Abstract
We aimed to compare amyloid-β (Aβ) accumulation rates between different tracers and investigate whether the relationship between changes in Aβ uptake and cognitive decline varies depending on tracer type. Two cohorts were analyzed: (1) a head-to-head longitudinal cohort using 18F-Florbetaben (FBB) and 18F-Flutemetamol (FMM) tracers (n = 13), and (2) separate longitudinal cohorts for each tracer (n = 174 for both FMM and FBB), matched by propensity score. Aβ uptake was measured using regional direct comparison of Centiloid (rdcCL) values. In the head-to-head cohort, subtracting changes in FMM rdcCL from FBB rdcCL yielded median values above zero in all regions except the cingulate. In the individual tracer cohorts, FBB rdcCL showed faster accumulation than FMM rdcCL in all cortical regions except the striatum (β [SE] = − 2.49 to − 1.56 [0.47–0.54], p < 0.001). Mini-Mental State Examination changes were associated with annualized FMM rdcCL changes in the temporal cortex (p = 0.02) and striatum (p = 0.01); however, no such differences were found in the FBB cohort. Our findings suggest that longitudinal Aβ positron emission tomography studies should consider the specific characteristics of tracers depending on the context of use.
Similar content being viewed by others
Introduction
The significance of β-amyloid (Aβ) positron emission tomography (PET) imaging has been emphasized with its ability to diagnose Alzheimer’s disease (AD) even in individuals with normal cognition1. The 11C-labelled Pittsburgh compound B (11C-PiB) tracer was initially used as an amyloid PET tracer2. Subsequently, several PET tracers labelled with fluorine-18(18F ), such as florbetapir (FBP)3, [18F]florbetaben (FBB)4, and [18F]flutemetamol (FMM)5, have been developed and clinically approved. In an effort to standardize these different tracers, a unit called Centiloid (CL) has emerged6. Recently, our group developed regional direct comparison methods for FBB and FMM CL (rdcCL) values in several regions, including the frontal, temporal, parietal, posterior cingulate, occipital, and striatal regions7.
Since Aβ deposition is one of the earliest pathological features of AD, understanding longitudinal changes in Aβ uptake is important for early diagnosis. The emergence of Aβ-targeted therapies underscores the importance of longitudinal studies of Aβ changes8,9. Additionally, Aβ PET studies have enabled the investigation of the relationship between longitudinal changes in Aβ uptake and cognitive changes in vivo, which was not possible with pathological studies. The emergence of various tracers has raised questions regarding differences in imaging characteristics. Understanding the unique properties of these tracers is essential for comparing and accurately interpreting results from diverse Aβ PET studies. A previous study conducted head-to-head comparisons of Aβ PET imaging using FBB and FMM tracers, suggesting that cortical uptakes with these two tracers were highly correlated10. However, FMM showed higher uptake in the striatum compared to FBB These findings highlight the importance of measuring subcortical Aβ deposition clinically, as Aβ staging burden according to striatal involvement predicts poorer clinical outcomes11,12,13. Differences in the longitudinal Aβ changes over time between different tracers and their association with cognitive changes have not yet been extensively evaluated.
In the present study, we aimed to investigate longitudinal changes in Aβ uptake using head-to-head longitudinal studies of FBB and FMM tracers and longitudinal studies for each tracer. First, we compared longitudinal changes in Aβ uptake between FBB and FMM tracers over time. Second, we determined whether the relationship between changes in Aβ uptake and cognitive decline might differ according to the tracer type.
Results
Demographics of the participants
Table 1 presents the demographic data of the participants. The FBB and FMM head-to-head longitudinal cohort consisted of 13 individuals. The propensity score-matched tracer longitudinal cohort consisted of 174 individuals for both FBB and FMM, with no significant differences observed between the two groups in terms of sex (p = 0.91), age (p = 0.42), education (p = 0.81), diagnosis (p = 0.59), or ApoE ε4 carrier ratio (p = 0.73) , or baseline rdcCL (p = 0.81), further supporting the successful matching. During the median follow-up period of 3.1 years, 5 of 53 initially Aβ negative participants (9.4%) in the FBB group and 4 of 50 initially Aβ negative participants (8%) in the FMM group converted to positive.
Longitudinal rdcCentiloid changes between FBB and FMM
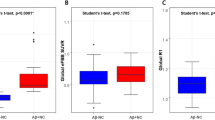
In the head-to-head longitudinal cohort, no significant difference was observed in the change of rdcCL for each tracer because of the small number of participants (Fig. 1a). However, subtracting the changes in FMM rdcCL from FBB rdcCL yielded median values above zero for all regions except the cingulate (Fig. 1b), suggesting that longitudinal rdcCL changes in FBB are larger than those in FMM.
Comparison between FBB rdcCL changes and FMM rdcCL changes in the head-to-head cohort. (a) Displays box plots illustrating the Centiloid change values measured for FBB and FMM across the global, frontal, parietal, cingulate, temporal, occipital cortex, and striatum regions. FBB is represented in red, while FMM is shown in sky blue. (b) Displays box plots representing the difference between FBB Centiloid change values and FMM Centiloid change values across each region. The values are displayed in a box plot format for each specific area. Abbreviation: ROI, region of interest.
In each tracer’s longitudinal studies (Fig. 2 and Supplementary Table S1), the accumulation rate of FBB rdcCL was faster than that of FMM rdcCL in the global cortex (β [SE] = − 1.89 [0.47], p < 0.001), frontal (β [SE] = − 2.49 [0.54], p < 0.001), parietal (β [SE] = − 1.56 [0.47], p = 0.001), cingulate (β [SE] = − 2.39 [0.52], p < 0.001), temporal (β [SE] = − 2.07 [0.47], p < 0.001) and occipital (β [SE] = − 2.09 [0.48], p < 0.001) cortex. However, there were no differences in the accumulation rates in the striatum between FBB and FMM (β [SE] = 0.19 [0.50], p = 0.69). We have presented individual participant data as spaghetti plots in Supplementary Fig. S1.
Comparison of longitudinal Centiloid changes between FBB and FMM. Differences in Aβ accumulation rate changes between FBB rdcCL and FMM rdcCL after propensity score matching. The Aβ accumulation rate of FBB was faster than that of FMM except in the striatum. Bayesian mixed-effects models were performed including age, sex, time (follow-up year), and an Aβ tracer by time interaction as fixed effects along with participant-specific random effects. This graph plots the mean of the predicted values for each follow-up year derived from the predicted model equation using a linear mixed effect model. Error bars are 95% confidence intervals. Colours represent FBB (blue) or FMM (red), lines and 95% CIs represent predicted values from a Bayesian mixed model.
Across all diagnostic subgroups, the direction of the differences in amyloid accumulation rates was largely consistent with the overall cohort, with FBB showing a faster accumulation rate than FMM in most cortical regions. However, statistical significance was only observed in the mild cognitive impairment (MCI) group (Supplementary Table S2).
Association between longitudinal centiloid changes and Mini-Mental State Examination (MMSE) changes
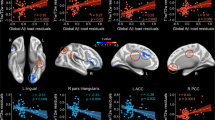
We conducted a stratified analysis and examined interactions based on the annualized rdcCL changes (rdcCL delta) and MMSE changes in the global cortex, frontal, parietal, cingulate, temporal, occipital cortex, and striatum for both FBB and FMM (Fig. 3 and Table 2). In the FMM longitudinal study, significant differences were observed in MMSE changes based on annualized Aβ changes (− 1SD, mean, + 1SD) in the temporal cortex (p = 0.02) and the striatum (p = 0.01). However, in the FBB longitudinal study, no significant differences were observed in MMSE changes based on annualized rdcCL changes (− 1SD, mean, + 1SD) across all regions. When examining the interaction between the two tracers and rdcCL changes over time, significant differences were observed in the temporal cortex (p = 0.03), occipital cortex (p = 0.02), and striatum (p = 0.02).
The longitudinal effect of Aβ accumulation differences between Florbetaben and Flutemetamol on cognition. A stratified analysis and interactions based on the annualized rdcCL changes (CL delta) and cognition (normalized MMSE) in the global cortex, frontal, parietal, cingulate, temporal, occipital cortex, and striatum for both FBB and FMM.
Discussion
In the present study, we compared longitudinal changes in Aβ uptake between FBB and FMM tracers in a head-to-head longitudinal study and the longitudinal studies for each tracer. We found that FBB showed greater uptake across the entire brain lesion, except the striatum, compared to FMM. However, changes in FMM were more reflective of cognitive deterioration compared with those in FBB. Our study suggests that longitudinal Aβ PET follow-up studies should consider tracers with different characteristics depending on the context of use.
Our first major finding was that FBB showed larger changes in uptake across the entire brain region, except the striatum, compared to FMM. This was supported by the following observations: (1) our head-to-head longitudinal study showed that subtracting the changes in FMM rdcCL from FBB rdcCL yielded median values above zero for all regions except the cingulate; (2) the longitudinal studies for each tracer showed that the Aβ accumulation rate of FBB rdcCL was faster than that of FMM rdcCL except in the striatum. Previously, a longitudinal head-to-head study between FBP and PiB showed that the annualized SUVR change in FBP was greater than in PiB14. Given that FBB and FBP were derived from Congo red and FMM and PiB were derived from thioflavin T15,16, our findings, along with previous studies, might be related to the nature of the staining reagents underlying the tracers. After stratification into three cognitive stages, statistical significance was only observed in the MCI group. This is likely due to the well-documented trajectory of amyloid accumulation, where the most substantial changes occur during the MCI stage, whereas cognitively unimpaired individuals (CU) exhibit slower accumulation, and dementia patients may have already reached a saturation point17. These findings suggest that differences in tracer-specific accumulation rates are most detectable in individuals undergoing active amyloid deposition, such as those in the MCI stage.
We found no significant difference in Aβ deposition rate between FBB and FMM in the striatum, unlike in other regions. In a previous head-to-head cross-sectional study for Aβ deposition in the striatum, FMM SUVR values were significantly higher than FBB SUVR values10. Recent studies have shown that FMM PET can accurately detect histologically-proven Aβ deposits in the striatum, where diffuse plaques with low fibrillarity of Aβ aggregates may be predominant18,19. Considering that Aβ PET is commonly used to measure the presence of neuritic plaques with high fibrillarity of Aβ aggregates20, our findings suggested that there were no differences in detecting longitudinal changes of diffuse plaques between FMM and FBB.
Another major finding was that changes in FMM better reflected cognitive deterioration compared with those in FBB. Previous pathological studies had suggested a marginal or restricted effect of Aβ on cognitive decline and AD progression21. However, the subsequent availability of Aβ PET imaging revealed that Aβ ( +) changes were associated with cognitive decline15,22. In the present study, greater increases in the FMM rdcCL values, but not FBB rdcCL, in the temporal and striatum regions were associated with more pronounced cognitive decline. Recent Aβ PET studies have demonstrated that striatal involvement of Aβ predicts a fast cognitive decline11,12,13. Therefore, our findings suggested that longitudinal studies with FMM PET would be more beneficial in monitoring cognitive trajectories along with Aβ changes, compared with FBB.
Based on these findings, the choice of tracer for Aβ PET longitudinal tracking should be based on the context of use. If longitudinal changes in Aβ uptake over time are important, such as in disease modifying therapy (DMT) trials8,23, FBB appears to be more appropriate. On the other hand, if longitudinal changes in Aβ uptake over time and cognitive changes are to be assessed together, FMM seems to be more appropriate, especially in scenarios such as follow-up visits or prognostic assessment in observational cohort studies.
The strength of our study lies in its ability to assess the characteristics of FBB and FMM tracers in a direct head-to-head comparison study and larger longitudinal studies of each tracer, providing a comprehensive understanding of the features of each tracer. However, there are some limitations. First, we could not determine whether changes in Aβ uptake on PET reflect true changes in Aβ burden or an overestimation of amyloid pathology because we did not have the standard pathological confirmation. While pathological studies provide critical insights, they are inherently cross-sectional and cannot directly capture longitudinal changes in Aβ burden. Second, due to the small sample size in the head-to-head cohort, we could only present preliminary results. To address this limitation, we calculated effect sizes to better illustrate differences between tracers. Larger cohorts will be needed to confirm these findings. Third, these findings should be interpreted with caution, as MMSE changes are influenced by multiple factors, such as tau burden and vascular pathology, which were not accounted for in this study. Fourth, while the Centiloid scale standardizes inter-tracer differences, longitudinal changes may reflect technical artifacts from SUVR-to-Centiloid conversion or non-biological factors rather than true biological differences. These limitations emphasize the need for careful interpretation of our findings, as the Centiloid scale inherently relies on cerebellar gray matter as a reference, limiting flexibility in accounting for tracer-specific characteristics. Fifth, alternative reference regions, such as white matter or composite regions, have been proposed for greater stability in longitudinal analyses. However, these approaches are incompatible with the Centiloid framework. Thus, our findings are constrained by the standardized methodology of the Centiloid scale, and future studies outside this framework may help better understand tracer-specific behavior and biological differences. Finally, our study utilized real-world data without fixed follow-up intervals, limiting the applicability of certain analyses, such as Bland–Altman plots, which assume uniformly distributed time points. Instead, we employed propensity score matching and mixed effects models to account for variability in follow-up timing and ensure robust estimates of tracer-specific differences. Nevertheless, our study is crucial in providing insights into the distinctive characteristics and discrepancies among Aβ tracers, which would be valuable for future research in DMT and beyond.
In conclusion, we examined the longitudinal characteristics of FBB and FMM Aβ tracers using head-to-head and individual tracer longitudinal cohorts. FBB was found to be more sensitive than FMM in reflecting Aβ accumulation in most brain regions except the striatum, while changes in FMM were more reflective of cognitive decline. Our findings highlight the importance of considering the characteristics of different tracers based on the context of use when tracking Aβ deposition.
Methods
Participants
The FBB and FMM head-to-head longitudinal cohort consisted of a total of 13 individuals, including cases of CU (n = 5), MCI (n = 4), and dementia (n = 4). Additionally, the longitudinal cohorts consisted of 189 individuals for FBB and 211 individuals for FMM. After propensity score matching (Supplementary Fig. S2) for age, sex, apolipoprotein E (APOE4) carrier status, clinical stage, and baseline global rdcCL value, 174 individuals remained in both groups. CU individuals over 55 years had (1) no history of neurologic or psychiatric disorders and (2) normal cognitive function, determined using neuropsychological tests24. MCI diagnosis was based on Petersen’s criteria involve patient or caregiver reports of cognitive issues, cognitive test scores that are more than 1.0 standard deviation (SD) below the norms adjusted for age, sex, and education in at least one cognitive area, preserved ability to perform daily activities, and no signs of dementia25. Dementia included Alzheimer’s type (DAT) and subcortical vascular dementia (SVaD). DAT followed the National Institute on Aging and Alzheimer’s Association research criteria for probable AD26, while SVaD included patients with subcortical vascular features and significant ischemia seen on magnetic resonance imaging (MRI) scans, defined as white matter hyperintensities on T2-weighted or fluid-attenuated inversion recovery images27.
All participants underwent clinical interviews, neurological and neuropsychological examinations, and laboratory tests such as complete blood count, blood chemistry, vitamin B12/folate, syphilis serology, and thyroid function tests. Brain MRI scans were conducted to ensure the absence of structural abnormalities like cerebral infarctions, brain tumors, hippocampal sclerosis, and vascular malformations.
The institutional review board of Samsung Medical Center (No. 2020–12-177) approved this study. Informed consent was obtained from all subjects and/or their legal guardian(s). All methods were performed in accordance with the relevant guidelines and regulations, including the Declaration of Helsinki.
Brain MRI acquisition
All participants underwent standardized three-dimensional T1 turbo field echo imaging using the same 3.0-T MRI scanner (Philips Achieva; Philips Healthcare, Andover, MA, USA). The imaging parameters were as follows: a sagittal slice thickness of 1.0 mm with contiguous slices and 50% overlap, no gap between slices, a repetition time of 9.9 ms, an echo time of 4.6 ms, a flip angle of 8°, and a matrix size of 240 × 240 pixels, reconstructed to 480 × 480 over a field of view of 240 mm.
Aβ PET acquisition
Participants underwent FBB PET and FMM PET scans at Samsung Medical Center using a Discovery STe PET/computed tomography (CT) scanner (GE Medical Systems, Milwaukee, WI, USA) in three-dimensional scanning mode, covering 47 slices with a thickness of 3.3 mm across the entire brain. The scans were performed on separate days, with no significant differences in the mean interval times (4.0 ± 2.5 months across all groups, p = 0.89). The median longitudinal follow-up interval was 2.98 years for FBB and 2.87 years for FMM. CT images were obtained using a 16-slice helical CT (140 keV, 80 mA; 3.75-mm section width) for attenuation correction. The PET scans were conducted 90 min after injection of mean doses of 311.5 MBq FBB and 185 MBq FMM, respectively, following protocols recommended by the tracer manufacturers. The emission PET scans lasted 20 min in dynamic mode (consisting of 4 × 5 min frames). Three-dimensional PET images were reconstructed using the ordered-subsets expectation maximization algorithm in a 128 × 128 × 48 matrix with a voxel size of 2 mm × 2 mm × 3.27 mm (FBB iterations = 4 and subset = 20; FMM iterations = 4 and subset = 20).
Aβ PET quantification using rdcCL methods
We used a methodology to direct comparison Centiloid (dcCL) units for FBB and FMM PET scans without the need to convert to the PiB SUVR28, and data were processed using Statistical Parametric Mapping version 8 (SPM8) for normalization. Global and regional Centiloid volumes of interest (VOIs) were created using the whole cerebellum mask from The Global Alzheimer’s Association Interactive Network as the reference region. The common area of Aβ accumulation in FMM and FBB PET images was determined using images from 25 Aβ-positive and 18 Aβ-negative patients in a comparative cohort. For each participant, four average images were generated for both positive and negative groups using each tracer. Positive-dominant FBB and FMM cortical (CTX) VOIs were defined by subtracting negative from positive images for each tracer. The FBB-FMM CTX VOI comprised the top 20% of voxels with the strongest intensity among the overlapping regions of these VOIs28.
The process of calculating MRI-based regional CL values involved obtaining standardized uptake value ratios (SUVR) from normalized PET images using VOIs derived from the whole cerebellum mask and specific VOIs for five subregions (Frontal, Parietal, Posterior Cingulate, Temporal, and Striatum) based on the Automated Anatomical Labeling atlas. Intensity of each subregion was divided by the intensity from the whole cerebellum VOI, acting as the reference region, to determine regional SUVR values. The sum of these five subregion VOIs constituted the VOI for the whole cortex. Similarly, SUVR values for the entire cortex were obtained using the same method. These SUVR values for the whole cortex were converted into CL values using the CL conversion formula.
\(SUVR_{ind}\) refers to individual SUVR values, while \(SUVR_{ADCI - 100}\) and \(SUVR_{YC - 0}\) represent the average SUVR values of AD participants and young healthy controls, respectively, corresponding to CL values of 100 and 0 from our cohort in previous study28. This process resulted in an equation to transform SUVR values from the entire cortex into CL values. The same approach was used to derive regional CL values by applying the steps to regional SUVR values. Aβ PET positivity was defined using a global rdcCL threshold of 25.5, obtained using the Gaussian mixture model.
Statistical analysis
For propensity score matching, we used the matchit function in the MatchIt R package. We included age, sex, initial disease stage, number of APOE ε4 alleles, and baseline global rdcCL value as matching variables. We tested different caliper sizes from 0.05 to 0.2 to find the optimal caliper size, resulting in matching with all variables showing absolute standardized mean differences less than 0.1 (Supplementary Fig. S2).
To compare demographic characteristics, analysis of variance testing was performed for continuous variables, and chi-square tests were performed for categorical variables.
To compare longitudinal Aβ deposition according to radiotracers, we used Bayesian mixed-effects models considering the limited number of PET scans per individual. We included age, sex, time, and an Aβ radiotracer (FBB and FMM) by time interaction as fixed effects along with participant-specific random effects.
To investigate the effect of annualized change in rdcCL values on longitudinal cognition as measured by MMSE, we utilized the NormPsy R package to transform MMSE raw scores into normalized scores between 0 and 100. We used random slope and random intercept linear mixed-effects models including baseline rdcCL, age, sex, education, time, and the interaction between annualized rdcCL and time as fixed effects, in addition to participant-specific random effects. To investigate differences between radiotracers based on this association, we additionally included a radiotracer by annualized CL by time interaction term. To investigate the longitudinal effect of Aβ deposition differences between FBB and FMM on cognition, we conducted a stratified analysis. We used R v4.3.2 (Institute for Statistics and Mathematics, Vienna, Austria; www.R-project.org).
Data availability
Derived data is available from the corresponding author on request by any qualified investigator subject to a data use agreement.
References
Chapleau, M., Iaccarino, L., Soleimani-Meigooni, D. & Rabinovici, G. D. The role of amyloid PET in imaging neurodegenerative disorders: A review. J. Nucl. Med. 63, 13S-S19 (2022).
Ikonomovic, M. D. et al. Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer’s disease. Brain 131, 1630–1645 (2008).
Doraiswamy, P. M. et al. Florbetapir F 18 amyloid PET and 36-month cognitive decline: A prospective multicenter study. Mol. Psychiatry 19, 1044–1051 (2014).
Villemagne, V. L. et al. Amyloid imaging with (18)F-florbetaben in Alzheimer disease and other dementias. J. Nucl. Med. 52, 1210–1217 (2011).
Nelissen, N. et al. Phase 1 study of the Pittsburgh compound B derivative 18F-flutemetamol in healthy volunteers and patients with probable Alzheimer disease. J. Nucl. Med. 50, 1251–1259 (2009).
Klunk, W. E. et al. The Centiloid Project: Standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement. 11, 1–15 (2015).
Kim, S. J. et al. Development and clinical validation of CT-based regional modified Centiloid method for amyloid PET. Alzheimers Res. Ther. 14, 157 (2022).
van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21 (2023).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: The TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527 (2023).
Cho, S. H. et al. Head-to-head comparison of 18F-Florbetaben and 18F-Flutemetamol in the cortical and striatal regions. J. Alzheimers Dis. 76, 281–290 (2020).
Cho, S. H. et al. Amyloid involvement in subcortical regions predicts cognitive decline. Eur. J. Nucl. Med. Mol. Imaging 45, 2368–2376 (2018).
Teipel, S. J. et al. In vivo staging of regional amyloid deposition predicts functional conversion in the preclinical and prodromal phases of Alzheimer’s disease. Neurobiol. Aging 93, 98–108 (2020).
Hanseeuw, B. J. et al. PET staging of amyloidosis using striatum. Alzheimers Dement. 14, 1281–1292 (2018).
Chen, C. D. et al. Longitudinal head-to-head comparison of (11)C-PiB and (18)F-florbetapir PET in a Phase 2/3 clinical trial of anti-amyloid-beta monoclonal antibodies in dominantly inherited Alzheimer’s disease. Eur. J. Nucl. Med. Mol. Imaging 50, 2669–2682 (2023).
Timmers, T. et al. Amyloid PET and cognitive decline in cognitively normal individuals: The SCIENCe project. Neurobiol. Aging 79, 50–58 (2019).
Barthel, H. et al. Cerebral amyloid-beta PET with florbetaben (18F) in patients with Alzheimer’s disease and healthy controls: A multicentre phase 2 diagnostic study. Lancet Neurol. 10, 424–435 (2011).
Jagust, W. J. & Landau, S. M. Temporal dynamics of β-amyloid accumulation in aging and Alzheimer disease. Neurology 96, e1347–e1357 (2021).
Beach, T. G., Thal, D. R., Zanette, M., Smith, A. & Buckley, C. Detection of striatal amyloid plaques with [18F]flutemetamol: Validation with postmortem histopathology. J. Alzheimers Dis. 52, 863–873 (2016).
Ikonomovic, M. D., Fantoni, E. R., Farrar, G. & Salloway, S. Infrequent false positive [(18)F]flutemetamol PET signal is resolved by combined histological assessment of neuritic and diffuse plaques. Alzheimers Res. Ther. 10, 60 (2018).
Jack, C. R. Jr., Barrio, J. R. & Kepe, V. Cerebral amyloid PET imaging in Alzheimer’s disease. Acta Neuropathol. 126, 643–657 (2013).
Karran, E., Mercken, M. & Strooper, B. D. The amyloid cascade hypothesis for Alzheimer’s disease: An appraisal for the development of therapeutics. Nat. Rev. Drug Discov. 10, 698–712 (2011).
Donohue, M. C. et al. Association between elevated brain amyloid and subsequent cognitive decline among cognitively normal persons. JAMA 317, 2305–2316 (2017).
Mintun, M. A. et al. Donanemab in early Alzheimer’s disease. N. Engl. J. Med. 384, 1691–1704 (2021).
Ahn, H. J. et al. Seoul Neuropsychological Screening Battery-dementia version (SNSB-D): A useful tool for assessing and monitoring cognitive impairments in dementia patients. J. Korean Med. Sci. 25, 1071–1076 (2010).
Petersen, R. C. et al. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 56, 303–308 (1999).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 7, 263–269 (2011).
Kim, G. H. et al. Seoul criteria for PiB(-) subcortical vascular dementia based on clinical and MRI variables. Neurology 82, 1529–1535 (2014).
Cho, S. H. et al. A new Centiloid method for (18)F-florbetaben and (18)F-flutemetamol PET without conversion to PiB. Eur. J. Nucl. Med. Mol. Imaging 47, 1938–1948 (2020).
Acknowledgements
This research was supported by the Korea Dementia Research Project through the Korea Dementia Research Center(KDRC), funded by the Ministry of Health & Welfare and Ministry of Science and ICT, Republic of Korea (grant number: RS-2020-KH106434); the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (RS-2019-NR040057); Institute of Information & communications Technology Planning & Evaluation (IITP) grant funded by the Korea government(MSIT) (No.RS-2021-II212068, Artificial Intelligence Innovation Hub); Future Medicine 20*30 Project of the Samsung Medical Center [#SMX1250081]; the “Korea National Institute of Health” research project(2024-ER1003-01).
Author information
Authors and Affiliations
Contributions
This paper is co-authored by Soo Hyun Cho, Heekyoung Kang, Hongki Ham, SeungHwan Moon, Hyemin Jang, Jihwan Yun, Eun Hye Lee, Daeun Shin, Sohyun Yim, Hee Jin Kim, Duk L. Na, and Sang Won Seo. S.H.C, J.P.K and S.W.S. contributed to conceptualization of the study, analysis, data interpretation, and drafting of the manuscript. H.K and H.H were responsible for imaging data analysis and technical support. H. J, J.Y, E.H.L, D.S, S.Y, H.J.K and D.L.N contributed to data interpretation and acquisition. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cho, S.H., Kang, H., Ham, H. et al. Comparison of accumulation rates of beta-amyloid tracers and their relationship with cognitive changes. Sci Rep 15, 7072 (2025). https://doi.org/10.1038/s41598-025-90642-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-90642-8