32−, indicating carbonization of the hydration product Ca(OH)2. The absorption peaks with wave numbers near 1643 cm− 1 and 3444 cm− 1 were characteristic peaks of H-OH bonding indicating the presence of bound water in the product42. The presence of these bands was due to crystalline H2O in C-(A)-S-H structure. The absorption peak with wave number near 3638 cm− 1 was the peak characterizing the O-H chemical bond in Ca(OH)2, which increases along with the increase in the dosage of CCS, indicating that CCS provides an alkaline environment for the gelling system, which was in agreement with the XRD analysis.
FITR of 5% and 25% CCS dosing for different cementitious systems. (a) CCS-Slag system. (b) CCS-Slag + FA system. (c) CCS-FA system.
In conclusion, CCS effectively boosted the hydration of both slag and FA. The FTIR spectra of the six samples shown similar wave forms, indicated fundamental consistency in hydration products among the three systems. Differences in compressive strength were attributed to variations in hydration product polymerization degree rather than differences in product type.
SEM and EDS
Figure 8 shows the SEM images of three gelling systems with 5%, 25% and 30% CCS added at 6% Na2SO4 content during 28 days of maintenance. As shown in Fig. 8a, b, the addition of CCS accelerated the hydration reaction, with the amount of CCS increased from 5 to 25%, more hydration products were generated, cemented and stacked with each other, and the micro structure shown a dense state. When the dosage of CCS exceeds 25% and reaches 30%, as shown in Fig. 8b, c, and the original hydration products are replaced by a large number of foil-like products, which generates a part of the hollow structure and reduces the compressive strength43. The microscopic results were consistent with the experimental data, confirming that the moderate amount of CCS dosing on the C-S was favorable, and shown a first increase and then decrease in the law, in 25% to achieve the best results.
SEM images of the gelling systems with 5%, 25% and 30% CCS dosage. (6% Na2SO4 content). (a) CCS-Slag system (5% CCS dosage). (b) CCS-Slag system (25% CCS dosage). (c) CCS-Slag system (30% CCS dosage). (d) CCS-Slag + FA system (5% CCS dosage). (e) CCS-Slag + FA system (25% CCS dosage). (f) CCS-Slag + FA system (30% CCS dosage). (g) CCS-FA system (5% CCS dosage). (h) CCS-FA system (25% CCS dosage). (i) CCS-FA system (30% CCS dosage). (j) EDS mapping of point (1) (k) EDS mapping of point (2) (l) EDS mapping of point 3.
As illustrated in Fig. 8d, with a lower dosage of CCS, the pore spaces within the cementitious system contained numerous unhydrated spherical FA particles. At this stage, only a minimal degree of FA had reacted, producing a limited amount of hydration producted that partially fill the pore spaces, resulting in relatively low compressive strength. As the CCS dosage increased to 25%, the spherical FA particles are nearly absent. The hydration reaction of FA generated an amorphous C-(A)-S-H gel, which, along with unhydrated gelling material particles, cements together to form a denser microstructure. This transition corresponded to a significant increase in compressive strength. However, when the CCS dosage was further increased to 30%, as shown in Fig. 8f, the previously dense microstructure is largely replaced by inter-stacked flakes and rods. This change leads to the formation of a more open, grid-like structure, resulting in a decrease in compressive strength.
Analyzing Fig. 8g, at this dosage, the microstructure of the cementitious system exhibits a small proportion of C-(A)-S-H gel that encapsulates unhydrated FA and Slag powder. Spherical unhydrated FA particles remain visible within the microstructure, which otherwise appears relatively dense. However, the presence of cracks is noticeable. Upon increasing the dosage of CCS to 30%, as depicted in Fig. 8h, i, the cementitious material undergoes significant hydration. A substantial amount of hydration products fills the pore spaces; however, the emergence of needle-like and rod-like structures within these products disrupts the previously dense microstructure. This disruption results in a more open, loose morphology, and the development of additional cracks, which contributes to a reduction in compressive strength.
Figure 9 shows the SEM images of specimens of three cementitious systems with 25% CCS and 2%, 6% and 10% Na2SO4 content, respectively, which were cured for 28 days. Figure 9a illustrates that in the C-S with 2% Na2SO4 content, there are numerous flaky Ca(OH)2 crystals and a small amount of CaCO3. As the Na2SO4 content increases to 6%, the flaky Ca(OH)2 is largely replaced by hydrated gel, resulting in a denser microstructure. At 10% Na2SO4 content, the microstructure shows the formation of gypsum clusters and a significant amount of Na2SO4 crystals on the surface of the hydration products. These Na2SO4 crystals absorb moisture, expand, and compromise the structural stability of the cementitious system. Figure 9d shows that with 2% Na2SO4 content in the C-F filamentous flocculent products appear on the surface of the FA particles, indicating a weak hydration reaction. As Na2SO4 content increases, the hydration of the FA particles progresses, with the resulting hydrated C-(A)-S-H gel enveloping the unhydrated FA particles and forming a dense matrix that fills the pores. At 10% Na2SO4 content, numerous rod-like and granular products appear, accompanied by the formation of cracks, which indicates damage to the cement structure. Figure 9h, i reveal that at 10% Na2SO4 content, the microstructure is characterized by a high density of needle-like and rod-shaped products, alongside an increase in porosity. The various hydration products are scattered and fail to bond effectively, leading to severe structural damage.
SEM images of gelling system at 2%, 6% and 10% Na2SO4 content. (25% CCS dosage). (a) CCS-Slag system (2% Na2SO4 content). (b) CCS-Slag system (6% Na2SO4 content). (c) CCS-Slag system (10% Na2SO4 content). (d) CCS-Slag + FA system (2% Na2SO4 content). (e) CCS-Slag + FA system (6% Na2SO4 content). (f) CCS-Slag + FA system (10% Na2SO4 content). (g) CCS-FA system (2% Na2SO4 content). (h) CCS-FA system (6% Na2SO4 content). (i) CCS-FA system (10% Na2SO4 content). (j) EDS mapping of point (1) (k) EDS mapping of point (2) (l) EDS mapping of point 3.
The Ca(OH)2 in the CCS provided Ca2+ and OH− ions. Na2SO4 contributes Na⁺ ions and SO₄²⁻ ions, which reacted to form NaOH. This NaOH, in turn, interacted with Slag and FA to produce additional hydration products such as C-(A)-S-H, which filled the pores, optimize pore structure, and improve compressive strength.
However, excessive Na2SO4 leads to the formation of more Aft and the precipitation of Na₂SO₄ crystals, which expand and damage the pore structure, ultimately reducing compressive strength. Similarly, an excess of CCS may result in unreacted Ca(OH)₂ precipitating as crystals, which weakens the crystal connection layer and decreases compressive strength.
Compressive strength prediction model based on projection-seeking regression
PPR model
PPR can objectively analyze and describe the structural characteristics and laws of each data sample, and achieve high-precision simulation calculations without making any assumptions based on the data information of each factor44. This method employs computer technology to project multi-dimensional spatial data into a low-dimensional subspace. By minimizing a specific projection index, it identifies a projection that captures the underlying laws and characteristics of the original data, facilitating the analysis of multi-dimensional datasets45. Let there be a set of random variables (X, Y), where Y is a Q-dimensional random variable. X is a P-dimensional random variable. PPR can be based on the results of n observations of (X, Y) (Xi, Yi) (i = 1,2,3,…,n). The model X, Y PPR can be expressed as follows: the regression function f (x) = E (Y | X = x) is approximated in the form of a weighted sum of multiple ridge functions:
Where: fm -mth ridge function; M -upper limit of the number of ridge functions; βm -ridge function contribution weight coefficient; αjm -mth projection value in the jth direction(j = 1, 2, 3, ···,M); \(\:\sum\:_{j=1}^{P}{\alpha\:}_{jm}^{2}=1\)
Minimization criteria:
Where: Mu -optimal number of ridge functions; Wi -weight coefficient of the dependent variable46.
PPR projects multidimensional data to a lower dimension, gradually optimizes, iteratively calculates in hierarchical groups, and thereby calculates the ridge function fm, the optimal number of ridge functions mu, the projection direction αjm and the weight coefficient wi of each factor, determines the regression function, and thereby makes L2 satisfy the minimum value. For details of the basic principles and theories of PPR, see the literature44.
Compressive strength prediction model
The PPR model was developed by training the compressive strength data for different CCS dosage, Na2SO4 content and cementitious systems using the methodology presented in Sect. 3.3.1. The established predictive model was then used to fit the model samples, and the accuracy of the predictive model was assessed by analyzing the differences between the anticipated values and the samples. Nevertheless, the model’s expected values did not quite match its inherent advantages and disadvantages. As a result, as Table A shows, the test data was split into training and retention samples. The model’s accuracy is validated by predicting values for the retained samples using simulated training data. From the table, it can be seen that the independent variables are CCS dosage (X1), Slag dosage (X2), FA dosage (X3) and Na2SO4 content (X4), and the predictor is compressive strength (Y1). The model parameters were configured as follows: P = 4, Q = 1, N = 60, M = 5, Mu = 3, and Span = 0.2. Table 3 presents the influence weights of variables after constructing the PPR model. Here, P denotes the count of independent variables, Q represents the number of dependent variables, and N signifies the training sample size. M and Mu denote the maximum number of ridge functions and the optimal number of ridge functions, which are crucial for improving the prediction accuracy. Span refers to the smoothing coefficient, which ranges from 0 to 1.0, influencing the model’s sensitivity; lower values indicate heightened sensitivity47.Since the parameter values of the PPR model are objectively verifiable, the problem of non-uniqueness of the model caused by manual parameter assignment is avoided.
PPR model precision analysis
The precision of the PPR model was assessed using a 15% threshold for relative error between calculated and test values. Samples were considered qualified if their relative error was below 15%. Table 4 displays qualification rates and average relative errors for modeled and tested samples. The model demonstrates excellent fit with test data, showing robust stability and high accuracy in predicting compressive strength across various mixing ratios.
Contour plots based on the PPR model show the effect of CCS dosage, Slag dosage, FA dosage and Na2SO4 content on the compressive strength of the different cementitious systems as shown in Fig. 10. The PPR theory can be used to determine the effects of CCS dosage and Na2SO4 content on the compressive strength of the three cementitious systems, providing a feasible method for proportioning design and mechanical strength prediction of all-solid waste cementitious systems.
PPR compressive strength prediction contour plot. (a) CCS-Slag system.
(b) CCS-Slag + FA system. (c) CCS-FA system.
Conclusions
In this paper, the preparation of CCS-sulfate-geopolymer composite cementitious system, the study of CCS, Na2SO4 on the mechanical properties of the composite cementitious system, the establishment of the composite cementitious system 28d strength prediction model. The main conclusions are as follows:
-
1.
CCS and Na2SO4 have a great influence on the mechanical properties of the composite cementitious system, the optimum dosage of CCS is 25% and the optimum content is 6%. CCS and Na2SO4 have the most significant stimulating effect on the C-F system.
-
2.
The CCS provides alkaline environment as well as Ca2+ for the hydration reaction of the cementitious system, which provides sufficient conditions for the hydration reaction of geopolymer, and accelerates the rate of hydration reaction. When the dosage of CCS exceeds 25%, a large number of Ca(OH)2 saturated precipitation, precipitated Ca(OH)2 crystals filled in the gel, destabilizing the gel, forming part of the stress weak zone, resulting in a reduction of compressive strength.
-
3.
The mechanical effect of Na2SO4 content on the gel system is divided into two stages: Na2SO4 content is less than < 6%, Na2SO4 and geopolymer hydration products react to form AFt, filling the pores and improving the strength of the gel system; Na2SO4 content > 6%, react with Ca2+ to form gypsum, and there is precipitation of Na2SO4, and these expansion products destroy the microstructure of the gel system, affecting the mechanical properties of the gel system.The test results showed that the idea of “turning danger into safety” was feasible.
-
4.
Based on PPR theory, a strength prediction model of cementitious composite system was established. Practice has proved that PPR can be used to determine the 28d compressive strength of cementitious composite system at different dosages of CCS, Na2SO4, Slag and FA, which provides a feasible method for designing and predicting the strength of cementitious composite materials with different ratios.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Haddad, R. H. & Alshbuol, O. Production of geopolymer concrete using natural pozzolan: A parametric study. Constr. Building Mater. J. 114, 699–707 (2016).
Li, L. Mitigation of China’s carbon neutrality to global warming. Nat. Commun. J. 13(1), 5315 (2022).
Qaidi, S. M., Dinkha, Y. Z., Haido, J. H., Ali, M. H. & Tayeh, B. A. Engineering properties of sustainable green concrete incorporating eco-friendly aggregate of crumb rubber: A review. Clean. Prod. J. 324, 129251 (2021).
Gökçe, H. S., Tuyan, M. & Nehdi, M. L. Alkali-activated and geopolymer materials developed using innovative manufacturing techniques: A critical review. Constr. Building Mater. J. 303, 124483 (2021).
Sharma, V., Dash, S. & Gupta, P. Comprehensive review of fly ash: Environmental impact and applications. Environ. Qual. Manage. J. 34(2), e22338 (2024).
Peng, W., Xianjun, L., Shugang, H. & Lei, Z. Research progress on the activation of the cementitious properties of granulated blast furnace slag. Metal Mine (in Chinese) J. 10, 157–161 (2012).
Jun, C., Xiaoying, J., Yuxiao, Z. & Liang, L. Phase evolution and quantitative analysis of fly ash-cement mortar in sulfate-chloride environment. Mater. Rep. (in Chinese) J. 22, 161–167 (2024).
Rashad, A. M., Bai, Y., Basheer, P. A. M., Milestone, N. B. & Collier, N. C. Hydration and properties of sodium sulfate activated slag. Cem. Concrete Compos. J. 37, 20–29 (2013).
Wu, P., Liu, X., Liu, X., Zhang, Z. & Wei, C. Effect of Industrial byproduct gypsum on the mechanical properties and stabilization of hazardous elements of cementitious materials. Rev. Mater. J. 17(17), 4183 (2024).
Huang, S. Y., Li, Z. H. & Cheng, J. P. Reaction kinetics in the FA-Ca(OH)2-H2O system. J. Chin. Ceramic Soc. J. 14(1), 191–197 (1986).
Lin, X. Q., Wang, D. M., Xu, C. Y., Zhao, J. H. & Han, F. H. Effect of sulfate and chloride exciters on FA activity. Coal Ash J. 24(1), 4–7 (2012).
Peng, R. Design and performance study of tailings filler prepared from sodium sulfate-inspired cementitious materials. Wuhan University of Technology. D. Wuhan, China (2019).
Wei, J. & Chun, H. Mechanical properties of sisal fiber-reinforced fly ash cement mortar activated by sodium sulfate. Constr. Building Mater. J. 445, 137925 (2024). ISSN 0950 – 0618.
Yu, Z. et al. Preparation and microscopic characteriz-ation of typical sulfate solid waste composite cementitious materials. Inorg. Chemicals Ind. J. 56(04), 90–97 (2024).
Li, Z. Effect of alkali and sulfate on the hydration characteristic of cement-based materials containing coal gasification slag. Mater. J. 15(24), 8868 (2022).
Huang, X. Enhancement of hydration product AFt in soft ground reinforcement. J. Chin. Ceramic Soc. J. 28(4), 299–302 (2000).
Deng, D. H., Xiao, J., Yuan, Q., Liu, Z. Q. & Zhang, W. E. Effect of limestone powder on the sulfate erosion resistance of cementitious materials and its mechanism. J. Chin. Ceramic Soc. J. 10, 1243–1248 (2006).
Deng, Y., Zhang, C. & Wei, X. Influence of lithium sulfate addition on the properties of Portlandcement paste. Constr. Build. Mater. J. 50, 457–462 (2014).
Zhao, L. W., Zhu, G. Y., Li, S. P., Meng, Z. H. & Mou, X. J. Characteristics of Calcium Carbide Slag and comprehensive utilization research progress. Clean. Coal Technol. J. 27(03), 13–26 (2021).
An, S., Wang, B. M., Chen, W. X. & Zhao, Q. X. Mechanism of action of CS to stimulate Slag-FA composite cementitious materials. Bull. Chin. Ceramic Soc. J. 42(04), 1333–1343 (2023).
Wan, Z. H., Zhang, W. Q., Liu, Z. C. & Wang, F. Z. Study on the performance of CS-Slag composite cementitious material. Bull. Chin. Ceramic Soc. J. 41(05), 1704–1714 (2022).
Abdelbaset, A. M., Katunský, D., Zeleňáková, M. & El-Feky, M. H. Mechanical properties stabilization of low plasticity kaolin soil using fly ash and hydrated lime. Case Studi. Construct. Mater. J. e03662 (2024).
Lam, T. V. & Nguyen, M. H. Incorporating Industrial by-products into geopolymer mortar: Effects on strength and durability. Mater. J. 16(12), 4406 (2023).
Ling, Y. H., Xu, D. Q. & Zhao, X. H. Status and prospect of utilization of calcium carbide slag and carbon dioxide resource utilization. Bull. Chin. Ceramic Soc. J. 38(09), 2876–2881 (2019).
Seo, J. Utilization of calcium carbide residue using granulated blast furnace slag. Mater. J. 12(21), 3511 (2019).
Wang, Y., Huo, H., Chen, B. & Cui, Q. Development and optimization of phosphogypsum-based geopolymer cement. Constr. Buildi. Mater. J. 369, 130577 (2023).
Feng, Y. Effects of phosphogypsum substitution on the performance of ground granulated blast furnace slag/fly ash-based alkali-activated binders. J. Buildi. Eng. J. 70, 106387 (2023).
Li, H. Investigation on mechanical properties of excess-sulfate phosphogypsum slag cement: from experiments to molecular dynamics simulation. Constr. Build. Mater. J. 315, 125685 (2022).
JGJ/T 70-2009; Standard for test method of performance on building mortar. Industry Standards-Construction Industry. S. Chinese Standard: Beijing, China, (2009).
Babak, V. & Ali, G. Self-healing effect of hydrogels in cement slag and fly ash pastes. Constr. Build. Mater. J. 438 (2024). 137036, ISSN 0950 – 0618.
Zhang, Y. et al. Understanding the changes in engineering behaviors and microstructure of FA-GBFS based geopolymer paste with addition of silica fume. J. Building Eng. J. 70, 106450 (2023).
Gao, X. et al. Calcium carbide residue as auxiliary activator for one-part sodium carbonate-activated slag cements: Compressive strength, phase assemblage and environmental benefits. Construct. Buildi. Mater.. J. Volume 308 125015 (2021), ISSN 0950 – 0618.
Lodeiroa, P. & Fernandez, J. Compatibility studies between N-A-S-H and C-(A)-S-H gels. Study in the ternary diagram Na2O-CaO-Al2O3-SiO2-H2O. Cem. Concr Res. j. 41 (2011).
Zhao, X. Investigation into the effect of calcium on the existence form of geopolymerized gel product of fly ash based geopolymers. Cem. Concrete Compos. J. 103, 279–292 (2019).
Shaqour, F., Ismeik, M. & Esaifan, M. Alkali activation of natural clay using a ca (OH)2/Na2CO3 alkaline mixture. Clay Minerals J. 52(4), 485–496 (2017).
Walkley, B. Phase evolution of C-(N)-ASH/NASH gel blends investigated via alkali-activation of synthetic calcium aluminosilicate precursors. Cem. Concr. Res. 89, 120–135 (2016).
Mozgawa, W., Król, M., Dyczek, J. & Deja, J. Investigation of the coal fly ashes using IR spectroscopy. Spectrochimica Acta Part. A: Mol. Biomol. Spectrosc. J. 132, 889–894 (2014).
Yu, P., Kirkpatrick, R. J., Poe, B., McMillan, P. F. & Cong, X. Structure of calcium silicate hydrate (C-S‐H): Near‐, mid‐, and far‐infrared spectroscopy. J. Am. Ceramic Soc. J. 82(3), 742–748 (1999).
Yousuf, M., Mollah, A., Hess, T. R., Tsai, Y. N. & Cocke, D. L. An FTIR and XPS investigations of the effects of carbonation on the solidification/stabilization of cement based systems-Portland type V with zinc. Cem. Concrete Res. J. 23(4), 773–784 (1993).
Xia, M., Yao, Z., Ge, L., Chen, T. & Li, H. A potential bio-filler: the substitution effect of furfural modified clam shell for carbonate calcium in polypropylene. J. Compos. Mater. J. 49(7), 807–816 (2015).
Chu, D. H. CO2 mineralization into different polymorphs of CaCO3 using an aqueous-CO2 system. Rsc Adv. J. 3(44), 21722–21729 (2013).
Li, W. & Yi, Y. Use of carbide Slag from acetylene industry for activation of ground granulated blast-furnace slag. Constr. Build. Mater. J. 6, 238 (2020).
Bai, Y., Guo, W. & Zhang, Y. Low carbon binder preparation from Slag-red mud activated by MSWI FA-carbide Slag: Hydration characteristics and heavy metals’ solidification behavior. J. Clean. Prod. J. 134007 (2022).
Zheng, Z. G., He, J. X. & Gong, J. W. Projection pursuit regression for complex systems without assumed modeling techniques and application examples. China Water Power Press. J. China pp.43 (2019).
Yang, H. H., Yang, W. & Liu, H. L. Fitting the stress-strain law of gravel based on PPR data modeling technology. Mater. Rep. J. 37(13), 1–13 (2023).
Zhu, X., Bai, Y., Chen, X., Tian, Z. & Ning, Y. Evaluation and prediction on abrasion resistance of hydraulic concrete after exposure to different freeze-thaw cycles. Constr. Building Mater. J. 316, 126055 (2022).
Nath, P. & Sarker, P. K. Effect of GGBFS on setting, workability and early strength properties of fly ash geopolymer concrete cured in ambient condition. Constr. Building Mater. J. 66, 163–171 (2014).
Funding
This research was funded by: 1.Natural Science Foundation of Xinjiang Uygur Autonomous Region (2022D01B98). 2. Xinjiang Key Laboratory of Hydraulic Engineering Security and Water Disasters Prevention (ZDSYS-JS-2021-01). 3. “Xinjiang Key Laboratory of Hydraulic Engineering Safety and Water Disaster Prevention” open project (6660946/2522GCCRC).
Author information
Authors and Affiliations
Contributions
Conceptualization, M. G. and Y. W.; Data curation, A. S.; Formal analysis, A. S.; Funding acquisition, M. G.; Methodology, A. S. and H. L.; Software, A. S. and Y. W.; Supervision, M. G.; Validation, H. L. and R. H.; Writing – original draft, A. S.; Writing – review & editing, M. G. and Y. W. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gong, M., Shen, A., Wang, Y. et al. Alkali and sulfate effects on mechanical properties and microscopic mechanisms of slag and fly ash geopolymers. Sci Rep 15, 3681 (2025). https://doi.org/10.1038/s41598-025-88194-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-88194-y