Abstract
This study aimed to explore the relationship between biochemical profiles and the risk of age-related macular degeneration (AMD) through a cross-sectional observational analysis. We examined data of U. S. population from the 2005–2008 National Health and Nutrition Examination Survey (NHANES) database. Student’s t-test, multivariable logistic regression, Pearson’s correlation, restricted cubic spline (RCS) model, and linear regression were applied to analyze the underlying relationship between biochemical profiles and the AMD risk, through comparing data between the non-AMD and AMD subgroups. Multivariable logistic regression, adjusted for age and demographic factors, showed no significant associations between the AMD risk and the levets of specific biochemical parameters (P > 0.05). Pearson’s correlation revealed a positive linear relationship between age and total bilirubin, uric acid in the non-AMD subgroup (P < 0.05), but no such liner association was found in the AMD subgroup (P > 0.05). The RCS model confirmed no non-linear relationships presented between these variables in the AMD subgroup. In addition, without age adjustment, significant associations were found between total bilirubin, uric acid, and the AMD presence (P < 0.05). Biochemical profiles, after adjusting for age, did not significantly influence the AMD risk. However, total bilirubin and uric acid might potentially be related to the AMD presence. Our findings suggest a need for further research to clarify the role of these biomarkers in AMD development.
Similar content being viewed by others
Introduction
Age-related macular degeneration (AMD) is a leading cause of vision loss in elderly individuals. It primarily affects the macula, the central portion of the retina responsible for sharp, central vision. The risk of developing AMD increases with age, of which the prevalence notably rising after the age of 501. AMD is a complex condition influenced by both genetic and environmental factors. One of the key challenges in treating AMD lies in its multifactorial pathophysiology and the lack of effective targeted therapies. While anti-vascular endothelial growth factor (anti-VEGF) agents have revolutionized the management of neovascular AMD, their efficacy varies among patients, and long-term treatment can be burdensome and costly. Moreover, treatment resistance of anti-VEGF and the need for frequent intravitreal injections pose significant challenges in clinical practice2. Innovative therapeutic strategies, such as gene therapy, stem cell therapy, and neuroprotection, hold promise for addressing of AMD and improving treatment outcomes3. However, translating these approaches from preclinical studies to clinical practice remains a formidable obstacle. Continued research efforts aimed at unraveling the complexities of AMD pathogenesis are crucial for addressing this growing public health concern.
Several studies suggest that dysregulation of lipid metabolism, or biochemical markers in the blood may contribute to AMD development and progression4. For example, elevated levels of certain lipids, such as low-density lipoprotein (LDL) cholesterol and triglycerides, have been associated with an increased risk of AMD5. Furthermore, chronic inflammation, characterized by elevated levels of inflammatory cytokines and C-reactive protein (CRP) in the blood, has been implicated in AMD pathophysiology6. Other biochemical markers, such as homocysteine, have also been studied in relation to the AMD risk7. In addition, elevated homocysteine levels, for instance, were argued to be associated with increased oxidative stress and endothelial dysfunction, which could contribute to AMD development. While these biochemical markers in blood showed some associations with AMD, their detailed causal relationship and predictive value in clinical practice remain unclear, calling for ongoing research8.
Hence, our study aimed to investigate the detailed relationship between biochemistry profiles and the risk of AMD, focusing on examining whether biochemistry profiles influenced the presence of AMD. By analyzing the relevant data on the biochemistry profiles of the U.S. population from the National Health and Nutrition Examination Survey (NHANES) database spanning from 2005 to 2008, where the data on AMD were available, we conducted a population-based analysis study to provide a reference dataset for related researches and to further explore the underlying relationship between biochemistry profiles and the risk of AMD.
Research design and methods
Study participants
NHANES, a comprehensive study conducted by the National Center of Health Statistics of the Centers for Disease Control and Prevention, serves as a representative survey of the non-institutionalized population in the United States. The NHANES study protocols obtained approval from the Institutional Review Board. For access to the dataset and additional information, please visit https://www.cdc.gov/nchs/nhanes/about_nhanes.htm9.
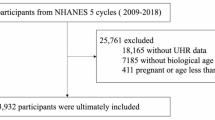
In order to analyze the AMD data, we focused our attention on participants involved in the NHANES survey campaigns from 2005 to 2008 (n = 20497), during which time the relevant epidemiological data and biochemical indicators related to AMD became available. After excluding participants who lacked data on retinal imaging grading data on AMD, we were left with a total of 5604 participants. Subsequently, 548 participants who had incomplete or missing information on demographics data were eliminated. Furthermore, 225 individuals lacking information on biochemistry profiles were also removed from the dataset, leaving us with 4831 participants with complete and required data for final analysis. Among them, 4458 participants were defined as having no AMD and 373 as AMD. The entire process of data integration was illustrated in Fig. 1. In the NHANES surveys, all participants were asked to provide written informed consent before being enrolled.
Definition of AMD severity
Retinal photographs of the final sample were collected. The assessment of the retinal photographs has been completed and provided on the NHANES website10. The diagnosis and classification of AMD was conducted by at least two experienced experts following a strict procedure. The definition of AMD was defined as the presence of drusen and/or pigmentary abnormalities, exudative or geographic atrophy signs in any eye.
Definition of biochemistry profiles
In our study, the detailed biochemistry profiles from NHANSE included albumin (g/L), alanine aminotransferase (ALT) (U/L), asparate aminotransferase (AST) (U/L), alkaline phosphotase (U/L), blood urea nitrogen (mmol/L), total calcium (mmol/L), cholesterol (mmol/L), bicarbonate (mmol/L), creatinine (µmol/L), gamma glutamyl transferase (U/L), serum glucose (mmol/L), refigerated iron, (umol/L), lactate dehydrogenase (U/L), phosphorus (mmol/L), total bilirubin (umol/L), total protein (g/L), triglycerides (mmol/L), uric acid (umol/L), sodium (mmol/L), potassium (mmol/L), chloride (mmol/L), and globulin (g/dL). The analyses of all the above biochemistry profiles were performed with a Beckman Synchron LX20. Specimens were processed, stored and shipped to Collaborative Laboratory Services in Ottumwa, Iowa. Detailed specimen collection and processing instructions were discussed in the NHANES Laboratory/Medical Technologists Procedures Manual (LPM). The analytical methods were described in the Description of the Laboratory Methodology section of NHANES11.
Definitions of covariates
From the demographic information of NHANSE, we selected covariates including age, gender, race/ethnicity (Mexican American, other Hispanic, non-Hispanic White, non-Hispanic Black, and other race), education level (less than 9th grade, 9th–11th grade, high school, college, and graduate), body mass index (BMI, calculated as weight in kilograms divided by height in meters squared), hypertension (an average systolic blood pressure above 140 mmHg/ diastolic blood pressure above 90 mmHg or self-reported as taking antihypertensive medication), income (family income-to-poverty ratio), smoking status (having smoked at least 100 cigarettes during their lifetime or not), alcohol use (having consumed at least 12 alcoholic drinks per year or not), and drinking habits9. All of the above covariates were carefully selected and accounted for in order to evaluate our targeted associations.
Statistical analysis
For analysis of all the data in our study, continuous variables were expressed as means ± standard deviations (SD). Meanwhile, categorical variables were summarized as the counts or percentages (%). Comparisons of the data between the non-AMD and AMD groups were performed using either the student’s t-test for continuous variables or the chi-square test for categorical variables. Furthermore, the multivariable logistic regression analysis was employed to evaluate the strength of association between the biochemistry profiles and the AMD status. In greater details, three models were evaluated for the adjusted odd ratios (OR) and their 95% confidence intervals (CIs) using multivariable logistic regression. Model 1 was unadjusted for any factors, while Model 2 was adjusted for race, BMI, family income-to-poverty ratio, and hypertension. Model 3 included additional adjustments for age, race, BMI, family income-to-poverty ratio, and hypertension. In the subgroup analysis, Pearson’s correlation analysis and linear regression were conducted to explore the associations between age and the specific parameters from biochemistry profiles in subgroups of both the AMD and non-AMD participants, respectively. In addition, the restricted cubic spline (RCS) model was further applied to explore the potential non-linear associations. All statistical analysis was conducted using the R software (version 4.4.2, R Foundation for Statistical Computing Austria, https://www.r-project.org/). A two-sided P value less than 0.05 was considered statistically significant.
Results
Baseline demographics and characteristics of AMD and non-AMD participants
The demographic and individual characteristics of the study population were analyzed and summarized in Table 1. A total of 4831 participants were included in the final analysis, with 373 having AMD and 4458 without AMD. The average age was 70.3 ± 11.2 years for the AMD group and it was 58.3 ± 12.0 years for the non-AMD group. In the AMD group, 51.7% were male and 48.3% were female, while in the non-DM group, 50.3% were male and 49.7% were female. Non-Hispanic whites made up the largest proportion in both the AMD and non-AMD groups, accounting for 71.3% and 53.9%, respectively. The most common education level was High school in the AMD group with a distribution of 29.2%, whereas it was Some college or AA degree with a distribution of 25.9% in the non-AMD group. The family income-to-poverty ratio was 1.95 ± 1.33 for the AMD group and 2.22 ± 1.53 for the non-DM group. The average BMI was 28.4 ± 5.68 in the AMD group and 29.3 ± 6.51 in the non-AMD group. The percentages of hypertension, smoking and alcohol drinking were 55.5%, 55.0%, and 65.1% in the AMD group, respectively, while the corresponding figures were 44.4%, 52.6%, and 69.0% in the non-AMD group.
Significant differences were observed in terms of age (P < 0.001), race (P < 0.001), family income-to-poverty ratio (P < 0.001), BMI level (P = 0.006) and hypertension (P = 0.001) between the AMD and non-DM participants. Meanwhile, no significant differences were found in the other variables mentioned above between the two groups.
Flow diagram for the selection of eligible participants included in the analysis of the association between biochemistry profiles and the risk of age-related macular degeneration (AMD) (NHANES: national health and nutrition examination survey, US, 2005–2008). AMD: age-related macular degeneration.
Associations between the biochemistry profiles and AMD status
By comparison and analysis, our results showed that the levels of albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, total bilirubin, triglycerides, uric acid and potassium were significantly different between the non-AMD and AMD participants, with all the P values less than 0.05. The levels of albumin and triglycerides were lower in AMD group compared to those of non-AMD (AMD vs. non-AMD: 41.4 ± 3.17 g/L vs. 41.9 ± 3.10 g/L for albumin; 1.76 ± 1.01 mmol/L vs. 1.88 ± 1.39 mmol/L for triglycerides), while the other of the above mentioned variables were higher in AMD group. No significantly differences between the non-AMD and AMD groups were found in the other variables listed above, such as alanine aminotransferase (ALT), asparate aminotransferase (AST), alkaline phosphotase, total calcium, cholesterol, bicarbonat, gamma glutamyl transferase, serum glucose, refigerated iron, phosphorus, total protein, sodium, chloride, and globulin (all P > 0.05). (Table 2)
To evaluate the association between the specific parameters from biochemistry profiles to the presence of the AMD, a multivariate logistics regression with three adjusted models was implemented as stated in the methods. The analysis with Model1 revealed statistically significant differences in the odds ratios (OR) with 95% confidence intervals (CI) for AMD presence across the following parameters: albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, total bilirubin, uric acid, and potassium (all P < 0.05). In the analysis of Model 2, which was adjusted for potential confounding variables including race, BMI, family income-to-poverty ratio, and hypertension, statistically significant differences in the OR with 95% CI were found for albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, total bilirubin, triglycerides, uric acid, and potassium (all P < 0.05). By adding the confounding variable of age in Model 3, no statistical differences were found in the OR with 95% CI between the above variables and the presence of AMD (all P < 0.05). (Table 3)
Correlations between age and the specific parameters from biochemistry profiles in subgroup analysis for AMD and non-AMD participants
The results of Pearson’s correlation analysis revealed a positive association between age and the level of total bilirubin in individuals from the non-AMD subgroup, with a Pearson’s correlation coefficient (r) value of 0.095 and a P value less than 0.001. Besides, a positive association between age and the level of uric acid in the non-AMD subgroup was found with a r value of 0.149 and a P value less than 0.001. However, no such significant correlation was found between age and the level of total bilirubin (r = 0.081, P = 0.120), or between age and the level of uric acid (r = 0.063, P = 0.057) in the AMD subgroup (Table 4). Significant correlations between age and the levels of albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, triglycerides, and potassium were found in both the AMD and non-AMD subgroups (all P < 0.05) (data shown in the supplementary materials).
Furthermore, the linear regression analysis revealed a linear relationship between age and the level of total bilirubin, and between age and the level of uric acid in the non-AMD subgroup, while no such linear relationship was observed in the AMD subgroup (Fig. 2). Additionally, the P values for nonlinearity obtained from the restricted cubic spline (RCS) analysis were 0.079 and 0.820 for the relationships between age and the level of total bilirubin, and between age and the level of uric acid in the AMD subgroup, respectively, indicating no evidence of a nonlinear correlation in these relationships. (Table 5; Fig. 3).
The linear regression analysis of associations between age and the specific parameters from biochemistry profiles in subgroup analysis for AMD and non-AMD participants. A: the plot and the linear association between age and total bilirubin in the non-AMD group; B: the plot and the linear association between age and total bilirubin in the AMD group; C: the plot of the association between age and uric acid in the non-AMD group; D: the plot and the linear association between age and uric acid in the AMD group. AMD: age-related macular degeneration; the red line: indicating the presence of a statistically significant linear relationship.
The restricted cubic spline (RCS) analysis of association between age and the specific parameters from biochemistry profiles in subgroup analysis for AMD and non-AMD participants. A: The plot and the RCS association between age and total bilirubin in the non-AMD group; B: the plot and the RCS association between age and total bilirubin in the AMD group; C: the plot of the RCS association between age and uric acid in the non-AMD group; D: the plot and the RCS association between age and uric acid in the AMD group. AMD: age-related macular degeneration.
Discussion
To the best of our knowledge, our study represented the detailed attempt to investigate the relationship between biochemistry profiles and risk of age-related macular degeneration (AMD) in a large population, as well as the association between age and total bilirubin or uric acid in AMD population. Our findings indicated that the status of biochemistry profiles did not significantly affect the risk of AMD after confounding the factor of age. However, there were no direct correlations between age and total bilirubin, or uric acid in AMD populations, while other parameters from biochemistry profiles were related to age. Total bilirubin or uric acid was significantly associated with the AMD risk without adjustment for age, indicating as a possible underlying affecting role factor for AMD, in addition to age.
Firstly, we conducted a conventional comparative analysis of the demographic characteristics between individuals from the non-AMD group and the AMD group to identify significant differences in demographic factors. Preliminary comparisons revealed notable differences between the AMD and non-AMD groups in terms of age, race, BMI, family income-to-poverty ratio, and hypertension. Specially, we observed the significant difference in age between the two subgroups, which was significantly higher in the AMD individuals. This indirectly validated the reliability of the survey data used in our study and indicated a relatively good response credibility, as AMD has been well-known to be associated with the factor of age12,13,14. Other studies have also shown similar age differences when grouping individuals with AMD based on survey questionnaires15,16,17.
Furthermore, we conducted a comparative analysis of data on biochemistry profiles in both AMD and non-AMD groups. The results revealed differences in albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, total bilirubin, triglycerides, uric acid, and potassium levels between the two groups. After that, a multiple regression analysis was conducted to explore the association between differing indicators from the biochemistry profiles and the AMD risk with adjustment for demographic and epidemiological variables. In details, we employed three corresponding adjusting models for calibration regression. There were significant differences in the relationship between the differing indicators from the biochemistry profiles and the presence of AMD, both with and without adjustment for demographic factors such as race, BMI, family income-to-poverty ratio, and hypertension. However, after including age as a demographic variable in the regression analysis, our results indicated that there was no direct association between albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, total bilirubin, triglycerides, or potassium levels and the risk of AMD. In order to further explore the underlying potential associations, the linear Pearson regression analysis was applied to delve into the correlation between respective pairs of the continuous variables from biochemistry profiles within subgroups of AMD and non-AMD, respectively. The results revealed significant correlations between age and the levels of albumin, blood urea nitrogen, creatinine, lactate dehydrogenase, triglycerides, and potassium in the AMD subgroup, with similar correlations observed in the non-AMD subgroup. Interestingly, no significant correlation was found between age and the levels of total bilirubin or uric acid in the AMD group. In contrast, a positive association was observed between age and the levels of total bilirubin or uric acid in individuals from the non-AMD group. In addition, we addressed the potential nonlinearity in the aforementioned associations by applying the RCS model18. The utility of RCS has been demonstrated in earlier studies19, which investigated the relationship between aldehyde exposure and the risk of cardiovascular disease, and revealed a nonlinearity relationship. Our results from the RCS model confirmed that no non-linear associations existed between age and the levels of total bilirubin or uric acid in the AMD group. The RCS plot results supported the findings from the Pearson’s correlation and linear regression analyses, indicating no linear or nonlinear associations between age and the levels of total bilirubin or uric acid in the AMD subgroup.
Total bilirubin is a vital biomarker that reflects various physiological processes within the body20. It is a yellowish pigment formed during the breakdown of heme, a component of hemoglobin found in red blood cells. It is mainly produced in the liver through the metabolism of heme and plays several vital roles in the body. One of its key roles is in the excretion of waste products. After production in the liver, bilirubin is conjugated with glucuronic acid, making it water-soluble and enabling its excretion into bile21. From there, bilirubin is eliminated from the body through feces, leaving a characteristic brown color. Therefore, total bilirubin levels usually serve as an indicator of liver function and the body’s capacity to metabolize and eliminate waste products.While elevated levels of total bilirubin can indicate liver dysfunction or impaired bile flow, physiological levels are typically considered normal and may even confer certain health benefits, such as a reduced risk of cardiovascular disease and a protection against oxidative stress-related damage22,23. Our results indicated that age was not associated with total bilirubin levels in AMD participants, while total bilirubin was revealed to be associated with the presence of AMD without age adjustment.. The underlying reason for this association might be linked to the reduced liver function and increased oxidative stress damage related to totle bilirubin. These factors could potentially lead to dyfunction of the retina, particularly the retinal pigment epithelium (RPE) during AMD.
Uric acid is a metabolic waste product formed from the breakdown of purines, which are compounds found in many foods and also produced insides the body24. While it is primarily known for its role in gout, uric acid also serves several physiological functions in the body. For example, uric acid might play a role in regulating blood pressure. Research suggests that elevated uric acid levels may impair endothelial function and contribute to the development of hypertension25,26. Conversely, some evidence indicates that uric acid may also have vasodilatory effects under certain conditions, potentially influencing blood pressure regulation26. While uric acid has beneficial effects in moderate levels, elevated levels can lead to health problems such as gout, kidney stones, and cardiovascular disease27. Since age was not associated with uric acid levels in the AMD population, our data revealed that uric acid was potentially linked to the presence of AMD without age adjustment. The underlying reason might be related to the role of uric acid in blood pressure regulation, which could affect the status of choroidal vessels beneath RPE and contribute to the neovascularization observed in AMD.
AMD is a complex, multifactorial disease, influenced by both genetic and physicochemicalfactors within the body. In AMD patients, distinct underlying mechanisms might affect the biochemical metabolism, potentially leading to different biochemical correlations compared to the non-AMD population. For example, inflammatory pathways, oxidative stress, and retinal vascular changes in AMD might alter how these biochemical markers interact with age and other variables, like total bilirubin and uric acid. In the non-AMD population, these markers may reflect the typical age-related physiological changes; However, among AMD patients, their levels could be influenced by the retinal inflammation, ischemia, or oxidative stress, potentially disrupting the usual age-related trends. Further exploration in this area is needed to confirm the above associations and investigate the possible mechanisms. In our future research, we plan to use animal models to replicate the condition and assess these two indicators, total bilirubin and uric acid, at different stages of the AMD model to further validate the conclusions of this study. Additionally, based on our clinical work, we would investigate and compare the relationship between these two indicators and AMD progression among patients, which could provide further support for our findings.
There were certain limitations in our study. Firstly, it was a cross-sectional study, and the results only provided a snapshot of biochemical profiles at a specific time point. The impact of biochemical profiles on the risk of AMD may have cumulative effects, highlighting the need for long-term follow-up studies to observe their prolonged impact. Secondly, our study did not explore the underlying mechanisms or conduct further analysis on other biochemical indicators related to AMD. Additionally, although we adjusted for several covariates in our model, these adjustments might not have been sufficient. For example, genetic factors, which could also influence biochemical profiles, were not available in the NHANES database. Lastly, the data used in this study were restricted to the period only from 2005 to 2008. It would be important to incorporate more recent databases to validate our findings in the future.
Altogether, our results revealed that biochemistry profiles did not significantly affect the risk of AMD after adjustment for age. However, total bilirubin and uric acid, which were not in correlations with age in AMD population, might somewhat potentially be related to the presence of AMD. In this context, the innovation and significance of our research suggest that, beyond age-related factors, these two biochemical indicators might play a potential role during AMD. Future longitudinal research is needed to confirm the above relationship and deepen our understanding of the underlying mechanisms.
Data availability
Publicly available datasets were analyzed in our study (https://www.cdc.gov/nchs/nhanes/index.htm).
References
Kozhevnikova, O. Advanced Research in Age-related macular degeneration: special issue. Biomedicines 12(2) (2024).
Gao, H. et al. Injectable anti-inflammatory Supramolecular Nanofiber Hydrogel to promote Anti-VEGF therapy in Age-related macular degeneration treatment. Adv. Mater. 35(2), e2204994 (2023).
Ponnusamy, C., Ayarivan, P., Selvamuthu, P. & Natesan, S. Age-related macular degeneration - therapies and their delivery. Curr. Drug Deliv. 21(5), 683–696 (2024).
Lei, S., Hu, M. & Wei, Z. Identification of systemic biomarkers and potential drug targets for age-related macular degeneration. Front. Aging Neurosci. 16, 1322519 (2024).
Lin, J. B., Halawa, O. A., Husain, D., Miller, J. W. & Vavvas, D. G. Dyslipidemia in age-related macular degeneration. Eye (Lond). 36(2), 312–318 (2022).
Toomey, C. B., Johnson, L. V. & Bowes, R. C. Complement factor H in AMD: bridging genetic associations and pathobiology. Prog Retin Eye Res. 62, 38–57 (2018).
Tawfik, A., Elsherbiny, N. M., Zaidi, Y. & Rajpurohit, P. Homocysteine and Age-Related Central Nervous System diseases: role of inflammation. Int. J. Mol. Sci. 22(12) (2021).
Brantley, M. J. et al. Plasma biomarkers of oxidative stress and genetic variants in age-related macular degeneration. Am. J. Ophthalmol. 153(3), 460–467 (2012).
Yan, W. et al. The association of the serum levels of aldehydes with diabetes-related eye diseases: a cross-sectional population-based study. Environ. Sci. Pollut Res. Int. 30(47), 104713–104725 (2023).
Jiang, B. et al. Association between dietary consumption of fatty acids and age-related macular degeneration in the National Health and Nutrition Examination Survey. Sci. Rep. 14(1), 11016 (2024).
Li, X. et al. Relationship between depressive disorders and biochemical indicators in adult men and women. BMC Psychiatry. 23(1), 49 (2023).
Guymer, R. H. & Campbell, T. G. Age-related macular degeneration. Lancet 401(10386), 1459–1472 (2023).
Curcio, C. A., Kar, D., Owsley, C., Sloan, K. R. & Ach, T. Age-related macular degeneration, a mathematically tractable disease. Invest. Ophthalmol. Vis. Sci. 65(3), 4 (2024).
Kumbhar, P. et al. Treatment avenues for age-related macular degeneration: breakthroughs and bottlenecks. Ageing Res. Rev. 98, 102322 (2024).
Carr, A. C., Lunt, H., Wareham, N. J. & Myint, P. K. Estimating Vitamin C Intake Requirements in diabetes Mellitus: analysis of NHANES 2017–2018 and EPIC-Norfolk cohorts. Antioxid. (Basel) 12(10) (2023).
Vu, G. T., Shakib, S., King, C., Gurupur, V. & Little, B. B. Association between uncontrolled diabetes and periodontal disease in US adults: NHANES 2009–2014. Sci. Rep. 13(1), 16694 (2023).
Lei, S., Liu, Z. & Li, H. Sleep duration and age-related macular degeneration: a cross-sectional and mendelian randomization study. Front. Aging Neurosci. 15, 1247413 (2023).
Nakatochi, M., Sugishita, A., Watanabe, C., Fuchita, E. & Mizuno, M. U-shaped link of health checkup data and need for care using a time-dependent cox regression model with a restricted cubic spline. Sci. Rep. 13(1), 7537 (2023).
Liao, S. et al. Association of aldehyde exposure with cardiovascular disease. Ecotoxicol. Environ. Saf. 206, 111385 (2020).
Puerto-Carranza, E. et al. Total bilirubin and bilirubin-to-triglycerides ratio predict changes in glycated hemoglobin in healthy children. Front. Endocrinol. (Lausanne). 14, 1303597 (2023).
Ma, Y., Du, L., Zhou, S., Bai, L. & Tang, H. Association of direct bilirubin to total bilirubin ratio with 90-day mortality in patients with acute-on-chronic liver failure. Front. Med. (Lausanne). 10, 1286510 (2023).
Cao, Y. X. et al. Circulating total bilirubin and long-term prognosis in patients with previous myocardial infarction. JACC Asia. 3(2), 242–251 (2023).
Lv, J. M. et al. Association between serum total bilirubin and diabetic kidney disease in US diabetic patients. Front. Endocrinol. (Lausanne). 14, 1310003 (2023).
Nakanishi, K. & Morita, H. Uric acid. Int. Heart J. 63(3), 423–425 (2022).
Kuwabara, M. et al. Update in uric acid, hypertension, and cardiovascular diseases. Hypertens. Res. 46(7), 1714–1726 (2023).
Savale, L. et al. Serum and pulmonary uric acid in pulmonary arterial hypertension. Eur. Respir J. 58(2) (2021).
Lee, S. J., Oh, B. K. & Sung, K. C. Uric acid and cardiometabolic diseases. Clin. Hypertens. 26, 13 (2020).
Funding
This study was supported by the Shaanxi Provincial Administration of Traditional Chinese Medicine(NO. SZY-KJCYC-2023-097; Xi’an Science and Technology Project (24YXYJ0091); the General Cultivation Project of Municipal Health Commission in Xi’an, China (2024ms18); The Chinese Medicine Research Project of Municipal Health Commission in Xi’an, China (SZJ202406); Bethune·Lumitin Research Funding for the young and middle-aged Ophthalmologists (BJ-LM2021011J); Joint Funds for the innovation of science and Technology,Fujian province (Grant number:2024Y99020424).
Author information
Authors and Affiliations
Contributions
S.B. and L.Z. wrote the main manuscript text and all the authors prepared Figs. 1, 2 and 3. W.H. provided the funding. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
The studies involving human participants were reviewed and approved by NCHS Institutional Review Committee. The patients/participants provided their written informed consent to participate in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bai, S., Zhang, L., Yan, W. et al. A cross-sectional observational study of the association between biochemistry profiles and the risk of age-related macular degeneration. Sci Rep 15, 8588 (2025). https://doi.org/10.1038/s41598-025-89121-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89121-x
Keywords
This article is cited by
-
Association between age-related macular degeneration and osteoporosis in US
Scientific Reports (2025)