Abstract
The high rate of postencephalitic epilepsy (PE) contributes to the unfavorable clinical outcome of herpes simplex virus-1 encephalitis (HSE). We aimed to identify the risk factors and explore possible mechanisms of PE in childhood following HSE. We conducted a retrospective review of children diagnosed with HSE and patients were categorized into two groups based on the presence or absence of PE. Multivariate logistic regression analysis was used to analyze factors associated with PE. Furthermore, cytokine and albumin levels in paired cerebrospinal fluid (CSF) and blood samples during acute HSE were also retrospectively reviewed. 97 HSE patients were included in the study and PE was diagnosed in 46. On multivariate analysis, the features predictive of PE (presented as odds ratio [OR] with confidence intervals [CIs]) were status epilepticus (OR 9.38, CI 1.71–10.37), focal seizures (7.41, 1.42–16.97), and restricted diffusion on MRI (6.15, 1.16–20.31). The median QAlb value (CSF to serum albumin ratio, a marker of blood-brain-barrier [BBB] integrity), levels of interleukin (IL)-6 and IL-6:IL-10 ratio in CSF were higher in children with PE during acute HSE. However, CSF levels of IL-10 were higher in non-PE patients. Furthermore, greater CSF IL-6 levels were associated with higher QAlb. These results demonstrated that enhanced BBB impairment and exaggerated proinflammatory response may play a role in the pathogenesis of PE following HSE.
Similar content being viewed by others
Introduction
Infections of the central nervous system (CNS) are the main cause of acquired epilepsy1. Herpes simplex virus type 1 (HSV-1) encephalitis (HSE) is the most common cause of sporadic fatal encephalitis worldwide2. Despite antiviral treatment, HSE is associated with significant morbidity and frequently leads to persistent neurological deficits. Epilepsy secondary to HSE remains one of the most common neurological complications in pediatric patients and contributes significantly to the unfavorable clinical outcome of the disease3. However, little is known about the clinical risk factors and mechanisms for the development of epilepsy after HSE in children, and most studies mainly focus on the adult population4,5. To identify risk factors predictive of post-HSE epilepsy might contribute to better management of the sequelae of the disease.
There is mounting evidence that dysregulated brain inflammation and breakdown of the blood–brain barrier (BBB) contribute to the development of epilepsy6,7. Proinflammatory cytokines such as interleukin (IL)-1β, tumor necrosis factor-α (TNF-α), and IL-6, typically concentrated in low quantities within the brain, are upregulated during brain inflammation, which can cause detrimental synaptic changes as well as neuronal hyperexcitability8. Breakdown of the BBB leads to leakage of peripheral proteins such as albumin into the brain tissue, which can induce imbalance in brain homeostasis, trigger inflammation and lead to seizures9. HSE has significant morbidity partly because of exaggerated neuroimmune response and increased BBB permeability10. It has also been reported that proinflammatory cytokine response is associated with greater clinical severity in patients with HSE11. However, the role of brain inflammation and BBB damage in epileptogenesis after HSE remains unclear.
In this study, we aimed to identify predictors of epilepsy secondary to HSE in pediatric patients. Furthermore, laboratory results of inflammatory cytokines and albumin both in cerebrospinal fluid (CSF) and blood at acute stage were retrospectively analyzed. The disruption of BBB was evaluated by QAlb (CSF /serum albumin ratio). Then, we also assessed their relationship with subsequent development of epilepsy.
Methods
Patients
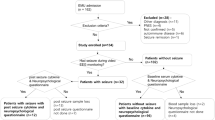
We retrospectively retrieved and reviewed the electronic medical records of all patients under 18 years of age who received a clinical diagnosis of encephalitis from January 2009 to February 2023 at Qilu Hospital of Shandong University, Qilu Children’s Hospital of Shandong University and Shandong Provincial Hospital Affiliated to Shandong First Medical University. Patients with all diagnostic codes for HSE were included (HSE, HSE + epilepsy, HSE + epileptic encephalopathy, HSE + seizure). HSE was confirmed by detection of HSV-1 DNA in CSF using polymerase chain reaction (PCR). From the 151 files collected, we excluded those without PCR confirmation of diagnosis of HSE (n = 16), with incomplete data (n = 14, lack of records of MRI, EEG or both) and lost to follow-up (n = 13). There were eleven patients who developed new-onset or worsening neurological symptoms after HSE. Clinical follow-up revealed that 2 of them had relapse of HSE, while 9 developed anti-NMDAR encephalitis. These 11 patients were also excluded from our study. At last, seizure and epilepsy outcome data after acute encephalitis were available in 97 patients in this study (Fig. 1). Details of the demographics, clinical manifestations, results of auxiliary examinations, treatment strategies, and outcomes were gathered. Patients were categorized by the presence or absence of postencephalitic epilepsy (PE). The Medical Ethics Committee of Qilu Hospital of Shandong University approved the study. We obtained written informed consent from all participant’s parents or guardians.
Flowchart of patients included in the study. HSE herpes simplex virus-1 encephalitis; PCR polymerase chain reaction; MRI magnetic resonance imaging; EEG electroencephalogram; anti-NMDAR anti–N-Methyl-D-Aspartate Receptor; PE postencephalitic epilepsy; QAlb CSF/serum albumin ratio.
Study definitions
Encephalitis was defined by presentation with altered mental status (decreased or altered level of consciousness, lethargy, or personality change) lasting ≥ 24 h, with at least three of the following associated manifestations: (1) temperature ≥ 38 °C within 72 h of presentation, (2) generalized or focal seizures not attributable to a pre-existing seizure disorder, (3) new-onset focal neurologic deficits, (4) CSF white blood count (WBC) count ≥ 5/mm3, (5) abnormal brain parenchyma on neuroimaging suggestive of encephalitis that is either new from prior studies or appears acute in onset, (6) abnormality on electroencephalography (EEG) consistent with encephalitis and not attributable to another cause12. A leukocyte count in CSF samples > 5 cells/ mm3 was defined as pleocytosis.
Currently, there is no consensus on the definition of postencephalitic epilepsy (PE) and the time thresholds vary among studies, with most studies applying the ongoing use of anti-seizure medication (ASMs) as their definition of PE13. In the present study, we used 12 months as our minimum “time threshold1. Thus, our operational definition of PE was defined by the requirement of continued and ongoing use of ASMs ≥ 12 months after recovery from the acute episode of HSE (> 21 days from onset of initial symptoms)3, including the continued and ongoing use of ASMs introduced after 12 months.
Seizures were defined clinically or by EEG confirmation. Status epilepticus (SE) was defined as continuous seizure activity lasting longer than 5 min or recurrent seizures without recovery to baseline between seizures for > 5 min1. Refractory SE (RSE) was defined as SE that failed to respond to adequately used first- and second-line antiepileptic drugs. Super-refractory SE (SRSE) occurred when SE persisted for 24 h or more after administration of anesthesia, or recurs after its withdrawal or reduction14. Mental status was graded using the Glasgow Coma Scale, with coma defined as a GCS score < 815.
Inclusion and exclusion criteria
Patients in our study fulfilled stringent inclusion criteria: the aforementioned definition of encephalitis, CSF PCR evidence of HSV-1 infection, with epilepsy following HSE and the age between 4 weeks and 18 years. All patients were treated with intravenous acyclovir for 14 to 21 days. All patients in the study were primary HSV-1 infection and none had relapse or chronic infection. We excluded cases with a previous diagnosis of epilepsy.
Patients were excluded from our study if they (1) had epilepsy prior to encephalitis; (2) had infection following surgical procedures; (3) had other concomitant risk factors for epilepsy, like tumor, head injury, toxins or metabolic causes; (3) had underlying neurologic disease; (4) had immunosuppressive therapy; (5) had relapse of HSE; (6) had autoimmne encephalitis following HSE.
Measurement of mediators
As a routine clinical practice, written informed consent was obtained for all patients from their parents or legal guardians before lumbar puncture. Paired serum and CSF samples were simultaneously collected during the acute phase of encephalitis and stored at 4 °C until analysis. To minimize potential blood contamination of the CSF, samples were centrifuged at 1000 r/min for 10 min prior to storage. The patient’s CSF routine and biochemical data were tested within 2 h after lumbar puncture. Within 2 h after collection, samples were measured for levels of IL-10, IL-6, IL-1β and TNF-a by enzyme-linked immunosorbent assay (ELISA, R&D Systems) according to the manufacturer’s instructions. The optical density was measured at 450 nm, and the cytokine concentration (pg/ml) was calculated using a standard curve. In addition, paired samples of serum and CSF were also processed for analysis of albumin within 24 h post-collection by nephelometry on a Beckman Image Immunochemistry system (Beckman Instruments, Beckman Coulter, Brea, CA, USA). As a marker of BBB permeability, the CSF/serum albumin ratio (QAlb) was determined. Normal individual age-related QAlb was determined as QAlb * = (4 + Age/15) ×10–3. BBB dysfunction was defined as QAlb > QAlb *16,17. Increased concentration of protein in the CSF was defined as > 45 mg/dL.
Paired CSF and serum samples from 65 patients (31 with PE and 34 without PE) were available for determining QAlb. Cytokine detection in paired CSF and serum samples were available in 66 patients (34 with PE and 32 without PE). Furthermore, overlapped QAlb and cytokine analysis were available in 47 patients (22 with PE and 25 without PE) (Fig. 1).
Radiological and EEG examination
Brain magnetic resonance imaging (MRI) was performed within the first week of admission in all patients. MRI acquisitions were performed with 1.5T or 3T MRI units using T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR), diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) sequences. Findings recorded were the presence of signal abnormalities, involvement of areas of the brain, and presence of hemorrhage. In addition, head computer tomography (CT) in some patients was also performed. The scans were reviewed and interpreted by two neurological radiologists, who were blinded to the clinical information.
Routine video EEG was performed for 4–24 h within 3 days after admission and repeated as clinically needed (e.g., in patients with fluctuating mentation or a seizure-like event), using the international 10–20 system with 21 electrodes. Indications for continuous EEG (cEEG) included ongoing clinical seizures, electrographic seizures, and unexplained disturbed consciousness potentially related to nonconvulsive seizures. All the recordings were interpreted by two board-certified clinical neurophysiologists. Recorded findings were background activity, degrees of slowing, the presence, location, and type of epileptiform discharges and presence and location of slowing.
Statistical analysis
Continuous variables are presented as medians and ranges, and categorical variables are expressed as numbers and percentages. Continuous variables were compared using Mann–Whitney U tests. Comparisons of categorical variables were analyzed using Pearson’s chi-squared test or Fisher’s exact test. Variables with p ≤ 0.05 in univariate analysis were retained in multivariate analysis. A multivariate logistic regression analysis adjusted for age and sex was employed to identify the independent predictors of PE, and the results are described as odds ratios (ORs) with 95% confidence intervals (CIs). Correlation between QAlb and IL-6 concentrations in CSF was assessed on the basis of Spearman’s rank correlation coefficient. Kaplan-Meier survival analysis was also performed to assess the incidence of epilepsy over time. The diagnostic values of CSF IL-6, IL-10, IL-6/IL-10 and QAlb for the prediction of PE were assessed by receiver operating characteristic (ROC) curves and the area under ROC curve (AUC). Cutoff values were identified following Youden’s index of the ROC curve. All reported p-values were two-tailed and considered statistically significant at ≤ 0.05. Data were analyzed using IBM SPSS version 16.0 and GraphPad Prism version 5.0.
Results
Demographic and clinical characteristics
In the present study, we included 97 patients with HSE (50 [51.5%] male and 47 [48.5%] female) (Table 1). The median (range) ages at onset of encephalitis were 1.8 (0.4–13.6) years for the patients with PE and 2.3 (0.8–12.1) years for the patients without PE. We observed no significant differences in the median age (p = 0.362) and distribution of age (p = 0.191; Fig. 2a) at onset of HSE between the two groups. The results showed that twenty-four patients (24.7%) presented with HSE before the age of 1 year, while fifty patients (51.6%) were between 1 year and 6 years of age. Only 23 children (23.7%) had HSE between the ages of 6 and 18 years (Fig. 2b). No significant differences in the duration of symptoms before hospital admission were found between the patients with PE and the patients without PE (median [range], 2.5 [0.5-6.0] vs. 3.0 [0.5-7.0] days; P = 0.347). We observed no significant differences in the median time from symptom onset to initiation of acyclovir (median [range], 4.5 [1.5–7.5] vs. 3.5 [2.5–8.5] days; p = 0.431) and treatment duration of acyclovir (median [range], 16 [14–21] vs. 17 [14–21] days; p = 0.346) between PE patients and non-PE patients. However, the percentage of patients treated with acyclovir started more than four days after symptom onset was higher in PE group than non-PE group (60.9% vs. 39.2%; p = 0.033). There was no statistically significant difference in the incidence use of ASMs (76.1% vs. 66.7%; p = 0.307) and steroid (71.7% vs. 78.4%; p = 0.446) between PE and non-PE groups. In addition, no difference was seen in the number of ASMs used between PE patients and non-PE patients (median [range], 1 [1–3] vs. 1 [1–2]; p = 0.665). The most common symptoms at presentation were fever in 91 cases (93.8%), altered consciousness in 83 (85.6%), focal seizures in 71 (73.2%), vomiting in 47 (48.5%), coma in 33 (34.0%), hemiparesis in 28 (28.9%), SE in 23 (23.7%), generalized seizures in 16 (16.5%), and abnormal behavior in 8 (8.2%). Many patients had more than one symptom at presentation. There was no statistically significant difference in the occurrence of seizures between PE and non-PE groups (87.0% vs. 78.4%; p = 0.270). Hemiparesis (39.1% vs. 19.6%; p = 0.034), coma (45.7% vs. 23.5%; p = 0.022), focal seizures (84.8% vs. 62.7%; p = 0.014) and SE (34.8% vs. 13.7%; p = 0.015) were more common in PE patients compared with non-PE patients. Though the proportion of patients with RSE (8.7% vs. 1.9%; p = 0.187) and SRSE (6.5% vs. 0%; p = 0.103) was higher in the PE group than in the non-PE group, there was no statistical difference, which may be attributed to the relatively small numbers of observed events.
Patient’s age distribution at HSE onset (a, b) and Kaplan-Meier curve showing the probability of survival free of seizure after the resolution of HSE (c). HSE herpes simplex virus-1 encephalitis; PE postencephalitic epilepsy.
Median time to follow-up was 5.6 years (range 1.2–12.5 years). The median time from resolution of HSE to the onset of PE was 0.5 years, with a wide range (from 0.1 up to 8.1 years) (Table 1). The Kaplan-Meier curve of survival free of epilepsy are shown in Fig. 2b. Among patients who developed epilepsy, 31 (67.4%) did so within the first year after HSE, and 38 (82.6%) during the first 2 years. The cumulative incidence of epilepsy in our cohort was 32.0% at 12 months after HSE, 39.2% at 2 years, and 47.4% at the last follow-up.
MRI findings
All patients underwent MRI scan (Table 2). There was no significant difference in the time from symptom onset to MRI imaging between PE and non-PE patients (median [range], 6.0 [0.5–12.0] vs. 6.5 [1.5–13.0] days; p = 0.626). On MRI examination, abnormalities were found in 45 PE patients (97.8%) and 46 non-PE patients (90.2%). Hemorrhage was noted in 25 cases (25.8%). Isolated temporal-insular region involvement was found in 19 patients (19.6%). Fifty-seven (58.8%) had temporal-insular and other regions involvement and 21 (21.6%) had exclusively extra-temporoinsular involvement. FLAIR/T2 signal abnormalities were identified in 85 patients (87.6%) and restricted diffusion was observed in 71 patients (73.2%). Compared with non-PE patients, restricted diffusion (84.8% vs. 62.7%; p = 0.014) was more common in PE patients.
EEG findings
EEG assessment was completed in all cases (Table 2) and EEG abnormalities were present in 93 patients (95.9%). EEG abnormalities consisted of epileptiform discharges in 75 (77.3%), lateralized periodic discharges (LPDs) in 10 (10.3%), focal slowing in 65 (67.0%), and diffuse slowing in 28 (28.9%) patients. No significant differences in EEG abnormalities were found between PE and non-PE patients.
CSF sample analysis
There was no significant difference in the time from symptom onset to CSF collection between the patients with PE and the patients without PE (median [range], 4.5 [0.5-7.0] vs. 5.0 [1.0-9.5] days; p = 0.179) (Table 2). Eighty-seven patients (89.7%) showed CSF pleocytosis. The median CSF leukocyte count was 51 cells/ mm3 (range 2–327) with ten patients having a count of less than 5 cells/mm3. Elevated CSF protein level was found in 37 patients (38.1%). There was no significant difference in CSF pleocytosis and levels of protein and glucose between the two groups.
Multivariate analysis of predictors of PE
The univariate analysis was performed to identify the variables associated with PE. Acyclovir started more than four days after symptom onset, hemiparesis, coma, focal seizures, SE, and restricted diffusion on brain MRI met the significance threshold of p ≤ 0.05 and were retained in the multivariate logistic regression model. On multivariate regression analysis adjusted for age and sex, the final predictors of PE were focal seizures (OR 7.41, CI 1.42–16.97, p = 0.018), SE (OR 9.38, CI 1.71–10.37, p = 0.005) and MRI restricted diffusion (OR 6.15, CI 1.16–20.31, p = 0.039) (Table 3).
Analysis of cytokines in CSF and serum
Levels of IL-6 in CSF during acute HSE were significantly higher in patients with PE than patients without PE (p = 0.003; Fig. 3a). However, CSF levels of IL-10 were lower in patients with PE (p = 0.005; Fig. 3b). In addition, the ratio of IL-6 to IL-10 in CSF was used to reflect the balance between pro- and anti-inflammatory responses in the brain. Patients with PE had higher IL-6:IL-10 ratio compared with patients without PE (p < 0.001; Fig. 3c), suggestive of exaggerated intrathecal proinflammatory cytokine response. There was no significant difference in CSF levels of IL-β (p = 0.603) and TNF-a (p = 0.097) between the two groups (Fig. 3d,e).
Higher CSF IL-6 levels and greater IL-6:IL-10 ratio but lower CSF IL-10 levels in PE patients compared with non-PE patients during acute HSE. CSF cerebrospinal fluid; PE postencephalitic epilepsy.
In most cases, serum levels of the four cytokines were below 10 pg/ml and lower than in CSF. No significant difference was observed in serum levels of all cytokines between PE and non-PE patients (Fig. 4).
No significant differences in serum cytokine levels between patients with and without PE during acute HSE. CSF cerebrospinal fluid; PE postencephalitic epilepsy.
BBB analysis
The CSF/serum quotient of albumin, QAlb, is the most reliable biomarker for the evaluation of BBB permeability. We found that the median QAlb during acute HSE was significantly higher in PE patients compared with non-PE patients (p = 0.025; Fig. 5a), indicating greater BBB leakage in patients with PE. No significant differences were found in the proportion of patients with BBB impairment between the two groups (p = 0.172; Fig. 5b).
Increased BBB permeability in PE patients compared with non-PE patients during acute HSE. BBB blood–brain barrier; HSE herpes simplex virus-1 encephalitis; PE postencephalitic epilepsy; QAlb CSF/serum albumin ratio.
Relationship between BBB permeability and CSF IL-6 levels
The results of the correlation analysis can be seen in Fig. 6. QAlb values were positively correlated to IL-6 levels in CSF in HSE patients (Spearman: r = 0.620, 95% CI 0.397 to 0.774; p < 0.001).
QAlb values were positively correlated with CSF IL-6 levels in HSE patients by Spearman correlation analysis. CSF cerebrospinal fluid; QAlb CSF/serum albumin ratio.
ROC curve analysis of CSF IL-6, IL-10 and IL-6/IL-10 and QAlb for predicting PE
The diagnostic values of CSF IL-6, IL-10, IL-6/IL-10 and QAlb for differentiating non-PE and PE cases to predict PE were assessed by receiver operating characteristic (ROC) curves and the area under ROC curve (AUC) (Fig. 7). Cutoff values were identified following Youden’s index of the ROC curve (Table 4).
ROC curve of CSF IL-6, IL-10, IL-6/IL-10 and QAlb for predicting PE. The AUC of IL-6 was the greatest among the four variables (AUC = 0.824). ROC receiver operating characteristic; CSF cerebrospinal fluid; IL interleukin; QAlb CSF/serum albumin ratio; PE postencephalitic epilepsy; AUC area under ROC curve.
The AUC of the CSF IL-6, IL-10, IL-6/IL-10 and QAlb was 0.716 (95% CI 0.590–0.842; p = 0.003), 0.703 (95% CI 0.577–0.828; p = 0.005), 0.824 (95% CI 0.726–0.922; p < 0.0001), and 0.646 (95% CI 0.508–0.785; p = 0.025), respectively. The cutoff values were 167.00 pg/mL (sensitivity, 84.3%; specificity, 58.8%), 11.15pg/mL (sensitivity, 90.6%, specificity, 41.2%), 5.19 (sensitivity, 75.0%; specificity, 79.4%), and 8.44 (sensitivity, 84.4%; specificity, 48.4%), respectively. These results showed that the ratio of IL-6/IL-10 in CSF was more accurate than the QAlb index for predicting PE, indicating the clinical usefulness of the measurement of CSF IL-6/IL-10 ratio with a threshold greater than 5.19 as a valuable diagnostic marker for epilepsy after HSE.
Discussion
A notable feature of our cohort is that most patients (76.3%) had HSE before age 6 years, with 24.7% before age 1 year. This finding is consistent with a previous study conducted in Taiwan, demonstrating that children were highly susceptible to HSV-1 before age 6 years18. In contrast, the peak incidence of HSE in adults is at age > 50 years and HSE incidence increases among the elderly. The age dependence of predisposition to HSV-1 in early childhood and elder adults may be due to genetic and epigenetic dysregulation of the immune system19. HSE is one of the most common causes of acquired epilepsy20. Previous data have shown that late unprovoked seizures occur in 40–60% of patients after HSE21. In our cohort, 46 of 97 children (47.4%) developed PE after HSE at last follow-up. The different incidence of PE could be attributed to difference in population, adults or children, and length of follow-up. In addition, we found that the risk of developing epilepsy was highest in the first few years, especially within the first 2 years after HSV-1 infection, a common feature of many acquired epilepsies5,22. Thus, we recommend that patients should be carefully monitored for seizure recurrence and better referred to an epileptologist for close outpatient follow-up, particularly during the first 2 years after HSE. Furthermore, we identified focal seizures, SE and radiographic presence of restricted diffusion on brain MRI during acute encephalitis as risk factors for PE. Notably, the marked elevation of CSF IL-6 and greater BBB leakage in the acute phase in patients with PE may indicate that exaggerated pro-inflammatory response to HSV-1 infection contributes to PE.
Focal neurological signs suggestive of impairments of cortical function, like seizures, are the common presentation of HSE23,24,25. In our study, 71 of 97 patients (73.2%) displayed focal seizures and 39 of 71 patients (54.9%) with focal seizures during acute encephalitis developed PE while focal seizures led to a 7.23-fold increase in the risk of subsequent epilepsy. This result confirmed previous findings which demonstrated that the presence of seizures in the acute stage of encephalitis was a major risk factor for PE1. Indeed, children with seizures on presentation may indicate focal cortical injury, which might constitute an epileptogenic focus and lead to epileptogensis26. In addition, SE during acute hospitalization was noted in 23.7% of HSE patients. In contrast, a previous study by Laurent Abel et al. reported a higher incidence (48%) of SE in French pediatric HSE patients19. The different incidence may be because many of their cases (44% of patients) presented with HSE before the age of one year, who tended to be younger at the onset of encephalitis. In fact, the brain is more susceptible to seizures in response to acute insults in the first year of life27. In this cohort, we found SE was associated with increased risk of PE. The mechanisms by which SE contributes to PE remain mysterious. Evidence has indicated that prolonged seizures may lead to augmented astrocytic IL-1β production in the hippocampus which may initiate molecular cascades promoting epileptogenesis28. In addition, prolonged or recurrent seizure activity can also irreversibly alter the way the immature brain develops and forms synapses29,30. All these alterations may initiate the epileptogenic process that ultimately leads to epilepsy.
Brain MRI abnormalities are common in HSE. Seventy-one patients (73.2%) in this study had restricted diffusion on MRI examination and restricted diffusion was significantly associated with the development of PE. As a matter of fact, histologic examination during acute HSE often shows edema as well as necrosis. Restriction of diffusion, a marker of cytotoxic edema which is often followed by neuronal death and necrosis in subacute and chronic stages31, may indicate more serious and irreversible structural brain injury which could provoke epileptogenesis during the postinfectious period. This result is consistent with previous studies demonstrating that restricted diffusion on MRI predicted worse functional outcome in pediatric and adult HSE patients4,32. The EEG in the acute stage of HSE can show a variety of abnormalities, including focal or generalized slow waves, epileptiform discharges, or electrical seizures33. It is highly probable for the diagnosis of HSE to be made if EEG records showed periodic discharges in adults in the setting of a rapidly progressive neurological disease associated with fever34,35. In our cohort, only 10 of 97 patients (10.3%) displayed LPDs on EEG tracings, which was similar to a previous research in pediatric patients, demonstrating that this abnormality has a lower incidence in childhood HSE36.
Although antiviral therapy with acyclovir in HSE is effective in reducing mortality, long-term neurological deficits in the survivors occur frequently3. Initiation of antiviral therapy more than four days after onset has been reported to be a risk factor for poor outcome in HSE37. Though acyclovir started more than four days after onset was associated with PE in univariate analysis, it was not found to be a risk factor for PE on multivariate analysis. Thus, delayed initiation of acyclovir may not be useful in the prediction of PE in HSE children. It’s worth noting that hemiparesis and coma were not identified as predictors of PE on the multivariate logistic regression analysis. The possible reasons for the lack of significance of these two factors may be, firstly, the small numbers of observed events might have been insufficient to allow detection as significant predictors, and secondly, hemiparesis and coma were partially influenced by other predictors, including focal seizures and SE. Consequently, neither of the two factors acted as independent predictors of PE on their own. In fact, evidence suggests that the propensity to develop late seizures after viral encephalitis is more closely related to the localization of the pathological lesions in the brain38.
The underlying mechanisms involved in the pathogenesis of epilepsy following HSE remain largely unknown. Intrathecal innate immune response characterized by production of proinflammatory cytokines such as IL-6 and TNF-α is necessary for controlling HSV-1 infection39,40,41. We also found significantly higher levels of IL-6 in CSF than in serum during the acute phase. However, as a proconvulsive and neurotoxic cytokine, enhanced production of CSF IL-6 can be harmful and contribute to brain tissue destruction and consequent neurologic sequelae40,42. Experimental data have shown that IL-6 can decrease long-term potentiation, dysregulate the neural connectivity and lead to increased neuronal network excitability that underlie epileptogenesis8. In contrast, anti-inflammatory cytokines are crucial for downregulating the excessive inflammatory response and maintaining homoeostasis for proper functioning of vital organs43. Elevated IL-10 level in CSF has been reported to be associated with a better coma score and a lesser degree of BBB permeability in HSE patients11. In the present study, patients with PE during acute encephalitis had higher levels of CSF IL-6 as well as IL-6:IL-10 ratio but lower CSF IL-10 levels, indicating that the balance between the proinflammatory and inhibitory mechanisms was skewed toward a pro-inflammatory state during acute HSE which may contribute to epileptogenesis.
The BBB is a physical barrier essential for the maintenance of a precisely regulated intracerebral microenvironment that is necessary for proper neuronal function44. Increased permeability of BBB can result in the subsequent leakage of albumin as well as immune cells into the brain parenchyma, influx of blood cells and pathogens, and cause local neuroinflammation and angiogenesis45,46. In addition, excessive production of proinflammatory mediators and repetitive seizures during acute encephalitis further lead to BBB malfunction in a positive feedback loop, which promotes excitability in surrounding neurons and ultimately causes chronic recurrent epileptic seizures as well as epilepsy progression47,48,49. In this study, we found QAlb was increased in most children who had evaluation of BBB penetration during acute HSE. Compared with non-PE patients, QAlb was significantly higher in patients with PE, indicating greater impairment of BBB integrity. Thus, it’s reasonable to suspect that enhanced BBB impairment may play a role in the pathogenesis of post-HSE epilepsy. Interestingly, we also found that greater CSF IL-6 levels were associated with higher QAlb values, suggesting that elevated CSF IL-6 contributes to BBB disruption. Moreover, ROC curve analysis revealed that the CSF IL-6/IL-10 ratio, with a threshold of > 5.19, was a valuable diagnostic marker for PE, exhibiting greater accuracy than the QAlb index. These results may imply that enhanced pro-inflammatory responses might play a greater role than the disruption of the BBB in the pathogenesis of post-HSE epilepsy. Though intravenous acyclovir effectively inhibits viral replication, it cannot stop the exaggerated neuroinflammatory processes triggered by HSV-150. In the future, the combination of anti-proinflammatory therapy and restoration of BBB function may hold promise as potential new therapeutic targets in the prevention of epileptogenesis following HSE. Notably, IL-6 receptor inhibitors like tocilizumab and satralizumab have been shown to exert therapeutic effects and improve the prognosis in some refractory autoimmune neurological diseases and new-onset refractory status epilepticus by suppressing excessive neuroinflammatory responses and preventing the disruption of the BBB51,52. Further prospective studies may be necessary to assess the efficacy and safety of this promising treatment and to optimize the immunomodulatory strategy for HSE.
Our study has several limitations. First, it was designed to include cases typically recognized as representative of HSE. As a result, we excluded patients without PCR confirmation, as well as those with relapsing HSE or secondary autoimmune encephalitis. Additionally, loss to follow-up is inevitable in epidemiological studies. These factors may lead to selection bias and limit the generalizability of our findings. Second, non-uniform EEG monitoring durations among patients due to the retrospective design may have affected the EEG results, potentially influencing the statistical analysis results to some extent. Despite these limitations, our study advances the understanding of risk factors for post-HSE epilepsy in children which may help in the careful surveillance of high-risk patients. Furthermore, the findings from this study support the involvement of dysregulated neuroinflammation in the pathogenesis of PE and may direct us toward new therapeutic interventions.
Conclusions
We found that the presence of focal seizures as well as SE in the acute hospital setting and restricted diffusion on brain MRI predicted the development of epilepsy following HSE. Enhanced proinflammatory response and disrupted BBB may play a role in the pathogenesis of PE. Further detailed neuropathological studies may lead to precise targets for therapeutic intervention.
The raw data, datasets used and/or analyzed during the current study available from the corresponding author on reasonable request by email (lbmqilu@gmail.com).
Data availability
The raw data, datasets used and/or analyzed during the current study available from the corresponding author on reasonable request by email (lbmqilu@gmail.com).
References
Singh, T. D., Fugate, J. E., Hocker, S. E. & Rabinstein, A. A. Postencephalitic epilepsy: clinical characteristics and predictors. Epilepsia56, 133–138. https://doi.org/10.1111/epi.12879 (2015).
Kennedy, P. G. & Chaudhuri, A. Herpes simplex encephalitis. J. Neurol. Neurosurg. Psychiatry. 73, 237–238. https://doi.org/10.1136/jnnp.73.3.237 (2002).
Sellner, J. & Trinka, E. Seizures and epilepsy in herpes simplex virus encephalitis: Current concepts and future directions of pathogenesis and management. J. Neurol.259, 2019–2030. https://doi.org/10.1007/s00415-012-6494-6 (2012).
Singh, T. D. et al. Predictors of outcome in HSV encephalitis. J. Neurol.263, 277–289. https://doi.org/10.1007/s00415-015-7960-8 (2016).
Zelano, J. & Westman, G. Epilepsy after brain infection in adults: A register-based population-wide study. Neurology95, e3213–e3220. https://doi.org/10.1212/WNL.0000000000010954 (2020).
van Vliet, E. A. et al. Blood-brain barrier leakage May lead to progression of Temporal lobe epilepsy. Brain130, 521–534. https://doi.org/10.1093/brain/awl318 (2007).
Shimada, T., Takemiya, T., Sugiura, H. & Yamagata, K. Role of inflammatory mediators in the pathogenesis of epilepsy. Mediators Inflamm.2014, 901902. https://doi.org/10.1155/2014/901902 (2014).
Rana, A. & Musto, A. E. The role of inflammation in the development of epilepsy. J. Neuroinflammation. 15, 144. https://doi.org/10.1186/s12974-018-1192-7 (2018).
Troscher, A. R. et al. Inflammation mediated epileptogenesis as possible mechanism underlying ischemic Post-stroke epilepsy. Front. Aging Neurosci.13, 781174. https://doi.org/10.3389/fnagi.2021.781174 (2021).
Michael, B. D. et al. Astrocyte- and Neuron-Derived CXCL1 drives neutrophil transmigration and Blood-Brain barrier permeability in viral encephalitis. Cell. Rep.32, 108150. https://doi.org/10.1016/j.celrep.2020.108150 (2020).
Michael, B. D. et al. The Interleukin-1 balance during encephalitis is associated with clinical severity, Blood-Brain barrier permeability, neuroimaging changes, and disease outcome. J. Infect. Dis.213, 1651–1660. https://doi.org/10.1093/infdis/jiv771 (2016).
Venkatesan, A. et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: Consensus statement of the international encephalitis consortium. Clin. Infect. Dis.57, 1114–1128. https://doi.org/10.1093/cid/cit458 (2013).
Pillai, S. C. et al. Postencephalitic epilepsy and drug-resistant epilepsy after infectious and antibody-associated encephalitis in childhood: Clinical and etiologic risk factors. Epilepsia57, e7–e11. https://doi.org/10.1111/epi.13253 (2016).
Vasquez, A., Farias-Moeller, R. & Tatum, W. Pediatric refractory and super-refractory status epilepticus. Seizure68, 62–71. https://doi.org/10.1016/j.seizure.2018.05.012 (2019).
Sarton, B. et al. Assessment of magnetic resonance imaging changes and functional outcomes among adults with severe herpes simplex encephalitis. JAMA Netw. Open.4, e2114328. https://doi.org/10.1001/jamanetworkopen.2021.14328 (2021).
You, X. et al. Disruption of blood-brain barrier integrity associated with brain lesions in Chinese neuromyelitis Optica spectrum disorder patients. Mult. Scler. Relat. Disord.. 27, 254–259. https://doi.org/10.1016/j.msard.2018.10.114 (2019).
Susse, M., Saathoff, N., Hannich, M. & von Podewils, F. Cerebrospinal fluid changes following epileptic seizures unrelated to inflammation. Eur. J. Neurol.26, 1006–1012. https://doi.org/10.1111/ene.13924 (2019).
Hsieh, W. B., Chiu, N. C., Hu, K. C., Ho, C. S. & Huang, F. Y. Outcome of herpes simplex encephalitis in children. J. Microbiol. Immunol. Infect.40, 34–38 (2007).
Abel, L. et al. Age-dependent Mendelian predisposition to herpes simplex virus type 1 encephalitis in childhood. J. Pediatr.157, 623–629. https://doi.org/10.1016/j.jpeds.2010.04.020 (2010). 629 e621.
Chen, S. F. et al. Seizure, neuron loss, and mossy fiber sprouting in herpes simplex virus type 1-infected organotypic hippocampal cultures. Epilepsia45, 322–332. https://doi.org/10.1111/j.0013-9580.2004.37403.x (2004).
Loscher, W. & Howe, C. L. Molecular mechanisms in the genesis of seizures and epilepsy associated with viral infection. Front. Mol. Neurosci.15, 870868. https://doi.org/10.3389/fnmol.2022.870868 (2022).
Pujar, S. S. et al. Long-term prognosis after childhood convulsive status epilepticus: A prospective cohort study. Lancet Child. Adolesc. Health. 2, 103–111. https://doi.org/10.1016/S2352-4642(17)30174-8 (2018).
Whitley, R. J. et al. Herpes simplex encephalitis. Clin. Assess. JAMA. 247, 317–320 (1982).
Armangue, T. et al. Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: A prospective observational study and retrospective analysis. Lancet Neurol.17, 760–772. https://doi.org/10.1016/S1474-4422(18)30244-8 (2018).
Bradshaw, M. J. & Venkatesan, A. Herpes simplex virus-1 encephalitis in adults: Pathophysiology, diagnosis, and management. Neurotherapeutics 13, 493–508. https://doi.org/10.1007/s13311-016-0433-7 (2016).
Lee, W. T., Yu, T. W., Chang, W. C. & Shau, W. Y. Risk factors for postencephalitic epilepsy in children: A hospital-based study in Taiwan. Eur. J. Paediatr. Neurol.11, 302–309. https://doi.org/10.1016/j.ejpn.2007.02.011 (2007).
Capovilla, G. et al. Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian league against epilepsy. Epilepsia54 Suppl 7, 23–34. https://doi.org/10.1111/epi.12307 (2013).
Dube, C. M. et al. Epileptogenesis provoked by prolonged experimental febrile seizures: Mechanisms and biomarkers. J. Neurosci.30, 7484–7494. https://doi.org/10.1523/JNEUROSCI.0551-10.2010 (2010).
Sloviter, R. S. Status epilepticus-induced neuronal injury and network reorganization. Epilepsia40 (Suppl 1). https://doi.org/10.1111/j.1528-1157.1999.tb00876.x (1999). S34–39; discussion S40-31.
Holmes, G. L. & Ben-Ari, Y. The neurobiology and consequences of epilepsy in the developing brain. Pediatr. Res.49, 320–325. https://doi.org/10.1203/00006450-200103000-00004 (2001).
Bani-Sadr, A. et al. Contribution of diffusion-weighted imaging to distinguish herpetic encephalitis from auto-immune encephalitis at an early stage. J. Neuroradiol.50, 288–292. https://doi.org/10.1016/j.neurad.2022.05.003 (2023).
Pillai, S. C. et al. Infectious and autoantibody-associated encephalitis: Clinical features and long-term outcome. Pediatrics135, e974–984. (2015). https://doi.org/10.1542/peds.2014-2702
Sutter, R. et al. Electroencephalography for diagnosis and prognosis of acute encephalitis. Clin. Neurophysiol.126, 1524–1531. https://doi.org/10.1016/j.clinph.2014.11.006 (2015).
Kim, Y. S. et al. Prognostic value of initial standard EEG and MRI in patients with herpes simplex encephalitis. J. Clin. Neurol.12, 224–229. https://doi.org/10.3988/jcn.2016.12.2.224 (2016).
Gilden, D. H., Mahalingam, R., Cohrs, R. J. & Tyler, K. L. Herpesvirus infections of the nervous system. Nat. Clin. Pract. Neurol.3, 82–94. https://doi.org/10.1038/ncpneuro0401 (2007).
Elbers, J. et al. (ed, M.) A 12-year prospective study of childhood herpes simplex encephalitis: Is there a broader spectrum of disease? Pediatrics119, e399–407. https://doi.org/10.1542/peds.2006-1494 (2007).
Kamei, S. et al. Evaluation of combination therapy using aciclovir and corticosteroid in adult patients with herpes simplex virus encephalitis. J. Neurol. Neurosurg. Psychiatry. 76, 1544–1549. https://doi.org/10.1136/jnnp.2004.049676 (2005).
Misra, U. K., Tan, C. T. & Kalita, J. Viral encephalitis and epilepsy. Epilepsia49 (Suppl 6), 13–18. https://doi.org/10.1111/j.1528-1167.2008.01751.x (2008).
Aurelius, E., Andersson, B., Forsgren, M., Skoldenberg, B. & Strannegard, O. Cytokines and other markers of intrathecal immune response in patients with herpes simplex encephalitis. J. Infect. Dis.170, 678–681. https://doi.org/10.1093/infdis/170.3.678 (1994).
Lind, L., Studahl, M., Persson Berg, L. & Eriksson, K. CXCL11 production in cerebrospinal fluid distinguishes herpes simplex meningitis from herpes simplex encephalitis. J. Neuroinflammation. 14, 134. https://doi.org/10.1186/s12974-017-0907-5 (2017).
Kamei, S. et al. Prognostic value of cerebrospinal fluid cytokine changes in herpes simplex virus encephalitis. Cytokine46, 187–193. https://doi.org/10.1016/j.cyto.2009.01.004 (2009).
Soltani Khaboushan, A., Yazdanpanah, N. & Rezaei, N. Neuroinflammation and Proinflammatory cytokines in epileptogenesis. Mol. Neurobiol.59, 1724–1743. https://doi.org/10.1007/s12035-022-02725-6 (2022).
Ng, P. C. et al. Proinflammatory and anti-inflammatory cytokine responses in preterm infants with systemic infections. Arch. Dis. Child. Fetal Neonatal Ed.88, F209–F213. https://doi.org/10.1136/fn.88.3.f209 (2003).
Moretti, R. et al. Blood-brain barrier dysfunction in disorders of the developing brain. Front. Neurosci.9, 40. https://doi.org/10.3389/fnins.2015.00040 (2015).
Bronisz, E., Cudna, A., Wierzbicka, A. & Kurkowska-Jastrzebska, I. Blood-brain barrier-associated proteins are elevated in serum of epilepsy patients. Cells. https://doi.org/10.3390/cells12030368 (2023).
Reiss, Y. et al. The neurovasculature as a target in Temporal lobe epilepsy. Brain Pathol.33, e13147. https://doi.org/10.1111/bpa.13147 (2023).
Liu, H., Qiu, K., He, Q., Lei, Q. & Lu, W. Mechanisms of blood-brain barrier disruption in herpes simplex encephalitis. J. Neuroimmune Pharmacol.14, 157–172. https://doi.org/10.1007/s11481-018-9821-6 (2019).
Vezzani, A., French, J., Bartfai, T. & Baram, T. Z. The role of inflammation in epilepsy. Nat. Rev. Neurol.7, 31–40. https://doi.org/10.1038/nrneurol.2010.178 (2011).
Baruah, J., Vasudevan, A. & Kohling, R. Vascular integrity and signaling determining brain development, network excitability, and epileptogenesis. Front. Physiol.10, 1583. https://doi.org/10.3389/fphys.2019.01583 (2019).
Rybak-Wolf, A. et al. Modelling viral encephalitis caused by herpes simplex virus 1 infection in cerebral organoids. Nat. Microbiol.8, 1252–1266. https://doi.org/10.1038/s41564-023-01405-y (2023).
Girardin, M. L. et al. Treatment of new onset refractory status epilepticus/febrile infection-related epilepsy syndrome with Tocilizumab in a child and a young adult. Epilepsia64, e87–e92. https://doi.org/10.1111/epi.17591 (2023).
Takeshita, Y. et al. New BBB model reveals that IL-6 blockade suppressed the BBB disorder, preventing onset of NMOSD. Neurol. Neuroimmunol. Neuroinflamm. 8 (2021). https://doi.org/10.1212/NXI.0000000000001076
Acknowledgements
We appreciate Xiafei Fu for polishing our paper.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
YP: Conceptualization, Data curation, Investigation, Methodology, Project administration, Writing – original draft, Writing – review & editing. PT: Data curation, Investigation, Methodology, Writing – original draft. XZ: Methodology, Supervision, Writing – original draft. DZ: Data curation, Writing – original draft. XY: Data curation, Writing – original draft. LY: Methodology, Writing – original draft. YW: Methodology, Writing – original draft. GL: Conceptualization, Investigation, Writing – review & editing. BL: Conceptualization, Investigation, Project administration, Writing – review & editing.All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study has been approved by the appropriate ethics committee and been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We obtained written informed consent from all participants’s parents or guardians.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yin, P., Tian, P., Zhang, X. et al. Clinical and pathological risk factors for postencephalitic epilepsy after herpes simplex virus-1 encephalitis in children. Sci Rep 15, 6471 (2025). https://doi.org/10.1038/s41598-025-91438-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-91438-6
This article is cited by
-
The synergistic role of viral infection and immune response in the pathogenesis of facial palsy
Journal of NeuroVirology (2025)