Abstract
Analyze the use of antibacterial drugs and bacterial resistance in psychiatric hospital during the epidemic. Using the hospital information system and the National Antibacterial Drug Clinical Application Monitoring Network, we retrospectively collected data on the use of antibacterial drugs and bacterial resistance in psychiatric hospitals during the 2022 epidemic. During the 2022 epidemic, our hospital had an antibiotic use rate of 5.00%, a usage intensity of 3.07, a combined medication rate of 11.11%, a cumulative DDDs of 12,039.04, and antibiotic costs accounting for 3.95% of total drug costs. These are much lower than the levels in Jiangsu Province and nationwide. However, the rate of microbiological submission for antibacterial drug use was 77.78%, higher than that of Jiangsu Province and nationwide. The main antibiotics used in our hospital were third-generation cephalosporins, penicillins, and quinolone antibiotics, with the most commonly used being cefodizime, amoxicillin, and piperacillin–tazobactam. The results showed that Gram-negative bacteria mainly exhibited resistance to penicillins, cephalosporins, and quinolones, especially ampicillin, amoxicillin–clavulanic acid, ceftazidime, ceftriaxone, amikacin, and ciprofloxacin. Gram-positive bacteria mainly resisted penicillins, macrolides, and quinolones, especially penicillin, benzylpenicillin, erythromycin, levofloxacin, and ciprofloxacin. This study reveals a complex relationship between the rational use of antibacterial drugs and bacterial resistance in the psychiatric hospital. Although antimicrobial usage during the pandemic was generally appropriate, increased use in psychiatric settings correlated with rising bacterial resistance, thereby impacting treatment outcomes and patient prognosis. Therefore, it is recommended to enhance monitoring of bacterial resistance and regularly analyze resistance data to optimize antimicrobial use in psychiatric hospitals. This approach aims to ensure effective treatment while minimizing the development of resistant strains, ultimately improving the overall value of healthcare services.
Similar content being viewed by others
Introduction
As the novel coronavirus (COVID-19) continues to break out globally, it poses significant challenges to healthcare institutions worldwide. COVID-19 belongs to the β subtype of viruses, to which the general population is highly susceptible1. Infections within medical institutions can lead to prolonged hospital stays, increased medical costs, and even fatalities in severe cases2,3. Psychiatric hospitals, as critical and unique medical institutions, must address both the mental and physical health of patients.
The 2020 World Health Organization (WHO) report indicated that 8% of COVID-19 patients had bacterial co-infections, primarily involving Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, and Pseudomonas aeruginosa4. As the number of COVID-19 patients rises, it is essential to address bacterial infections and the importance of antibacterial treatments. However, the widespread use of broad-spectrum antibiotics has resulted in global concerns over misuse and improper use, leading to increased antibiotic resistance and the prevalence of multi-drug-resistant strains5. Patients with mental illnesses often lack self-care abilities and have weaker hygiene and self-protection awareness6,7. Long-term use of antipsychotic drugs can lead to conditions like granulocytopenia or leukopenia, reducing the body’s immunity and making them more susceptible to bacterial infections8. Moreover, psychiatric hospitals often employ a closed management system, with a high patient population participating in group activities, increasing the risk of infections9,10. Thus, the use of antibacterial drugs plays a crucial role in the treatment of psychiatric hospital patients11,12,13.
During the epidemic, the use of antibacterial drugs in psychiatric hospitals has inevitably become a pressing issue. This study aims to analyze the usage of antibacterial drugs in psychiatric hospitals during the epidemic and to examine bacterial resistance patterns, providing a reference for rational clinical antibiotic use and infection control during the epidemic.
Materials and methods
Source of data
Data on the use of antibacterial drugs in 2022 from nationwide, Jiangsu Province, and our hospital was retrieved from the National Antibacterial Drug Clinical Application Monitoring Network (http://y.chinadtc.org.cn/login). Through the hospital information system, we extracted microbial test samples from our hospital in 2022, along with positive pathogen identification and drug sensitivity test data.
Methods
We employed a retrospective approach, using Excel to collect data on the rate of antibiotic use, antibiotic use intensity (AUD), combined medication rate, cumulative frequency of various types of antibiotic usage (DDDs), the percentage of antibiotic costs in total drug expenses, rate of microbiological submissions for antibiotics, and bacterial pathogen species and resistance patterns. Antibiotics (such as cefodizime, amoxicillin, and piperacillin–tazobactam) were among the most frequently used in terms of cumulative DDDs in 2022, based on the classification of antimicrobial agents by type. Our study follows the Clinical and Laboratory Standards Institute (CLSI) classification standards for multidrug-resistant (MDR) bacteria, with the primary categories being methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), extended-spectrum β-lactamase-producing bacteria (ESBLs), carbapenem-resistant Enterobacteriaceae (CRE), carbapenem-resistant Pseudomonas aeruginosa (CRPA), and multidrug-resistant Acinetobacter baumannii (MDRAB).
Statistical analysis
All data were analyzed descriptively using SPSS 20.0 software. Descriptive statistics were used to calculate frequencies (n) and percentages (%) for categorical variables, while continuous variables, such as the cumulative DDDs of antibiotics, were summarized using means where appropriate. T-tests were applied to assess differences between groups, and a significance threshold of P < 0.05 was considered statistically significant. Data were presented in tabular and graphical formats to facilitate comparison between our hospital’s antimicrobial usage patterns and the reported provincial and national averages.
Results
Antibacterial drugs usage
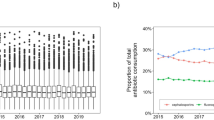
In 2022, the antibiotic drug usage rate in our hospital was 5.00%, with a usage intensity of 3.07, cumulative DDDs of 12,039.04, and the antibiotic costs accounting for 3.95% of total drug expenses. These metrics are significantly lower than those of Jiangsu Province and the national levels (all P < 0.01). The combined medication rate in our hospital was 11.11%, which was significantly lower than that of Jiangsu Province (P < 0.05) but not significantly different from the national level (P = 0.16). The microbiological submission rate for antibacterial drug use in our hospital reached 77.78%. Although higher than the levels in Jiangsu Province and nationwide, the differences were not statistically significant (P > 0.05) (Fig. 1).
Antibacterial Drugs Usage. (A) The percentage of antibiotic drug usage (%); (B) Antibiotic use intensity; (C) Antibiotic combination usage rate (%); (D) Cumulative DDD of antibiotics; (E) Proportion of antibiotic costs; (F) Microbial submission rate. Note Compared to Jiangsu Province and the national average, MHC has lower rates of antimicrobial usage, intensity of use, combination medication rate, cumulative DDDs, and proportion of antimicrobial costs. However, the microbiological submission rate is higher.
Accumulated DDDs of various types of antibiotics
Based on the classification of antibiotics, the accumulated DDDs in 2022 were mainly from third-generation cephalosporins (74.61%), penicillin class + enzyme inhibitors (9.52%), penicillin antibiotics (7.15%), and quinolone antibiotics (6.15%). By antibiotic variety ranking, the accumulated DDDs in 2022 mainly consisted of cefodizime (74.41%), amoxicillin (7.15%), and piperacillin–tazobactam (5.27%) (Fig. 2).
Accumulated DDDs of various types of antibiotics. (A) The percentage of classification of antibiotics; (B) By antibiotic variety ranking.
Bacterial resistance status
In 2022, our hospital submitted 5,373 specimens for testing. Of these, 2,737 positive bacterial strains were isolated, with a positive rate of 50.94%. The majority were Gram-negative bacteria, primarily Pseudomonas aeruginosa (23.20%), Escherichia coli (22.43%), and Klebsiella pneumoniae (15.45%). Among the key multi-drug-resistant bacteria detected, Staphylococcus aureus in Gram-positive strains had the highest proportion of resistance (70.69%), followed by Acinetobacter baumannii (59.26%) (Table 1).
Research has shown that the Gram-negative bacteria in our hospital primarily show high resistance rates to penicillin, cephalosporins, and quinolones. For instance, Pseudomonas aeruginosa has a 100% resistance rate to tetracyclines, ampicillin, amoxicillin–clavulanate, ceftriaxone, and ertapenem. Drugs with a resistance rate exceeding 50% include compound neomycin (66.46%) and tigecycline (66.46%). E. coli demonstrates a high resistance rate to cephalosporins (50.68%) and quinolones (73.53%), including specific drugs like ampicillin (92.92%), cefazolin (80.71%), ceftriaxone (77.91%), amikacin (75.29%), levofloxacin (72.31%), and ciprofloxacin (74.76%). K. pneumoniae shows high resistance to penicillins (57.31%) and quinolones (50%), including specific drugs like ampicillin (100%), cefazolin (60.16%), ceftriaxone (56.65%), amikacin (58.12%), and ciprofloxacin (54.83%). Acinetobacter baumannii displays high resistance to penicillins (95.77%), cephalosporins (70.42%), and carbapenems (57.84%), with specific drugs like ampicillin, amoxicillin–clavulanate, cefazolin, and amikacin all having a 100% resistance rate. Drugs with a resistance rate exceeding 50% include piperacillin–tazobactam (57.14%), ticarcillin-clavulanate (50.00%), cefepime (58.73%), ceftriaxone (59.77%), imipenem (59.26%), and ciprofloxacin (60.85%) (Table 2).
Among Gram-positive bacteria, Staphylococcus aureus has a high resistance rate to penicillins (82.76%), macrolides (53.45%), and quinolones (47.70%); resistance rates exceeding 50% were observed for penicillin, benzylpenicillin, erythromycin, levofloxacin, and ciprofloxacin. The resistance rate for vancomycin is low (1.79%), and no resistant strains were found for nitrofurantoin, linezolid, tigecycline, and rifampin (Table 3).
Discussion
During the epidemic in 2022, our hospital had an antimicrobial usage rate of 5.00%, an intensity of 3.07, a combination medication rate of 11.11%, a cumulative DDDs of 12,039.04, and antimicrobial expenses accounting for 3.95% of the total drug costs. The antibiotic usage rate in our hospital is lower than the rates reported in studies14,15,16, and it meets the national guidelines for the Clinical Application of Antimicrobial Agents17 and the Notice on Further Strengthening the Special Rectification of Clinical Use of Antimicrobial Agents Nationwide18, which set a target of ≤ 5% for psychiatric hospitals. In line with these policies, our hospital has actively implemented strict antibiotic management measures, such as enhancing the monitoring and evaluation of antimicrobial usage, providing training for medical staff to raise awareness of rational drug use, and promoting the rational use of antibiotics through multidisciplinary collaboration. Additionally, psychiatric patients often have other underlying conditions and complex medical needs, which may also lead to a more cautious approach to antibiotic use. Our hospital had a microbial submission rate for antimicrobial usage of 77.78%, higher than the levels in Jiangsu province and nationally, complying with the requirements of the Notice on Further Strengthening the Special Rectification of Clinical Use of Antimicrobial Agents Nationwide (2011) that the microbial submission rate before using antimicrobial agents should be ≥ 30%19. These results indicate that our hospital’s antimicrobial usage during the epidemic was generally rational, in line with the requirements for psychiatric clinical practice, and essentially provided appropriate antibiotic treatment for patients. However, it’s crucial to further strengthen the supervision, publicity, and training on the rational use of antimicrobials to enhance medical staff awareness about the proper use of antimicrobials and prevent misuse.
DDDs reflect the frequency of antimicrobial use. A higher DDD indicates a greater preference for that drug or category of drugs. During the epidemic, our hospital mainly used third-generation cephalosporins (74.61%), penicillin + enzyme inhibitors (9.52%), penicillin antibiotics (7.15%), and quinolone antibiotics (6.15%). Cefotaxime (74.41%) was the most used, followed by amoxicillin (7.15%) and piperacillin–tazobactam (5.27%). The 2022 WHO Global Antimicrobial Resistance and Use Surveillance System (GLASS) report showed that broad-spectrum penicillin and third-generation cephalosporins were the most commonly used. During the epidemic, respiratory infections, especially upper respiratory infections, were predominant in psychiatric specialties, primarily treated with penicillin and cephalosporin antibiotics17. Among the cephalosporins, third-generation cephalosporins have a stronger antibacterial effect against most G- bacteria than the first and second generations, making them a more popular choice in clinical settings.
Related to antibiotic resistance in 2022 revealed that out of 5,373 samples submitted, 2,737 strains of bacteria tested positive, a positivity rate of 50.94%. The majority were Gram-negative bacteria, consistent with many studies20,21. Pseudomonas aeruginosa (23.20%), Escherichia coli (22.43%), and Klebsiella pneumoniae (15.45%) were among the most common. Gram-negative bacteria showed high resistance rates to penicillins, cephalosporins, and quinolones. The most drug-resistant antibiotics include ampicillin, amoxicillin–clavulanate, ceftizoxime, ceftriaxone, norfloxacin, and ciprofloxacin. Gram-positive bacteria were primarily resistant to penicillins, macrolides, and quinolones, with high resistance rates against penicillin, benzylpenicillin, erythromycin, levofloxacin, and ciprofloxacin.
This study found that although the overall antibiotic usage in our hospital is relatively low, certain antibiotics are used excessively, mainly cephalosporins, penicillins and their enzyme inhibitors, and quinolones. Among these, cefodizime is used the most, followed by amoxicillin, piperacillin–tazobactam, and others. This correlates with the high antibiotic resistance rates of Gram-negative and Gram-positive bacteria in our hospital. The excessive frequency of use of these antibiotics is likely contributing to the generally high resistance rates. In addition to drug usage, environmental factors and infection control policies in psychiatric settings can indeed contribute to resistance. The closed environment and high-density patient gatherings in psychiatric hospitals increase the risk of infection transmission. Psychiatric patients generally have longer hospital stays, and some may be uncooperative, allowing resistant bacteria to spread rapidly through contact. This is an important factor in the increase in resistance rates22. Environmental factors, such as water sources, air quality, and surface cleanliness, may also affect the spread of resistant bacteria. Infection control strategies, including hand hygiene, isolation measures, and appropriate disinfection and cleaning protocols, are crucial to reducing the spread of resistant bacteria; strengthening infection monitoring and control policies can help limit their transmission23.
Additionally, psychiatric patients may suffer from other physical illnesses, which could increase their demand for antibiotics, especially in the treatment of complex infections. Furthermore, the immune systems of psychiatric patients may be weakened, and some psychiatric medications may affect their immune function, reducing their ability to resist infections and making them more susceptible to resistant bacteria24. Therefore, besides medication, environmental factors, infection control policies, and patients’ health status are all worth attention. To effectively address this issue, psychiatric hospitals need to adopt comprehensive measures, including strict infection control policies, rational antibiotic management, and education for both patients and healthcare workers, to reduce the development and spread of resistance.
Conclusion
This study offers an in-depth analysis of the use of antimicrobial agents and bacterial resistance in psychiatric specialty hospitals during the pandemic. It was found that the use of antimicrobials during the pandemic was generally reasonable. However, as the use of antimicrobials in psychiatric specialty hospitals increased, so did the rate of bacterial resistance. Therefore, there is a need to strengthen the management of antimicrobial use in psychiatric specialty hospitals including drug sensitivity testing and tailored antibiotic protocols based on resistance data, enhance the monitoring of bacterial resistance, and rationally select antimicrobials with low resistance rates based on drug sensitivity test results to reduce the occurrence of bacterial resistance. This study contributes to improving antibiotic management and reducing bacterial resistance in psychiatric specialty hospitals, providing experience and strategies for the rational use of antimicrobials in these settings. Measures include enhancing monitoring systems by developing and implementing regular surveillance of antibiotic usage and resistance patterns, which can collect and analyze data, identify trends, and adjust antibiotic strategies in a timely manner. Regular educational training for patients and healthcare staff on the importance of correct antibiotic use is also essential. Establishing a mechanism for periodic review of antibiotic prescribing patterns and setting up a dedicated antibiotic stewardship team to evaluate the appropriateness of prescriptions and provide feedback ensures that all antibiotic prescriptions undergo strict scrutiny. Implementing strict infection control policies, such as hand hygiene, environmental disinfection, and isolation measures, reduces the risk of bacterial and resistant strain transmission. Finally, promoting multidisciplinary collaboration among pharmacists, infectious disease specialists, and psychiatrists helps ensure the rationality and effectiveness of antibiotic use.
Data availability
The dataset generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Chinese Society for Preventive Medicine, COVID-19 Prevention and Control Expert Group. The latest understanding of the epidemiological characteristics of COVID-19. J. Viral Dis. China 10(2), 86–92 (2020). https://doi.org/10.16505/j.2095-0136.2020.0015.
Li, Q. et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 382(13), 1199–1207 (2020).
Kissler, S. M. et al. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 368(6493), 860–868 (2020).
Shen, N., Liu, B. & He, B. Discussion on the rational use of antibacterial drugs when COVID-19 is complicated with infection. Chin. J. Tuberc. Respir. Dis. 44(1), 4–7 (2021).
Jin, W. F. et al. Distribution and drug resistance analysis of bacteria in hospitalized psychiatric patients. Int. J. Lab. Med. 37(22), 3128–3131 (2016).
Almeida, M. & Fletcher, S. J. Serious mental illness in women. Curr. Opin. Psychiatry 35(3), 157–164 (2022).
Daugherty, B., Warburton, K. & Stahl, S. M. A social history of serious mental illness. CNS Spectr. 25(5), 584–592 (2020).
Feng, J. C. & Yang, G. Y. Distribution of 317 strains of infectious pathogens in a psychiatric hospital in 2019 and their impact on the rational use of antimicrobial drugs. Antimicrob. Pharm. 18(06), 856–859 (2021).
Choi, K. R. et al. Mental health conservatorship among homeless people with serious mental illness. Psychiatr. Serv. 73(6), 613–619 (2022).
Demler, T. L. & Mulcahy, K. B. Implications of infection and trends of antibiotic prescribing in hospitalized patients diagnosed with serious mental illness. Int. Clin. Psychopharmacol. 33(1), 49–55 (2018).
Ye, F., Wu, L. & Qi, M. H. Analysis of hospital infections and the use of antimicrobial drugs in psychiatric inpatients. North. Pharm. 19(09), 22–25 (2022).
Zhang, F. M., Zhou, J. Q. & Zhou, F. Survey and analysis of irrational use of antimicrobial drugs in inpatients of a psychiatric specialty hospital. Antimicrob. Pharm. 18(2), 214–215. https://doi.org/10.13493/j.issn.1672-7878.2021.02-017 (2021).
Wan, H. Analysis of the use of antimicrobial drugs in a psychiatric specialty hospital in 2018. Chin. Pharm. 29(02), 34–36 (2020).
Klein-Petersen, A. W., Kohler-Forsberg, O. & Benros, M. E. Infections, antibiotic treatment, and the microbiome in relation to schizophrenia. Schizophr. Res. 234, 71–77 (2021).
Tieri, J. J. et al. Antibiotic prescribing in mental health units across the Veterans’ Health Administration: How much and how appropriate?. Infect. Control Hosp. Epidemiol. 44(2), 308–311 (2023).
Wang, H. W. et al. Investigation of hospital infections and the use of antimicrobial drugs in a tertiary psychiatric specialty hospital. Chin. J. Hosp. Infect. 15, 2377–2381 (2023).
Clinical Application Guideline of Antimicrobial Drugs Revision Group. Clinical application guideline of antimicrobial drugs (2015 edition) 174 (People’s Health Publishing House, 2015).
National Health and Family Planning Commission of the People’s Republic of China. Notice on further conducting the national clinical application of antimicrobial drugs special rectification activity. Beijing, 2013.
National Health and Family Planning Commission of the People’s Republic of China. Notice on further conducting the national clinical application of antimicrobial drugs special rectification activity. Beijing, 2011.
Chen, J. J. et al. Analysis of bacterial resistance monitoring results in the First Affiliated Hospital of China Medical University in 2019. J. China Med. Univ. 50(3), 245–249 (2021).
Hu, F. P. et al. 2018 CHINET China bacterial resistance monitoring. Chin. J. Infect. Chemother. 20(01), 1–10. https://doi.org/10.16718/j.1009-7708.2020.01.001 (2020).
Harris, M., Fasolino, T., Ivankovic, D., Davis, N. J. & Brownlee, N. Genetic factors that contribute to antibiotic resistance through intrinsic and acquired bacterial genes in urinary tract infections. Microorganisms 11(6), 1407. https://doi.org/10.3390/microorganisms11061407 (2023).
Jamrozik, E. & Selgelid, M. J. Drug-resistant infection: Causes, consequences, and responses. In: Jamrozik, E. & Selgelid, M. (eds) Ethics and Drug Resistance: Collective Responsibility for Global Public Health. Public Health Ethics Analysis, vol 5. Springer, Cham, 2020. https://doi.org/10.1007/978-3-030-27874-8_1.
Okeke, I. N., Lamikanra, A. & Edelman, R. Socioeconomic and behavioral factors leading to acquired bacterial resistance to antibiotics in developing countries. Emerg. Infect. Dis. 5(1), 18–27. https://doi.org/10.3201/eid0501.990103 (1999).
Funding
The work is supported by the National Natural Science Foundation of China (82104244), Wuxi Municipal Science and Technology Bureau (K20231039 and K20231049), Top Talent Support Program for young and middle-aged people of Wuxi Health Committee (HB2023088), Scientific Research Program of Wuxi Health Commission (Q202457), Wuxi Taihu Talent Project (WXTTP2021), Medical Key Discipline Program of Wuxi Health Commission (FZXK2021012).
Author information
Authors and Affiliations
Contributions
Zhiqiang Du and Haohao Zhu conceived the study; Ying Jiang, Rongrong Lu, Qin Zhou, Yuan Shen and Mengmeng Ou collected the report; Ying Jiang, Zhiqiang Du and Haohao Zhu wrote the manuscript and edited the manuscript. All authors have approved publishment of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All experimental protocols were approved by Wuxi Mental Health Center. The ethics approval and informed consent have been waived by the Ethics Committee of Wuxi Mental Health Center. All methods were performed in accordance with relevant guidelines and regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jiang, Y., Lu, R., Shen, Y. et al. Analysis of antibacterial drug use and bacterial resistance in psychiatric hospital in the epidemic. Sci Rep 15, 4984 (2025). https://doi.org/10.1038/s41598-025-88260-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-88260-5