Abstract
Affective temperaments are inherited parts of personality determining mood and activity, affecting the management of somatic conditions. We aimed to investigate the association between affective temperaments, depressive symptoms, and self-care (physical activity, smoking, alcohol consumption), and their effect on glycemic control, among patients with type 2 diabetes (T2DM) in general practice, in a cross-sectional study enrolling 338 consecutive patients from six primary care practices in Hungary. A self-administered questionnaire (history, anthropometric, socioeconomic, laboratory parameters), the Beck Depression Inventory (BDI), the Hamilton Anxiety Scale, and the Temperament Evaluation of Memphis, Pisa, Paris, and San Diego Autoquestionnaire were used. Cyclothymic affective temperament determined HbA1c levels in regression analysis (p = 0.002), and the BDI score (p = 0.048). In causal mediation analyses, cyclothymic affective temperament was directly associated with higher HbA1c (p = 0.008). Hyperthymic affective temperament was indirectly associated with lower HbA1c, mediated by BDI (p = 0.034). Depressive, anxious, and irritable affective temperaments, and lifestyle factors were not associated with HbA1c neither in regression nor in mediation analysis as direct or mediating factors. Among primary care patients with T2DM, cyclothymic temperament correlates with worse glycemic control, independently of depressive symptoms. Hyperthymic temperament reduces depressive symptoms, thereby improving glycemic control. Identifying affective temperaments may improve diabetes care.
Similar content being viewed by others
Introduction
In somatic medicine, the role of psychological factors in the development, treatment, course, and outcome of illnesses, as well as their role as possible targets for improving the management and well-being of patients is increasingly recognised. Diabetes mellitus is a major health issue in today’s population because of its high prevalence and complications. Diabetes has become one of the 10 leading causes of death, with a significant increase of 70% since 20001. Between 1990 and 2019, the disability-adjusted life years (DALY) attributable to diabetes increased by 24.4%2.
Personality and individual psychological characteristics may have an important role in patients’ behaviour. Temperaments have a strong biological basis and are stable parts of the personality, which are inherited and determine the emotional mood, reactivity, and the level of activity. Temperaments are manifested in the early years and are stable throughout life3. While several temperamental models were derived on theoretical bases to describe human reactivity in general, the model of affective temperaments was developed based on observations of affective disorder patients and their healthy first-degree relatives, and it was extrapolated to healthy functioning. The affective temperaments (depressive, cyclothymic, hyperthymic, irritable and anxious) are subclinical, trait-related manifestations and are common precursors of affective mood disorders, like unipolar major depression or bipolar (I) and (II) disorders and suicide risk4. In addition to their association with psychiatric disorders, numerous studies found a relationship between temperaments and somatic diseases. For example, several studies pointed to string associations between affective temperaments and multiple aspects of cardiovascular diseases including risk, complications and disease course. Robust significant associations have been reported between cyclothymic affective temperament and risk of hypertension5, history of acute coronary syndrome and myocardial infarction in hypertensive patients6, brachial systolic blood pressure7, earlier hypertension onset in women8accelerated vascular aging9and higher resistant hypertension10, while hyperthymic temperament was negatively associated in earlier studies with pulse wave reflection augmentation index7, and both of these temperaments played a role in predicting severe coronary artery disease with opposite direction11,12. These studies confirmed in new patient populations the pathophysiological role of cyclothymic temperament on the cardiovascular system and the protective role of hypertymic temperament.
Affective temperaments were thus found to play a major role in highly prevalent, chronic, and long-term illnesses, where identification of temperamental factors influencing both risk of development of the disease and its successful long-term treatment would provide an important tool for prevention and better management as well.
Despite being highly prevalent, the relationship between affective temperaments and diabetes mellitus is still elusive and partly controversial. Gois et al. found that patients with excessive depressive temperaments had worse glycemic control13. In another study, depressive and cyclothymic temperaments positively correlated with fasting glucose but not with HbA1c values among obese individuals with type 2 diabetes14, while both positive14and inverse15 associations have been reported between anxious temperament and HbA1c.
The aim of our study was:
-
1.
To assess the possible association between affective temperaments and glycemic control among patients with type 2 diabetes in the general practice setting.
-
2.
To investigate whether temperaments affect metabolic control either through depression or by impairing self-care (physical activity, smoking, alcohol consumption).
Materials and methods
Patients
A total of 338 adult patients (over 18 years of age) with type 2 diabetes mellitus were included in our study, with the help of accredited general practitioners of the Department of Family Medicine at Semmelweis University, from six primary care practices in Budapest, between September 2018 and February 2020. Consecutive patients attending to the practices were included. The general practitioners were provided with verbal and written information and were consulted if they had any questions. Participation in the study was voluntary. Patients were informed verbally and in written form about the study and gave written informed consent. We excluded patients with type 1 and gestational diabetes mellitus, patients with type 2 diabetes mellitus who had psychiatric outpatient follow-up, or had hospitalization with a psychiatric diagnosis, or cognitive impairment, and patients who were taking antidepressants or antipsychotics. Patients were not rewarded for participating in the study. One patient was excluded because of type 1 diabetes and 5 patients declined to participate, 3 due to lack of time and 2 did not give a reason. The data of the patients were used anonymously.
Our study was approved by the national ethical committee under the number 44677-2/2018/EKU. The research follows the ethical guidelines of the Declaration of Helsinki.
Measures
The self-report general questionnaire recorded sociodemographic data, addictions, history of suicide attempt and physical activity. The remaining part of the questionnaire was completed by the general practitioners (GPs), based on the patient’s medical records. GPs recorded any co-morbidities, complications, the patient’s medication history and their blood glucose and HbA1c levels. We used the WHO recommended classification for BMI categories (normal BMI: 18.5–24.9 kg/m2, overweight: 25.0–29.9 kg/m2, obese: ≥ 30.0 kg/m2).
Temperament evaluation of Memphis, Pisa, Paris and San Diego Autoquestionnaire (TEMPS- A)
We used the Temperament Evaluation of Memphis, Pisa, Paris and San Diego Autoquestionnaire (TEMPS-A) to assess affective temperaments on depressive, cyclothymic, hyperthymic, irritable and anxious subscales16. The TEMPS-A contains 110 items (109 in the version for males), requiring ‘yes’ (score 1) or ‘no’ (score 0) answers. The mean and standard deviation of each subscale are calculated. The Hungarian version of the questionnaire used in this study was validated in 2006 by Rózsa and coworkers17.
Beck Depression Inventory (BDI) and Hamilton Anxiety Scale (HAM-A)
We chose the Beck Depression Inventory (BDI) to assess the presence and severity of depressive symptoms because it is recommended by the American Diabetes Association for screening symptoms of depression among patients with type 2 diabetes mellitus18,19. The BDI is a 21-item, four-point (0–3) self-report questionnaire20,21.
Although the American Diabetes Association recommends the 21-item Beck Anxiety Inventory for screening anxiety among patients with type 2 diabetes mellitus, we chose the Hamilton Anxiety Scale (HAM-A), which is still one of the most widely used assessment scales today22. According to a study, the specificity and sensitivity of the Beck Anxiety Inventory and the HAM-A were found to be almost identical23. During our research, we asked patients for information based on several aspects, so it was important to keep the burden on patients manageable to ensure they did not skip completing the questionnaire. The HAM-A questionnaire was preferred because it is quick to complete and consists of 14 questions in total, as opposed to the 21 questions of the Beck Anxiety Inventory. The HAM-A questionnaire items can be scored from 0 (no symptoms) to 4 (very severe symptoms)24,25.
Statistical analysis
Analyses were conducted using the R statistical software package rms v.6.3 and IBM SPSS Statistical software version 25.026,27. The significance threshold was set at p < 0.05. Continuous variables are presented as mean ± standard deviation, categorical variables are presented as count (percentage).
Univariate linear regression analyses were performed to examine the relationship between HbA1c and affective temperaments, BDI, HAM-A, physical activity, smoking, alcohol, age and sex. The analyses used change in TEMPS-A scores, BDI, HAM-A, smoking, physical exercise, alcohol intake, sex and age as predictor variables and HbA1c as dependent variable. After that, causal mediation analysis was applied to examine whether any of the observed variables that significantly predicted HbA1c based on the univariate analysis mediated the effect of affective temperaments on HbA1c. Causal mediation analysis was run with affective temperaments as exposure, HbA1c as response, and any significant predictors of HbA1c as mediator variables.
In mediation analysis, nonparametric bootstrap confidence intervals were calculated with the percentile method28. Results are presented as estimated regression coefficients, with 95% confidence intervals. The significance threshold was set at p< 0.05. Analyses were conducted using the R statistical software package mediation v.4.5 and IBM SPSS Statistical software version 25.027,29.
Results
Characteristics of patients
Descriptive parameters for 338 patients with type 2 diabetes are shown in Table 1.
The mean age of patients was 63.98 ± 11.51 years, 67.2% were 60 years or older, and 61.2% were female. Sociodemographically, nearly one-third had completed only primary education (32.7%), one third had tertiary education (30.1%) and 37.2% had secondary education. Half of the patients lived in the capital city (51.2%). The consumption of alcohol was 35.5%, 13.3% of patients smoked and 21.1% were physically active. More than half of the patients were obese (53.4%) and 33.2% were overweight. 3.1% of patients had a history of suicide attempt. Mean HbA1c was 7.23 ± 1.26% and fasting blood glucose was 8.39 ± 4.11 mmol/l.
The mean BDI score was 8.49 ± 7.65, the mean Hamilton score was 11.08 ± 8.65. The mean depressive affective temperament score was 7.98 ± 3.75 point, cyclothymic 5.8 ± 4.93, hyperthymic 11.3 ± 4.77, irritable 4.91 ± 4.04 and anxious 7.92 ± 6.43.
Regression analysis
Univariate linear regression analyses showed that cyclothymic affective temperament and BDI were associated with HbA1c. A 1-point increase in cyclothymic score was associated with a 0.043% increase in HbA1c (p = 0.002), while a 1-point increase in BDI score was associated with a 0.017% increase in HbA1c (p = 0.048). Depressive, anxious, irritable, and hyperthymic affective temperaments were not associated directly with HbA1c. Lifestyle factors, such as smoking, physical activity, and alcohol intake were not predicting HbA1c. Table 2. presents the results of the regression analyses.
Causal mediation analysis
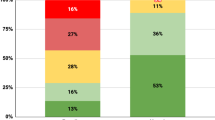
Since besides affective temperaments, only BDI was a significant predictor of HbA1c based on univariate regression analyses, causal mediation analysis was run with affective temperaments as exposure, BDI as mediator and HbA1c as response variables. Based on causal mediation analyses, a 1-point higher cyclothymic affective temperament score was directly associated with a 0.04% higher HbA1c score (p = 0.008), with the effect not mediated by BDI, while a 1-point higher hyperthymic affective temperament score was indirectly associated with a 0.011% lower HbA1c score, mediated by BDI (p = 0.034) (Fig. 1). Depressive, anxious, and irritable affective temperaments were not associated with HbA1c neither directly nor indirectly. Table 3. presents the results of the causal mediation analyses.
Mediation analysis results. (A) Direct effect of cyclothymic affective temperament on HbA1c. (B) Indirect effect of hyperthymic affective temperament on HbA1c, mediated by BDI. Key: AT: affective temperament. *indicates significant values.
Discussion
In our study focusing on the association between affective temperaments and glycemic control, we found a significant direct association between cyclothymic affective temperament and HbA1c. Furthermore, mediation analyses revealed that the effect of cyclothymic affective temperament on HbA1C (a temperament score 1 point higher was directly associated with an HbA1c score increase of 0.04% (p= 0.008)), was not mediated by BDI. On the other hand, while hyperthymic temperament was not directly associated with HbA1c, we found a significant effect mediated by BDI. Our results suggest that cyclothymic temperament directly affects glycemic control independently of depression (although depression had a significant direct effect on glycemic control, and cyclothymic temperament was also significantly associated with depression scores), while hyperthymic temperament exerts its effect via reduced BDI scores and thus lowering their deleterious effect on glycemic control, as already demonstrated in our previous study30. More severe depressive symptoms may impair self-care in people with diabetes31(diet, physical activity and medication adherence), which plays an important role in metabolic control32. Therefore, it is important to understand the complex association between affective temperaments and depression in their relationship with diabetes, to identify intervention targets that could improve disease management and glycemic control.
Previous studies reported significant results on the relationship between affective temperaments and diabetes mellitus confirming their role, however, results concerning the specific temperaments were divergent. Gois et al. examined the association between affective temperaments and metabolic control among patients with type 2 diabetes. In their relatively small sample (90 patients) they chose to investigate only anxious and depressive temperaments, making it difficult to compare to our study where all temperaments were included13. Another study investigated the relationship between affective temperaments and glycemic control among 185 obese patients with type 2 diabetes mellitus. Beside HbA1c the researchers used the less reliable fasting glucose as dependent variable as well. The reported correlation between depressive, cyclothymic and anxious temperaments and fasting glucose should be interpreted cautiously. However, they found a significant relationship between HbA1c values and anxious temperament14.
This finding is conflicting with the results of Hall et al., who reported an inverse association between anxious temperament and HbA1c values among newly diagnosed patients with type 2 diabetes15. Individual differences in anxious temperament were assessed using the Behavioral Inhibition Scale (BIS), the Behavioral Inhibition/Behavioral Approach Scales (BIS-BAS) questionnaire and other temperament types were not measured. Omitting other temperaments in these studies might have lead to attaching too much importance to anxious and depressive temperaments, while glossing over the effect of other temperaments.
While sample size13, questionnaire used, and not including all temperaments13,15probably affected findings, some other factors, including gender ratio or age of the patients33 could also explain contradicting results.
Cyclothymic temperament is associated with instability, both in terms of activity and mood, unstable self-esteem, socialisation and energy levels as well as rapid mood swings34,35, which may significantly impact therapeutic adherence both in terms of medication, diet, and other recommended lifestyle changes, independently of depressed mood. However, other psychological and behavioural factors, as well as biological, neurobiological and even genetic contributors may play a role in the association between cyclothymic temperament and worse glycaemic control, which warrants further studies to improve our understanding and put findings to clinical use.
Variability in personality or temperament can affect lifestyle choices, self-care, adherence to treatment, and other factors that can affect glycemic control36. Modifiable risk factors such as exercise, smoking and alcohol consumption may play a role in the relationship between cyclothymic temperament and glycemic control. In our study, we did not find a significant association between lifestyle factors and glycemic control, and thus they are not considered as mediators, although the literature suggests that they may be involved in mediation. Individuals with cyclothymic temperament are known to smoke more often and consume alcohol at higher rates37,38. Cyclothymic temperament can also affect glucose metabolism through adherence to therapy. Yamamoto et al. investigated the link between affective temperaments and poor glycemic control due to insufficient self-care among 77 outpatients with type 2 diabetes mellitus. Significant associations were found between poor glycemic control and stress-induced overeating and poor medication adherence. Furthermore, the cyclothymic temperament was significantly associated with stress-induced overeating and poor medication adherence. Path analysis found a possible causal link between cyclothymic temperament and poor glycemic control through disordered eating and poor medication adherence39. In contrast, Shamsi et al. found a relationship between irritable temperament and medication compliance in 207 patients with type 2 diabetes. Irritable temperament has a negative effect on medication compliance and through this, on glycemic control40. Szabó et al. recent meta-analysis (9 studies, n= 1138) also supports the results of the above studies, as they found that cyclothymic, depressive and irritable temperaments may influence adherence to therapy41.
Another important finding of our study is the association between hyperthymic temperament and glycemic control mediated by depression scores. Hyperthymic temperament, associated with a more stable mood, a positive and optimistic attitude and increased energy levels as well as easier and better adaptation to stress34,42and has a beneficial role on glucose metabolism by reducing depressive symptoms. Our finding is also in part confirms previous results reporting a negative correlation with HbA1c among obese patients with type 2 diabetes mellitus14. It must be noted, however, that our study also managed to show that the relationship is not direct but is rather mediated through decreased depression. Understanding by which other mechanisms hyperthymic temperament is protective against impaired glycemic control and how they can be exploited in the management of these patients is also warranting further studies.
Our research has some limitations, which should always be taken into account when interpreting the results and applying them more widely. We used validated questionnaires, but we could not exclude misunderstandings and errors by patients. In addition, the cross-sectional nature of the study does not allow us to explore causal relationships. The lack of a healthy, non-diabetic control group also limits the generalizability of our results. The subjects in the study were all of Caucasian origin, and as race-specific differences may be present in the affective temperament patterns, this may also be a limitation.
Conclusion
Among patients with type 2 diabetes mellitus in primary care, cyclothymic temperament correlates with worse glycemic control, with the effect not mediated by BDI, but through a different route. Further studies are needed to explore which factors may be involved. Another important finding of our study is that hyperthymic temperament reduces depressive symptoms as expected, thereby improving glycemic control. Altogether, our results suggest that screening for temperamental and personality factors may help predict treatment outcome in Type 2 diabetes patients and also identify those, where limited adherence may compromise efficacy.
Data availability
Raw data are available upon request to the corresponding author.
References
WHO. World Health Organization. The top 10 causes of death.
GBD 2019 Diseases and Injuries Collaborators. (2020) Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222
Bouchard, T.J., Genes, environment, and personality. Science, 264, 1700–1701 (1994).
Rihmer, Z., Akiskal, K.K., Rihmer, A. & Akiskal, H.S. Current research on affective temperaments. Curr. Opin. Psychiatry 23, 12–18 (2010).
Eőry, A., et al. Personality and cardiovascular risk: association between hypertension and affective temperaments-a cross-sectional observational study in primary care settings. Eur. J. Gen. Pract. 20, 247–252 (2014).
Eőry, A., et al. Affective temperaments contribute to cardiac complications in hypertension independently of depression. Psychother. Psychosom. 83, 187–189 (2014).
László, A., et al. Association of affective temperaments with blood pressure and arterial stiffness in hypertensive patients: A cross-sectional study. BMC Cardiovasc. Disord. 16, 158 (2016).
Kőrösi, B., et al. Association between cyclothymic affective temperament and age of onset of hypertension. Int. J. Hypertens. 2019, 9248247 (2019).
Vecsey-Nagy, M., et al. The association between accelerated vascular aging and cyclothymic affective temperament in women. J. Psychosom. Res. 145, 110423 (2021).
Kőrӧsi, B., et al. Evaluation of affective temperaments and arterial stiffness in different hypertension phenotypes. Hypertens. Res. 44, 47–54 (2021).
Vecsey-Nagy, M., et al. Association between affective temperaments and severe coronary artery disease. J. Affect. Disord. 295, 914–919 (2021).
Nemcsik, J., et al. Inverse association between hyperthymic affective temperament and coronary atherosclerosis: A coronary computed tomography angiography study. J Psychosom Res 103, 108–112 (2017).
Gois, C., et al. The role of affective temperaments in metabolic control in patients with type 2 diabetes. J. Affect. Disord. 134, 52–58 (2011).
Lesiewska, N., et al. Affective temperament and glycemic control - The psychological aspect of obesity and diabetes mellitus. Diabetes Metab. Syndr. Obes. 14, 4981–4991 (2021).
Hall, P.A., Rodin, G.M., Vallis, T.M. & Perkins, B.A. The consequences of anxious temperament for disease detection, self-management behavior, and quality of life in Type 2 diabetes mellitus. J. Psychosom. Res. 67, 297–305 (2009).
Akiskal, H.S., Akiskal, K.K., Haykal, R.F., Manning, J.S. & Connor, P.D. TEMPS-A: Progress towards validation of a self-rated clinical version of the Temperament Evaluation of the Memphis, Pisa, Paris, and San Diego Autoquestionnaire. J. Affect. Disord. 85, 3–16 (2005).
Rózsa, S., et al. Affective temperaments: Psychometric properties of the Hungarian TEMPS-A. Psychiatr Hung. 21, 147–160 (2006).
ADA. American Diabetes Association Professional Practice Committee. 5. Facilitating positive health behaviors and well-being to improve health outcomes: Standards of Care in Diabetes—2024. Diabetes care 47, S77-S110 (2024).
Young-Hyman, D., et al. Psychosocial care for people with diabetes: A position statement of the american diabetes association. Diabetes Care 39, 2126–2140 (2016).
Beck, A.T., Ward, C.H., Mendelson, M., Mock, J. & Erbaugh, J. An inventory for measuring depression. Arch. Gen. Psychiatry 4, 561–571 (1961).
Pető, Z., Hunya, P. & Eller, J. Application of psychometric methods for the investigation of mood life. Clin. Neurosci. 40, 537–546 (1987).
Thompson, E. Hamilton Rating Scale for Anxiety (HAM-A). Occupational Med. 65, 601 (2015).
Leentjens, A.F.G., et al. Anxiety rating scales in Parkinson’s disease: A validation study of the Hamilton anxiety rating scale, the Beck anxiety inventory, and the hospital anxiety and depression scale. Movement Disord. 26, 407–415 (2011).
Hamilton, M. The assessment of anxiety states by rating. Br. J. Med. Psychol. 32, 50–55 (1959).
Tringer, L. Hamilton anxiety scale hungarian version. Clin. Neurosci. 26, 1–18 (1973).
Harrell, F.E. rms: Regression modeling strategies.
IBM Corporation. SPSS Statistics for Windows, Version 25.0., Armonk, NY. (2017).
Imai, K., Keele, L. & Tingley, D. A general approach to causal mediation analysis. Psychol Methods 15, 309–334 (2010).
Tingley, D., Yamamoto, T., Hirose, K., Keele, L. & Imai, K. Mediation: R package for causal mediation analysis. J. Stat. Softw. 59, 1–38 (2014).
Hargittay, Cs., et al. Severity of depressive but not anxiety symptoms impacts glucose metabolism among patients with type 2 diabetes in primary care. Front. Med. (Lausanne) 9, 944047 (2022).
Schmitt, A., et al. Depression is linked to hyperglycaemia via suboptimal diabetes self-management: A cross-sectional mediation analysis. J.Psychosom. Res. 94, 17–23 (2017).
Lin, K., et al. Effects of depression, diabetes distress, diabetes self-efficacy, and diabetes self-management on glycemic control among Chinese population with type 2 diabetes mellitus. Diabetes Res. Clini. Pract. 131, 179–186 (2017).
Eőry, A. & Rihmer, Z. The clinical importance of affective temperaments. Psychiatr Hung. 26, 144–157 (2011).
Eőry, A., Gonda, X., Torzsa, P., Kalabay, L. & Rihmer, Z. Affective temperaments: From neurobiological roots to clinical application. Orv. Hetil. 152, 1879–1886 (2011).
Rihmer, Z., et al. Association between affective temperaments and season of birth in a general student population. J. Affect. Disord. 132, 64–70 (2011).
van Dooren, F.E., et al. Psychological and personality factors in type 2 diabetes mellitus, presenting the rationale and exploratory results from The Maastricht Study, a population-based cohort study. BMC Psychiatry 16, 17 (2016).
Bisol, L.W., Soldado, F., Albuquerque, C., Lorenzi, T.M. & Lara, D.R. Emotional and affective temperaments and cigarette smoking in a large sample. J. Affect. Disord. 127, 89–95 (2010).
Pacini, M., et al. Affective temperaments in alcoholic patients. Alcohol 43, 397–404 (2009).
Yamamoto, T., et al. Cyclothymic temperament is associated with poor medication adherence and disordered eating in Type 2 diabetes patients: A case-control study. Diabetes Ther. Res. Treatment Educ. Diabetes Related Disord. 12, 2611–2624 (2021).
Shamsi, A., Khodaifar, F., Arzaghi, S.M., Sarvghadi, F. & Ghazi, A. Is there any relationship between medication compliance and affective temperaments in patients with type 2 diabetes? J. Diabetes Metab. Disord. 13, 96 (2014).
Szabó, G., Fornaro, M., Dome, P., Várbíró, S. & Gonda, X. A bitter pill to swallow? Impact of affective temperaments on treatment adherence: a systematic review and meta-analysis. Transl .Psychiatry 12, 360 (2022).
Akiskal, K.K. & Akiskal, H.S. The theoretical underpinnings of affective temperaments: implications for evolutionary foundations of bipolar disorder and human nature. J. Affect. Disord. 85, 231–239 (2005).
Acknowledgements
We thank the support of the general practitioners and patients participating in this study.
Funding
Open access funding provided by Semmelweis University.
Author information
Authors and Affiliations
Contributions
CsH: writing the manuscript. XG and PT: study design. AE, AL, CsH, BM, BT, PT, and KV: data collection. GSz: provide statistical analysis. AE, AL, XG, BM, BT, GSz, KV, ZR, and PT: review and edit the manuscript. XG, PT, and ZR: critical revision of the article. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study involving human participants was reviewed and approved by the Semmelweis University Ethics Committee. All patients/participants provided their written informed consent to participate in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hargittay, C., Vörös, K., Eőry, A. et al. The influence of cyclothymic and hyperthymic affective temperaments on glycemic control in patients with type 2 diabetes. Sci Rep 15, 5845 (2025). https://doi.org/10.1038/s41598-025-90292-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90292-w