Abstract
The shale reservoir has the characteristics of small rock skeleton particles, large specific surface area, micro-nano pore development and mixed wetting. Previous studies have fully studied the influence of pore size of nano-scale confined space on the critical parameters of fluid phase state, but the influence mechanism of confined space wall wettability on fluid critical temperature/pressure needs to be further studied. Aiming at the problem that the interface effect of micro-nano pores affects the critical properties of n-pentane, the grand canonical Monte Carlo (GCMC) simulation method was used to construct slits with different nano-sizes and different wettability. The phase change of n-pentane in silica nanopores under different temperature and pressure conditions was simulated, and the quantitative relationship between nano-pore size, wettability and critical properties of n-pentane was obtained. Studies have shown that the critical temperature/pressure of n-pentane is lower than that of the bulk phase under restricted conditions. When the nanopore size reaches 11 nm, the critical temperature of n-pentane in slits with different wettability is close to the critical temperature of the bulk phase. When the wetting contact angle of the slit wall is 27.3° and the pore size reaches 30 nm, the critical pressure of n-pentane is close to that of the bulk. However, when the wettability of the wall changes from hydrophilic to lipophilic, and the wetting contact angle is 105.1°, n-pentane with a pore size of 110 nm can break away from the confinement effect of the slit, and its critical pressure is consistent with the bulk space. The stronger the lipophilicity, the greater the van der Waals force between the pore wall and the fluid molecules. The molecules are more likely to be attracted by the wall surface and gather and the fluid is easily undergo phase transition.This study deepens our understanding of oil and gas phase transitions in underground micro nano pores, which helps us better understand the occurrence state of underground oil and gas and facilitates us to adopt more effective development methods.
Similar content being viewed by others
Introduction
The global shale oil reserves are about 11 trillion to 13 trillion tons. It has great potential in development and has become a hot spot in current oil and gas exploration and development. For shale reservoirs, crude oil fluids are ubiquitous in micro-nano pores1,2. When the pore size of oil and gas is smaller than the molecular free path, the assumption of continuous medium fails. At this time, the nano-space will limit the movement of molecules, and a special ' confinement effect ' will appear, which will affect the phase change law of fluid in micro-nano porous media. The special occurrence form of determines the importance of micro-nano pore phase state research in promoting the development of tight oil and gas. At present, there are three main methods to study the phase behavior of hydrocarbons under confined conditions, including theoretical prediction, experiment and molecular simulation3,4,5,6. Tan and Piri modified the equation of state of confined fluid by empirical method, but the main controlling factors of fluid phase behavior in confined space have not been clearly analyzed7. Meanwhile, Zuo et al. utilized an equation of state (EOS) and a modified Young–Laplace equation8. Their study considered the influence of phase behavior and capillary pressure on the phase envelope. They successfully developed a general framework comprising theoretical models and algorithms for predicting the phase envelope of shale gas and shale oil inside nanopores. Feng et al. conducted a study where they extended the Peng-Robinson equation of state model to explore the impact of wettability on the phase behavior and interfacial tension of hydrocarbons confined at the nanoscale9. Their findings revealed that the nanoconfinement effect was not solely dependent on pore size, but also influenced by the wettability of the pore wall. The theoretical approach employed proved effective in predicting the phase behavior of hydrocarbon fluids in nanoconfinement. However, analyzing the microscopic mechanisms underlying the interaction between the pore wall and the fluid remains challenging due to the intricate nature of wall wetting. As laboratory chip technology continues to advance, physical simulation methods are gradually emerging as an invaluable tool for investigating unconfined domains. Luo et al. conducted experimental measurements to determine the bubble point temperature of hydrocarbon mixtures confined within silica nanopores of sizes 4.3 nm and 38.1 nm10. Their findings indicate that the confinement effect is not noteworthy for the larger pore size of 38.1 nm. However, at the smaller pore size of 4.3 nm, there is a significant deviation of up to 15 K. Similarly, Lowry et al. investigated the confined phase behavior of propane and n-butane in both surface-modified and unmodified silica using experimental methods11. Their research revealed that the effect of surface modification on the phase state of alkanes cannot be overlooked. Furthermore, Qiao et al. examined the critical point of ethylene and carbon dioxide in MCM-41 and studied the significant influence of capillary pressure, temperature, and pore size on the adsorption capacity and relative pressure during fluid phase transition12.
These studies have demonstrated that the critical temperatures of confined gases are significantly lower than their respective bulk critical temperatures. Zhuet al. specifically examined the phase behavior of methane in numbers and observed that the interaction between methane and kerogen primarily involves van der Waals forces13. This suggests that the adsorption of fluids onto kerogen is a form of physical adsorption, and the van der Waals forces between the fluid and minerals might influence the phase transition characteristics. Both theoretical equation prediction methods and experimental approaches have been employed to investigate the phase behavior of fluids within nano-scale spaces. Nevertheless, these two approaches have not provided a comprehensive understanding of the microscopic mechanisms governing the interactions between fluids and mineral surfaces. Additionally, they have not adequately quantified the influence of mineral surface wettability on fluid phase transitions. Hence, the utilization of molecular simulation methods becomes crucial in investigating the alterations in critical properties of alkanes within micro-nano pores exhibiting varying degrees of wettability, as well as unraveling the underlying microscopic mechanisms at play.
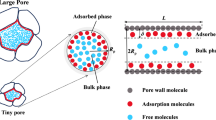
As computers continue to evolve, molecular simulation has become a prevalent theoretical research method for investigating fluid phase behavior in nanopores14,15,16,17,18. Piri et al. employed molecular simulation techniques to explore the impact of surface wettability on the phase behavior of propane19. Their findings revealed that in nanopores with a high degree of oil-wetness, the fluid exhibited a more pronounced deviation from its critical behavior. Falk et al. conducted simulations to investigate the phase behavior of methane and carbon dioxide in illite slits using the Grand Canonical Monte Carlo (GCMC) method20. They found that van der Waals forces play a crucial role in the interactions between solid and gas molecules. Furthermore, they observed that the confinement effect has a more pronounced influence on the thermodynamic and kinetic properties of the adsorbed phase when larger molecules are involved. Hence, considering the strength of the interaction between the surface-modifying molecules and the pore wall, it becomes crucial to examine its influence on the critical properties of the fluid. Gubbins et al. addressed this by introducing wetting parameters to simulate these interactions21. In a related study, Muñoz et al. investigated the effects of pore size and aminopropyl surface modification22. The findings highlighted that surface chemistry significantly influenced the loading and desorption processes, with pore size being of lesser significance. Moreover, Collell et al. conducted a molecular simulation study on the phase behavior of methane/ethane mixtures within artificially created spherical pores embedded in a dense kerogen matrix15. As shown in Fig. 1, the fluid phase in nanopores is mainly affected by temperature, pressure, pore size, wettability, and capillary force. Previous studies have analyzed the influencing factors alone and have not quantitatively characterized the influencing mechanism by combining all factors. This study comprehensively considers the effects of temperature, pressure, pore size, wettability, and capillary force, and quantitatively characterizes the influence of various factors on the phase of n-pentane under limited conditions. The researchers noted the impact of nanopore size confinement, leading to deviations from classical equations of state. Prior molecular simulation studies on hydrocarbon fluids within micro-nano pores primarily concentrated on minerals such as kerogen23,24,25, montmorillonite26,27, illite17, kaolinite28, and calcite29. However, quartz, being a significant constituent of shale minerals30, holds considerable research significance. While earlier investigations mainly focused on small molecules like methane and ethane31,32,33,34,35,36, relatively larger molecules received comparatively less attention. Consequently, as a key component of shale gas, n-pentane has significant research value. Hence, this study utilizes the grand canonical Monte Carlo simulation method to examine the impact of mineral surface wettability on the phase transition behavior of n-pentane within silica nanopores, taking into account various nanopore sizes, temperature levels, and pressure conditions. Furthermore, a detailed analysis of the underlying microscopic mechanisms is presented. Building upon the simulation outcomes, the quantitative relationship between nanopore size and wettability affecting the critical temperature and pressure of n-pentane was established. The limitation of this method is that it cannot be simulated for larger pore sizes. We use fitting methods to predict the changes in the phase state of n-pentane in larger pore sizes, which may result in some errors. The in-depth study of the phase state of oil and gas in micro nano pores can provide reliable theoretical basis and technical support for oil and gas exploration and development, reduce development costs, and improve the efficiency of oil and gas exploration and development.
Factors affecting fluid phase transition in confined spaces.
Methodology
Modelling
In this study, the adsorption law and phase change of n-pentane in slits with different pore sizes (2, 3, 5, 7, 9, 11 nm) and silica nanoslits modified with different functional groups (100%-OH, 50%-OH, 25%-OH, 100%-CH3, 100%-(CH2)4CH3) and unmodified were studied. We first took the 5 nm slit as an example. We selected α-quartz single cell as the base model unit structure. The lattice parameters were as follows: a = b = 4.913 Å, c = 5.4052 Å, α = β = 90°, and γ = 120°. The (1 0 0) surface was cleaved and supercell 10 × 10 was used as the quartz mineral floor to build the slit model. The surface functional groups of mineral models with different wettability in this work are relatively dense, and the influence of mineral surface roughness on wettability is relatively small, which can be regarded as a smooth model37. Adjust the slit spacing to 5 nm, and finally get the slit model modified by each group as shown in Fig. 2. The contact angle refers to the angle(θ) between the tangent line of the gas–liquid interface and the solid–liquid boundary line at the intersection of gas, liquid, and solid phases. The larger the wetting contact angle, the stronger the lipophilicity of the surface wall. The wetting contact angle of the slit model modified by each group is referred to this literature38.
The initial configuration of each group modified slit at 5 nm (Red circle: Oxygen atom in SiO2, Yellow circle: Silicon atom, Blue circle: Oxygen atom in -OH, Violet circle: Hydrogen atom in -OH, Black circle: Carbon atom, Green circle: Hydrogen atom in –CH3).
Potential models
In this study, the NERD39 molecular model of n-pentane, which defines the non bonding interactions of n-pentane as including non bonding interactions between different atoms of different molecules and between atoms with a distance greater than 3 covalent bonds in the same molecule. The Lennard–Jones parameters and charge parameters are shown in Table 1.
The corresponding parameters can be determined by Lorentz-Berthelot combining rule, as shown in the following formula40. M is the molar mass of the molecular cluster. Q is the charge of the molecular cluster. The L-J potential energy parameters of silica substrate atoms are shown in Table 2:
Non bonding interactions refer to non covalent bonding interactions between different molecules or atoms within a molecule, including Coulomb interactions and van der Waals interactions. Van der Waals interactions describe the repulsive or attractive interactions between atoms caused by non covalent bonds and non electrostatic forces. The intermolecular interaction between SiO2 nanoslits and n-pentane is represented by the Lennard–Jones 12-6 potential as:
Where VLJ(rij) is the van der Waals interaction between atom i and atom j; rij is the distance between two virtual atoms in molecule i and molecule j; σij is the scale parameter of L-J potential energy between two virtual atoms in i and j molecules. εij is the energy parameter of L-J potential energy between two virtual atoms in i and j molecules.
Simulation details
In this study, Monte Carlo simulation method is used to study the phase change of n-pentane in different wettability and pore size slits (confined phase). Periodic boundary conditions(PBC) are used in the X, Y, and Z directions. The cut-off radius of the L-J force between the particles is 1.2 nm. Nano slits were fixed in the simulation to reduce computational complexity without affecting the calculation results. The total number of simulation steps is 2 million steps to obtain the results after full simulation equilibrium. And the integration step of this simulation is 1 fs. The position coordinate information of the particles is output every 2000 steps, and the simulation data is calculated by LAMMPS software.
Results and discussions
As the main basis for calculating other physical property data, the critical temperature and pressure of fluid are important parameters in the development process of oil and gas fields. In order to solve the problem that the confinement effect affects the critical properties of n-pentane, a nano-slit model with different wettability (100%-OH, 50%-OH, 25%-OH, 100%-CH3, 100%-(CH2)4CH3) and pore size (2, 3, 5, 7, 9, 11 nm) was established. The grand canonical Monte Carlo (GCMC) simulation method was used to simulate the adsorption law and phase change of n-pentane in silica nanopores under different temperature (400-460 K) and pressure (0–3.6 MPa) conditions. The quantitative relationship between wettability, nano-pore size and critical properties of n-pentane was obtained, and its microscopic mechanism was analyzed.
The influence of different wettability on the phase transition law of n-pentane
To confirm the effect of wettability on the phase transition of n-pentane, the isothermal adsorption-density plot of n-pentane in a 5 nm slit modified with methyl and hydroxyl groups was first simulated at a temperature of 420 K, as shown in Fig. 3. The density calculation is based on Formula (3). The adsorption amount of n-pentane in the methyl-modified slit is always higher than that in the hydroxyl-modified slit, indicating that n-pentane is more easily adsorbed on the lipophilic wall. This is because the adsorption between alkanes and minerals mainly relies on van der Waals forces, just like the principle of similar solubility. The stronger the lipophilicity of minerals, the greater the attraction to alkanes, resulting in a higher adsorption capacity. The adsorption capacity of n-pentane increases with the increase of pressure, and the jump of adsorption capacity occurs at a certain point. At the same temperature, the n-pentane phase transition pressure in the methyl-modified slit is lower, because the interaction between the lipophilic wall and n-pentane is stronger, which has a greater impact on the critical properties of n-pentane, so n-pentane has a gas–liquid phase transition at a lower pressure. In order to further study the law between wettability and n-pentane phase transition, the isothermal adsorption-density plot of n-pentane in slits with different wettability were calculated. The wettability of mineral surface was quantified by wetting contact angle, and the law of wettability, n-pentane adsorption and phase transition was obtained as shown in Fig. 4. It is not difficult to see that with the increase of lipophilicity, the adsorption amount of n-pentane also increases. The phase transition pressure of n-pentane in hydrophilic slits is higher than that in non-hydrophilic slits at the same temperature. As hydrophilic slits or non-hydrophilic slits, wettability has little effect on n-pentane phase transition. However, in general, with the increase of the lipophilicity of the slit surface, the phase transition pressure of n-pentane at the same temperature gradually decreases. This phenomenon will be more obvious when the wettability of the mineral surface changes from lipophilic to hydrophilic.
where N is the adsorption quantity; M is the molar mass of n-pentan, g/mol; NA is avogadro constant; V is the pore volume of slit, g/cm3.
Adsorption-density plot of hydroxyl and methyl modified n-pentane.
Adsorption-density plot of n-pentane modified by different wettability.
The isothermal adsorption lines at different temperatures were calculated, and the P–T phase diagram of n-pentane in slits with different wettability was obtained by using the isothermal adsorption lines as shown in Fig. 5.The bulk properties of n-pentane are obtained from the National Institute of Standards and Technology website41,42. As shown in Table 3, the critical temperature of n-pentane is 469.7 K, the critical pressure is 3.396 MPa, and the critical density is 0.236 g/cm3. The wettability has a significant effect on the phase change of n-pentane. The P–T phase diagram of n-pentane in the hydrophilic slit is above, but it is smaller than the bulk phase condition. Because the interaction force between the hydrophilic wall and n-pentane is small, the phase transition temperature/pressure of n-pentane is higher than that of the lipophilic wall. With the increase of lipophilicity, the P–T phase diagram of n-pentane shifts downward, which proves that wettability has a great influence on the critical pressure of n-pentane in nanopores, and has little effect on the critical temperature. With the increase of lipophilicity, the critical pressure of n-pentane gradually decreases.
P–T phase diagram of n-pentane in 5 nm slits with different wettability.
The critical properties of n-pentane in slits with different wettability vary with pore size
The critical properties of n-pentane in nano-slits with different wettability are calculated as shown in Fig. 6. The calculation method for critical properties refers to the method provided in this literature43. The critical temperature changes with the nano-aperture and the wettability of the slit wall are shown in Fig. 6(a). It is not difficult to see that the critical temperature increases with the increase of the nano-aperture, but the rate of increase in critical temperature decreases with the increase of pore size. When the nano-aperture reaches 11 nm, the restriction effect of the slit on the fluid is obviously weakened. At this point, the critical temperature of n-pentane can be considered as its volume critical temperature. With the increase of the lipophilicity of the slit wall, the critical temperature of n-pentane in different nano-apertures basically shows an upward trend, but the wettability has little effect on the critical temperature of n-pentane. When the nano-aperture reaches 11 nm, the critical temperature of n-pentane in different wettability nano-slits can be close to the critical temperature of the bulk phase.
The change of critical properties of n-pentane with pore size in slits with different wettability and pore size.
In order to describe the influence of wettability on the phase transition of confined fluid more conveniently, a new physical quantity, the critical pore size of phase transition, is introduced in this study. Which is the critical pore size when the critical temperature/pressure of confined fluid reaches the bulk phase. Due to the significant computational complexity caused by the increase in pore size, the critical properties of n-pentane in pores larger than 11 nm are fitted by a formula. The variation of critical pressure with wettability and pore size is different from that of critical temperature. The variation of the critical pressure with the nanopore size and the wettability of the slit wall is calculated as shown in Fig. 6b. The critical pressure increases with the increase of the pore size, but the critical pore size of the n-pentane critical pressure in the slits with different wettability is different. When the wetting contact angle of the slit wall is 27.3° and the aperture reaches 30 nm, the critical properties of n-pentane in the slit are the same as those in the bulk. When the wetting contact angle of the wall reaches 105.1°, the wettability of the wall has changed from hydrophilic to lipophilic. Under the restricted conditions, n-pentane needs to reach 110 nm in order to break away from the restriction of the slit and achieve the bulk phase properties. With the increase of the lipophilicity of the slit wall, the interaction between the pore wall and the fluid molecules increases. The greater the critical pressure of n-pentane is affected, the larger the nanopore size required to reach the bulk phase condition. The wettability of the slit wall affects the interaction force between the fluid and the wall, and the interaction force affects the phase behavior of the fluid molecules and leads to the change of the fluid properties. These simulation results are consistent with the previous assumptions. However, when the pores are large enough, the effect is negligible. Therefore, for the slit with strong lipophilicity, the interaction force between the slit and the wall surface is greater, and larger pores are needed to be regarded as bulk fluid.
The change of the critical pore size of n-pentane phase transition with the wettability of the slit is shown in Fig. 7. For slits exceeding 11 nm, regardless of the wettability, the influence of the confined phase on the critical temperature of n-pentane can be ignored and approximated to the bulk phase treatment. However, with the increase of wall lipophilicity, the critical pore size at which the critical pressure reaches the bulk phase has been on the rise. It can be seen that the confinement effect in nanopores depends not only on the pore size but also on the wettability. Combined with the simulation results, the influence of pore size on n-pentane phase transition under confined conditions is greater than that of mineral surface wettability. However, for real shale reservoirs, if the reservoirs are water wet and oil wet respectively, the change of wettability may lead to a great change in oil and gas phase.
Variation of critical pore size of n-pentane phase transition with wettability of slits.
The wettability has little effect on the critical temperature. As can be seen from Fig. 6(a), the critical temperature difference of pentane with different wettability is basically less than 10 K, and the critical pore size of pentane critical temperature change under each wettability condition is consistent. Therefore, a line is selected for fitting. The fitting of the critical temperature of n-pentane in the original silica slit with the change of pore size is shown in Fig. 8. The change of the critical temperature with the pore size is shown in Formula (4). The fitted formula can ensure that the difference of the critical temperature of n-pentane under different wettability conditions is less than 5 K, which can be used to calculate the change of the critical temperature of n-pentane with the pore size under different wettability conditions.
where Tc is critical temperature, K; x is aperture, nm.
Fitting diagram of critical temperature of n-pentane with different wettability changing with pore size.
The wettability has a great influence on the critical pressure, so the wetting degree must be taken into account in the critical pressure fitting formula. Firstly, the variation of the critical pressure of pentane in the original silica slit with the pore size is fitted as shown in Fig. 9. Where r is the nano pore size. When the wetting contact angle is 58.5°, the fitting formula of the critical pressure of pentane with the change of pore size is as follows (5):
Fitting diagram of the critical temperature of pentane in the original silica slit with the change of pore size.
According to Fig. 9, the relationship between the critical pore size of n-pentane phase transition and the wettability of the slit is shown in Formula (6):
where θ is the wetting angle.
According to Formula (5), when the wetting angle is 58.5°, the relationship between r and critical pressure can be obtained as Formula (7):
Because when the wetting angle is 58.5°, the critical pore size of n-pentane phase transition is calculated to be 73 nm, it can be obtained that:
Therefore, the variation of the critical pressure of pentane with pore size and wettability can be obtained as shown in Eq. (9):
where Pc is critical pressure, MPa; r is aperture, nm; rc is critical pore size, nm; θ is wetting contact angle, °.
The formula ensures that the critical pore size of pentane phase transition remains unchanged under various wettability conditions. Other phase transition pressure points are equivalent to adding a coefficient according to the critical pore size parameters. The calculated value has a slight error with the simulated value, but it is also within the acceptable range. According to the formula, the comparison of the critical pressure of different wettability with the simulated value is shown in Fig. 10. It is not difficult to see that the fitting value is in good agreement with the simulated value under the condition that the critical pore size is constant, and the formula can be used to calculate the change of the critical pressure of pentane with the pore size under different wettability conditions.
Comparison of calculated and simulated critical pressure of pentane with different wettability.
Analysis of the microscopic mechanism of wettability affecting pentane phase transition
The confinement effect leads to the abnormal phase behavior of the fluid in the nanopore. The previous calculations show that in addition to the nano-pore size, the shift of the critical properties is also affected by the wettability of the mineral surface. In this section, the microscopic interaction between the pore wall and the fluid is analyzed in depth to further explore the effect of confinement on the n-pentane phase transition process and the microscopic action principle.
Shale is abundant in nanoscale pores, and the confinement effect is highly significant. The interaction between the wall and fluid, also known as wall force or wall/fluid interaction, plays a crucial role in the transfer of momentum and energy44. In the actual nanopores of shale gas reservoirs, the attractive effect of the pore wall surface restricts the free movement of gas molecules, while the wall force (wall potential field) also influences the trajectory of these molecules45. Please refer to Fig. 11 for visual representation. The impact of the wall force on the molecular free path primarily manifests in the direction and trajectory of molecular motion: as the wall force increases, the angle between the molecular motion direction and the wall normal decreases. When the wall force surpasses the intermolecular force within the gas, the gas molecules within the pores tend to be attracted towards the wall. Consequently, the gas molecules tend to collide with the wall along a shorter path and at a smaller angle, thereby affecting the fluid properties. It is not difficult to see that the greater the interaction force between the mineral surface and the fluid, the more severe the displacement of fluid molecules, and thus the greater the impact on their physical properties.
Schematic diagram of collision between gas molecules and pore wall under the influence of wall potential field.
The density distribution map of pentane in pores can intuitively reflect the effect of wall action on fluid adsorption. In order to better understand the influence of different wettability slits on the pentane adsorption process, the adsorption density distribution of n-pentane in different wettability nanopores under 420 K and 1 MPa conditions was calculated as shown in the Fig. 12. The total simulation steps are two million steps. We obtained the adsorption simulation data by averaging the data between 1.5 million and 2 million steps. At this point, the system has reached stability, and the average processed data is smoother. The distribution of pentane adsorption density inside the slit is mainly divided into the adsorbed state on both sides of the slit and the free state in the middle. Firstly, it can be observed that the density of adsorbed pentane gradually increases with the increase of slit lipophilicity. The reason is that the interaction force between the lipophilic wall and pentane is greater, which not only increases the total amount of adsorbed pentane, but also greatly increases the total amount of adsorbed pentane molecules in the slit. Secondly, the variation pattern of free state pentane is the same as that of adsorbed state. With the increase of lipophilicity, the maximum can increase to 0.55 g/cm3, and the minimum hydrophilic hydroxyl modified wall is 0.4 g/cm3. Due to the confinement effect of pentane in the 5 nm slit, the density of intermediate free states is greater than that of bulk pentane.
Changes in local density of pentane adsorption with wettability.
The diffusion behavior of n-pentane in different wetting gaps at a temperature of 420 K was studied, as shown in Fig. 13. The diffusion coefficient increases with the increase of slit lipophilicity and decreases with the increase of pressure. Subsequently, the diffusion coefficient of pentane decreases uniformly with increasing pressure. Wettability also has a significant impact on the diffusion ability of pentane. As the lipophilicity of the slit increases, the diffusion ability of pentane gradually weakens, indicating that pentane molecules have stronger interaction energy with the lipophilic surface. This interaction energy limits the action of pentane molecules on the quartz slit wall, increasing with the increase of lipophilicity.
Variation of pentane diffusion coefficient with pressure in slits with different wettability.
To further investigate the impact of the wettability of the slit surface on the phase transition process of n-pentane and its underlying microscopic mechanisms, the relationship between the van der Waals force between the slit and n-pentane with varying wettability is calculated at 420 K and 1.5 MPa, as depicted in Fig. 14. It is evident that the van der Waals force between n-pentane and the slit progressively increases as the lipophilicity of the slit wall intensifies. This observation further confirms that the interaction between the pore wall and fluid molecules escalates with greater lipophilicity of the slit wall. Consequently, the critical pressure of n-pentane is subject to greater influence, which means a larger nanopore size is required to reach the bulk condition. The van der Waals force fluctuation between the hydrophilic slit wall and n-pentane is relatively pronounced due to the phase transition of n-pentane under the specified temperature and pressure conditions. The alternating changes in n-pentane within the system result in a significant energy range. Conversely, the van der Waals force between n-pentane and the lipophilic slit wall remains relatively stable as n-pentane has already undergone phase transition, further affirming the increased propensity for phase transition within the lipophilic slit. The joint atom model is employed in this study, and the van der Waals force, serving as the primary force between n-pentane and the slit, significantly affects the phase transition of n-pentane within the nanopore. In nano-slit reservoirs, a larger wetting contact angle and stronger lipophilicity correspond to greater interaction between the pore wall and fluid molecules, facilitating fluid phase transition and requiring a larger critical pore size for the transition to occur. This understanding can enhance our comprehension of the subsurface occurrence of oil and gas.
The relationship between van der Waals force and wettability between slit and n-pentane.
Conclusion
In this study, there are many complex interface effects in shale reservoirs, which lead to the complex phase change law of fluid in micro-nano pore media. Based on molecular simulation, the wetting mechanism of micro-nano pore surface and the coupling effect of complex interface effects in micro-nano pores are systematically studied. The characteristics of fluid phase change and its microscopic action principle, the main conclusions are as follows:
-
(1)
The confinement effect in nanopores is affected by both pore size and mineral surface wettability. Under this confinement condition, the critical temperature/pressure of n-pentane is lower than that of bulk phase.
-
(2)
The critical temperature is less affected by wettability. When the nanopore size reaches 11 nm, the critical temperature of n-pentane in the confined space is almost the same as that in the bulk phase. At this time, the influence of various interface effects on the critical temperature of the fluid under confined conditions can be ignored.
-
(3)
The critical pressure is greatly affected by wettability. When the wetting contact angle of the slit wall is 27.3° and the pore size reaches 30 nm, the critical pressure of n-pentane is close to the bulk phase. However, when the wettability of the wall changes from hydrophilic to lipophilic, and the wetting contact angle is 105.1°, n-pentane under confined conditions needs to reach 110 nm to break away from the confinement effect of the slit and reach the bulk critical pressure.
-
(4)
The main factor affecting the phase state of n-pentane in the slit is the van der Waals force between them, which is attractive. The stronger the lipophilicity is, the greater the van der Waals force between the pore wall and the fluid molecules is. The molecules are more easily attracted by the wall surface and gather near the wall surface, which is more likely to change from the gas phase to the liquid phase, and the larger the critical pore size of the phase transition is.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Nagarajan, N. R., Honarpour, M. M. & Arasteh, F. The critical role of rock and fluid in unconventional shale reservoir performance. In Unconventional Resources Technology Conference 1230–1247 (Society of Exploration Geophysicists, American Association of Petroleum Geologists, Society of Petroleum Engineers, 2013).
Chalmers, G. R., Bustin, R. M. & Power, I. M. Characterization of gas shale pore systems by porosimetry, pycnometry, surface area, and field emission scanning electron microscopy/transmission electron microscopy image analyses: Examples from the Barnett, Woodford, Haynesville, Marcellus, and Doig units. AAPG Bull. 96(6), 1099–1119 (2012).
Liu, X. & Zhang, D. A review of phase behavior simulation of hydrocarbons in confined space: Implications for shale oil and shale gas. J. Nat. Gas Sci. Eng. 68, 102901 (2019).
Feng, D. et al. Water adsorption and its impact on the pore structure characteristics of shale clay. Appl. Clay Sci. 155, 126–138 (2018).
Wang, T. et al. Molecular simulation of CO2/CH4 competitive adsorption on shale kerogen for CO2 sequestration and enhanced gas recovery. J. Phys. Chem. C 122(30), 17009–17018 (2018).
Song, Y. et al. Phase behavior of hydrocarbon mixture in Shale nanopores considering the effect of adsorption and its induced critical shifts. Ind. Eng. Chem. Res. 59(17), 8374–8382 (2020).
Tan, S. P. & Piri, M. Equation-of-state modeling of associating-fluids phase equilibria in nanopores. Fluid Phase Equilib. 405, 157–166 (2015).
Zuo, J. Y. et al. Impact of capillary pressure and nanopore confinement on phase behaviors of shale gas and oil. Energy Fuels 32(4), 4705–4714 (2018).
Feng, D. et al. Wettability effects on phase behavior and interfacial tension in shale nanopores. Fuel 290, 119983 (2021).
Luo, S., Nasrabadi, H. & Lutkenhaus, J. L. Effect of confinement on the bubble points of hydrocarbons in nanoporous media. AIChE J. 62(5), 1772–1780 (2016).
Lowry, E. & Piri, M. Effect of surface chemistry on confined phase behavior in nanoporous media: an experimental and molecular modeling study. Langmuir 34(32), 9349–9358 (2018).
Qiao, S. Z., Bhatia, S. K. & Nicholson, D. Study of hexane adsorption in nanoporous MCM-41 silica. Langmuir 20(2), 389–395 (2003).
Zhu, Y. Q., Su, H., Jing, Y., Guo, J. & Tang, J. Methane adsorption on the surface of a model of shale: a density functional theory study. Appl. Surf. Sci. 387, 379–384 (2016).
Zhao, J., Wang, Z. & Guo, P. Microscopic simulation of methane adsorption in organic matter. Ind. Eng. Chem. Res. 58(8), 3523–3530 (2019).
Collell, J. et al. Molecular simulation and modelisation of methane/ethane mixtures adsorption onto a microporous molecular model of kerogen under typical reservoir conditions. Microporous Mesoporous Mater. 197, 194–203 (2014).
Rogacka, J. et al. High-throughput screening of metal–organic frameworks for CO2 and CH4 separation in the presence of water. Chem. Eng. J. 403, 126392 (2021).
Chen, G. et al. Research of CO2 and N2 adsorption behavior in K-illite slit pores by GCMC method. Sci. Rep. 6(1), 1–10 (2016).
Zhang, L., Liu, C. & Li, Q. Molecular simulations of competitive adsorption behavior between CH4-C2H6 in K-illite clay at supercritical conditions. Fuel 260, 116358 (2020).
Lowry, E. & Piri, M. Effect of surface chemistry on confined phase behavior in nanoporous media: an experimental and molecular modeling study. Langmuir 34, 9349–9358 (2018).
Falk, K. et al. Effect of chain length and pore accessibility on alkane adsorption in kerogen. Energy Fuels 29(12), 7889–7896 (2015).
Gubbins, K. E., Long, Y. & Asliwinska-Bartkowiak, M. Thermodynamics of confined nano-phases. J. Chem. Thermodyn. 74, 169–183 (2014).
Muñoz, B. et al. MCM-41 organic modification as drug delivery rate regulator. Chem. Mater. 15, 500–503 (2003).
Liu, Y. et al. Molecular simulation of CO2 adsorption in micro-and mesoporous carbons with surface heterogeneity. Int. J. Coal Geol. 104, 83–95 (2012).
Xiaoqi, W. et al. Molecular simulation of CO2/CH4 competitive adsorption in organic matter pores in shale under certain geological conditions. Pet. Explor. Dev. 43(5), 841–848 (2016).
Zhang, T. et al. Molecular simulation of shale gas adsorption in organic-matter nanopore. J. Nat. Gas Geosci. 2(5–6), 323–332 (2017).
Chen, G. et al. Keys to linking GCMC simulations and shale gas adsorption experiments. Fuel 199, 14–21 (2017).
Jian, X., Xiangjun, L. & Lixi, L. Molecular simulation on the adsorption behaviors of methane in montmorillonite slit pores. Acta Pet. Sin. 37(8), 1021 (2016).
Xiong, J. et al. Adsorption behavior of methane on kaolinite. Ind. Eng. Chem. Res. 56(21), 6229–6238 (2017).
Wang S, Feng Q, Javadpour F, et al. Multiscale modeling of shale apparent permeability: an integrated study of molecular dynamics and pore network model[C]//SPE Annual Technical Conference and Exhibition. OnePetro. (2017).
Ross, D. J. K. & Bustin, R. M. Characterizing the shale gas resource potential of Devonian-Mississippian strata in the Western Canada sedimentary basin: Application of an integrated formation evaluation. AAPG Bull. 92(1), 87–125 (2008).
Zhao, J. et al. Molecular level investigation of methane and carbon dioxide adsorption on SiO2 surface. Comput. Mater. Sci. 168, 213–220 (2019).
Chen, H. et al. The synergistic effects of surface functional groups and pore sizes on CO2 adsorption by GCMC and DFT simulations. Chem. Eng. J. 415, 128824 (2021).
Yang, Z. et al. Construction of Buertai coal macromolecular model and GCMC simulation of methane adsorption in micropores. ACS Omega 6(17), 11173–11182 (2021).
Zeng, K. et al. Methane adsorption capacity measurement in shale matrix nanopores at high pressure by low-field NMR and molecular simulation. Chem. Eng. J. 430, 133151 (2022).
Zhang, D. et al. Molecular simulation of methane adsorption in nanoscale rough slits. J. Nat. Gas Sci. Eng. 102, 104608 (2022).
Dai, X. et al. Methane/carbon dioxide adsorption and diffusion performances at different mineral compositions and buried depth conditions. Energy Fuels 35(19), 15567–15578 (2021).
Liu, J. et al. A new wetting mechanism based upon triple contact line pinning. Langmuir 27(1), 196–200 (2011).
Wang, Z. et al. Molecular dynamics simulation for quantitative characterization of wettability transition on silica surface. J. Mater. Res. Technol. 19, 4371–4380 (2022).
Nath, S. K., Escobedo, F. A. & de Pablo, J. J. On the simulation of vapor–liquid equilibria for alkanes. J. Chem. Phys. 108(23), 9905–9911 (1998).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: An N⋅log (N) method for Ewald sums in large systems. J. Chem. Phys. 98(12), 10089–10092 (1993).
Alexandrov, I., Gerasimov, A. & Grigorev, B. Generalized fundamental equation of state for the normal alkanes. Int. J. Thermophys. 34(10), 1865–1905 (2013).
Scholz, C. W. et al. Speeds of sound in n-pentane at temperatures from 233.50 to 473.15 K at pressures up to 390 MPa. J. Chem. Eng. Data 65(7), 3679–3689 (2020).
Yu, C. et al. Vapor-liquid phase equilibrium of n-pentane in quartz nanopores by grand canonical Monte Carlo calculation. J. Mol. Liq. 365, 120075 (2022).
Li, J. et al. Thickness and stability of water film confined inside nanoslits and nanocapillaries of shale and clay. Int. J. Coal Geol. 179, 253–268 (2017).
Shindo, Y. et al. Gas diffusion in microporous media in knudsen’s regime. J. Chem. Eng. Japan 16(2), 120–126 (1983).
Acknowledgements
This work was supported by National Natural Science Foundation of China (52074236, 51836003).
Author information
Authors and Affiliations
Contributions
Cao Yu and Zhouhua Wang wrote the main manuscript text. Jianfei Zhao, Kunpeng Fan, Ping Guo and Hongxian Kuang were responsible for method research. Huang Liu, Hanmin Tua, Yisheng Hu and Zhao Su were responsible for data simulation verification, and all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Yu, C., Zhao, J. et al. The phase transition characteristics of n-pentane in silica slits with different wettability by Monte Carlo method. Sci Rep 15, 7332 (2025). https://doi.org/10.1038/s41598-025-89207-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89207-6