Abstract
Veno-venous extracorporeal membrane oxygenation (VV-ECMO) plays an important role in the perioperative care of critically ill lung transplant patients. However, the factors predicting prognosis are unclear. This study assessed the association between static respiratory compliance (Crs) and outcomes of lung transplant patients receiving VV-ECMO in terms of 90-day mortality. Data were retrospectively collected for patients that underwent lung transplantation with VV-ECMO support during 2022–2023. Patients were divided into two groups according to the early postoperative Crs: lower Crs (Crs < 25 ml/cmH2O) and higher Crs (Crs ≥ 25 ml/cmH2O). Differences in patient characteristics and prognosis were then compared between the two groups. Receiver operating characteristic (ROC) curve analysis was used to evaluate the value of Crs for predicting 90-day mortality and univariate Cox proportional hazard model analysis was performed to estimate risk of Crs. Data were available for a total of 85 patients, including 50 (58.8%) patients in the higher Crs group and 35 (41.2%) patients in the lower Crs group. A lower Crs was significantly associated with a longer postoperative ECMO duration (hours, 42 vs. 24; P = 0.022), longer postoperative ventilator time (days, 3.7 vs. 2.0; P = 0.003), higher application of continuous renal replacement therapy (CRRT) (20.0% vs. 6.0%; P = 0.049), higher incidence of pneumonia (42.9% vs. 20.0%; P = 0.023), and higher 90-day mortality (22.9% vs. 6.0%; P = 0.023). The area under the curve of Crs for predicting 90-day mortality was 0.661 (P = 0.034). A higher Crs was a protective factor (hazard ratio = 0.925 [0.870–0.984)] P = 0.014). For lung transplant patients receiving VV-ECMO support, Crs < 25 ml/cmH2O is associated with more complications and higher 90-day mortality. As Crs is easily obtained at the bedside, it may be useful for predicting prognosis and guiding patient management.
Similar content being viewed by others
Introduction
Lung transplantation is an effective procedure for benign end-stage lung disease. However, patients with poor underlying condition are prone to insufficient cardiopulmonary function to maintain the necessary oxygenation and circulatory stability during surgery. Thus, veno-venous extracorporeal membrane oxygenation (VV-ECMO) is a reliable and effective form of support that can expand the patient population suitable for lung transplantation and support the transition to the perioperative period1. According to the International Society of Heart and Lung Transplantation, ECMO is performed in 29% of all lung transplants; however, ECMO does not reduce the risk of death2. Thus, predictors are needed to indicate prognosis at an early stage to guide clinical practice. Static respiratory compliance (Crs) is a measure that can be easily determined at the bedside. Prior studies have reported a significant correlation between Crs and prognosis in patients with acute respiratory distress syndrome (ARDS) undergoing VV-ECMO3. Primary graft dysfunction (PGD) occurs 72 h after lung transplantation and has many clinical features in common with ARDS4. Notably, both PGD and ARDS can affect lung compliance. In this study, we aimed to investigate the relationship between Crs and the prognosis of lung transplant patients with VV-ECMO.
Methods
Study design
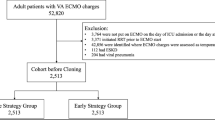
This retrospective study investigated patients who underwent lung transplantation with VV-ECMO between 2022 and 2023 at the Affiliated Wuxi People’s Hospital of Nanjing Medical University. The inclusion criteria were (1) age > 18 years and (2) underwent lung transplantation with VV-ECMO. The exclusion criteria were (1) lung retransplantation; (2) incomplete Crs records. Figure 1 shows the flow diagram of patients in this study.
The flow diagram.
This retrospective study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by the Ethics Commission of the Affiliated Wuxi People’s Hospital of Nanjing Medical University (No. KY24059). Due to the retrospective nature of this study, the need for informed consent was waived by the Committee.
ECMO strategy
The pre-ECMO evaluation is performed by both surgeons and anesthesiologists according to the Extracorporeal Life Support Organization (ELSO) guidelines5. In cases requiring one-lung ventilation, if the hemodynamics were stable and the percutaneous oxygen saturation (SpO2) was maintained > 90%, ECMO treatment was not considered; if the hemodynamics were stable and SpO2 was continuously < 90%, VV-ECMO support was used. VV-ECMO support was established intraoperatively in all cases that met the indications for ECMO.
Peripheral cannulation was the preferred method of support with VV-ECMO. The most common cannulation sites were femoral-internal jugular, with the tip of a femoral drainage cannula at the inferior vena cava-right atrium junction and an internal jugular return cannula with the top either at the superior vena cava-right atrium junction or the right atrium. ECMO management and weaning were performed following the ELSO guidelines6.
Measurement of Crs
Within 2 h of admission to the ICU, patients were in a supine position without spontaneous respiration; otherwise, safe doses of sedative, analgesic, and muscle relaxants were used to prevent spontaneous breathing. Patients received volume-controlled ventilation delivered using a square waveform flow. The initial parameter settings were: tidal volume (VT) at 6 mL/kg of predicted body weight (PBW), PBW was calculated as 50 + 0.91 × (height [cm] – 152.4) for men and 45.5 + 0.91× (height [cm] – 152.4) for women, positive end-expiratory pressure (PEEP) at 5 cmH2O, respiratory rate (RR) at 12/min. Next, we recorded the plateau pressure (Pplat); Crs was calculated as VT/(Pplat-PEEP). The ventilator parameters during the non-measurement period were set by clinicians according to the lung-protective ventilation strategy and each patient’s condition7.
PGD definition
PGD was diagnosed according to the latest recommendation of the ISHLT working group4. Patients were graded on the basis of the ratio of the partial pressure of oxygen (PaO2) to fraction of inspired oxygen (FiO2) (P/F ratio) and chest radiographs. The PGD grade at index ICU admission (0 h), 24 h, 48 h, and 72 h was assessed. Patients with a chest x-ray indicating pulmonary infiltrates and P/F ratio < 200 were defined as PGD3, while patients on ECMO were defined as PGD3 with any P/F ratio.
Data collection
The following data were collected from the medical records of lung transplant patients: age, body mass index (BMI), gender, primary disease, chronic disease, preoperative cardiopulmonary function and laboratory parameters, Acute Physiology and Chronic Health Evaluation II (APACHEII) score, Sequential Organ Failure Assessment (SOFA) score, cold ischemia time, surgical condition, ventilator parameters and ECMO settings, lac, P/F, and PaCO2 within 2 h after surgery. The primary outcome was 90-day survival after lung transplantation. The secondary outcomes were postoperative ECMO time, postoperative ventilator time, ICU stay, hospital stay, PGD3, continuous renal replacement therapy (CRRT), and pneumonia.
Statistical analysis
Normally distributed continuous variables are presented as the mean ± standard deviations and Student’s t test was used for group comparisons. Non-normally distributed continuous variables are presented as the median (interquartile range) and the Mann-Whitney U test was used for group comparisons. Categorical variables are expressed as numbers (percentages) and analyzed using the Chi-square test or Fisher’s exact test. Receiver operating characteristic (ROC) curve analysis was used to calculate the area under the curve (AUC) values to assess the predictive ability of Crs for 90-day mortality. Cutoff points were calculated by obtaining the best Youden index (sensitivity + specificity − 1). The Kaplan-Meier method was used to plot the cumulative curves of ECMO and mechanical ventilation times between the two groups. For the survival analysis, the effect of a lower Crs was assessed using the log-rank test. Univariate Cox regression was used to examine the correlation between Crs and 90-day mortality. Statistical analysis was performed using SPSS 25.0, GraphPad Prism 6.0, and R version 4.3.0 with the KMsurv, survival, and survminer packages. Results with a two-tailed P value < 0.05 were considered to be statistically significant.
Results
Clinical characteristics
Data were available for a total of 85 lung transplant patients who received VV-ECMO. In the ROC analysis, the AUC of Crs for predicting 90-day mortality was 0.661 (P = 0.034). The best Youden index was obtained when Crs = 25 ml/cmH2O. Based on Crs values, patients were assigned to the higher Crs group (n = 50, 58.8%) or lower Crs group (n = 35, 41.2%). As shown in Table 1, there were no significant group differences in the baseline data (P > 0.05)。.
Postoperative outcomes
Compared to the higher Crs group, the lower Crs group had a higher driving pressure (14 ± 1.2 vs. 12 ± 0.8 cmH2O, P<0.001) and Pplat (20 ± 1.3 vs. 18 ± 1.1 cmH2O, P = 0.015). There were no significant group differences in terms of ventilator parameter settings or VV-ECMO flow rates at the initial postoperative procedure (Table 2). Figure 2A shows the change in P/F over time. The P/F at 24 h was higher than that at 0 h, while the 48 h and 72 h P/F values tended to be stable. Compared to the lower Crs group, the higher Crs group had a higher 0 h P/F (274 ± 39.3 vs. 197 ± 33.1, P = 0.006) and 24 h P/F (348 ± 28.2 vs. 259 ± 23.9, P<0.001). Moreover, the higher Crs group had a lower 0 h PaCO2 (32.5 ± 1.9 vs. 38.2 ± 3.6 mmHg, P = 0.003) and 24 h PaCO2 (35.9 ± 2.1 vs. 39.7 ± 2.6 mmHg, P = 0.020) compared to the lower Crs group, as shown in Fig. 2B.
A P/F recorded from 0 h to 72 h in two groups; B PaCO2 recorded from 0 h to 72 h in two groups.
For all patients, the 24 h, 48 h, and 72 h PGD proportions were 51.8%, 35.3%, and 31.8%, respectively. The incidence of grade 3 PGD at 48 h was significantly lower than that at 24 h (P = 0.030); however, there was no significant difference between 72 h and 48 h (Fig. 3). The lower Crs group tended to include more patients with 24 h PGD3 (P = 0.087). Compared with the higher Crs group, the lower Crs group had increased pneumonia (42.9% vs. 20.0%, P = 0.023), CRRT (20.0% vs. 6.0%, P = 0.049), postoperative ECMO time (42 h vs. 24 h, P = 0.022), and postoperative MV time (3.7 days vs. 2.0 days, P = 0.003) and decreased 90-day survival (77.1% vs. 94.0%, P = 0.023).
Proportion of postoperative PGD3 occurrence at 24 h, 48 h and 72 h.
The cumulative curve of ECMO weaning and MV showed a significant difference between the two groups (Fig. 4). The difference in K-M survival curves between the lower and higher Crs groups was statistically significant (P = 0.023, Fig. 5).
A Postoperative ECMO time curves in two groups ; B Ventilator time curves in two groups.
Kaplan–Meier survival curves in patients.
The effect of Crs remained significant in the univariate Cox regression model [HR 0.925 (0.870–0.984), P = 0.014], indicating that higher Crs was a protective factor for 90-day mortality.
Discussion
VV-ECMO during lung transplantation is considered a safe and effective form of life support that is increasingly used8. Critically ill patients often require ECMO support due to preoperative respiratory failure, pulmonary hypertension, or cardiac dysfunction during lung transplantation9. The V-V mode offers a main pulmonary support effect10. ECLS has reported survival rates of 56–100% for ECMO support as a bridge to lung transplantation11,12,13. The use of ECMO for intraoperative support management varies considerably across centers across the world. Its use in the perioperative setting of lung transplantation is associated with favorable outcomes14. Christian et al. reported that ECMO as a bridge to lung transplantation is associated with higher perioperative mortality, but acceptable mid-term survival, in carefully selected patients15. In another larger study, Ius et al. found no differences in long-term complications or outcomes in ECMO recipients16. Sef et al. reported a similar 30-day mortality between bridge to transplantation and non‐bridge to transplantation patients (4.6% vs. 6.6%, p = 0.083) despite a higher incidence of early postoperative complications (e.g., need for ECMO, delayed chest closure, and acute kidney injury)17. In their study, the 90-day survival rate of patients treated with VV-ECMO lung transplantation was 87%. Takahashi et al. retrospectively analyzed clinical data of 204 patients with PGD3 after lung transplantation from 2010 to 2020, finding no significant survival difference between patients with and without perioperative ECMO. Thus, the authors concluded that perioperative ECMO did not increase the risk of mortality18. Zhao et al. reported that the use of intraoperative ECMO support reduced ischemia-reperfusion injury due to the avoidance of hyperperfusion in double sequential or lobar lung transplantation, improved surgical exposure and reduced operative time19. Notably, the donor lung is prone to the development of pulmonary edema by ischemia reperfusion injury. Prolonging the use of VV-ECMO thus enables “lung rest” and restoration of lung function. ECMO respiratory support reduces lung injury by ultraprotective lung ventilation strategies and the potential harm of high FiO2 to lungs in PGD3 patients. During the postoperative period, ECMO may also affect right ventricular afterload by reducing pulmonary resistance. ECMO may reduce the pulmonary edema on lung injury to lower the PGD grade. A previous controlled study at our center found that delayed VV-ECMO weaning was associated with lower complications and shorter hospital stay20. However, delayed ECMO weaning will impact the incidence of PGD3 at 0 h. Thus, further research is needed to analyze ECMO strategies in lung transplantation patients.
The main findings of the present study are that, using Crs = 25 ml/cmH2O as the cut-off point, the lower Crs group had a longer ECMO time, longer MV time, higher incidence of complications, and decreased 90-day survival rate, suggesting that Crs might be a prognostic factor. Notably, there are relatively few known prognostic factors for lung transplantation patients with VV-ECMO support21. As Crs is easily measured at the bedside, increased clinical attention and management of patients with lower Crs may improve outcomes. In a study of COVID-19 patients with VV-ECMO divided into three groups based on Crs (Crs ≤ 11 cmH2O, Crs 11–20 cmH2O, and Crs >20 cmH2O), Crs was associated with 180-day survival3. PGD is an acute lung injury due to ischemia-reperfusion during lung transplantation. The incidence of patients with ECMO was further increased, with 57% of patients developing PGD3 within 72 h after surgery22. PGD shares many clinical features and radiographic findings with ARDS, including decreased lung function, increased elastic resistance, and ventilation/flow imbalance23. Decreased lung compliance results in low PaO2 and decreased excretion of CO2, which delay the weaning of ECMO and MV and increase the incidence of ventilator-associated pneumonia and other complications24. Patients with lower Crs are more susceptible to ventilator-associated lung injury, even when low-tidal volume lung protective ventilation is used25. Ischemia reperfusion injury is not only a risk factor for PGD, but can also lead to inflammatory damage in other organs, and the kidney, one of the affected organs, is prone to acute kidney injury. In addition, the use of nephrotoxic drugs also increases the incidence of acute kidney injury in lung transplantation. These high-risk factors will require increased use of CRRT26. These risk factors can interact, further affecting prognosis. In the present study, there was no significant difference between groups in the incidence of PGD3 at 24 h, 48 h, and 72 h. There are several possible explanations for this finding. Different PGD phenotypes may have had different effects on lung compliance. Alternatively, it may relate to the sample size. The incidence of PGD3 in the two groups gradually approached similar levels over time, which might relate to PGD treatment and progression. The lower Crs group showed a delayed rise in P/F and a high initial carbon dioxide content. Although there were differences between P/F and carbon dioxide, the overall trend was similar. Future studies should explore whether different PGD phenotypes can be distinguished according to Crs in order to provide individualized clinical management, and the relationship between changes in Crs and PGD warrants further research.
Our study is subject to several limitations. First, it is a retrospective single-center study. Although the lung transplantation capacity of our center is relatively high, multi-center validation is needed. Second, the dynamic change in lung compliance was not monitored. The relationship between dynamic change and prognosis should be further studied. Third, as data on donor lungs were not collected, we cannot exclude the influence of donor lungs on prognostic confounding factors. Fourth, as ventilatory management of lung transplantation patients was relatively individualized, this may have influenced outcomes.
Conclusions
In lung transplant patients with VV-ECMO support, Crs < 25 ml/cmH2O is associated with higher complications and 90-day mortality. Furthermore, the lower Crs group tended to have an increased incidence of PGD3. The use of Crs, which is easily accessible at the bedside, may help predict prognosis and guide patient management. The relationship between Crs and the prognosis of lung transplantation patients with VV-ECMO needs to be confirmed through multi-center prospective studies in the future.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Laskey, D. et al. Intraoperative extracorporeal support during lung transplantation: not just for the High-Risk patient. J. Clin. Med. ; 13(1). (2023).
Kirklin, J. K. et al. First annual IMACS report: A global international society for heart and lung transplantation registry for mechanical circulatory support. J. Heart Lung Transpl. 35 (4), 407–412 (2016).
Valentin, S. et al. Prognostic value of respiratory compliance course on mortality in COVID-19 patients with vv-ECMO. Ann. Intensive Care. 13 (1), 54 (2023).
Snell, G. I. et al. Report of the ISHLT working group on primary lung graft dysfunction, part I: definition and grading-A 2016 consensus group statement of the international society for heart and lung transplantation. J. Heart Lung Transpl. 36 (10), 1097–1103 (2017).
Tonna, J. E. et al. Management of adult patients supported with venovenous extracorporeal membrane oxygenation (VV ECMO): guideline from the extracorporeal life support organization (ELSO). ASAIO J. 67 (6), 601–610 (2021).
Gattinoni, L. et al. Extracorporeal gas exchange: when to start and how to end? Crit. Care. 23 (Suppl 1), 203 (2019).
Barnes, L. et al. MECHANICAL VENTILATION FOR THE LUNG TRANSPLANT RECIPIENT. Curr. Pulmonol. Rep. 4 (2), 88–96 (2015).
Hartwig, M. et al. The American association for thoracic surgery (AATS) 2022 expert consensus document: the use of mechanical circulatory support in lung transplantation. J. Thorac. Cardiovasc. Surg. 165 (1), 301–326 (2023).
Zhang, Y. et al. A risk score for predicting extracorporeal membrane oxygenation support before lung transplantation. J. Thorac. Dis. 16 (1), 231–240 (2024).
Patterson, C. M. et al. Extracorporeal life support as a Bridge to lung transplantation: where are we now? J. Heart Lung Transpl. 41 (11), 1547–1555 (2022).
Biscotti, M. et al. Awake extracorporeal membrane oxygenation as Bridge to lung transplantation: A 9-Year experience. Ann. Thorac. Surg. 104 (2), 412–419 (2017).
Rando, H. J. et al. Extracorporeal membrane oxygenation as a Bridge to lung transplantation: practice patterns and patient outcomes. J. Heart Lung Transpl. 43 (1), 77–84 (2024).
Todd, E. M. et al. Extracorporeal membrane oxygenation as a Bridge to lung transplantation: A single-center experience in the present era. J. Thorac. Cardiovasc. Surg. 154 (5), 1798–1809 (2017).
Martin, A. K. et al. Extracorporeal membrane oxygenation in lung transplantation: analysis of techniques and outcomes. J. Cardiothorac. Vasc Anesth. 35 (2), 644–661 (2021).
Bermudez, C. A. et al. Extracorporeal membrane oxygenation as a Bridge to lung transplant: midterm outcomes. Ann. Thorac. Surg. 92 (4), 1226–1231 (2011).
Ius, F. et al. Long-term outcomes after intraoperative extracorporeal membrane oxygenation during lung transplantation. J. Heart Lung Transpl. 39 (9), 915–925 (2020).
Sef, D. et al. Midterm outcomes of venovenous extracorporeal membrane oxygenation as a Bridge to lung transplantation: comparison with nonbridged recipients. J. Card Surg. 37 (4), 747–759 (2022).
Takahashi, T. et al. Outcomes of extracorporeal membrane oxygenation for primary graft dysfunction after lung transplantation. Ann. Thorac. Surg. 115 (5), 1273–1280 (2023).
Zhao, Y. et al. Extracorporeal membrane oxygenation support for lung transplantation: initial experience in a single center in China and a literature review. Front. Med. (Lausanne). 9, 950233 (2022).
Li, L. J. et al. Impact of delayed veno-venous extracorporeal membrane oxygenation weaning on postoperative rehabilitation of lung transplantation: a single-center comparative study. J. Artif. Organs. 26 (4), 303–308 (2023).
Faccioli, E. et al. Validated prognostic scores to predict outcomes in ECLS-Bridged patients to lung transplantation. Transpl. Int. 36, 11609 (2023).
Hoetzenecker, K. et al. Extracorporeal life support as a Bridge to lung transplantation-experience of a high-volume transplant center. J. Thorac. Cardiovasc. Surg. 155 (3), 1316–28e1 (2018).
Li, D. et al. Atelectasis in primary graft dysfunction survivors after lung transplantation. Clin. Transpl. 35 (7), e14315 (2021).
Clauseni, E. & Cantu, E. Primary graft dysfunction: what we know. J. Thorac. Dis. 13 (11), 6618–6627 (2021).
Assouline, B., Combes, A. & Schmidt, M. Setting and monitoring of mechanical ventilation during venovenous ECMO. Crit. Care. 27 (1), 95 (2023).
Kim, N. E. et al. Risk factors and mortality of acute kidney injury within 1 month after lung transplantation. Sci. Rep. 11 (1), 17399 (2021).
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Hongyang Xu, Jingyu Chen, and Chenhao Xuan participated in the design of the study. Chenhao Xuan wrote the application for the ethical approval. Chenhao Xuan collected the data. Chenhao Xuan and Jingxiao Gu analyzed the data. Chenhao Xuan drafted the manuscript. Chenhao Xuan prepared Figs. 1, 2, 3, 4 and 5; Table 1, and 2. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This retrospective study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by Ethics Commission of the Affiliated Wuxi People’s Hospital of Nanjing Medical University (No. KY24059), and the need for informed consent was waived by the Committee due to the retrospective nature of this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xuan, C., Gu, J., Chen, J. et al. Respiratory compliance related to prognostic of lung transplant patients with veno‑venous extracorporeal membrane oxygenation support. Sci Rep 15, 8421 (2025). https://doi.org/10.1038/s41598-025-93396-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93396-5