Abstract
Solid-solid phase change materials (SS-PCMs) hold promise for energy storage/dissipation in batteries and energetic materials. Yet, phase change kinetics for SS-PCMs undergoing metastable to semi-stable/stable phase transformations remain relatively ill-studied because trapping metastable phases remain challenging. Recently, we demonstrated the kinetic entrapment and stabilization of a highly disordered and amorphous Al-oxide phase m-AlOx@C (x~2.5-3.0) via laser ablation synthesis in solution (LASiS). We report here, to our knowledge, the first chemical kinetics analysis for S-S phase transition of the m-AlO3@C nanocomposites (< 5–8 nm sizes) into semi-stable equilibrium alumina phases (θ/γ-Al2O3) via disproportionation reaction, while releasing excess trapped gases. Our results indicate the atomic density of the AlO3 structures to be ~5–10 times less than that of the final Al2O3 phases, which led to the hypothesis of a volume shrinkage process during their phase transition. Temperature-dependent X-ray diffraction studies reveal the high-temperature phase transition for m-AlO3 → θ/γ-Al2O3 to follow contracting volume kinetics model, thereby validating our earlier hypothesis. Using the geometric volume contraction model, reaction kinetics analyses from Arrhenius plots reveal the activation energy barrier for the phase transition to be ~270±11 kJ/mol. This makes the activation energy barrier nearly identical to the oxidation of micron-sized Al particles.
Similar content being viewed by others
Introduction
Efficient energy conversion and storage via enabling materials technology holds the key to the design and optimization of myriad engineering products in the rapidly evolving landscape of human-technology interfaces. Accelerated developments in technologies such as mobile devices, batteries, rocket propulsion, and electric vehicles have propelled the demands for innovative functional materials that can overcome the current limitations in energy consumption, dissipation, and storage1. Disordered amorphous metal-oxide nanostructures in metastable states could potentially become revolutionary materials in these fields, with their properties and applications only now being investigated and understood2,3,4.
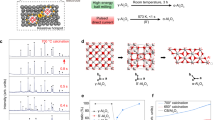
Schematic for reaction progression depicting the kinetic entrapment of metastable metal-oxide phases
It can be postulated that kinetically trapped intermediate metastable metal-oxide phases, perched above the ground state equilibrium en route to their final stable oxide phases (see Fig. 1), can plausibly offer novel routes for energy release (thermal/non-thermal) via structural bond rearrangements under external perturbations. Such molecular bond rearrangements can result in solid-state phase transitions to lowest energy equilibrium states via direct nucleation/crystallization processes. Solid-solid phase change materials (SS- PCMs) have received increasing interest for thermal energy storage because of their high energy-storage density, solid-state processing, small volume changes during phase transition, less phase segregations, and long cyclic life. Specifically, inorganic SS-PCMs are able to store and release thermal energy in solid phase via various energy dissipation routes including crystallographic structure transformations (crystalline to crystalline), magnetic transformations, disorder-to-order transitions, as well as amorphous to crystalline structural transformations1,5,6. The usefulness of such SS-PCMs is highly dependent upon two parameters: (1) how large is the relative thermal energy release or storage and (2) what are the related kinetics for such release7. An example of this would be the well-studied diamond to graphite transition, with a prohibitively high amount of energy required for the perturbation and a relatively slow kinetics (reported activation energy barrier ~ 700–1160 kJ/mol), thereby making it unsuitable for use as a SS-PCM8. However, our design concept here was the development of an SS-PCM with a relatively low perturbation energy or activation energy barrier and fast relative kinetics that can offer higher chemical stability under ambient conditions. This would allow for the safe long-term storage and on-demand release of potentially large amounts of trapped energy either, during a single metastable to stable transition or, through reversible amorphous to crystalline transitions5.
Phase transitions from one semi-stable state to stable crystal state have been extensively studied by past works and are fairly well understood. However, in-situ study of solid-state phase transitions of amorphous and disordered structures trapped in a metastable state at nano-scale to a semi-stable ordered crystalline state have only been theorized9,10,11. Before any detailed chemical kinetics analyses on such phase transitions can be carried out, the critical bottleneck has been the first step of kinetically trapping and stabilizing nanomaterials in metastable phases in localized free-energy minima via suitable non-equilibrium synthesis routes. Experimentally viable non-equilibrium synthesis routes to kinetically trap and stabilize metastable metal/meta oxide phases remain elusive with most attempts employing ultra-fast quenching, physical/chemical deposition, high-pressure compression, and mechanical attrition techniques12,13,14,15. The similarity in these systems being their non-equilibrium nature that increases the metal’s internal entropy through rapid energy dumping and quenching before transitioning to a final stable oxide form. Laser Ablation Synthesis in Solution (LASiS) is one of the few methods known that can effectively trap these forms of nanoscale metastable oxides; however, the mechanistic underpinning of such trapping process in high-T/high-P plasmas is still an active area of investigation. It has been long-postulated that the highly non-equilibrium LASiS processes can promote this kinetic trapping of metastable states, via rapid laser energy dumping and liquid-phase quenching; however, this has rarely been seen experimentally12,13,16,17. To that end, our group has been working to develop and demonstrate the versatility of a specific class of reactive LASiS technique termed as Laser Ablation Synthesis in Solution-Galvanic Replacement Reaction (LASiS-GRR) (Pat.: US 10,326,146 B2 (2019); Div. Pat.: 11,127,956 B2 (2021))18,19 that interfaces laser ablation under solutions with desired solution-phase reaction chemistry to synthesize disparate functional composite nanoparticles (NPs). Specifically, we have employed LASiS-based techniques in the past to synthesize a diverse library of functional nanomaterials for electrocatalytic, supercapacitive, and energetic applications that include, morphologically tailored CoOx NPs20, binary and ternary nanoalloys/nanocomposites,20,21,22,23,24 hybrid GO-interfaced MOx NCs25, and graphitic shell coated Al NPs26 for their use as superior electrocatalysts as well as high-energy density energetic solid propellants. Recently, Davis et al. has implemented LASiS to kinetically trap metastable hyper-oxidized amorphous-AlOx nanostructures uniquely phase-stabilized by ordered carbon monolayers in carbonaceous matrices (m-AlOx@C; x ~ 2.5-3.0)15. These highly disordered amorphous-AlOx nanostructures become the primary material of focus as their SS-PC characteristics are investigated.
Specifically, in-situ high-temperature quantitative X-ray diffraction (HTXRD) studies are used to carry out the detailed chemical kinetics analyses of solid-solid phase transitions of highly disordered (amorphous) and metastable AlOx (m-AlOx) NPs, stabilized by ordered C-shells into semi-stable θ/γ-Al2O3 polymorphic phases via disproportionation reaction. HTXRD provides a direct characterization method that can probe the kinetics of phase transformations in real time by rapidly collecting diffraction data during heating under non-isothermal and isothermal conditions. This allows for the identification of the LASiS-synthesized metastable and non-equilibrium phases and the kinetic pathways to their final conversion to lower energy stable equilibrium phases. The kinetics of the phase transition can be tracked by analyzing the growth via the peak area which corresponds to the time/temperature-dependent crystallization progress for the θ/γ-Al2O3 polymorphic phase. For isothermal studies, the evolution of crystallization can be tracked as a function of time for a specific temperature. The ensuing extent of conversion for the m-AlOx to the stable crystalline θ/γ-Al2O3 phase measured over time generates the characteristic time-dependent iso-conversion curves that are subsequently used to determine the reaction model that best fits the phase transition kinetics. The following section presents the detailed results for our kinetics study from in-situ HTXRD measurements that indicates a distinct volume shrinkage process during the phase transformation. These kinetic measurements provide crucial information on two SS-PCM parameters as discussed earlier - the relative thermal energy storage and related activation energy barriers during solid-state m-AlOx → θ/γ-Al2O3 phase transitions.
Experimental methods
The particles used via laser ablation synthesis in organic solvents or LASiS. A Q-switched Nd-YAG pulsed laser operating at 1064 nm with 4 ns pulse width, 10 Hz repetition rate and a maximum energy of 180 mJ/pulse was used for the breakdown of acetone and ablation of a pure aluminum target purchased from Kurt J. Lesker Company (99.95% purity, 0.65 cm diameter × 0.65 cm height). The Al target was submerged in acetone (> 99.9% pure, purchased from Sigma-Aldrich with the main impurity being < 0.1% water by volume). Ablation took place at ambient temperature once the solvent had been bubbled with nitrogen to minimize the concentration of diffused oxygen. All samples were ablated in a constant volume of solvent (35 ml) for a time of 10 min. Subsequently, after ablation, the NPs were collected with centrifugation at 4700 rpm, gathered, and then dried overnight under vacuum. The dried materials were used for the following characterization experiments. The high temperature X-ray diffraction studies (HTXRD) were carried out on a Malvern PANalytical Empyrean X-ray diffractometer with Cu radiation and a PIXcel detector. HTXRD data were collected using an Anton Paar HTK1200N environmental chamber. For all five HTXRD isothermal condition tests, a fresh sample was placed in the environmental chamber on a heating stage. The samples were then heated to their respective test condition temperatures of 750 oC, 760 oC, 770 oC, 780 oC and 790 oC at a ramp rate of ~50 oC/min. Once at the desired temperature, the samples were monitored until the θ/γ-Al2O3 peak no longer increased in intensity and the environmental chamber was allowed to cool in air roughly one hour after no growth occurred. Once collected, the data was analyzed utilizing a Malvern Panalytical HighScore data analysis package with a batch processing program written to fit and calculate the peak area growth throughout time for each temperature step. The transmission electron microscopy (TEM) imaging coupled with electron energy loss spectroscopy (EELS) used for the areal density measurements were carried out on a Zeiss Libra 200 MC Electron Microscope operating at 200 keV. The in-situ heating S/TEM measurements were conducted using a Protochips heating holder on a JEOL NEOARM operating at 200 kV at the Center for Nanophase Materials Sciences at Oak Ridge National Laboratory (ORNL). The sample was first heated in the S/TEM to 550 oC while placed under a beam shower condition to remove excess hydrocarbons. Once the sample was cleaned, the temperature was increased at ~100 oC/Min to ~710 oC where the desired phase transition was observed.
Results
AlO3 density measurements: (a) Chosen NP for density measurement at the center of the AlO3 NP indicated as “beam”. (b) EELS spectra for chosen particle in (a) marked in blue with model fit as orange.
Phase-stabilized structures of the amorphous and highly disordered m-AlOx@C nanocomposites, as synthesized via LASiS in our previous work, were studied here using HRTEM to carry out areal density measurements15. Having previously reported on the structure-composition properties of the m-AlOx NPs trapped in a non-equilibrium phase and stabilized by C-shell monolayers, this work focuses on the material’s phase change characteristics. Using HRTEM EELS, the electron-beam scattering probability (ppm/eV) as a function of the electron energy loss could be estimated from the Al and O spectrum. The EELS spectra were then used to calculate the approximate number of Al and O atoms. By fitting a model of scattering probability vs. eV to the raw data, the number of Al and O atoms for each particle could be extrapolated27,28. Figure 2b shows the model fit compared to the EELS spectra of the particle shown in Fig. 2a with the electron beam location marked as “Beam”. Knowing both the number of atoms present as well as the particle’s respective size, the effective areal density of the NPs could be calculated and compared to that of standard Al2O3 NPs of the same particle size. This was done for five different particles of similar sizes. The average areal density of Al atoms in the 100 nm thick Al2O3 standard was found to be roughly ~1,500 atoms/nm2. Table 1 provides the average volumetric density ratios for five NPs measured compared to the average Al2O3 NP areal volumetric density. The particles on average are ~6–7 times less dense than an Al2O3 NP of the same size.
HTXRD data and analysis of AlO3 nano composites: (a) HTXRD taken at a temperature of ~700oC and ~800oC showing a broad peak for theta/gamma alumina. (b) Amorphous to theta/gamma HTXRD at ~750oC showing the θ/γ-Al2O3 peak intensity increase as a function of time.
The phase transition from θ/γ-Al2O3 to α-Al2O3 has been well studied throughout other publications. However, the study of an amorphous metastable material phase transitioning to its first semi-stable crystal structure has been less researched till date11,29,30. For this reason, the focus of this work is solely on the first phase transition from m-AlOx/C to θ/γ-Al2O3. In our previous publication, we observed that the AlOx/C nanocomposites began to transition from their initial amorphous phase to θ/γ-Al2O3 (700 oC < T < 900 oC) and finally to α-Al2O3 (T > 900 oC)15. During the initial transition to the semi-stable θ/γ-Al2O3, the material undergoes a disproportionation reaction, where AlOx with an intermediate oxidation state (O-1) converts to Al2O3 with a higher oxidation state (O− 2) and O2 (g) with a lower oxidation state (0) as shown in the reaction equation below (Eq. 1). Eq. 2 shows that the O anion must be -1 to charge balance AlOx (x~3) and Eq. 3 (Eq. 3) shows that the O anion must be -2 to charge balance Al2O3. This also corroborates the observations from our earlier work that had indicated a large percentage of low coordinated O− species, thereby allowing us to ascribe the − 1 oxidation state to the oxygen in the AlOx NPs13,15,31
This makes the m-AlOx@C phase transition fairly unique since the majority off SS-PCMs undergo oxidation or simply bond rearrangements resulting in matching oxidation states for both reactant and product. The only other disproportionation reactions in SS-PC materials are known to occur in peroxide phase transitions31,32.
Conversion factor alpha (α) vs. time for isothermal temperatures of: (a) 750 oC, and (b) 790 oC compared for a set of known solid phase change models.
In-situ high-temperature quantitative X-ray diffraction (HTXRD) studies are used to determine the detailed chemical kinetics analyses of solid-solid phase transitions of the highly disordered (amorphous) and metastable AlOx (x~2.5-3.0) stabilized by ordered C-shells into the semi-stable θ/γ-Al2O3 phase via disproportionation reaction. HTXRD provides a characterization method that can probe the kinetics of phase transformations in real time by rapidly collecting diffraction data during heating under both non-isothermal and isothermal conditions. This allows for identification of metastable and non-equilibrium phases and kinetic pathways. The kinetic phase transition can be tracked by analyzing the increase in peak area which corresponds to crystallization. For isothermal experiments the evolution of crystallization can be tracked as a function of time for a particular temperature. The ensuing extent of conversion, denoted as α, can be experimentally measured as the peak area measured over time divided by the maximum crystalized area or α=Aobserved/Amax. Focusing on the strongest intensity for the visible θ/γ-Al2O3 peak, centered around 2~45.5o , as shown in Fig. 3a, for five separate isothermal conditions, between 750 and 790oC, HTXRD data were collected until the peak reached its maximum intensity and no longer increased. Figure 3b displays the emergence and increase in intensityof the θ/γ-Al2O3 peak as time progresses, illustrating the material’s amorphous to polycrystalline transition with the increase in intensity relating to the concentration of crystallites nucleated. Even after allowing for the appropriate time to achieve the maximum transformation to θ/γ-Al2O3 crystals, the peaks for all temperatures retain low relative intensity and high broadening (see figure S1). This could be due to their polycrystalline nature with a small crystallite size of ~3–4 nm15. Due to the small crystallite sizes, peak overlaps, and short count times needed for the in-situ HTXRD, the analysis of the data for each isothermal data set was challenging. However, even at the onset of crystallization, the peaks were discernible from the background and care was taken to keep the data analysis procedure consistent. This made the comparisons for the change in peak areas plausible and the deviations between measured areas relatively small.
In this case the α vs. time curve can be used to determine the kinetic model which best describes the kinetic phase transition. The most common kinetic models can be broken into four main categories, nucleation, geometrical contraction, diffusion, and reaction-order models33,34,35. In the case of most solid-state crystallization, the reaction is described by nucleation models or Avrami models. In the case of crystal nucleation, the reaction governing rate is driven by the nucleation rate at heterogeneities within the lattice known as nucleation sites. Nucleation models can be driven by exponential, linear, power or instantaneous equations and are therefore often quite fast in comparison to other transition models. For diffusion-controlled phase transitions, the mobility of constituents through the lattice is the rate limiting factor. In diffusion driven reactions such as metallic oxidation, the rate of product formation along the reaction wall decreases the mobility of constituents as the thickness of the product barrier layer increases. Phase boundary-controlled reactions such as geometric contraction models assume that nucleation of the phase change takes place at the surface and subsequent growth is the rate limiting factor. Reaction order models are simplistic models that assume that the reaction rate is proportional to concentration or number of reactants present and decrease as reactants convert to product. The areas for the resulting peaks throughout each isothermal test condition were obtained by fitting each peak using the HighScore data analysis package and then normalizing to the largest peak area at the conclusion of each isothermal data collection. This gives us the effective extent of conversion, α for each isothermal temperature measured throughout time. Figure 4 shows α vs. time for the lowest and highest isothermal temperatures. The total conversion time decreases as the temperature increases. The slope, with higher slopes correlating to faster conversion rates, increases as temperature increases, shown in Fig. 5a, thereby promoting the phase transition.
By comparing the extent of conversion data to the possible conversion models a suitable phase transition model could be chosen33,34,35. For all isothermal cases, the volumetric contraction model was found to fit most appropriately (figure S2) and hence, was compared for 750 and 790oC in Fig. 5a. The equation for the volumetric contraction model can be seen below, where f(α) is a mathematically derived representation (Eq. 4) of the reaction model and g(α) is the integral form of f(α) (Eq. 5). In the following equations dα/dt is the rate of conversion with respect to time where k is the conversion rate constant36. The calculated fits in Fig. 5a were obtained by varying the k values to obtain the best agreement to the data resulting in, k = 0.0023 (s− 1) and k = 0.008 (s− 1), for 750 and 790oC, respectively.
Isothermal kinetics analysis of AlOx nanocomposites: (a) HTXRD measured at ~750oC and ~790oC over time fitted to the volumetric contraction model using the fitting rate constants (k). (b) data linearized using the integral expression for volumetric contraction model, g(α).
Once a suitable model is chosen, the two SS-PCMs parameters discussed earlier - the relative thermal energy storage and related kinetics - can be approximated. In the context of using the volumetric contraction model, and given the extreme porosity of the material, the AlOx/C NPs will have to contract considerably to achieve the large change in density in the transitioned θ/γ- Al2O3 polycrystals. This can be seen in the size reduction from the ~10 nm NPs to the ~3–4 nm θ/γ-Al2O3 polycrystals. After determining the appropriate crystallization model, Eq. 5 with the data was used to calculate g(α) as a function of time and compared to a linear fit in the ,region of the data shown in Fig. 5b, where there was minimal scatter and the slope remained constant. Here, the slope for each isothermal plot represents the respective k value or reaction rate constant at the corresponding temperature, agreeing well with the reaction rate constants predicted using the function in Fig. 5a33,35,36. With the reaction rate constant determined, the Arrhenius expression can be used in the Eq. 6 below:
where A is the pre-exponential factor, Ea is the activation energy, R is the ideal gas constant and T is the isothermal temperature considered. The reaction activation energy barrier Ea (kJ/mol) could be approximated by comparing the natural log of k with respect to the isothermal temperature of each reaction and taking the linear fit of the points. The resulting slope of the linear fit divided by the gas constant is equal to the average activation energy barrier or the perturbation energy needed to induce the phase transition.
Activation energy plots for AlO3 nano composites: (a) Arrhenius plot used for calculating the average activation energy needed to induce the phase transition. (b) Schematic of activation energy vs. reaction progression highlighting the barrier for AlOx stuck in localized energy well.
For the AlOx/C nanocomposite the activation energy was found to be ~270±11 kJ/mol (Fig. 6a) approximately 8x of that reported by Park et al., where NPs of similar sizes required only~32 kJ/mol to induce diffusion driven oxidation36, making the AlOx nanocomposites roughly as stable as a micron sized Al particle. The extreme stability of the AlOx/C nanocomposites is likely due to the monolayer carbon atom shell stabilizing this metastable material, as discussed in our earlier work, which in effect increases the material’s activation energy15. Similar to Fig. 1, we can construct a proposed depiction for the kinetic entrapment of the AlO3 nanocomposites as based on our experimental observations and as depicted in Fig. 6b. Eac1 represents the amount of energy required to trap the metastable structure while Eac2 is the energy required to perturb the material enough to induce the SS-PC. ΔE1 is the depth of the energy well that the material is trapped or the formation energy of the AlO3 in the LASiS-induced plasma and ΔE2 is the amount of energy released upon perturbation which in this case is taken as the difference between the formation energies of AlO3 and γ-Al2O3, i.e, ~2 eV/atom (193 kJ/mol).
To investigate the structural evolution during the high-temperature phase transition of the as-synthesized amorphous m-AlOx phase in the NPs into the semi-stable θ/γ-Al2O3 polymorphic phase, in-situ STEM imaging on a high-temperature stage was carried out at a fixed temperature of ~710oC. Due to the STEM environment (high vacuum under electron beam interaction), the low initial temperature was chosen to ensure that the onset of the phase transition and crystallization to the intermediate θ/γ-Al2O3 phase was slow enough to be observed at (Fig. 7). This also ensured that once crystalized the intermediate semi-stable phase, observed in HAADF STEM images and confirmed via d-spacing measurements (Fig. 8), could be verified before the NPs could transition to the final α-Al2O3 phase. Using in-situ STEM, the volume changes for two separate spherical particles were tracked, as indicated in Fig. 7, wherein the starting amorphous volume of the particle was noted to be ~524 nm3 (diameter~10 nm). After ~20 s of isothermal heating at ~710oC, the inspected particles are observed to have decreased to ~45 nm3 (diameter~4.4 nm), thereby undergoing a ~12x volume contraction. This correlates well with our previous analysis for the areal density measurements of the AlOx NPs which notably found the density to be ~10 times less dense than standard alumina. These results further confirmed that the initial metastable to semi-stable phase transition of amorphous m-AlOx to crystalline θ/γ-Al2O3 indeed follows a volume contraction process wherein the size of the particle does in fact decrease in order for the densification to occur and facilitate the crystal growth.
In-situ heating STEM showing the decrease in size of the AlOx NPs during transition from amorphous to crystalline.
STEM analysis of post-heated NP shown in Fig. 7 indicating: (a) clear crystalline lattice spacings of roughly 0.28 nm matching the γ-Al2O3 phase in (220) crystal orientation. (b) line plot for the the crystal lattice spacings accross the NP shown in (a).
After the tracked volume contraction, detailed inspection of the NP in the final frame of Fig. 7 (time = 44 s) revealed that the particle indicated a measurable d-spacing of ~0.28 nm (shown in Fig. 8a, b) which matches that of the (220) crystal orientation for the γ-Al2O3 polymorphic phase37,38.
Discussion
The first assumption made about the structure of this unique meta-stable hyperoxide was that it should be fairly dense due to the stabilization and the synthesis methods. LASiS-made NPs from the hot plasma conditions are thrown into the surrounding solvent to rapidly quench and at this time almost instantaneously phase stabilize into the meta-stable structure. After quenching, the material is held at this state by an ultra-thin atomic monolayer of carbon at the NP’s outer most edge due to the high dissociation energies of diatomic covalent C − O (~ 11.2 eV) and C − C (~ 6 eV) bonds39,40. It is for this reason that our initial hypothesis was that the AlO3 NPs would be trapped under high internal compression in a densified form, with the carbon monolayer containing the internal structure like a balloon. However, what we instead found was a highly porous material (~3.8 atoms/nm3), several times less dense than common alumina. (~14.6 atoms/nm3). This could mean that as the NPs exit the plasma, they instead undergo rapid expansion before phase stabilizing due to them transitioning from an ultra high-pressure plasma to the vastly lower pressure (atmospheric) solvent. The pressure difference going from the plasma conditions to atmospheric solvent can be as high as GPa in magnitude41.
Due to the starting composition and final product composition, we could confidently state that oxidation was not occurring during phase transition as tracked by isothermal HTXRD, so diffusion limited transformation models are not applicable. However, a phase transition boundary could be present which would resemble a diffusion transition. To rule this out a two-model system which would assume a fast nucleation followed by a slowed boundary layer diffusion driven transition was attempted. This would result in a two-reaction rate and a two-activation energy approximation. When compared to the data, however, the two-model system did not easily fit with the determined slopes at all isothermal temperatures. Hence, it was determined that the three-dimensional contracting volume model fit better in all the cases and was concluded to be the simplest and most direct method for the aforesaid phase transition. Even before the direct visualization of the volumetric shrinkage, the three-dimensional contracting volume model made logical sense given the reaction steps that would be needed during this phase transition. The starting material in question, AlO3, was known to be hyperoxidized and substantially less dense than the γ-Al2O3 phase product. This meant that the AlO3 NPs would first need to release the excess trapped oxygen before shrinking to accommodate the denser lattice structures for γ-Al2O3 phase. This leads us to conclude that the release of the excess trapped oxygen and the nucleation of the γ-Al2O3 crystals must be relatively quick when compared to the shrinkage of the starting NPs, otherwise those would be the transition rate determining processes.
Ourfinding of the high activation energy barrier makes the AlO3 nanocomposites an extremely stable form as a solid energy storage material. However, as discussed earlier, this could make it difficult to tap into the estimated ~2 eV/atom due to its high stability. Past works have focused primarily on the phase transition between the semi-stable θ/γ-Al2O3 to ground state α-Al2O3. In this study we focused solely on the metastable to semi-stable phase change; this was because the formerly mentioned phase changes have been extensively studied and they tend to be the least energy releasing step of the process. As shown in Figure 6b, the formation energy difference for the phase transition from θ/γ-Al2O3 to α-Al2O3 is relatively small, relinquishing at the most ~0.4 eV/atom with a small activation energy barrier, making it unstable during storage. Current aluminum air (Al-air) batteries function using the reaction driven formation of Al(OH)3 with the addition of an electrolyte, typically potassium hydroxide (KOH), with a formation energy difference of ~2 eV/atom. The phase change material presented here has the potential to have similar energy densities to that of current Al-air batteries with the main difference being the reaction kinetics. Additionally, the oxygen release during reaction could allow for a secondary reaction with a conventional Al anode allowing for a closed loop, fully closed cell design. More study is required, though the potential for such revolutionary metastable nanocomposites could hold the key to advancing a plethora of new and emerging technologies.
Summary
In conclusion, we have implemented HTXRD and HRTEM in analyzing the phase change of a metastable AlOx nanocomposite to the slightly more globally stable alumina polymorph γ/θ-Al2O3. The premise for the phase change and the materials studied were briefly discussed in our previous publication15. HRTEM EELS, spectral data for 5 separate NPs were gathered and their approximate Al/O atomic areal densities to calculated areal densities of Al2O3 of roughly the same size were compared. The synthesized particles measured between 5 and 10 times less dense than standard Al2O3. The AlOx/C nanocomposite was then examined using isothermal HTXRD and crystallization was tracked throughout time for temperatures ranging from 750 oC to 790 oC. The resultant phase extent of conversion was compared to known theoretical models with the three-dimensional constricting volume model matching best for all temperatures. Once linearized using the chosen model, the Arrhenius parameters were determined, that led to the activation energy of ~270 ± 11 kJ/mol being significantly higher than that of similar sized Al particles. Finally, we use in-situ heating STEM to track the predicted shrinkage and visually confirm our model. During STEM heating the particle volumetric shrinkage ratio was measured to be roughly ~12 which was close to the volumetric density analysis which predicted that it would need to around 5 to 10 times smaller before being dense enough to crystalize to θ/γ-Al2O3.
Data availability
The data used throughout the publication was collected in whole by the authors and will be provided upon fair request to the corresponding author.
References
Fallahi, A., Guldentops, G., Tao, M., Granados-Focil, S. & Van Dessel, S. Review on solid-solid phase change materials for thermal energy storage: molecular structure and thermal properties. Appl. Therm. Eng. 127, 1427–1441. https://doi.org/10.1016/j.applthermaleng.2017.08.161 (2017).
Esposito, V. & Castelli, I. E. Metastability at defective metal oxide interfaces and nanoconfined structures. Adv. Mater. Interfaces. 7 (13), 1902090 (2020).
Liu, C. et al. Metastable Crystalline Phase in two-Dimensional Metallic Oxide Nanoplates. Angew. Chem. 131 (7), 2077–2081 (2019).
Huang, J. C. et al. -Y. A simple approach to achieve a metastable metal oxide derived from carbonized metal–organic gels. Chem. Commun. 55 (31), 4475–4478 (2019).
Blatter, A. & von Allmen, M. Reversible amorphization in laser-quenched Titanium alloys. Phys. Rev. Lett. 54 (19), 2103–2106. https://doi.org/10.1103/PhysRevLett.54.2103 (1985).
Raj, C. R., Suresh, S., Bhavsar, R. R. & Singh, V. K. Recent developments in thermo-physical property enhancement and applications of solid solid phase change materials. J. Therm. Anal. Calorim. 139 (5), 3023–3049. https://doi.org/10.1007/s10973-019-08703-w (2020).
Pogatscher, S., Leutenegger, D., Schawe, J. E. K., Uggowitzer, P. J. & Löffler, J. F. Solid–solid phase transitions via melting in metals. Nat. Commun. 7 (1), 11113. https://doi.org/10.1038/ncomms11113 (2016).
Davies, G. & Evans, T. Graphitization of diamond at zero pressure and at a high pressure. Proc. Royal Soc. Lond. Math. Phys. Sci. 328 (1574), 413–427 (1972).
Lamouri, S. et al. Control of the γ-alumina to α-alumina phase transformation for an optimized alumina densification. Boletín De La. Sociedad Española De cerámica Y Vidrio. 56 (2), 47–54 (2017).
Macêdo, M. I. F., Bertran, C. A. & Osawa, C. C. Kinetics of the γ → α-alumina phase transformation by quantitative X-ray diffraction. J. Mater. Sci. 42 (8), 2830–2836. https://doi.org/10.1007/s10853-006-1364-1 (2007).
Brazhkin, V. V. Metastable phases, phase transformations, and phase diagrams in physics and chemistry. Phys. Usp. 49 (7), 719 (2006).
Scaramuzza, S., Agnoli, S. & Amendola, V. Metastable alloy nanoparticles, metal-oxide nanocrescents and nanoshells generated by laser ablation in liquid solution: influence of the chemical environment on structure and composition. Phys. Chem. Chem. Phys. 17 (42), 28076–28087. https://doi.org/10.1039/c5cp00279f (2015).
Li, Z. et al. Aluminum Oxide Nanoparticle films deposited from a Nonthermal plasma: synthesis, characterization, and crystallization. ACS Omega. 5 (38), 24754–24761. https://doi.org/10.1021/acsomega.0c03353 (2020).
Rondinone, A. J., Pawel, M., Travaglini, D., Mahurin, S. & Dai, S. Metastable tetragonal phase CdWO4 nanoparticles synthesized with a solvothermal method. J. Colloid Interface Sci. 306 (2), 281–284. https://doi.org/10.1016/j.jcis.2006.10.064 (2007).
Davis, E. M., Duscher, G., Wen, J. & Mukherjee, D. Laser-Induced Trapping of Metastable Amorphous AlOx/C (2.5 < x ≤ 3.5) nanocomposites: implications for Use as Solid Phase Gas Generators. ACS Appl. Nano Mater. 6 (13), 10977–10985. https://doi.org/10.1021/acsanm.3c00473 (2023).
Amendola, V. & Meneghetti, M. What controls the composition and the structure of nanomaterials generated by laser ablation in liquid solution? Phys. Chem. Chem. Phys. 15 (9), 3027–3046. https://doi.org/10.1039/c2cp42895d (2013).
Amendola, V. et al. Coexistence of plasmonic and magnetic properties in Au89Fe11 nanoalloys. Nanoscale 5 (12), 5611–5619. https://doi.org/10.1039/C3NR01119D (2013).
Mukherjee, D. Compositions, Systems and Methods for Producing Nanoalloys and/or Nanocomposites Using Tandem Laser Ablation Synthesis in solution-galvanic Replacement Reaction (US, 2019).
Mukherjee, D. Compositions, Systems and Methods for Producing Nanoalloys and/or Nanocomposites Using Tandem Laser Ablation Synthesis in solution-galvanic Replacement Reaction (US, 2021).
Hu, S., Melton, C. & Mukherjee, D. A facile route for the synthesis of nanostructured oxides and hydroxides of cobalt using laser ablation synthesis in solution (LASIS). Phys. Chem. Chem. Phys. 16 (43), 24034–24044. https://doi.org/10.1039/c4cp03018d (2014).
Hu, S. et al. A facile and surfactant-free route for nanomanufacturing of tailored ternary nanoalloys as superior oxygen reduction reaction electrocatalysts. Catal. Sci. Technol. 7 (10), 2074–2086. https://doi.org/10.1039/c7cy00073a (2017).
Hu, S. et al. Correction: a facile and surfactant-free route for nanomanufacturing of tailored ternary nanoalloys as superior oxygen reduction reaction electrocatalysts. Catal. Sci. Technol. 7 (14), 3163–3163. https://doi.org/10.1039/c7cy90063e (2017).
Hu, S., Goenaga, G., Melton, C., Zawodzinski, T. A. & Mukherjee, D. PtCo/CoO x nanocomposites: Bifunctional electrocatalysts for oxygen reduction and evolution reactions synthesized via tandem laser ablation synthesis in solution-galvanic replacement reactions. Appl. Catal. B. 182, 286–296. https://doi.org/10.1016/j.apcatb.2015.09.035 (2016).
Hu, S., Tian, M., Ribeiro, E. L., Duscher, G. & Mukherjee, D. Tandem laser ablation synthesis in solution-galvanic replacement reaction (LASiS-GRR) for the production of PtCo nanoalloys as oxygen reduction electrocatalysts. J. Power Sources. 306, 413–423. https://doi.org/10.1016/j.jpowsour.2015.11.078 (2016).
Hu, S., Erick; Davari, S. A., Tian, M., Mukherjee, D. & Khomami, B. Hybrid nanocomposites of nanostructured Co3O4 interfaced with reduced/nitrogen-doped graphene oxides for selective improvements in electrocatalytic and/or supercapacitive properties. RSC Adv. 7 (53), 33166–33176. https://doi.org/10.1039/c7ra05494g (2017).
Davari, S. A. et al. Graphitic coated Al nanoparticles manufactured as superior energetic materials via laser ablation synthesis in organic solvents. Appl. Surf. Sci. 473, 156–163. https://doi.org/10.1016/j.apsusc.2018.11.238 (2019).
Tian, M., Dyck, O., Ge, J. & Duscher, G. Measuring the areal density of nanomaterials by electron energy-loss spectroscopy. Ultramicroscopy 196, 154–160. https://doi.org/10.1016/j.ultramic.2018.10.009 (2019).
Hofer, F. Determination of inner-shell cross-sections for EELS-quantification. Microsc Microanal Microstruct. 2 (2–3), 215–230 (1991).
Stølen, S., Glöckner, R. & Grønvold, F. Nearly stoichiometric iron monoxide formed as a metastable intermediate in a two-stage disproportionation of quenched wüstite. Thermodynamic and kinetic aspects. Thermochim. Acta. 256 (1), 91–106. https://doi.org/10.1016/0040-6031(94)02166-L (1995).
Ren, X., Zhao, M., Feng, J. & Pan, W. Phase transformation behavior in air plasma sprayed yttria stabilized zirconia coating. J. Alloys Compd. 750, 189–196. https://doi.org/10.1016/j.jallcom.2018.03.011 (2018).
Dupin, J. C., Gonbeau, D., Vinatier, P., Levasseur, A. & Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2 (6), 1319–1324. https://doi.org/10.1039/A908800H (2000).
Luo, Y., Tan, W., Suib, S. L., Qiu, G. & Liu, F. Dissolution and phase transformation processes of hausmannite in acidic aqueous systems under anoxic conditions. Chem. Geol. 487, 54–62. https://doi.org/10.1016/j.chemgeo.2018.04.016 (2018).
Khawam, A. & Flanagan, D. R. Solid-state kinetic models: basics and Mathematical fundamentals. J. Phys. Chem. B. 110 (35), 17315–17328. https://doi.org/10.1021/jp062746a (2006).
Salasin, J. R. & Rawn, C. In-Situ Kinetic Investigation of Calcium Aluminate formation. Ceramics 1 (1), 175–197 (2018).
Gotor, F. J., Criado, J. M., Malek, J. & Koga, N. Kinetic Analysis of Solid-State reactions: the universality of Master plots for analyzing Isothermal and Nonisothermal experiments. J. Phys. Chem. A. 104 (46), 10777–10782. https://doi.org/10.1021/jp0022205 (2000).
Park, K., Lee, D., Rai, A., Mukherjee, D. & Zachariah, M. R. Size-resolved kinetic measurements of aluminum nanoparticle oxidation with single particle Mass Spectrometry. J. Phys. Chem. B. 109 (15), 7290–7299. https://doi.org/10.1021/jp048041v (2005).
Nelson, A. E., Sun, M. & Adjaye, J. Reply to comments on ‘Examination of spinel and Nonspinel Structural models for γ-Al2O3 by DFT and Rietveld Refinement simulations’. J. Phys. Chem. B. 110 (41), 20724–20726. https://doi.org/10.1021/jp0616720 (2006).
Sun, M., Nelson, A. E. & Adjaye, J. Examination of spinel and Nonspinel Structural models for γ-Al2O3 by DFT and Rietveld Refinement simulations. J. Phys. Chem. B. 110 (5), 2310–2317. https://doi.org/10.1021/jp056465z (2006).
Zhang, M. et al. In-situ conversion of amorphous carbon to graphene enhances the oxidation resistance of dendritic copper powder. Diam. Relat. Mater. 120, 108695 (2021).
Beattie, J. M. A., Goss, J. P., Rayson, M. J. & Briddon, P. R. Electron-affinity and surface-stability of aluminium-oxide terminated diamond surfaces. Diam. Relat. Mater. 94, 137–145. https://doi.org/10.1016/j.diamond.2019.02.009 (2019).
Yang, G. W. Laser ablation in liquids: applications in the synthesis of nanocrystals. Prog. Mater. Sci. 52 (4), 648–698. https://doi.org/10.1016/j.pmatsci.2006.10.016 (2007).
Acknowledgements
We acknowledge the core XRD and Microscopy facilities and the Institute for Advanced Materials and Manufacturing (IAMM) at the University of Tennessee, Knoxville, for access to and assistance with various instruments. We would also like to acknowledge the technical and research assistance from Dr. Gerd Duscher the Institute for Advanced Materials and Manufacturing (IAMM). We acknowledge the use of in-situ heating S/TEM at the Center for Nanophase Materials Sciences at Oak Ridge National Laboratory (ORNL) through a user proposal access. The in situ S/TEM work performed at the Center for Nanophase Materials Sciences, a U.S. Department of Energy Office of Science User Facility at Oak Ridge National Laboratory, was supported by the U.S. DOE, Office of Basic Energy Sciences.
Author information
Authors and Affiliations
Contributions
Author Initials: Elijah M. Davis (ED), Claudia Rawn (CR), Matthew G. Boebinger (MB), Dibyendu Mukherjee (DM). ED and DM carried out the experimental designs, set-up and discussions. ED, DM, CR, and MB carried out the data collection and analysis. ED and DM wrote the main manuscript. ED, DM, CR, and MB edited the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
There are no conflicts of interest to declare for this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Davis, E.M., Rawn, C., Boebinger, M.G. et al. Kinetic analyses for solid-state phase transition of metastable amorphous-AlOx (2.5 < x ≤ 3.0) nanostructures into crystalline alumina polymorphs. Sci Rep 15, 7854 (2025). https://doi.org/10.1038/s41598-025-90586-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90586-z