Abstract
To evaluate the effect of recombinant human thrombopoietin (rhTPO) on platelet engraftment after autologous stem cell transplantation (ASCT) in newly diagnosed multiple myeloma (NDMM) patients. Clinical data of 40 NDMM patients who underwent high-dose melphalan conditioning and ASCT were analyzed, with 26 receiving rhTPO and 14 in the control group. The primary study endpoint was the median time to platelet engraftment. The secondary study endpoints included the median time to neutrophil engraftment, number of blood cells transfused, treatment-related adverse events, long-term hematopoietic reconstitution, and economic cost-effectiveness. The median time to neutrophil engraftment was 10.92 and 11.14 days in the rhTPO and control groups, respectively (p = 0.405). The median time to platelet engraftment (11.5 vs. 11.36 days, p = 0.776), median number of erythrocyte infusions (0.23 vs. 0 units, p = 0.326), and median number of platelet infusions (1.615 vs. 1.5 units, p = 0.721) did not significantly differ between the groups. The median hospitalization duration was 27.08 and 25.07 days in the rhTPO and control groups, respectively (p = 0.193). The median hospitalization cost was RMB 73,846 and RMB 54,136 in the rhTPO and control groups, respectively (p < 0.001). At 180 days post-transplantation, the neutrophil count, hemoglobin level, and platelet count did not significantly differ between the groups. Administering rhTPO after ASCT for NDMM did not accelerate platelet engraftment or reduce platelet transfusions, but increased hospitalization costs.
Similar content being viewed by others
Introduction
The prevailing therapeutic approach for newly diagnosed multiple myeloma (NDMM) patients eligible for transplantation is induction therapy, followed by high-dose chemotherapy and autologous stem cell transplantation (ASCT) as a consolidation treatment1. Although the incidence of early treatment-related mortality (TRM) following ASCT in NDMM patients is lower than 5%, it warrants careful consideration2,3. Conditioning regimens prior to ASCT commonly employ melphalan at a high dose of 200 mg/m2 (MEL-200), resulting in substantial neutropenia and thrombocytopenia. These hematologic complications increase the risk of infections and hemorrhages, thereby contributing to early TRM3. Delayed platelet engraftment is a critical complication associated with both autologous and allogeneic hematopoietic stem cell transplantation (HSCT). Enhancement of the rate of platelet engraftment is of paramount importance to mitigate the incidence of severe hemorrhagic events, which contribute significantly to TRM. Recent studies provided limited evidence suggesting that recombinant human thrombopoietin (rhTPO) enhances platelet engraftment following ASCT in patients with NDMM and aggressive lymphoma4,5.
Thrombopoietin (TPO) is a hematopoietic growth factor that plays a critical role in stimulating all phases of megakaryopoiesis6. rhTPO retains amino acid sequence identity with native TPO and facilitates platelet recovery in conditions such as primary immune thrombocytopenia, intermediate-to-high-risk myelodysplastic syndromes (MDS), acute myeloid leukemia (AML), aplastic anemia (AA), and chemotherapy-induced thrombocytopenia in patients with solid tumors7,8,9,10. The safety profile of rhTPO appears to be favorable, with no reported incidences of liver or kidney function abnormalities or thrombosis, including among pregnant women11. However, the efficacy of rhTPO in promoting platelet engraftment following HSCT remains a subject of debate. Two phase I clinical studies demonstrated that rhTPO neither increases the bone marrow megakaryocyte content nor enhances the platelet recovery time following HSCT12,13. Conversely, numerous studies demonstrated that rhTPO facilitates platelet engraftment following cord blood and haploidentical HSCT14,15,16,17. Consequently, it is imperative to assess the impact of rhTPO on platelet engraftment following ASCT for NDMM in a real-world study.
Materials and methods
Patients
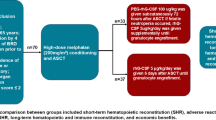
This study performed a retrospective analysis at a single center, encompassing 40 patients with NDMM who underwent ASCT between January 2018 and June 2024 at the Department of Hematology and Oncology, China-Japan Union Hospital of Jilin University. Patients were stratified according to administration of rhTPO following ASCT, resulting in 26 patients in the rhTPO group from January 2020 to June 2024 and 14 patients in the control group from January 2018 to December 2019. Before undergoing ASCT, all patients provided written informed consent, permitting the use of their data for scientific research. Patients in the treatment group signed an informed consent for the use of rhTPO for supra-indications. The study was approved by the Ethics Committee of China-Japan Union Hospital of Jilin University. All methods were performed in accordance with the relevant guidelines and regulations. Patient data were retrieved from the hospital’s electronic health record management system. Participants were required to meet the following inclusion criteria: a diagnosis of NDMM; an age of 65 years or younger; completion of four cycles of induction therapy with the VRD (bortezomib, lenalidomide, and dexamethasone) regimen to achieve a complete response, very good partial response, or partial response prior to transplantation; absence of relapse or refractory disease; adequate organ function (defined as a left ventricular ejection fraction of ≥ 40%, an exertional expiratory volume in 1 s or exertional lung volume in 1 s of ≥ 40% of the predicted value, and a creatinine clearance rate of ≥ 40 mL/min); and an Eastern Cooperative Oncology Group performance status score of ≤ 2 (Fig. 1).
Processes for grouping NDMM patients suitable for ASCT who participated in this retrospective study. NDMM newly diagnosed multiple myeloma; ASCT autologous hematopoietic stem cell transplantation; VRD bortezomib, lenalidomide, and dexamethasone; rhG-CSF recombinant human granulocyte-stimulating factor; rhTPO recombinant human thrombopoietin; TRAEs treatment-related adverse events; ECOG Eastern Cooperative Oncology Group; PLT platelet; ANC absolute neutrophil count.
Conditioning regimen
All patients were administered intravenous melphalan at a dose of 100 mg/m2 on days − 3 and − 2 preceding ASCT.
Supportive care
All patients received a regimen consisting of 50 mg of fluconazole daily for candida infection prophylaxis, 800 mg of cotrimoxazole daily for Pneumocystis carinii infection prevention, and 200 mg of acyclovir twice daily for herpesvirus prevention. Additionally, hepatitis B virus-infected individuals were administered 0.5 mg of entecavir daily to prevent hepatitis B virus activation. Kangfuxin liquid, a traditional Chinese medicine, was routinely administered during pretreatment and hematopoietic reconstruction to prevent oral mucosal ulcers. In cases of oral mucosal ulcers, the treatment regimen included a combination of oral gel and supplementation with multivitamins and micronutrients. Furthermore, 5 µg/kg/day granulocyte-stimulating factor was administered from day 6 after infusion of autologous stem cells until the absolute neutrophil count exceeded 0.5 × 109/L on three consecutive days. In the rhTPO group, rhTPO was administered at a daily dose of 15,000 units starting from the first day post-ASCT and continued for 14 days or until platelet engraftment. The control group received solely platelet transfusion support until platelet engraftment was achieved. A red blood cell suspension was administered when the hemoglobin level fell below 60 g/L, while platelets were transfused when the platelet count dropped below 20 × 109/L. Throughout the process of hematopoietic reconstitution, patients experiencing febrile neutropenia were prescribed anti-bacterial and anti-fungal medications on a personalized basis.
Criteria for engraftment
Neutrophil engraftment was characterized as the initial occurrence of three consecutive days during which the absolute neutrophil count was equal to or greater than 0.5 × 109/L, without any subsequent decrease. Similarly, platelet engraftment was defined as the first of seven consecutive days with a platelet count equal to or exceeding 20 × 109/L, in the absence of platelet transfusion18.
Safety assessment
Adverse events were determined by referring to the Common Terminology Criteria for Adverse Events, version 5.0. TRM was defined as death from any cause other than disease recurrence or progression within the first 100 days after ASCT.
Economic benefits
The median duration of the hospital stay and median hospitalization expenses (in RMB) were compared between the two groups.
Statistical analysis
All statistical analyses were performed using Statistical Package for Social Science (SPSS 29.0) software. The Student’s t-test was used to compare the medians and standard deviations of the variables between the two groups. The chi-square test was used to compare the incidences of specific variables between the two groups. All p-values were two-sided and p < 0.05 was considered statistically significant.
Results
Patient characteristics
Median age, sex ratio, initial Durie-Salmon staging, International Staging System (ISS) staging, Revised ISS staging, risk stratification, and pre-transplantation disease status did not significantly differ between the two groups (p > 0.05) (Table 1).
Hematopoietic reconstruction
The median number of infused CD34 + cells was 3.619 × 106/kg and 3.557 × 106/kg in the rhTPO and control groups, respectively (p = 0.934) (Table 2; Fig. 2A). The platelet engraftment rate is 100% in both groups. The median number of red blood cell infusions was 0.23 and 0 units in the rhTPO and control groups, respectively (p = 0.326) (Fig. 2B). The median number of platelet infusions was 1.615 and 1.5 units in the rhTPO and control groups, respectively (p = 0.721) (Fig. 2C). The median time to neutrophil engraftment was 10.92 and 11.14 days in the rhTPO and control groups, respectively (p = 0.405) (Fig. 2D). The median time to platelet engraftment was 11.5 and 11.36 days in the rhTPO and control groups, respectively (p = 0.776) (Fig. 2E). The median time to platelet count > 50 × 109/L was 13.96 and 14 days in the rhTPO and control groups, respectively (p = 0.966). At 15 days after ASCT, the proportion of patients with platelet counts > 50 × 109/L in the rhTPO and the control groups was 73.1% and 64.3%, respectively (p = 0.720). The duration of granulocyte deficiency was comparable between the two groups (6.269 and 6 days, p = 0.504). The post-transplantation granulocyte and platelet recovery curves indicated a consistent pattern of recovery in both groups. Specifically, granulocytes and platelets showed an upward trend on the tenth day following ASCT, with no significant differences in the implantation trends (Fig. 2F and G).
Hematopoietic reconstitution after ASCT in the rhTPO and control groups. (A) Median number of CD34 + cells infused. (B) Median number of red blood cell suspensions transfused during hematopoietic reconstitution. (C) Median number of platelets transfused during hematopoietic reconstitution. (D) Median duration to neutrophil engraftment. (E) Median duration to platelet engraftment. (F) Curve illustrating neutrophil counts post-ASCT. (G) Curve illustrating platelet counts post-ASCT. rhTPO recombinant human thrombopoietin, RBC red blood cell, PLT platelet, ANC absolute neutrophil count.
Treatment-related adverse events (TRAEs)
TRAEs were similar in the two groups (Table 3). The most prevalent hematologic adverse events were grade ≥ 3 neutropenia and thrombocytopenia, with similar incidences in the two groups. Anemia was predominantly classified as mild-to-moderate, and its incidence did not significantly differ between the two groups. The majority of non-hematologic TRAEs were graded as ≤ 2 and included nausea and vomiting (65.4% vs. 78.6%, p = 0.613), elevated transaminases (15.4% vs. 14.3%, p = 1.000), elevated bilirubin (23.1% vs. 21.4%, p = 1.000), elevated creatinine (7.7% vs. 7.1%, p = 1.000), and fever (50% vs. 57.1%, p = 0.666), with no significant differences between the rhTPO and control groups. The incidence of diarrhea was 96.1% and 85.8% in the rhTPO and control groups, respectively. Specifically, grade 1–2 diarrhea was observed in 42.3% (n = 11) and 42.9% (n = 6) of patients in the rhTPO and control groups, respectively, while grade 3–4 diarrhea was observed in 53.8% (n = 14) and 42.9% (n = 6) of patients in the rhTPO and control groups, respectively (p = 0.732). The median duration of diarrhea was marginally longer in the rhTPO group (9.03 days) than in the control group (6.27 days); however, this difference was not statistically significant (p = 0.088). The median duration of nausea and vomiting (2.73 vs. 3.57 days, p = 0.422) and the median number of days that patients received intravenous nutrition (6.15 vs. 9.28 days, p = 0.097) did not significantly differ between the two groups. Furthermore, no rhTPO-related adverse events, including allergy, thrombosis, and bone pain, were observed in the rhTPO group.
Short-term efficacy
Following ASCT, the immediate therapeutic efficacy was similar in the two groups. In the rhTPO and control groups, the complete remission rate was 73.1% and 92.9%, respectively, the very good partial remission rate was 23.1% and 7.1%, respectively, and the partial remission rate was 3.8% and 0%, respectively (p = 0.484) (Table 4).
Hematopoietic reconstruction follow-up
Following ASCT, the median follow-up duration was 45 and 42 months in the rhTPO and control groups, respectively (Table 5). At 180 days post-ASCT, two patients from both groups were lost to follow-up. The median neutrophil count of patients who remained in the study was 3.003 × 109/L and 3.241 × 109/L in the rhTPO and control groups, respectively (p = 0.405). The median hemoglobin level was 122 and 129.3 g/L in the rhTPO and control groups, respectively (p = 0.306). The median platelet count was 134 × 109/L and 158.9 × 109/L in the rhTPO and control groups, respectively (p = 0.174). These results indicate that hematopoietic reconstitution did not significantly differ between the two groups.
Economic benefits
The median length of hospitalization did not significantly differ between the rhTPO group (27.08 days) and the control group (25.07 days) (p = 0.193). Conversely, the median hospitalization cost was significantly higher in the rhTPO group (RMB 73,846) than in the control group (RMB 54,136) (p < 0.001). The median additional expenditure in the rhTPO group was RMB 19,710.
Discussion
ASCT is the established consolidation treatment strategy for NDMM19. Despite the relatively low risk of hemorrhage, it remains a significant event contributing to early TRM and warrants careful consideration. In addition to platelet transfusion, investigation of accelerated platelet implantation within a safe profile is a significant area of research. Limited research has assessed the impact of rhTPO on platelet engraftment in patients with NDMM. The selection of NDMM patients deemed appropriate for ASCT is an important issue in observational studies. In the current study, the induction protocols were uniformly selected as standard VRD regimens, the quantity of infused CD34 + cells did not significantly differ among the patients, and the conditioning regimen employed was standard high-dose melphalan. These measures collectively ensured maximal homogeneity of the study population.
In this real-world study, we demonstrated for the first time that administration of rhTPO following ASCT in patients with NDMM does not promote platelet engraftment. Our findings are consistent with the outcomes of two phase I clinical trials12,13. Although administration of rhTPO in these two studies differed from that in our study and these two studies did not stratify the effects of patient age, disease type, number of CD34 + cells reinfused, and type of hematopoietic stem cells reinfused on hematopoietic reconstruction post-transplantation, the results are highly consistent with our findings. Therefore, we hypothesize that use of rhTPO primarily increases financial expenditures for patients compared with the no-intervention control group.
Recent studies notably demonstrated that rhTPO facilitates platelet recovery following allogeneic HSCT in patients with hematologic conditions such as AA, MDS, and AML14,16. This discrepancy with the findings of our study may be attributable to several factors. First, the determinants influencing platelet engraftment following allogeneic and autologous HSCT are not identical. In particular, platelet engraftment post-allogeneic HSCT is additionally affected by factors such as graft-versus-host disease and early viral infections16. Second, platelet engraftment is influenced by the specific type of primary hematopoietic disorder. Notably, platelet engraftment occurs more rapidly in patients with lymphoma and myeloma than in those with AML and MDS17. Finally, the mechanisms underlying hematopoietic reconstitution exhibit fundamental differences between allogeneic and autologous HSCT20,21. Therefore, while rhTPO may expedite platelet engraftment following allogeneic HSCT, its impact on hematopoietic reconstitution post-ASCT in patients with NDMM appears to be controversial.
Recently, a clinical study administered rhTPO following ASCT in patients with NDMM. Regrettably, the results of that study do not align with the findings of our research4. The variability in ISS staging, induction protocols, and preconditioning regimens among the patients included in that study may have contributed to the inconsistent findings. Interestingly, in that study, patients who received an infused-back CD34 + cell count greater than 2 × 106/kg (consistent with the standard clinical dose) did not exhibit enhanced platelet engraftment, regardless of whether they were administered rhTPO. This finding is corroborative evidence supporting the conclusions of our study.
From a safety perspective, no allergic reactions, pain, or thrombotic events were observed in the rhTPO group. Consistent with the findings of previous studies, adjunctive use of rhTPO did not increase TRAEs.
Our study has several limitations. First, the limited sample size and lack of random sampling may introduce selection bias into our findings. This may also explain the slightly higher incidence of diarrhea in the rhTPO group and the slightly lower platelet counts during long-term follow-up, despite the lack of statistical significance. Second, given the heterogeneous and unique characteristics of patients with NDMM, we attempted to enhance the uniformity of the study cohort by retrospectively selecting individuals eligible for transplantation who received four cycles of VRD induction therapy and excluding those who were treated with alternative regimens such as VCD, VTD, and D-VRD. The generalizability of our results to different patient populations remains uncertain. Finally, retrospective studies are intrinsically affected by subjective factors, and the accuracy of medical records is not governed by the study’s design, which can introduce potential confounding variables and bias. Consequently, future research should prioritize prospective clinical studies with large, multi-center samples.
Conclusion
This study determined that rhTPO did not facilitate platelet engraftment following ASCT for NDMM. Additionally, patients who received rhTPO incurred higher median hospitalization costs. Consequently, rhTPO may be a more costly option for supportive care post-ASCT in patients with NDMM.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Rajkumar, S. V. Multiple myeloma: 2024 update on diagnosis, risk-stratification, and management. Am. J. Hematol. (2024).
Gertz, M. A. et al. Autologous stem cell transplant in 716 patients with multiple myeloma: low treatment-related mortality, feasibility of outpatient transplant, and effect of a multidisciplinary quality initiative. Mayo Clin. Proc. 83, 1131–1138 (2008).
Jantunen, E. et al. Early treatment-related mortality in adult autologous stem cell transplant recipients: a nation-wide survey of 1482 transplanted patients. Eur. J. Haematol. 76, 245–250 (2006).
Gu, J. et al. Recombinant human thrombopoietin improved platelet engraftment after autologous hematopoietic stem cell transplantation in patients with newly diagnosed multiple myeloma. Cancer Med. 10, 7641–7649 (2021).
Qiu, X. et al. Recombinant human thrombopoietin improves platelet engraftment after autologous hematopoietic stem cell transplantation in patients with aggressive lymphoma. Transfus. Apher. Sci. 63, 103972 (2024).
Kaushansky, K. Thrombopoietin: the primary regulator of platelet production. Blood 86, 419–431 (1995).
Yu, Y. et al. High-dose dexamethasone plus recombinant human thrombopoietin vs high-dose dexamethasone alone as frontline treatment for newly diagnosed adult primary immune thrombocytopenia: a prospective, multicenter, randomized trial. Am. J. Hematol. 95, 1542–1552 (2020).
Chen, X. et al. Recombinant human thrombopoietin promotes platelet recovery in DCAG-treated patients with intermediate-high-risk MDS/hypoproliferative AML. Medicine (Baltimore) 102, e33373 (2023).
Yang, Y. et al. High- vs regular-dose recombinant human thrombopoietin plus cyclosporine A in patients with newly diagnosed non-severe aplastic anemia: a retrospective cohort study. Hematology 29, 2298523 (2024).
Vadhan-Raj, S. Clinical experience with recombinant human thrombopoietin in chemotherapy-induced thrombocytopenia. Semin Hematol. 37, 28–34 (2000).
Lin, J. et al. Recombinant human thrombopoietin therapy for primary immune thrombocytopenia in pregnancy: a retrospective comparative cohort study. BMC Pregnancy Childbirth. 23, 820 (2023).
Wolff, S. N. et al. Recombinant human thrombopoietin (rhTPO) after autologous bone marrow transplantation: a phase I pharmacokinetic and pharmacodynamic study. Bone Marrow Transpl. 27, 261–268 (2001).
Nash, R. A. et al. A phase I trial of recombinant human thrombopoietin in patients with delayed platelet recovery after hematopoietic stem cell transplantation. Biol. Blood Marrow Transpl. 6, 25–34 (2000).
Fu, A. et al. Recombinant human thrombopoietin promotes platelet engraftment in severe aplastic anemia patients following treatment with haploid hematopoietic stem cell transplantation using modified post-transplantation cyclophosphamide. Transpl. Cell. Ther. 30, 500–509 (2024).
Tang, B. et al. Recombinant human thrombopoietin promotes platelet engraftment after umbilical cord blood transplantation. Blood Adv. 4, 3829–3839 (2020).
Wang, H. et al. Recombinant human thrombopoietin promotes platelet engraftment and improves prognosis of patients with myelodysplastic syndromes and aplastic anemia after allogeneic hematopoietic stem cell transplantation. Biol. Blood Marrow Transpl. 23, 1678–1684 (2017).
Han, T. T. et al. Recombinant human thrombopoietin promotes platelet engraftment after haploidentical hematopoietic stem cell transplantation: a prospective randomized controlled trial. Ann. Hematol. 94, 117–128 (2015).
Wen, B. et al. Oral eltrombopag versus subcutaneous recombinant human thrombopoietin for promoting platelet engraftment after allogeneic stem cell transplantation: a prospective, non-inferiority, randomized controlled trial. Hematol. Oncol. 40, 777–786 (2022).
Lin, C. M. et al. Treatment benefit of upfront autologous stem cell transplantation for newly diagnosed multiple myeloma: a systematic review and meta-analysis. BMC Cancer. 23, 446 (2023).
Wiegering, V. et al. Comparison of immune reconstitution after allogeneic versus autologous stem cell transplantation in 182 pediatric recipients. J. Pediatr. Hematol. Oncol. 41, e302–e307 (2019).
Schwinger, W. et al. Immune reconstitution after purified autologous and allogeneic blood stem cell transplantation compared with unmanipulated bone marrow transplantation in children. Br. J. Haematol. 135, 76–84 (2006).
Acknowledgements
The study was carried out by a multidisciplinary team of clinicians associated with the Department of Oncology and Hematology, the Central Research Laboratory, and the Medical Laboratory Center of the China-Japan Union Hospital at Jilin University.
Funding
This work was supported by grants from the Science and Technology Development Project of Jilin Province (20210401174YY and YDZJ202201ZYTS092 to WZ).
Author information
Authors and Affiliations
Contributions
Conception and design: W.Z.Provision of study materials or patients: D.C., Y. B., W.Z.Collection and assembly of data: R.W. and Jing C.Data analysis and interpretation: R.W. and Jing C.Manuscript writing: R.W., Z.C. and W.Z.Final approval of manuscript: All authorsAccountable for all aspects of the work: All authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, R., Cai, J., Chen, Z. et al. Recombinant human thrombopoietin does not promote platelet engraftment in newly diagnosed multiple myeloma patients following autologous stem cell transplantation. Sci Rep 15, 5393 (2025). https://doi.org/10.1038/s41598-025-89535-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89535-7