Abstract
The endometrial receptive status is critical for a successful embryo implantation, but it only last for a short period of time, and the window of implantation (WOI) of each woman changes individually, which will lead to poor outcomes of assisted reproduction. This so-called WOI can be detected by molecular diagnostic method, endometrial receptivity analysis (ERA), which was gradually applied to clinical practice. This study aimed to evaluate the clinical efficacy of personalized embryo transfer (pET) guided by ERA in patients with and without RIF. A total of 3605 patients with previous failed embryo transfer (ET) cycle in the reproductive medicine center of Renmin Hospital Hubei University of Medicine from January 2016 to October 2022 were retrospectively analyzed. The 3605 patients were divided into non-RIF group and RIF group, among which 782 patients who received ERA test underwent personalized embryo transfer (pET). The decision whether to accept ERA test or not was based on the patient’s voluntary choice. We divided the 782 patients into normal WOI group and displaced WOI group according to the result of ERA test. The pregnancy outcomes were compared between the different groups. The age, number of previous ET cycle and serum E2/P ratio were mainly analyzed to investigate its relationship with displaced WOI. The clinical pregnancy rate and live birth rate in non-RIF with pET group were higher than that of non-RIF with non-personalized embryo transfer (npET) group (64.5% vs 58.3%, P = 0.025; 57.1% vs 48.3%, P = 0.003). The clinical pregnancy rate and the live birth rate in RIF with pET group were significantly higher than that of RIF with npET group (62.7% vs 49.3%, P < 0.001; 52.5% vs 40.4%, P < 0.001) after propensity score matching (PSM). The early abortion rate in the non-RIF with pET group was lower than that in the non-RIF with npET group (8.2% vs 13.0%, P = 0.038). There was a significant difference in age and the number of previous failed ET cycle between the normal WOI group and displaced WOI group (age: 32.26 vs 33.53 years, P < 0.001; the number of previous failed ET cycle: 1.68 vs 2.04, P < 0.001). Logistic regression analysis also showed that the age and number of previous failed ET cycles were positively correlated with displaced WOI. The displaced WOI rate increased gradually with the increase of age and number of previous failed ET cycle; the displaced WOI rate in the median group (4.46 < E2/P ≤ 10.39 pg/ng) was significantly lower than that in the other two groups (54.8% vs 40.6% vs 58.5%, P < 0.001). The clinical pregnancy rate and live birth rate of patients with previous failed ET cycle was improved after pET guided by ERA, especially in RIF patients; the early abortion rate of the non-RIF patient was reduced after pET guided by ERA. An appropriate E2/P ratio was beneficial for maintaining endometrial receptivity state, and the age and number of previous failed ET cycle was correlated with increased displaced WOI.
Similar content being viewed by others
Introduction
The relationship between embryo quality and endometrial receptivity is similar to “seed” and “soil” in the process of assisted reproductive technology (ART). Successful embryo implantation requires capable embryo, receptive endometrium, and synchronous dialogue between maternal and embryonic tissue1. With the improvement of laboratory technology in invitro fertilization-embryo transfer (IVF), great progress has been made in culturing and screening high-quality embryos at present2, but some patients still face the thorny situation of implantation failure with high-quality embryo3. Another important factor to successful embryo implantation is endometrial receptivity, which has attracted more and more attention. Endometrial receptivity refers to the period of endometrial maturation during which the trophectoderm of the blastocyst can attach to the endometrial epithelial cells and subsequently invade the endometrial stroma and vasculature4. The endometrium undergoes a series of dynamic and complex changes during the menstrual cycle in humans, and blastocysts are allowed to implant in a brief period. This period is called window of implantation (WOI), which is usually around 19–24 days of the menstrual cycle5. However, the WOI of each woman has an individual variation, and some women with displace-WOI may lead to embryo-endometrium asynchrony, which usually leads to implantation failure or even recurrent implantation failure (RIF)6,7. If we can accurately detect the normal WOI and make a personalized embryo transfer, it will provide new possibilities to improve pregnancy rate of ART.
The WOI was mainly judged by transvaginal ultrasound, serum estrogen and progesterone levels, endometrial biopsy and other methods in the past. But these methods have certain limitations and poor repeatability8,9. With the continuous development of modern molecular gene diagnosis technologies such as gene transcriptome analysis based on high-throughput sequencing technology, ERA technology developed by combining computer programs10.
However, the current studies on ERA were basically based on small sample sizes, with no classification of RIF and non-RIF for comparison. Furthermore, because the expense and invasive nature of ERA continue to be obstacle for the infertile patients, not every patient is willing to undergo ERA test. Therefore, it is also important to analyze the clinical factors correlated with displaced WOI and screen patients who are more likely to have a displaced WOI and need to undergo ERA test. In this study, we conducted a retrospective analysis on a large sample of 3065 patients to evaluate the clinical efficacy of personalized embryo transfer guided by ERA in patients with and without RIF, and for the first time we analyzed the effect of clinical indicators on the outcomes of ERA test and displaced WOI.
Materials and methods
Study setting and patients
3605 patients with previous failed embryo transfer (ET) cycle in the department of reproductive medicine center of Renmin Hospital Hubei University of Medicine from January 2016 to October 2022 were retrospectively analyzed. The patient was only be included once in the study if they underwent more than one ET cycle, and patients whose embryos were tested for aneuploidy using PGT-A screening were not included in this study. Among the 3605 patients, 782 patients received ERA and underwent personalized embryo transfer (pET) in accordance with the ERA test results in next hormone replacement therapy (HRT) cycle. In the same period, there were 2823 patients who did not received ERA and underwent non-personalized embryo transfer (npET) at a standardized FET time of P + 5d in HRT cycle. According to the definition of RIF, we divided these 3605 patients into non-RIF group (including 301 cases who received ERA and 1744 cases who did not received ERA) and RIF group (including 481 cases who received ERA and 1079 cases who did not received ERA). We furtherly divided the 782 patients who received ERA into normal WOI group and displaced WOI group according to the result of ERA test.
The exclusion criteria were as follows: (i) Patients with uterine and endometrial factors that may affect endometrial receptivity, such as chronic endometritis, severe intrauterine adhesions, endometrial tuberculosis, endometrial hyperplasia, uterine malformations, severe hydrosalpinx, severe endometriosis (stage III-IV) etc.; (ii) Other patients with severe underlying diseases which are not suitable for pregnancy (including tumor, hypertension, diabetes, etc.).
RIF was defined as failure to achieve a clinical pregnancy after receiving at least 4 morphologically high-quality cleavage embryos or 2 high quality blastocysts in a minimum of 2 fresh or frozen transfer cycles11,12. The criteria for embryos of high quality were as follows: (i) Blastocysts: ≥ 3BB13; (ii) Cleavage-stage embryo: number of cells ≥ 8, fragmentation < 20% and uniform size of blastomeres14.
Endometrial sampling and preparing
HRT protocol was used in the endometrial preparing. Estrogen (oral or external use, and both if necessary) should be pretreated for 16 days from the third day of menstruation, the ultrasound was used to assess the thickness of the endometrium, if the thickness is greater than six millimeters, then progesterone (P)60 mg is injected intramuscularly. The first day of progesterone supplementation is regarded as P + 0d, lasting for 5 days, and endometrial biopsy should be performed at P + 5d.
Endometrial receptivity testing
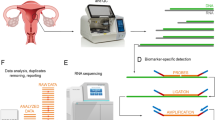
Endometrial receptivity test in our center is based on endometrial receptivity gene chip which is consisted of a customized array containing 238 genes expressed at different stages of the endometrial cycle15. Endometrial receptivity status can be predicted through computer analysis. Specific steps and criteria for ERA testing are described in the supplemental materials. The detailed process of endometrial receptivity test can be seen in Fig. 1
The detailed process of endometrial receptivity test.
The primary outcome measures
Clinical pregnancy was defined as ultrasonography showing a pregnancy sac after receiving embryo transfer four weeks later. Clinical pregnancy rate = number of pregnancy cycles/number of transplantation cycles × 100%. Live birth was defined as giving birth to at least one live infant, and the gestational age was longer than 28 weeks. Live birth rate = number of births of live babies/number of total transplantation cycles × 100%.
Statistical analysis methods
Our data collection and analysis method are shown in Fig. 2. SPSS26.0 software was used for statistical analysis. The measurement data conform to the normal distribution is expressed by the mean and standard deviation (Mean ± SD), and the comparison between groups was made by one-way ANOVA. The counting data is expressed by the rate (%), and the comparison between groups adopts χ2 test; Because there had significant differences in characteristics between the RIF patient with pET and npET group, we used propensity score matching (PSM) to adjust for potential differences in characteristics, and nearest neighbor matching method was use for PSM. The variables included in PSM are those with differences between the two groups: Age, Infertility type, Basal serum FSH, Basal LH. ERA patients with RIF were matched to non-ERA patients with RIF in a 1:2 matching ratio, the caliper value was set to 0.1. The majority of ERA patients with RIF were successfully matched to 2 non-ERA patients with RIF. Logistic regression analysis was used to exclude the influence of confounding factor and analyze the correlation between related factors and displace WOI. P < 0.05 is the difference with statistical significance.
The detailed process of data collection and analysis method.
Ethical approval
This study was approved by the Ethics Committee of Renmin Hospital, Hubei University of Medicine (reference number: syrmyy2023-052) and conducted in conformity with the Helsinki Declaration. Informed consent was waived by the Ethics Committee of Renmin Hospital, Hubei University of Medicine. This was not required as this study was based on the retrospective clinical data.
Results
Comparison of baseline characteristics between non-RIF patients and RIF patients
We found that the average age, infertility duration, and baseline FSH of non-RIF group are lower than that of RIF group (age: 32.53 ± 4.44 vs 33.79 ± 4.50 years, P < 0.001; infertility duration: 5.09 ± 3.13 vs 5.80 ± 3.31 years, P < 0.001; baseline FSH: 7.28 ± 2.58 vs 7.65 ± 2.84 IU/L). The proportion of blastocysts transferred in non-RIF group is higher than that in RIF group (21.1% vs 16.9%, P = 0.002). And the average number of previous failed ET cycle for RIF patients was 2.5 times, compared to 1 time for non-RIF patients. There is no statistical difference in other indicators. See Table 1 for details.
Comparison of baseline characteristics and pregnancy outcomes in non-RIF patients with pET and npET
The comparison of basic characteristics showed no significant difference between the non-RIF with pET and non-RIF with npET groups, which indicated the comparability between the two groups. The clinical pregnancy rate and live birth rate in non-RIF with pET group were higher than that of non-RIF with npET group (64.5% vs 58.3%, P = 0.025; 57.1% vs 48.3%, P = 0.003). Moreover, the early abortion rate in the non-RIF with pET group was lower than that in the non-RIF with npET group (8.2% vs 13.0%, P = 0.038). There was no difference in the ectopic pregnancy rate between the two groups. See Table 2, Fig. 3 for details.
Comparison of pregnancy outcomes of pET and npET in non-RIF and RIF patients. (A) Clinical pregnancy; (B) Live birth rate; (C) Early abortion rate.
Comparison of basic characteristics and pregnancy outcomes in RIF patients with pET and npET
There were significant differences in basic characteristics of female age, infertility type, BMI, basal serum PRL between the RIF with pET and RIF with npET groups. We used propensity score matching (PSM) to adjust for potential differences in characteristics between the two groups. The matching ratio was set to 1:2, and 434 patients in RIF with pET group were matched to755 patients in RIF with non-pET group. The clinical pregnancy rate and the live birth rate in RIF with pET group were significantly higher than that in RIF with npET group after PSM (62.7% vs 49.3%, P < 0.001; 52.5% vs 40.4%, P < 0.001). There is no difference in the early abortion rate and ectopic pregnancy rate between the two groups. See Table 3, Fig. 3 for details.
Endometrial receptivity testing results
Among the 782 patients who underwent ERA test on P + 5d, 381 (48.7%) patients were found to be receptive, and diagnosed with normal WOI; 401 patients (51.3%) were found to be non-receptive, and diagnosed with displaced WOI. All the 401 patients with a displaced WOI were biopsied again at the recommended time of the ERA test in subsequent cycles until the receptivity state is found. Finally, among the 401patients with a displaced WOI, 245 patients went through two ERA test to find the receptive state; 156 patients went through three ERA test to find the receptive state.
Comparison of normal WOI and displaced WOI group in patients who underwent ERA test
There was a significant difference in age and the number of previous failed ET cycle between the normal WOI group and displaced WOI group (age:32.26 vs 33.53 years, P < 0.001; the number of previous failed ET cycle:1.68 vs 2.04, P < 0.001). There was no significant difference in other factors including serum E2 and P levels on P + 5d between the normal WOI group and displaced WOI group. See Table 4 for details.
The logistic regression analysis of the correlation between related factors and displaced WOI
In order to evaluate the correlation between related factors and displaced WOI, the logistic regression analysis was performed with a enter regression method. The results showed that age and number of previous failed ET cycles were positively correlated with displaced WOI, while other variables, including serum E2 and P, were not in linear correlation with endometrial receptivity. See Table 5 for details.
Correlation analysis between E2/P ratio, age, number of previous failed ET cycle and displaced WOI
We divided 782 patients who received ERA test into three groups according to their age (age ≤ 30 years group, 30 years < age ≤ 35 years group, age > 35 years group), the number of previous failed ET cycles (number of previous failed ET cycle = 1 group, = 2 group, and ≥ 3 group), the results showed that the displaced WOI rate increased gradually with the increase of age group (42.0% vs 51.6% vs 61.0%, P < 0.001) (Table 6, Fig. 4A); and the displaced WOI rate increased gradually with the increase of the number of previous failed ET cycle (40.9% vs 54.6% vs 65.3%, P < 0.001) (Table 6, Fig. 4B).
Group comparison to analysis correlation between related factors and displaced. (A) Age group; (B) no. of previous failed ET cycle group; (C) E2/P ratios group.
We also divided 782 patients with ERA test into tertiles for analysis according to the E2/P ratios: E2/P ≤ 4.46 pg/ng group, 4.46 < E2/P ≤ 10.39 pg/ng group, E2/P > 10.39 pg/ng group. The results showed that the displaced WOI rate of the median group 4.46 < E2/P ≤ 10.39 pg/ng was significantly lower than that of the other two groups (54.8% vs 40.6% vs 58.5%, P < 0.001) (Table 6, Fig. 4C).
Discussion
Traditionally, the WOI period is considered quite wide, but the optimal window has been proved to be much narrower, which may last for only 12 ~ 48 h16. The embryo transplantation outside the window will lead to poorer outcomes of ART treatment6,7. Therefore, it is very important to detect the normal WOI objectively for pET. It is reported that nearly 1/3 to 2/3 of implantation failures are caused by the endometrial non-receptivity status17,18. It has been reported that the pregnancy outcomes of patients who received ERA test significantly increased compared with control group, especially in patients with RIF19,20. However, there are also recent studies hold the opposite opinions that ERA can’t improve the pregnancy outcome in RIF patients21,22. Other investigators considered that the value was rather limited when applied ERA in the first embryo transfer cycles or on the patient with good prognosis, ERA were not recommended as a routine test for unselected patient populations23,24. Because some factors can affect endometrial gene expression, such as chronic endometritis, severe intrauterine adhesions, endometrial tuberculosis, endometrial hyperplasia4,25,26, so we excluded some factors that may affect endometrial receptivity to eliminate these biases. In this study, we divide the population into non-RIF and RIF, and we found that non-RIF with pET group had higher clinical pregnancy rates and live birth rate than non-RIF with npET group. The pregnancy rate in RIF with pET group and the live birth rate are significantly higher than RIF with non-pET group after PSM matching, and the gap of pregnancy outcomes between the RIF with pET group and npET group is larger than that of non- RIF with pET and npET group. This study further confirmed the clinical value of ERA in infertile patients with previous failed ET cycle, especially in RIF patients.
It is known that in normal implantation, the establishment of endometrial tolerance to the developing embryo relies on the active recognition of trophoblasts by maternal immune cells within the decidua27. This immune response facilitates and supports ongoing implantation, and is important for determining pregnancy success28,29. Therefore, the immune system is thought to be intricately involved in mediating endometrial receptivity and facilitating implantation, abnormal immunological responses and the disruption of cytokine networks during the window of implantation (WOI) has long been suspected to explain RIF in certain patients30,31. Evidence shows that different amounts of immune cell types such as CD4T cells, Myeloid-Derived Suppressor Cells (MDSC), regulatory T (Treg) cells, NK cell subtypes, and CD8 T cells have been reported in RIF in comparison to the healthy women, and the abnormal production of these immunological factors may have a detrimental effect on implantation and ultimately lead to RIF32,33,34. However, in this study, we did not completely exclude the effect of immunological factors on the statistical results of RIF patients, which is also a limitation of this study. On the other hand, many patients who have experienced more implantation failure may be tested for endometrial or immune problems and treated accordingly, which may lead to a better outcome for RIF patients. And, some RIF patients may have already received additional treatments, such as granulocyte colony-stimulating factor (G-CSF), hysteroscopy, or hormone optimization, which also affect the outcome for RIF patients35,36. Not only that, we also found that pET guided by ERA has a certain effect on reducing the early spontaneous abortion rate for patients with non-RIF. This is a new discovery, indicating that the displacement of WOI may be related to early abortion. However, pET did not have such an effect for RIF patients, we believe the possible reasons are that RIF patients have numerous other factors that can lead to miscarriage, including factors such as aneuploidy and immunological factors37,38,39. Furthermore, we need to pay attention to another point of endometrial scraping, which is an intentional damage to endometrium to improve outcomes of fertility treatment or the chance of natural conception40,41. So, the improvement in pregnancy outcomes may be related to endometrial scraping caused by endometrial biopsy during ERA test, but we cannot rule out the influence of this confounding factor, which is another limitation of this article.
In the complex physiological process of embryo implantation, numerous of molecular mediators participate in the initial maternal–fetal interaction under the coordination of ovarian steroid hormones42. Despite the ovaries produce many steroids throughout the menstrual cycle, only E2 and P have been proven to be essential for endometrial receptivity5,43. Studies have shown that the disorder of sex hormone will affect the endometrial receptivity44,45. Progesterone in advance or low serum progesterone on day of embryo transfer will have a negative impact on pregnancy rates of ART46,47. Research has confirmed that exposure of human endometrial epithelial cells to high levels of estrogen can lead to abnormal expression of many key implantation molecules48. Estradiol reduction adversely affect the embryo quality and clinical outcomes of IVF-ET49. However, these studies did not combine estrogen and progesterone with gene expression profile. There are currently no reports linking endometrial receptivity testing with hormone analysis for research. In this study, we compared the estrogen and progesterone levels of P + 5d between the normal and displaced WOI groups, but there is no difference between the two groups, and the logistic regression analysis also showed that the estrogen and progesterone levels were not linearly correlated with the displaced WOI. Therefore, we divided the E2/P values into tertiles for analysis, we found that the displaced WOI rate of the median group (4.46 < E2/P ≤ 10.39 pg/ng) was significantly lower than that of the other two groups. Estrogen and progesterone exert transcriptional regulation through estrogen receptor α/β and progesterone receptor A/B expressed in the uterus. At the same time, the co-regulation of other cytokines, transcription factors and growth factors are also required to promote the normal establishment of endometrial receptivity1,42. In humans, ERα is the dominant subtype expressed in uterus. Estrogen induces epithelial proliferation and differentiation through growth factor-mediated paracrine action via ERα50. Progesterone induces stromal cell proliferation through the joint involvement of PR expressed on the epithelium and stroma, while antagonizing estrogen-induced epithelial proliferation51. Therefore, both estrogen and progesterone regulate uterine receptivity in a spatiotemporal specific manner. Our study indicated that an appropriate E2/P ratio value was important for maintaining the endometrial receptivity state.
Age is an important influencing factor on pregnancy rate of assisted reproduction. There have some studies reported the relationship between advanced maternal age and decline of endometrial receptivity52,53,54, but all these researches only studied the endometrial receptivity from the standpoint of pregnancy outcomes. So far, there has no research combined age with the gene expression test of endometrial receptivity. Our study showed that there has a significant difference in age between the normal WOI and displaced WOI patients, and the results of logistic regression analysis also showed that age was positively correlated with displaced WOI. We divided all patients who received ERA test into three groups according to their age, we found that the displaced WOI rate increased gradually with the increase of age group, which indicated that as age increases, the incidence of displaced WOI also gradually increases. It is reported that the decrease of endometrial receptivity is the major cause for age-related decline of fecundity. The decrease in endometrial receptivity with age is responsible for the higher rate of implantation failure in older women. Patients with nonfunctioning ovaries have a better outcome in ovum donation programs55. In the progress of aging, senescent cells secrete inflammatory factors inducing a chronic, low-degree, systemic, uncontrolled inflammatory state, termed “inflammaging”. Accumulated inflammatory mediators such as interleukin-10 (IL-10), interleukin-6 (IL-6), leukemia inhibitory factor (LIF) and tumor necrosis factor-a (TNF-a) in peripheral circulation and local uterus, together with estrogen and progesterone, lead to the abnormal expression and function of endometrial receptivity markers such as homeobox A10 (HOXA10), integrin av03, mucoprotein (MUC1) and matrix metalloproteinase (MMP), which disturbs the endometrial homeostasis and contributes to the age-related decline of endometrial receptivity56. Furthermore, advanced age influences epigenetic regulation in the endometrium, thus altering the relation between the epigenetic and chronological age57. These may be the mechanisms by which age affects endometrial receptivity.
We also found that there was a significant difference in the number of previous failed ET cycle between the normal WOI and displaced WOI patients, and the results of logistic regression analysis also showed that the number of previous failed ET cycle was positively correlated with the displaced WOI. According to the number of previous failed ET cycle, we divided the patients into three groups for detailed analysis, and found that the displaced WOI rate increased gradually with the increase of number of previous failed ET cycle. Our result showed that the more the number of previous failed cycle, the higher the rate of displaced WOI in patients. Psychological pressure and loss of hope for success are factors that must be taken seriously in the in ART clinical practice58. It was reported that a surprising number of couples stop continuing treatment after at least one failed IVF cycle59. The cause of implantation failure is partially attributed to to individual differences of WOI. According to our study, we suggest that positive actions including endometrial receptivity assessments are recommended to improve the pregnancy outcomes for elderly patients and patients who have experienced one or more failed ET cycles.
As this article is a retrospective study, we know that when patients are voluntarily included, selection bias may be introduced. Our large sample size may mask subtle differences between groups, especially if some influencing variables are not controlled. In addition, PSM was used in the analysis of RIF groups in our study. As some individuals may not be able to find suitable matches, which will reduce the effective RIF sample size in the analysis and potentially decrease the statistical power of the study, this is also a limitation of this article. In future work, we hope to classify RIF according to etiology and conduct prospective studies on ERA if the conditions permit, so as to make the results more reliable and credible. As our study suggests that the endometrial receptivity is influenced by some maternal factors, including age-time changes, hormonal changes, and there maybe has some other unknown factors. Due to the invasive examination of endometrial biopsy, embryo transfer cannot be performed during the biopsy cycle. So, the endometrial receptivity status during the biopsy cycle may differ from the actual embryo transfer cycle, which may also lead to different outcomes. We believe this may also be a reason for controversy regarding the application of ERA. Therefore, it is necessary to search for new non-invasive methods for ERA.
Conclusions
The clinical pregnancy rate and live birth rate of patients with previous failed ET cycle was improved after pET guided by ERA, especially in RIF patients; the early abortion rate of the non-RIF patient was reduced after pET guided by ERA. Moreover, we firstly found that an appropriate E2/P ratio is beneficial for maintaining endometrial receptivity state, and the probability of displacement WOI will increase with age and number of previous failed ET cycle.
Data availability
The datasets generated and analyzed during the current study are available in patients’ records attending the Reproductive Medicine Center, Renmin Hospital, Hubei University of Medicine. The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request and also with the permission of the Reproductive Medicine Center, Renmin Hospital, Hubei University of Medicine.
References
Awonuga, A. O. et al. Determinants of embryo implantation: Roles of the endometrium and embryo in implantation success. Reprod. Sci. 30(8), 2339–2348 (2023).
Massarotti, C., Makieva, S. & Stigliani, S. Editorial: Challenges in fertilization and implantation success. Front. Cell Dev. Biol. 11, 1268635 (2023).
Cimadomo, D. et al. Leave the past behind: women’s reproductive history shows no association with blastocysts’ euploidy and limited association with live birth rates after euploid embryo transfers. Hum. Reprod. 36(4), 929–940 (2021).
Hiraoka, T., Osuga, Y. & Hirota, Y. Current perspectives on endometrial receptivity: A comprehensive overview of etiology and treatment. J. Obstet. Gynaecol. Res. 49(10), 2397–2409 (2023).
Sebastian-Leon, P. et al. Transcriptional changes through menstrual cycle reveal a global transcriptional derepression underlying the molecular mechanism involved in the window of implantation. Mol. Hum. Reprod. 27(5), 27 (2021).
Bui, A. H., Timmons, D. B. & Young, S. L. Evaluation of endometrial receptivity and implantation failure. Curr. Opin. Obstet. Gynecol. 34(3), 107–113 (2022).
de Ziegler, D., Scott, R. T. Jr. & Pirtea, P. If recurrent implantation failure (RIF) is rare, what could be the cause of repeat unsuccessful ARTs?. Fertil. Steril. S0015–0282(23), 02036–02038 (2023).
Urban, A., Warzecha, D., Laudanski, P., Pietrzak, B., & Wielgos, M. Personalized embryo transfer (pET) guided by endometrial receptivity (ER) assessment—a possibility to increase effectiveness of IVF procedures. Review of available methods. Ginekol Pol. (2023).
Wu, J., Sheng, J., Wu, X. & Wu, Q. Ultrasound-assessed endometrial receptivity measures for the prediction of in vitro fertilization-embryo transfer clinical pregnancy outcomes: A meta-analysis and systematic review. Exp. Ther. Med. 26(3), 453 (2023).
Rubin, S. C. et al. Review of endometrial receptivity array: A personalized approach to embryo transfer and its clinical applications. J. Pers. Med. 13(5), 749 (2023).
Polanski, L. T. et al. What exactly do we mean by “recurrent implantation failure”? A systematic review and opinion. Reprod. Biomed. Online. 28(4), 409–423 (2014).
Chinese Association of Reproductive Medicine; Professional Committee of Reproductive Medicine, China Medical Women’s Association. [Expert consensus on diagnosis and treatment of recurrent implantation failure]. Zhonghua Yi Xue Za Zhi. 103(2), 89–100 (2023).
Ziebe, S. et al. Embryo morphology or cleavage stage: How to select the best embryos for transfer after in-vitro fertilization. Hum. Reprod. 12(7), 1545–1549 (1997).
Gardner, D. K. et al. Reprint of: Blastocyst score affects implantation and pregnancy outcome: Towards a single blastocyst transfer. Fertil. Steril. 112(Suppl 1), e81–e84 (2019).
Díaz-Gimeno, P. et al. A genomic diagnostic tool for human endometrial receptivity based on the transcriptomic signature. Fertil. Steril. 95(1), 50–60 (2011).
Sun, B. & Yeh, J. Non-invasive and mechanism-based molecular assessment of endometrial receptivity during the window of implantation: Current concepts and future prospective testing directions. Front. Reprod. Health. 4, 863173 (2022).
Liu, Z. et al. The clinical efficacy of personalized embryo transfer guided by the endometrial receptivity array/analysis on IVF/ICSI outcomes: A systematic review and meta-analysis. Front. Physiol. 13, 841437 (2022).
Simón, C. et al. A 5-year multicentre randomized controlled trial comparing personalized, frozen and fresh blastocyst transfer in IVF. Reprod. Biomed. Online. 41(3), 402–415 (2020).
Ohara, Y. et al. Clinical relevance of a newly developed endometrial receptivity test for patients with recurrent implantation failure in Japan. Reprod. Med. Biol. 21(1), e12444 (2022).
Chen, J. et al. The RNA-seq based endometrial receptivity test (rsERT) compared to pinopode: A better diagnostic tool for endometrial receptivity for patients with recurrent implantation failure in Chinese population. Front. Endocrinol. (Lausanne). 13, 1009161 (2022).
Hessami, K., Khatibi, A. & Arian, S. E. Response letter to Endometrial receptivity array before frozen embryo transfer cycles: A systematic review and meta-analysis. Fertil. Steril. 119(5), 892 (2023).
Richter, K. S. & Richter, M. L. Personalized embryo transfer reduces success rates because endometrial receptivity analysis fails to accurately identify the window of implantation. Hum. Reprod. 38(7), 1239–1244 (2023).
Riestenberg, C. et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil. Steril. 115(4), 1001–1006 (2021).
Glujovsky, D. et al. Personalized embryo transfer guided by endometrial receptivity analysis: A systematic review with meta-analysis. Hum. Reprod. 38(7), 1305–1317 (2023).
Liu, Z. et al. Overexpression of hypoxia-inducible factor 1α and excessive vascularization in the peri-implantation endometrium of infertile women with chronic endometritis. Front. Endocrinol. (Lausanne). 13, 1001437 (2022).
Shan, J., Li, D. J. & Wang, X. Q. Towards a better understanding of endometriosis-related infertility: A review on how endometriosis affects endometrial receptivity. Biomolecules. 13(3), 430 (2023).
Liu, S. et al. The role of decidual immune cells on human pregnancy. J. Reprod. Immunol. 124, 44–53 (2017).
Mori, M., Bogdan, A., Balassa, T., Csabai, T. & Szekeres-Bartho, J. The decidua-the maternal bed embracing the embryo-maintains the pregnancy. Semin. Immunopathol. 38(6), 635–649 (2016).
Moffett, A. & Colucci, F. Uterine NK cells: active regulators at the maternal-fetal interface. J. Clin. Invest. 124(5), 1872–1879 (2014).
Amjadi, F. et al. The uterine immunological changes may be responsible for repeated implantation failure. J. Reprod. Immunol. 138, 103080 (2020).
Lédée, N. et al. The uterine immune profile may help women with repeated unexplained embryo implantation failure after in vitro fertilization. Am. J. Reprod. Immunol. 75(3), 388–401 (2016).
Mukherjee, N., Sharma, R. & Modi, D. Immune alterations in recurrent implantation failure. Am. J. Reprod. Immunol. 89(2), e13563 (2023).
Genest, G. et al. Immunomodulation for unexplained recurrent implantation failure: Where are we now?. Reproduction. 165(2), R39–R60 (2023).
Ebrahimi, F., Omidvar-Mehrabadi, A., Shahbazi, M. & Mohammadnia-Afrouzi, M. Innate and adaptive immune dysregulation in women with recurrent implantation failure. J. Reprod. Immunol. 164, 104262 (2024).
Ma, J., Gao, W. & Li, D. Recurrent implantation failure: A comprehensive summary from etiology to treatment. Front. Endocrinol. (Lausanne). 13, 1061766 (2023).
ESHRE Working Group on Recurrent Implantation Failure; Cimadomo, D., de Los Santos, M. J., Griesinger, G. et al. ESHRE good practice recommendations on recurrent implantation failure. Hum. Reprod. Open. 3, hoad023 (2023).
Von Woon, E. et al. Number and function of uterine natural killer cells in recurrent miscarriage and implantation failure: A systematic review and meta-analysis. Hum. Reprod. Update. 28(4), 548–582 (2022).
Vomstein, K. et al. Uterine microbiota plasticity during the menstrual cycle: Differences between healthy controls and patients with recurrent miscarriage or implantation failure. J. Reprod. Immunol. 151, 103634 (2022).
Stevens Brentjens, L. et al. Endometrial factors in the implantation failure spectrum: Protocol of a MUltidisciplinary observational cohort study in women with Repeated Implantation failure and recurrent Miscarriage (MURIM Study). BMJ Open. 12(6), e056714 (2022).
Palomba, S. & Vitagliano, A. Is the era of the endometrial scratching finished?. Fertil. Steril. 118(3), 603 (2022).
Wong, T. Y. et al. Effect of endometrial scratching on unassisted conception for unexplained infertility: a randomized controlled trial. Fertil. Steril. 117(3), 612–619 (2022).
Stevens Brentjens, L. B. P. M. et al. An integrative analysis of endometrial steroid metabolism and transcriptome in relation to endometrial receptivity in in vitro fertilization patients. F S Sci. 4(3), 219–228 (2023).
Perry, G. A., Ketchum, J. N. & Quail, L. K. Importance of preovulatory estradiol on uterine receptivity and luteal function. Anim. Reprod. 20(2), e20230061 (2023).
Al-Lamee, H. et al. Altered endometrial oestrogen-responsiveness and recurrent reproductive failure. Reprod. Fertil. 3(1), 30–38 (2022).
Parisi, F., Fenizia, C., Introini, A., et al. The pathophysiological role of estrogens in the initial stages of pregnancy: Molecular mechanisms and clinical implications for pregnancy outcome from the periconceptional period to end of the first trimester. Hum. Reprod. (2023).
Shekhar, B. et al. Low serum progesterone on day of transfer adversely impacts ongoing pregnancy rates in hormonally prepared single blastocyst frozen embryo transfer cycles. Eur. J. Obstet. Gynecol. Reprod. Biol. 289, 55–59 (2023).
Gurunath, S. Premature progesterone elevation in in vitro fertilisation cycles—current perspectives. J. Hum. Reprod. Sci. 15(4), 325–336 (2022).
Ullah, K. et al. Serum estradiol levels in controlled ovarian stimulation directly affect the endometrium. J. Mol. Endocrinol. 59(2), 105–119 (2017).
Cheng, J., Yang, S., Ma, H., Liang, Y. & Zhao, J. Estradiol (E2) Reduction adversely affect the embryo quality and clinical outcomes of in vitro fertilization and embryo transfer (IVF-ET). J. Healthc. Eng. 2022, 2473876 (2022).
Yu, K. et al. Estrogen receptor function: Impact on the human endometrium[J]. Front. Endocrinol. 13, 827724 (2022).
Tan, J. et al. Differential uterine expression of estrogen and progesterone receptors correlates with uterine preparation for implantation and decidualization in the mouse[J]. Endocrinology 140(11), 5310–5321 (1999).
Pathare, A. D. S., Loid, M., Saare, M. et al. Endometrial receptivity in women of advanced age: An underrated factor in infertility. Hum. Reprod. Update. 2023 Jul 19:dmad019.
Shao, Y., Feng, H., Li, L., Zong, Y. & Liang, J. Predictive value of endometrial receptivity for pregnancy outcomes of in-vitro fertilization embryo transfer for patients of different ages. Altern. Ther. Health Med. 29(4), 210–217 (2023).
Zhao, J. et al. Relationship between advanced maternal age and decline of endometrial receptivity: A systematic review and meta-analysis. Aging (Albany NY). 15(7), 2460–2472 (2023).
Yaron, Y. et al. Endometrial receptivity: The age-related decline in pregnancy rates and the effect of ovarian function. Fertil. Steril. 60(2), 314–318 (1993).
Zhao, X., Song, J. & Xia, T. Progress in the study of the effect of inflammaging on endometrial receptivity. Chin. J. Reprod. Contracept. 40, 684–688. https://doi.org/10.3760/cma.j.cn101441-20190721-00314 (2020).
Pathare, A. D. S. et al. Endometrial receptivity in women of advanced age: An underrated factor in infertility. Hum. Reprod. Update. 29(6), 773–793 (2023).
Sharma, A. & Shrivastava, D. Psychological problems related to infertility. Cureus. 14(10), e30320 (2022).
Kulkarni, G. et al. Survey of reasons for discontinuation from in vitro fertilization treatment among couples attending infertility clinic. J. Hum. Reprod. Sci. 7(4), 249–254 (2014).
Funding
This study was supported by a research grant (No. 81771543) from the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Contributions
S.Y.X contributed to statistical analysis and interpretation of data and drafting of the manuscript. Y.Z and Y.Z.Z conceived and designed this study, performed statistical analysis and participated in the discussion. H.L.D and Y.X were involved in acquisition, analysis and interpretation of data. S.Y.X, YZ, C.J.Z and Y.Z.Z participated in the discussion and critically revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, S., Diao, H., Xiong, Y. et al. The study on the clinical efficacy of endometrial receptivity analysis and influence factors of displaced window of implantation. Sci Rep 15, 7326 (2025). https://doi.org/10.1038/s41598-025-91745-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-91745-y
Keywords
This article is cited by
-
Endometrial receptivity–guided embryo transfer: a systematic review and meta-analysis of the evidence
Journal of Assisted Reproduction and Genetics (2026)
-
Interaction between embryo transfer timing (D3/D4) and duration of progesterone exposure (P3/P4) on live birth rate in hormone replacement therapy cycles of frozen embryo transfer
Reproductive Biology and Endocrinology (2025)