Abstract
Atrial fibrosis is associated with atrial fibrillation (AF) and stroke. Tumor endothelial marker 1 (TEM1/CD248) is a transmembrane protein that appears in mesenchymal lineage-derived cells only during embryogenesis. Re-upregulation of TEM1 in fibroblasts plays an important role in organ fibrosis. We evaluated TEM1 expression in atrial fibrosis of AF patients and its physiological significance. Left atrial (LA) appendages were collected from 30 AF patients (mean age 64.0 years, 76.7% male) who underwent indicated cardiac surgery. Immunofluorescence staining showed TEM1 expression in the atrial cardiac fibroblasts in AF but not in normal atrial tissue. Western blot could detect TEM1 expression in the LA tissues of all 30 AF patients. There was a positive correlation between the levels of TEM1 expression in western blot with the severity of atrial fibrosis. In animal experiment, angiotensin II (Ang II) infusion induced TEM1 expression and atrial fibrosis in mice. Atrial fibrosis was less severe in TEM1-deficient transgenic mice after Ang II infusion. TEM1 activated cardiac fibroblast and increased its proliferation, survival and migration. Our study results indicate that TEM1 re-expression in cardiac fibroblasts in AF is associated with severity of atrial fibrosis. TEM1 changes the cell behaviors of cardiac fibroblasts and contributes to atrial fibrosis.
Similar content being viewed by others
Introduction
Atrial fibrosis is a hallmark of maladaptive remodeling of the atria and produces the substrate to promote atrial fibrillation (AF) by causing local conduction disturbance1,2. Atrial fibrosis not only induces AF but also causes atrial chamber enlargement and contractile dysfunction, increasing the risk of atrial thrombosis and embolic stroke3,4. Atrial cardiac fibroblast is the major cell type that is responsible for the process of atrial fibrosis1,5. Atrial cardiac fibroblasts proliferate and undergo a transformation into activated cardiac myofibroblasts during cardiac stress, secreting excessive amount of collagen and other extracellular matrix (ECM), thereby promoting fibrosis5. Angiotensin II (Ang II), connective tissue growth factor (CTGF), platelet-derived growth factor (PDGF) and transforming growth factor-beta (TGF-β) are the major profibrotic stimuli of atrial fibrosis1,5. The profibrotic stimuli work on their cell membrane receptors on cardiac fibroblasts and activate the downstream signaling pathways, such as the Smad proteins, inducing a range of transcription factors to increase the ECM production.
Tumor endothelial marker 1 (TEM1), also known as CD248 or endosialin, is a type I transmembrane glycoprotein which belongs to a C-type lectin domain protein family6. The protein structure of TEM1 includes six domains which are extracellular N-terminal C-type lectin domain, a sushi domain, epidermal growth factor (EGF) domain, a mucin-like region, transmembrane domain, and cytoplasmic tail6,7. TEM1 expression is found in cells derived from the mesenchymal lineage during embryo development and plays a role in tissue formation. Its expression level in the normal tissues of healthy adults after birth becomes very low or undetectable8,9. However, TEM1 expression can be re-upregulated under certain pathological conditions, such as in the pericytes of tumor blood vessels10,11. During renal, hepatic, pulmonary fibrosis and wound healing, TEM1 also expresses in activated fibroblasts and contributes to the fibrotic process12,13,14,15. Expression of TEM1 in these pathological conditions suggests that TEM1 plays a role in tissue remodeling and repairing process. Previously, we found TEM1 expression can be detected in the ventricular cardiomyocytes and cardiac fibroblasts in patients with heart failure16,17. The cell composition in atria and ventricles is quite different. Cardiomyocytes are the most prevalent cardiac cells and comprise higher percentages in ventricles than atria. On the contrary, the percentage of fibroblasts is much higher in atria than ventricles18. Atrial fibroblasts also behave differently and show a more reactive response than ventricular fibroblasts to pro‑fibrotic stimuli19. TEM1 expression and its physiological role in atrial fibrosis are unknown. A deeper understanding of the pathophysiology of atrial fibrosis may pave the way for developing new strategies to prevent AF and stroke. We hypothesized that TEM1 expression is increased in atrial cardiac fibroblasts during atrial fibrosis and participates in atrial fibrosis by regulating cell behaviors of cardiac fibroblasts. The aim of this study was to evaluate TEM1 expression in atrial tissue in AF patients and examine its association with atrial fibrosis. The influences of TEM1 expression on cell behaviors of cultured cardiac fibroblasts were also explored.
Methods
Human atrial tissue
Adult patients (≥ 18 years) with AF who were admitted for indicated cardiac surgery were consecutively included in our hospital. Echocardiography was performed before surgery and the CHA2DS2-VASc score was calculated. Because the left atrial (LA) appendage has been proven to be a source of cerebral or systemic emboli in AF, LA appendage resection or closure is proposed to prevent postoperative embolism in AF patients undergoing cardiac surgery for other indications20. Our patients underwent both the Maze procedure for AF treatment and surgical resection of LA appendage during the indicated cardiac surgery. Informed consent was obtained from each patient and the excised LA appendages were collected for analysis. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Institutional Review Board of our hospital (IRB number: A-ER-110-031). Normal human LA paraffin tissue section slide (category number HuFPT046) was purchased from US Biomax, Inc., Derwood, USA.
Immunofluorescence staining
To identify TEM1 expression in atrial tissue, double immunofluorescence staining was performed using primary antibodies for troponin (cardiomyocyte marker, 1:1000 Abcam), fibroblast activation protein (FAP, cardiac fibroblast marker, 1:1000 Abcam) and TEM1 (1:1000, ATLAS for human and Proteintech for mouse). Secondary antibodies were goat anti-mouse IgG Alexa Fluor 546 and goat anti-rabbit IgG Alexa Fluor 488 (Invitrogen). Each slide was incubated with primary antibodies at 4 °C overnight, then secondary antibodies for 1 h at room temperature. Nuclei were counterstained with 4ʹ, 6-diamidino-2-phenylindole (DAPI, Sigma). Confocal microscope (Olympus FV3000) was used for observation.
Western blot
Atrial tissues or cultured cardiac fibroblasts were treated with lysis buffer and protein samples were prepared. The proteins were loaded and separated using SDS-PAGE in phosphate-buffered saline (PBS). Samples were transferred to polyvinylidene difluoride-plus membrane, blocked with 5% skim milk and incubated with antibodies. Protein blots were visualized with the method of chemiluminescence and the band intensity was quantified using AlphaImager 2200 software (Alpha Innotech, San Leandro, CA). To evaluate the expression levels of TEM1 in atrial tissues, the band intensity of TEM1 was obtained and normalized with respect to the band intensity of the loading internal control of housekeeping protein, β-actin, detected on the same blot.
Collagen assay
Sirius red staining was used to visualize collagen fibers in atrial tissue. The slides were deparaffinized, washed and stained with 0.1% Sirius red solution (Abcam, ab246832) for 60 min. Under a light microscope (Olympus BX 63), collagen fibers were stained in red color, and muscle fibers in yellow color. Quantitative assessment of fibrosis was performed using a pixel-based method with Adobe Photoshop (Adobe Corporation, San Jose, CA, USA) in 10 randomly selected sections in each patient. Red pixels were counted and summed for a total number representing collagen. Yellow pixels and red pixels were summed for the total number of pixels. Severity of atrial fibrosis was evaluated by calculating the absolute total numbers of red pixels and the percentage of red pixels to total pixels.
Human atrial cardiac fibroblast
After excision by a cardiac surgeon, the LA appendages were placed in cardioplegic solution for immediate isolation of cardiac fibroblasts. The tissue samples were finely minced with scalpel blades and then subjected to a series of enzymatic digestion with collagenase according to the previously described methods21. The digests were pooled and centrifuged. The cell pellets were pooled, resuspended and seeded onto in culture plates containing Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin (Sigma). The isolated cells were confirmed to be cardiac fibroblasts by immunofluorescence staining with vimentin-positive (fibroblast marker, 1:2000 Abcam) and CD31-negative (endothelial cell marker, 1:1000 Abcam) immunoreactivity. TEM1 expression in cardiac fibroblasts were evaluated with the primary antibody for TEM1 (1:1000, ATLAS). Secondary antibodies were goat anti-mouse IgG Alexa Fluor 546 and goat anti-rabbit IgG Alexa Fluor 488. Nuclei were counterstained with DAPI. The slides were evaluated by confocal microscope (Olympus FV3000). Normal human cardiac fibroblasts were purchased from ScienCell (No. 6330, ScienCell, CA, USA). TEM1 expression (positive staining area of vimentin and TEM1) and whole cell area were traced and measured using a software (MetaMorph, Molecular Devices, Sunnyvale, CA). Five cells from the normal or patients were measured for the positive staining area and whole cell area.
Mouse atrial tissue
C57BL/6 mice (12–16 weeks, weight 18–25 g) were purchased from the National Laboratory Animal Center, Taipei, Taiwan. TEM1-deficient transgenic mice (TEM1lacZ/lacZ mice), in which TEM1 exon was disrupted and replaced by lacZ gene driven by TEM1 promoter, were bred in our laboratory as previously described8. The animals were anesthetized with intraperitoneal injection of zoletil/xylazine (50 mg/kg and 10 mg/kg). An osmotic mini-pump (Alzet 2004) was implanted for subcutaneous infusion of Ang II (1000 ng/kg/min, Sigma) or saline for 28 days. At 28 days after pump implantation, the mice were anesthetized and euthanized by placing in the chamber and introducing CO2 at a rate of 30% to 70% of the chamber volume per minute. The atrial tissues were taken for further studies. All animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. The study result was reported in accordance with ARRIVE guidelines. The protocol was approved by the Institutional Animal Care and Use Committee of our hospital (IACUC approval number 112044). Primary mouse cardiac fibroblasts were obtained from the atrial tissue of the mice with the same method used to isolate human cardiac fibroblasts.
Rat cardiac fibroblast cell culture
Rat cardiac fibroblasts (No. 6300, ScienCell, CA, USA) were maintained in DMEM supplemented with 10% fetal bovine serum in humidified atmosphere with 5% CO2 at 37 °C. The cultured cardiac fibroblasts received Ang II (Sigma) or other indicated treatment and western blot was then performed.
Recombinant TEM1 protein
Recombinant TEM1 protein (rTEM1) containing the extracellular domains were produced in our laboratory as previously described17. In brief, the pSecTag2-A vector containing human TEM1 with amino acid residues 1–639 was constructed. This vector was transfected into HEK293 cell line, and rTEM1 was induced by insulin and purified from the conditioned medium using nickel-chelating sepharose. Cultured cardiac fibroblasts were treated with rTEM1, and we then evaluated changes in their behavior.
TEM1 siRNA knockdown
Rat cardiac fibroblasts in culture were silenced for TEM1 expression and the changes of cell behaviors were observed. Briefly, the cells were grown to reach 60–70% confluency and then transfected with either TEM1 siRNA (Rat CD248, ID 293669, Dharmacon) or control siRNA (sc-37007, Santa Cruz) using lipid-based transfection reagents (Dharmacon) according to the manufacturer’s instructions. After transfection, the cells were harvested to evaluate the knockdown efficiency of TEM1.
Cell proliferation
Cell proliferation was evaluated with 5-bromo-2-deoxyuridine (BrdU) assay. After receiving rTEM1 treatment, 10 mM BrdU was added and followed by incubation for 2 h. The nuclear incorporation of BrdU was measured using a cell proliferation ELISA kit (Roche Diagnostics, Mannheim, Germany).
Cell apoptosis
Cell apoptosis was induced by starvation with 0.5% fetal bovine serum. The severity of apoptosis was measured by quantification of DNA fragments using a cell death ELISA kit (Roche Diagnostics, Mannheim, Germany).
Cell migration
A Transwell migration assay was used to evaluate cell migration. The cells (2.5 × 104 per chamber) were seeded into the upper chamber of the Transwell plates (Costar, Corning, New York, NY). Either rTEM1 or PDGF was added to the lower chamber. After incubation for 6 h at 37 °C, the cells on the upper membrane were removed with a cotton swab. The cells that migrated to the lower side of the membrane were then fixed and visualized by crystal violet stain. The number of migrated cells on the membrane was counted using ImageJ software in ten randomly selected views under × 100 magnification.
Statistical analysis
Data were presented as mean ± standard error. Comparisons between two groups were made by Mann Whitney U test. For multiple comparisons of groups, one-way ANOVA was used and followed by Bonferroni post hoc analysis. Correlation analyses were performed with Pearson correlation analysis. All statistical analyses were performed using SPSS 22 (SPSS Inc. Chicago, IL, USA). A p value < 0.05 was considered to be statistically significant.
Results
TEM1 expression in human atrial tissue and atrial fibrosis
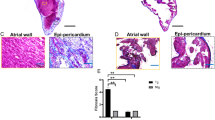
Overall, LA appendages were taken from 30 AF patients (mean age 64.0 ± 1.6 years, male 76.7%) who underwent indicated cardiac surgery (Table 1). Most patients (96.7%) received mitral and/or tricuspid valve surgery. The patients had normal left ventricular systolic function (ejection fraction 62.9 ± 1.6%) and dilated LA (LA dimension 5.27 ± 0.17 cm). The mean CHA2DS2-VASc score was 2.77 ± 0.29. Figure 1 shows the results of double immunofluorescence staining in normal control and 3 AF patients. Normal human LA tissue showed no significant TEM1 expression. But TEM1 expression was found in atrial cardiac fibroblasts in all 3 patients (Fig. 1A). Minimal TEM1 expression was observed in cardiomyocytes of patient B only, not in the other 2 patients (Fig. 1B). Western blot could detect TEM1 expression in the LA tissues of all 30 AF patients (Fig. 1C, Supplementary Fig. 1). Sirius red staining revealed more atrial fibrosis in AF patients compared with normal control (total number of red pixels: 157,811 ± 4064 vs. 50,863 ± 6357, percentage of red pixels to total pixels: 11.3 ± 0.3 vs. 3.7 ± 0.5%, all p < 0.001) (Fig. 1D). There was a significant correlation between the band intensity of atrial TEM1 expression (ratio of TEM1/β-actin) in western blot with the severity atrial fibrosis, including the absolute total number of red pixels (r = 0.47, p = 0.01) and the percentage of red pixels to total pixels (r = 0.48, p = 0.01) in Sirius red staining. The expression of TEM1 also had a positive correlation with the expression of galectin-3, another biomarker of atrial fibrosis and Smad3, a critical mediator of fibrosis (Fig. 1E). There were no significant correlations of atrial TEM1 expression with the LA diameter and CHA2DS2-VASc score.
TEM1 expression in human atrial tissue and atrial fibrosis. (A) Immunofluorescence staining of TEM1 of cardiac fibroblasts in the atrial tissue. Representative images of double staining of TEM1 (green) and fibroblast activation protein (red, FAP, cardiac fibroblast marker). White arrows indicate TEM1 staining (yellow) in cardiac fibroblasts in atrial tissue from 3 AF patients (f, i, and l). Nucleus was stained with DAPI. Scale bar = 20 μm. (B) Immunofluorescence staining of TEM1 of cardiomyocytes in the atrial tissues. Representative images of double staining of TEM1 (green) and troponin (red, cardiomyocyte marker). Minimal TEM1 expression was observed in cardiomyocytes of patient B (i). Nucleus was stained with DAPI. Scale bar = 20 μm. (C) Western blot could detect TEM1 expression in the atrial tissues of all 30 AF patients. The expression level of TEM1 in each patient was determined by calculating the ratio of the TEM1 signal intensity to the control signal intensity of β-actin. (D) Sirius red staining of the atrial tissues from normal control and AF patients. Representative images show collagen fibers stained in red color and myocardial muscle fibers stained in yellow color. Severity of atrial fibrosis was evaluated by quantification of the total number of red pixels and the percentage of red pixels to total pixels in normal and 30 AF patients (n = 10 sections in normal and each patient). ***p < 0.001 compared with normal. Scale bar = 100 µm. (E) Pearson correlation analysis for the association of atrial TEM1 expression levels (ratios of TEM1/β-actin expression levels in western blot) with total number of red pixels (upper panel, left), percentage of red pixels to total pixels (upper panel, right) and expression levels of galectin-3 (lower panel, left) or pSMAD (lower panel, right) in AF patients (n = 30). (F) Immunofluorescence staining of TEM1 in the atrial cardiac fibroblasts isolated from AF patients. Normal human cardiac fibroblasts (HCF) were purchased from ScienCell (6330, ScienCell, CA, USA). Representative images show double staining of TEM1 (green) and vimentin (red, cardiac fibroblast marker) in the atrial cardiac fibroblasts isolated from 2 AF patients. Nucleus was stained with DAPI. Scale bar = 50 μm.
Primary human cardiac fibroblasts were directly isolated from the LA tissue of AF patients. Cardiac fibroblast was identified by its spindle shape and confirmed by immunofluorescence study with negative stain of CD31 (endothelial cell marker) (Supplementary Fig. 2) and positive stain of vimentin (fibroblast marker). Immunofluorescence study showed there was increased TEM1 expression in the atrial fibroblasts isolated from AF patients compared to the normal control (TEM1 staining area ratio, 14.8 ± 2.1 vs. 4.4 ± 0.9%, p < 0.001) (Fig. 1F).
TEM1 expression in mouse atrial tissue and atrial fibrosis
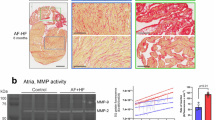
Since Ang II is a profibrotic stimulus leading to atrial fibrosis, we evaluated TEM1 expression in the mice atria after Ang II treatment. In wild-type mice, there was no significant TEM1 expression in the mouse atrial tissue after saline infusion, but TEM1 expression was detected in cardiac fibroblasts in atrial tissue after Ang II infusion (Fig. 2A). There was also minimal TEM1 expression in atrial cardiomyocytes (Fig. 2B). No TEM1 expression was found in TEM1-deficient mice after Ang II treatment. Sirius red staining showed increased atrial fibrosis in Ang II-treated wild-type mice compared with saline-treated wild-type mice or Ang II-treated TEM1-deficient mice (Ang II-treated wild-type mice vs. saline-treated wild-type mice vs. Ang II-treated TEM1-deficient mice, total number of red pixels: 173,371 ± 9798 vs. 58,504 ± 3109 vs. 81,927 ± 8736, percentage of red pixels to total pixels: 12.5 ± 0.7 vs. 4.2 ± 0.2 vs. 5.9 ± 0.6%, all p < 0.001) (Fig. 2C). Western blot shows that there was increased TEM1 expression and collagen production in the hearts of Ang II-treated wild-type mice. Collagen production was decreased in the hearts of Ang II-treated TEM1-deficient mice (Fig. 2D, Supplementary Fig. 3). Primary cardiac fibroblasts were directly isolated from the mouse atrial tissue with the same methods used to isolate human cardiac fibroblasts. Cardiac fibroblast was identified by its spindle shape and confirmed by immunofluorescence study with negative stain of CD31 (endothelial cell marker) (Supplementary Fig. 4) and positive stain of vimentin (fibroblast marker). Immunofluorescence staining showed TEM1 expression was increased in the cardiac fibroblasts isolated from the atrial tissue in Ang II-treated wild-type mice, but not in those received saline-treated mice (Ang II-treated vs. saline-treated wild-type mice, TEM1 staining area ratio: 11.2 ± 1.7 vs. 1.3 ± 0.3, p < 0.001) (Fig. 2E). When cultured rat cardiac fibroblasts received Ang II treatment, western blot showed there was an increased TEM1 expression in cardiac fibroblasts (Ang II/saline treatment western blot density ratio: saline vs. Ang II 100 nM vs. 500 nM vs. 1000 nM vs. 2000 nM vs. 4000 nM: 1 vs. 1.50 ± 0.08 vs. 1.52 ± 0.09 vs. 1.61 ± 0.18 vs. 1.74 ± 0.08 vs. 1.78 ± 0.09, p < 0.01). There was a progressively increased TEM1 expression at 24 h after Ang II 1000 nM treatment (Fig. 2F, Supplementary Fig. 5).
TEM1 expression in mouse atrial tissue and atrial fibrosis. (A) Immunofluorescence staining of TEM1 of cardiac fibroblasts in the atrial tissue of mice. Representative images show double staining of TEM1 (green) and fibroblast activation protein (red, FAP, cardiac fibroblast marker). White arrows indicate TEM1 staining (yellow) in cardiac fibroblasts in the atrial tissue of mice received Ang II (f). There was no TEM1 expression in the wild-type mice received saline (c) and TEM1-deficient mice (TEM1lacZ/lacZ mice) received Ang II (i). Nucleus was stained with DAPI. Scale bar = 20 μm. (B) Immunofluorescence staining of TEM1 of cardiomyocytes in the atrial tissue of mice. Representative images show double staining of TEM1 (green) and troponin (red, cardiomyocyte marker). There was minimal TEM1 expression in atrial cardiomyocytes of wild-type mice received Ang II infusion (f). Nucleus was stained with DAPI. Scale bar = 20 μm. (C) Sirius red staining in the atrial tissues of wild-type mice received saline (left, n = 3), wild-type mice received Ang II (middle, n = 3) and TEM1-deficient mice (TEM1lacZ/lacZ mice, n = 3) received Ang II (right). Severity of atrial fibrosis was evaluated by quantification of the total number of red pixels and the percentage of red pixels to total pixels (n = 5 sections in each mouse). ***p < 0.001 compared with wild-type mice received saline infusion and TEM1-deficient mice received Ang II. Scale bar = 100 µm. (D) Western blot of TEM1 and collagen-3 in the hearts of wild-type mice received saline or Ang II and TEM1-deficient mice (TEM1lacZ/lacZ mice) received Ang II. Representative image shows decreased collagen-3 expression in the TEM1-deficient mice after Ang II. (E) Immunofluorescence staining of TEM1 in the atrial cardiac fibroblasts isolated from mice. Representative images show double staining of TEM1 (green) and vimentin (red, cardiac fibroblast marker). Significant TEM1 staining (yellow) was found in the cardiac fibroblasts isolated from 2 wild-type mice received Ang II. Nucleus was stained with DAPI. Scale bar = 50 μm. (F) Western blot of TEM1 in cultured cardiac fibroblasts received Ang II treatment at different concentrations and time points. Representative images show increased TEM1 expression after Ang II treatment. The expression levels were expressed as a ratio to baseline before Ang II treatment (n = 3). *p < 0.05 and **p < 0.01 compared to normal at baseline before Ang II treatment.
TEM1 and cell behaviors of cardiac fibroblasts
Because up-regulation of alpha-smooth muscle actin (α-SMA) is a hallmark of activated fibroblasts, we first looked at the effect of TEM1 on α-SMA expression. Figure 3A shows rTEM1 treatment increased α-SMA expression and knockdown of TEM1 decreased its expression in cardiac fibroblasts showing that TEM1 activates cardiac fibroblasts (Fig. 3A, Supplementary Fig. 6). Treatment with rTEM1 increased proliferation of cardiac fibroblasts. The absolute BrdU absorbance value increased significantly after rTEM1 treatment compared with saline (saline vs. rTEM1 10 nM vs. 50 nM vs. 100 nM vs. PDGF 20 ng/mL: 0.55 ± 0.02 vs. 0.62 ± 0.04 vs. 0.73 ± 0.04 vs. 0.74 ± 0.04 vs. 0.77 ± 0.01, p < 0.01) showing increased proliferation induced by rTEM1 (Fig. 3B). TEM1 knockdown reduced the spontaneous proliferation of cardiac fibroblasts (scrambled- vs. TEM1-siRNA transfected cells, BrdU absorbance: 24 h 1.84 ± 0.05 vs. 1.50 ± 0.07, 48 h 0.62 ± 0.02 vs. 0.44 ± 0.04, all p < 0.05) (Fig. 3C, Supplementary Fig. 7). Treatment with rTEM1 decreased starvation-induced apoptosis of cardiac fibroblasts (cell death ELISA absorbance, saline vs. rTEM1 10 nM vs. 50 nM vs. 100 nM: 1.06 ± 0.09 vs. 0.61 ± 0.05 vs. 0.49 ± 0.11 vs. 0.33 ± 0.02, p < 0.05). TEM1 knockdown increased starvation-induced cell apoptosis (scrambled- vs. TEM1-siRNA transfected cells, cell death ELISA absorbance: 24 h 0.69 ± 0.11 vs. 2.88 ± 0.05, 48 h 1.03 ± 0.003 vs. 2.08 ± 0.08, all p < 0.01) (Fig. 3D). In cell migration assay, rTEM enhanced migration of cardiac fibroblasts (number of migrated cells per field: saline vs. rTEM1 10 nM vs. 100 nM vs. PDGF 50 ng/mL: 140.5 ± 5.0 vs. 177.2 ± 5.8 vs. 226.5 ± 7 vs. 274.9 ± 7.6, p < 0.001) (Fig. 3E). TEM1 knockdown decreased migratory ability of cardiac fibroblasts (number of migrated cells per field: normal cells vs. scrambled- vs. TEM1-siRNA transfected cells: 161.0 ± 5.1 vs. 147.8 ± 4.4 vs. 63.9 ± 2.7, p < 0.001) (Fig. 3F). Finally, blocking TEM1 with antibody could prevent the effects of angiotensin II-induced collagen production in cardiac fibroblasts (Fig. 3G, Supplementary Fig. 8).
TEM1 influences cell behaviors of cardiac fibroblasts. (A) Western blot of α-SMA in cultured cardiac fibroblasts. Representative images show rTEM1 increased and knockdown of TEM1 decreased α-SMA expression. (B) Treatment of rTEM1 increased proliferation of cardiac fibroblasts. The data were presented as absolute values of absorbance of BrdU ELISA (n = 3). PDGF was used as positive control. *p < 0.05 and **p < 0.01 compared to control group without adding rTEM1. (C) Cardiac fibroblasts were transfected with siRNA specific for TEM1 or scrambled-siRNA. Representative image showed decreased TEM1 expression after TEM1-siRNA transfection compared with scrambled-siRNA transfection. Knockdown of TEM1 reduced spontaneous cell proliferation. The data were presented as absolute values of absorbance of BrdU ELISA (n = 3). *p < 0.05 compared to cells transfected with scrambled-siRNA. (D) Treatment of rTEM1 reduced starvation-induced apoptosis of cardiac fibroblasts. The data were presented as absolute values of absorbance of Cell Death ELISA (n = 3). *p < 0.05 and **p < 0.01 compared to control group without adding rTEM1. Knockdown of TEM1 increased starvation-induced apoptosis of cardiac fibroblasts (n = 3). **p < 0.01 compared to cells transfected with scrambled-siRNA. (E) Cell migration was determined by Transwell migration assay. Bar graph shows the number of migrated cells per field after adding saline (normal), rTEM1 10 nM, 100 nM or PDGF. The data were calculated according to the number of migrated cells in 10 randomly selected field under × 100 magnification. **p < 0.01 and ***p < 0.001 compared to normal. (F) Knockdown of TEM1 decreased cell migration of cardiac fibroblasts. Representative images showed less migrated cardiac fibroblasts in TEM1 knockdown cells (n = 3). ***p < 0.001 compared to normal cells or cells transfected with scrambled-siRNA. (G) Western blot of collagen-3 in cultured cardiac fibroblasts received Ang II or Ang II plus anti-TEM1 antibody treatment. Representative images show anti-TEM1 antibody decreased Ang II-induced collagen-3 expression.
TEM1 signaling pathway
Finally, we looked at the potential signaling pathways of TEM1 in cardiac fibroblasts. We found rTEM1 treatment activated the Akt/PKB (protein kinase B) kinase and extracellular signal-regulated kinase (Erk) pathways. Akt or Erk inhibitor treatment could partially suppress rTEM1-induced collagen production in cardiac fibroblasts (Fig. 4, Supplementary Fig. 9).
TEM1 signaling pathway in cardiac fibroblasts. Western blot of p-Akt, p-ERK and collagen-3 in cardiac fibroblasts after indicated treatment. Representative images show rTEM1-induced collagen-3 production was reduced after adding U0126 (Erk inhibitor) or LY294002 (Akt/PI3 kinase inhibitor). PDGF was used as a positive control.
Discussion
To the best of our knowledge, this is the first study investigating the association of TEM1 expression with atrial fibrosis. The present study demonstrates that atrial TEM1 expression levels are associated with the histologically proven extent of atrial fibrosis in patients with AF. The in vitro study shows that TEM1 may influence the cell behaviors of cardiac fibroblasts by enhancing their proliferation, survival and migration, thus supporting our hypothesis of TEM1’s contribution to atrial fibrosis.
Normally, TEM1 expression is restricted to embryonic development8,9. The re-emergence of TEM1 in cardiac fibroblasts within the atria of the heart in AF patients is quite like the conditions in other organ fibrosis. In the liver, TEM1 re-expression in hepatic stellate cells was found after hepatic injury and enhanced hepatic fibrosis13. Collagen production and accumulation in the liver was reduced in TEM1 knockout mice. Antibody‑drug conjugate was developed to bind and kill TEM1-positive hepatic stellate cells and this method provided anti-fibrotic effects in mice with liver fibrosis22. In pulmonary fibrosis, there was higher protein levels of TEM1 in the lung fibroblasts compared with the cells from normal lung tissue. The expression levels of TEM1 were positively correlated with the severity of pulmonary fibrosis14. Further study shows that some lung fibroblasts had high TEM1 expression and were usually localized in the collagen-rich connective tissue. TEM1 expression could be used as a marker to differentiate subpopulations of lung fibroblasts23. The drug is under development to inhibit pulmonary fibrosis by targeting and killing TEM1-positive lung fibroblasts. TEM1 was upregulated in the pericytes and stromal fibroblasts in patients with chronic kidney disease and contributes to renal fibrosis24. In the animal model of kidney injury using unilateral ureteral obstruction, TEM1 expression was found in the stromal fibroblasts. TEM1-deficient transgenic mice had significantly less fibroblast accumulation and renal fibrosis in this kidney injury model12. The drug targeting and killing TEM1-positive fibroblasts has excellent anti-fibrotic effect in renal fibrotic mice25. In atrial fibrosis, we found the similar phenomenon that TEM1-deficient transgenic mice have significantly less atrial fibrosis after Ang II treatment. TEM1-positive cells contribute to the fibrotic process and become the target of anti-fibrotic treatment in some organs. The potential effects of using drug that kills TEM1-positive cardiac fibroblasts to reduce atrial fibrosis in AF and prevent AF recurrence after antiarrhythmic drug or catheter ablation need further study.
Our study demonstrates that rTEM1 treatment or TEM1 knockdown alter the behavior of cardiac fibroblasts. The specific effects of TEM1 on cellular behaviors can vary depending on the cell type and the surrounding environment. The re-expression of TEM1 in pathological conditions and its influence on cellular behaviors have been reported in several mesenchyme-derived cell types, including pericyte in cancer, stellate cells in hepatic injury, smooth muscle cells in atherosclerosis, cardiomyocyte in heart failure, and dermal fibroblasts in wound healing. TEM1 was expressed in the pericytes of tumor-associated vessels. Its expression increased pericyte proliferation, enhancing cancer angiogenesis and growth11,26. Hepatic stellate cells are the pericytes found in perisinusoidal space of the liver. PDGF stimulation induced proliferation of the wild-type, but not the TEM1-deficient stellate cells13. In atherosclerosis, TEM1 expression in vascular smooth muscle cells induced the cells to switch to a more synthetic and proinflammatory state, thereby contributing to atherosclerosis progression27. In heart failure, TEM1 was upregulated in cardiomyocytes and TEM1 increased proliferation and hypertrophy of cardiomyocytes17. During wound healing, activated dermal fibroblasts have significant TEM1 expression. The migration, adhesion, and proliferation of cultured fibroblasts were suppressed following TEM1 knockdown15. Overall, TEM1 is a functional protein that carries significant influence on cellular behaviors in various contexts. Our findings added the functional role of TEM1 in atrial cardiac fibroblasts and its influence on their cell behaviors.
Our study has several limitations. First, we could not obtain the atrial tissue directly from subjects with sinus rhythm. Only commercially available normal human left atrial tissue sections can be used. Since the sections were from normal subjects, we think they can represent the atrial tissue from sinus rhythm. Second, we previously found soluble TEM1 levels had correlations with heart failure biomarkers of cardiac fibrosis, including growth differentiation factor-15 and galectin-316. In the current study, we also found the TEM1 and galectin-3 expression levels had a positive correlation in the atrial tissues of AF patients. The potential role of TEM1 as a prognostic biomarker in patients with AF or heart failure remains unknown and requires further investigation. Third, our current and previous study showed that both cardiac fibroblasts and cardiomyocytes could express TEM1 after stimulation17. The cellular function of TEM1 varies depending on the cell type and environment. Therefore, unlike its potential role in the other organs, such as liver or kidney, further research is needed to explore the role of TEM1 in the cell–cell interaction between cardiac fibroblasts and cardiomyocytes and its influence on the global cardiac function. Fourth, we demonstrated that TEM1 activates Erk and Akt signaling pathways in cardiac fibroblasts. We and others have found that TEM1 may function as a mediator of the PDGF receptor, modulating PDGF-induced signaling pathways and cellular functions in fibroblasts and cardiomyocytes15,17. Other potential molecular mechanisms involving TEM1 effect on cardiac fibroblasts might exist and need further study. Finally, previous study demonstrated that there is an intracellular pool of TEM1 in Golgi apparatus. It is possible that TEM1 might be trafficked to and from the cell surface and can be stained in the cytosol28.
Our findings suggest that there is increased TEM1 expression in atrial cardiac fibroblasts in AF patients and its expression level is associated with atrial fibrosis. TEM1 is not a bystander and its presence is linked to increased cardiac fibroblast proliferation, survival and migration contributing to the process of atrial fibrosis.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Dzeshka, M. S., Lip, G. Y., Snezhitskiy, V. & Shantsila, E. Cardiac fibrosis in patients with atrial fibrillation: Mechanisms and clinical implications. J. Am. Coll. Cardiol. 66, 943–959 (2015).
Nattel, S. Molecular and cellular mechanisms of atrial fibrosis in atrial fibrillation. JACC Clin. Electrophysiol. 3, 425–435 (2017).
Hirsh, B. J., Copeland-Halperin, R. S. & Halperin, J. L. Fibrotic atrial cardiomyopathy, atrial fibrillation, and thromboembolism: Mechanistic links and clinical inferences. J. Am. Coll. Cardiol. 65, 2239–2251 (2015).
Fonseca, A. C. et al. Patients with undetermined stroke have increased atrial fibrosis: A cardiac magnetic resonance imaging study. Stroke 49, 734–737 (2018).
Travers, J. G., Kamal, F. A., Robbins, J., Yutzey, K. E. & Blaxall, B. C. Cardiac fibrosis: The fibroblast awakens. Circ. Res. 118, 1021–1040 (2016).
Khan, K. A., McMurray, J. L., Mohammed, F. & Bicknell, R. C-type lectin domain group 14 proteins in vascular biology, cancer and inflammation. FEBS J. 286, 3299–3332 (2019).
Christian, S. et al. Molecular cloning and characterization of endosialin, a C-type lectin-like cell surface receptor of tumor endothelium. J. Biol. Chem. 276, 7408–7414 (2001).
Huang, H. P. et al. Gene targeting and expression analysis of mouse Tem1/endosialin using a lacZ reporter. Gene Expr. Patterns 11, 316–326 (2011).
Valdez, Y., Maia, M. & Conway, E. M. Cd248: Reviewing its role in health and disease. Curr. Drug Targ. 13, 432–439 (2012).
Simonavicius, N. et al. Endosialin (CD248) is a marker of tumor-associated pericytes in high-grade glioma. Mod. Pathol. 21, 308–315 (2008).
Hong, C. L. et al. CD248 regulates wnt signaling in pericytes to promote angiogenesis and tumor growth in lung cancer. Cancer Res. 82, 3734–3750 (2022).
Smith, S. W. et al. Genetic deletion of the stromal cell marker CD248 (endosialin) protects against the development of renal fibrosis. Nephron 131, 265–277 (2015).
Wilhelm, A. et al. CD248/endosialin critically regulates hepatic stellate cell proliferation during chronic liver injury via a PDGF-regulated mechanism. Gut 65, 1175–1185 (2016).
Bartis, D. et al. Role of CD248 as a potential severity marker in idiopathic pulmonary fibrosis. BMC Pulm. Med. 16, 51 (2016).
Hong, Y. K. et al. Tumor endothelial marker 1 (TEM1/Endosialin/CD248) enhances wound healing by interacting with platelet-derived growth factor receptors. J. Investig. Dermatol. 139, 2204-2214.e7 (2019).
Feng, W. H., Chen, P. S., Chung, H. C., Lin, Y. H. & Li, Y. H. Soluble tumor endothelial marker 1 in heart failure with reduced ejection fraction: A pilot study. Front. Cardiovasc. Med. 9, 1015471 (2022).
Chen, P. S. et al. Tumor endothelial marker 1 is upregulated in heart after cardiac injury and participates in cardiac remodeling. Sci. Rep. 12, 10532 (2022).
Litviňuková, M. et al. Cells of the adult human heart. Nature 588, 466–472 (2020).
Burstein, B., Libby, E., Calderone, A. & Nattel, S. Differential behaviors of atrial versus ventricular fibroblasts: A potential role for platelet-derived growth factor in atrial-ventricular remodeling differences. Circulation 117, 1630–1641 (2008).
Whitlock, R. P. et al. Left atrial appendage occlusion during cardiac surgery to prevent stroke. N. Engl. J. Med. 384, 2081–2091 (2021).
Sahadevan, P. & Allen, B. G. Isolation and culture of adult murine cardiac atrial and ventricular fibroblasts and myofibroblasts. Methods 203, 187–195 (2022).
Liu, S. et al. Antibody-drug conjugates targeting CD248 inhibits liver fibrosis through specific killing on myofibroblasts. Mol. Med. 28, 37 (2022).
Matsushima, S. et al. CD248 and integrin alpha-8 are candidate markers for differentiating lung fibroblast subtypes. BMC Pulm. Med. 20, 21 (2020).
Smith, S. W. et al. CD248+ stromal cells are associated with progressive chronic kidney disease. Kidney Int. 80, 199–207 (2011).
Xu, C. et al. Antibody-drug conjugates targeting CD248+ myofibroblasts effectively alleviate renal fibrosis in mice. FASEB J. 36, e22102 (2022).
Tomkowicz, B. et al. Endosialin/TEM-1/CD248 regulates pericyte proliferation through PDGF receptor signaling. Cancer Biol. Ther. 9, 908–915 (2010).
Hasanov, Z. et al. Endosialin promotes atherosclerosis through phenotypic remodeling of vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 37, 495–505 (2017).
MacFadyen, J. R. et al. Endosialin (TEM1, CD248) is a marker of stromal fibroblasts and is not selectively expressed on tumour endothelium. FEBS Lett. 579, 2569–2575 (2005).
Acknowledgements
This study was supported by Grants from the Ministry of Science and Technology, Taipei, Taiwan (109-2314-B-006-070-MY3 and 112–2314-B-006-096-MY3) to Dr. Yi-Heng Li.
Author information
Authors and Affiliations
Contributions
Y.H.L., Y.N.H., H.C.C. were responsible for performing experiments, aiding in collecting and analysis of data. Y.H.L., T.C.H., M.S.H., were responsible for acquisition, analysis and interpretation of data. Y.H.L. Y.N.H., T.C.H., M.S.H., and H.C.C. contributed to study design and critical revision of the manuscript; Y.H.L. was responsible for drafting of the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, YH., Hu, YN., Huang, TC. et al. Expression of tumor endothelial marker 1 in cardiac fibroblast contributes to atrial fibrosis in atrial fibrillation. Sci Rep 15, 8685 (2025). https://doi.org/10.1038/s41598-025-92719-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92719-w