Abstract
This study aimed to investigate the relationship between lipoprotein(a) [Lp(a)] levels and non-alcoholic fatty liver disease (NAFLD), and to analyze its linear association and subgroup differences. This cross-sectional analysis was based on data from 2308 participants in the National Health and Nutrition Examination Survey (NHANES) III. Multivariate logistic regression models were used to assess the association between Lp(a) and NAFLD, adjusting for demographic factors, lifestyle behaviors, comorbidities, and biomarkers. Subgroup analyses were conducted based on age, sex, body mass index (BMI), diabetes, and hypertension. Restricted cubic spline (RCS) regression model was used to explore the nonlinear relationship between Lp(a) and NAFLD. Higher Lp(a) levels were significantly associated with a lower risk of NAFLD. In the fully adjusted model, compared to the lowest quartile group (Q1), the third and fourth quartiles (Q3 and Q4) had significantly reduced risks of NAFLD [Q3: OR = 0.701, 95% CI 0.511, 0.961; P = 0.027; Q4: OR = 0.605, 95% CI 0.438, 0.835; P = 0.002]. Subgroup analysis showed that the association between higher Lp(a) levels and reduced NAFLD risk was significant in individuals aged 50 years and older, those with BMI ≥ 30 kg/m2, non-diabetics, and those with hypertension. RCS analysis further confirmed a linear negative association between Log10Lp(a) and NAFLD risk (P = 0.029, P nonlinearity = 0.888). There is a significant linear negative association between Lp(a) levels and the risk of NAFLD, suggesting that Lp(a) may serve as a potential biomarker for assessing NAFLD risk.
Similar content being viewed by others
Introduction
Non-alcoholic fatty liver disease (NAFLD) is a common chronic liver disease characterized by the accumulation of fat in the liver, which is not caused by excessive alcohol consumption1. NAFLD has become a significant global public health issue, especially in the context of the rising incidence of metabolic syndrome-related diseases such as obesity and type 2 diabetes2. According to global data, the prevalence of NAFLD has approached 25%, and this number is expected to further increase with changes in lifestyle and population aging3. NAFLD, as a liver disease, is also considered an independent risk factor for cardiovascular diseases (CVD), particularly in individuals without other significant risk factors, making the study of the pathophysiological mechanisms of NAFLD and its related risk factors particularly important4.
In the pathogenesis of NAFLD, lipid metabolism disorders and inflammatory responses are considered key factors5. Among them, lipoprotein(a) [Lipoprotein(a), Lp(a)] is a structurally complex lipoprotein that shares a similar structure with low-density lipoprotein (LDL) but contains the unique apolipoprotein(a)6. The distinctive role of Lp(a) in lipid metabolism has made it a focal point in many studies, especially regarding its association with CVD and mortality7,8,9. Compared with LDL, Lp(a) has several advantages as a research focus. Firstly, Lp(a) levels are primarily determined by genetic factors and are less influenced by external factors such as diet and metabolic state, making it a more stable and reliable biomarker for chronic diseases6,10,11. Secondly, the presence of apolipoprotein(a) confers unique biological functions, including a role in inflammation and fibrosis, which are critical processes in the progression of NAFLD to advanced stages such as liver fibrosis and cirrhosis11. Additionally, elevated Lp(a) levels are closely associated with lipid metabolism disorders, hepatic fat deposition, and chronic inflammation, all of which are central to the pathogenesis of NAFLD11,12,13. However, the relationship between Lp(a) and NAFLD remains unclear. Lp(a) levels are largely influenced by genetic factors and show high inter-individual variability, making Lp(a) a potential biomarker for metabolic diseases10. In this context, exploring the correlation between Lp(a) and NAFLD may help elucidate the pathophysiological mechanisms of NAFLD and potentially provide new biomarkers for early diagnosis and prevention of NAFLD. Currently, studies on the relationship between Lp(a) and NAFLD are relatively limited, and existing research results are somewhat controversial. The genetic background of different populations, environmental factors, and the heterogeneity of NAFLD may be among the reasons for the inconsistent results. Therefore, larger-scale epidemiological studies are needed to further clarify the relationship between Lp(a) and NAFLD.
The National Health and Nutrition Examination Survey (NHANES) is a nationally representative health survey that collects detailed, high-quality, and diverse health data, playing a crucial role, particularly in epidemiological studies of chronic diseases. Therefore, this study will analyze the correlation between Lp(a) levels and NAFLD based on NHANES data, aiming to clarify the potential relationship between the two and explore the possibility of Lp(a) as a predictive indicator of NAFLD risk. This study will not only contribute to a deeper understanding of the pathological mechanisms of NAFLD but also potentially provide new ideas for early clinical screening and prevention.
Methods
Study population
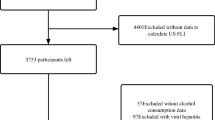
This study was a cross-sectional analysis based on the NHANES database. NHANES, conducted by the U.S. Centers for Disease Control and Prevention (CDC), is a nationwide survey designed to assess the health and nutritional status of the U.S. population. This study utilized data from NHANES III, primarily analyzing the correlation between Lp(a) levels and NAFLD. The NHANES III dataset includes 33,994 participants, and after excluding individuals under 18 years of age, those without archived gallbladder ultrasound images, individuals positive for hepatitis B surface antigen, hepatitis C antibodies, heavy drinkers (women consuming > 20 g/day and men > 30 g/day)14, and participants without Lp(a) data, a total of 2,308 participants were included in the study (Fig. 1). As the NHANES database provides publicly available anonymized data, this study did not require approval from an ethics committee. And the study scheme was in line with the Declaration of Helsinki.
Flowchart of study population enrollment. NHANES, National Health and Nutrition Examination Survey; Lp(a), lipoprotein(a).
Covariates collection and definitions
The covariate data in this study were derived from NHANES III and collected and defined based on standardized questionnaires, physical examinations, and laboratory test results. Age was calculated in years, and gender and race were self-reported by the participants. Race was categorized into non-Hispanic White, non-Hispanic Black, Mexican American, and other races. Educational attainment was classified into three categories: less than high school, high school graduate, and above. Marital status was divided into married and non-married. The family poverty income ratio (PIR) was based on the ratio of household income to the poverty threshold and categorized into ≤ 1.0, 1.0–3.0, and > 3.0. Ideal physical activity was classified based on whether participants met the World Health Organization’s recommended guidelines for weekly physical activity, defined as at least 150 min of moderate-intensity or 75 min of vigorous-intensity physical activity, or an equivalent combination of both15. Smoking status was divided into daily smokers, occasional smokers, and never smokers, and drinking status was divided into drinkers and non-drinkers.
Comorbidities included hypertension, diabetes, and dyslipidemia, determined through questionnaires or laboratory test results. The diagnostic criteria for hypertension were a systolic blood pressure (SBP) ≥ 140 mmHg, a diastolic blood pressure (DBP) ≥ 90 mmHg, or the current use of antihypertensive medication16. Diabetes was diagnosed based on a self-reported history of diabetes, fasting plasma glucose (FPG) ≥ 7.0 mmol/L, glycated hemoglobin (HbA1c) ≥ 6.5%, or current use of antidiabetic medication17. Dyslipidemia was defined as total cholesterol (TC) ≥ 5.17 mmol/L, low-density lipoprotein cholesterol (LDL-C) ≥ 3.37 mmol/L, high-density lipoprotein cholesterol (HDL-C) < 1.04 mmol/L, or triglycerides (TG) ≥ 1.7 mmol/L, or the current use of lipid-lowering medication18. Treatment data included the use of antihypertensive, antidiabetic, and lipid-lowering medications. Body mass index (BMI) was calculated from height and weight, and blood pressure was measured using standardized methods, including SBP and DBP. Lipid markers included TG, TC, LDL-C, and HDL-C. Lp(a) levels were measured by enzyme-linked immunosorbent assay (ELISA). Liver function markers included alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), and albumin (ALB). Kidney function markers included blood urea nitrogen (BUN), creatinine (CR), and uric acid (UA). Glucose control indicators included FPG and HbA1c. All data were collected according to NHANES’ standard operating procedures to ensure accuracy and consistency. These covariates will be used in subsequent analyses to adjust the models and control for potential confounding factors.
Ultrasound-based hepatic steatosis assessment and NAFLD classification protocol
During the assessment of hepatic steatosis via ultrasound, the initial gallbladder images were captured using Toshiba Sonolayer SSA-90 A equipment and a Toshiba video recorder. These recordings were later digitized through a SONY RDR-VX560 DVD-VHS recorder and saved onto recordable DVDs. The digitized images were then reviewed on a Dell flat panel monitor by trained ultrasound readers who followed standardized procedures. Under the guidance of a radiologist with expertise in hepatic imaging, the readers evaluated key parameters such as liver-to-kidney contrast, parenchymal brightness, deep beam attenuation, vessel wall definition, and gallbladder wall definition. Based on these criteria, the liver was classified into normal, mild, moderate, or severe hepatic steatosis. To ensure accuracy and consistency, rigorous quality control protocols were in place, including routine training, reliability assessments, and adherence to quality assurance standards throughout the study.
Given that controlled attenuation parameter (CAP) readings obtained through transient elastography (FibroScan® TE) have shown strong accuracy in gauging hepatic steatosis, a CAP threshold of ≥ 238 dB/m was used for diagnosis19. After excluding individuals who were heavy drinkers (women consuming > 20 g/day and men > 30 g/day) and those with other known causes of hepatic steatosis (e.g., positive serology for hepatitis B virus [HBV], and hepatitis C virus [HCV]), the remaining cases of hepatic steatosis were classified as NAFLD14.
Statistical analysis
All continuous variables were first tested for normality using the Kolmogorov-Smirnov test. For normally distributed continuous variables, the mean and standard deviation (mean ± SD) were reported, while for non-normally distributed continuous variables, the median and interquartile range [M (Q1, Q3)] were used. Categorical variables were expressed as frequencies and percentages [n (%)]. For the comparison between the NAFLD and non-NAFLD groups, normally distributed continuous variables were compared using the independent samples t-test, non-normally distributed continuous variables were compared using the Mann-Whitney U test, and categorical variables were compared using the chi-square test or Fisher’s exact test. For the four Lp(a) quartile groups, normally distributed continuous variables were compared using one-way analysis of variance (ANOVA), non-normally distributed continuous variables were compared using the Kruskal-Wallis test, and categorical variables were compared using the chi-square test or Fisher’s exact test. Multivariate logistic regression analysis was used to evaluate the association between Lp(a) and NAFLD. Model 1 was unadjusted; Model 2 was adjusted for age and sex; Model 3 was further adjusted for race, education level, marital status, family PIR, ideal physical activity, smoking status, drinking status, diabetes, hypertension, dyslipidemia, and hypoglycemic drug use; and Model 4 was additionally adjusted for BMI, SBP, DBP, TG, TC, HDL-C, ALT, AST, ALB, BUN, UA, FPG, and HbA1c based on Model 3. To check for multicollinearity among covariates included in the multivariate models, the variance inflation factor (VIF) was calculated, and a VIF value greater than 10 was considered indicative of multicollinearity (Table S1). Additionally, subgroup logistic regression analyses were conducted to explore the association between Lp(a) and NAFLD in different subgroups, such as age, sex, BMI, diabetes, and hypertension. Finally, restricted cubic spline (RCS) regression model was used to assess the nonlinear association between Lp(a) and NAFLD. Data analysis was performed using SPSS version 26.0 and R version 4.1.3 software. All statistical tests were two-sided, and a P-value of < 0.05 was considered statistically significant.
Results
Baseline characteristics
As shown in Table 1, participants were divided into Non-NAFLD (1505) and NAFLD (803) groups. The NAFLD group was older, had higher proportions of Mexican-Americans, less education, more married individuals, more frequent smokers, fewer drinkers, and higher rates of hypertension, diabetes, dyslipidemia, and more frequent use of hypotensive and hypoglycemic drugs (P < 0.05). They also had higher BMI, SBP, DBP, TG, TC, ALT, AST, UA, FPG, and HbA1c levels, but lower proportion of PIR > 3.0, and lower HDL-C, Lp(a), and ALB (P < 0.001).
As shown in Table 2, a total of 2,308 participants were divided into four groups based on the quartiles of Lp(a): Q1: ≤ 0.05, Q2: 0.05 < Q2 ≤ 0.19, Q3: 0.19 < Q3 ≤ 0.38, and Q4: > 0.38. Compared to individuals in the lowest quartile (Q1) of Lp(a), those in the highest quartile (Q4) exhibited the following characteristics: a lower proportion of males, a higher proportion of non-Hispanic Blacks, a greater percentage with a high school education or above, a higher proportion of unmarried individuals, a slightly higher proportion of drinkers, an increased prevalence of hypertension, and a decreased prevalence of diabetes. Additionally, the prevalence of NAFLD gradually decreased with increasing quartiles of Lp(a). Biochemical indicators revealed that as Lp(a) levels increased, these individuals exhibited higher DBP, TC, LDL-C, and HDL-C levels, as well as lower levels of TG, ALT, AST, TBIL, and ALB (P < 0.05).
Association between Lp(a) and NAFLD
Multivariate logistic regression analysis (Table 3) showed that higher Lp(a) levels were significantly associated with a lower risk of NAFLD. In the unadjusted model (Model 1), compared to the Q1 group, the risk of NAFLD was significantly reduced in the Q2, Q3, and Q4 groups, with ORs of 0.658 (95% CI 0.519, 0.835; P = 0.001), 0.628 (95% CI 0.495, 0.798; P < 0.001), and 0.557 (95% CI 0.437, 0.711; P < 0.001), respectively, showing a significant decreasing trend (P for trend < 0.001). In the model adjusted for age and sex (Model 2), the risk of NAFLD remained significantly lower in the Q2, Q3, and Q4 groups, with ORs of 0.668 (95% CI 0.524, 0.850; P = 0.001), 0.630 (95% CI 0.494, 0.803; P < 0.001), and 0.566 (95% CI 0.442, 0.724; P < 0.001), with a trend test result of P < 0.001. In the model further adjusted for race, education, marital status, lifestyle, and comorbidities (Model 3), the risk of NAFLD in the Q2, Q3, and Q4 groups remained significantly lower, with ORs of 0.703 (95% CI 0.521, 0.947; P = 0.021), 0.628 (95% CI 0.466, 0.847; P = 0.002), and 0.547 (95% CI 0.403, 0.742; P < 0.001). In the fully adjusted model (Model 4), which accounted for BMI, blood pressure, lipids, and other biochemical markers, the risk of NAFLD remained significantly lower in the Q3 and Q4 groups, with ORs of 0.701 (95% CI 0.511, 0.961; P = 0.027) and 0.605 (95% CI 0.438, 0.835; P = 0.002), respectively. Additionally, each 1-unit increase in Lp(a) was associated with a significant reduction in NAFLD risk across all models, with an OR of 0.626 (95% CI 0.421, 0.932; P = 0.021) in Model 4.
The subgroup analysis in Table 4 showed significant differences in the association between Lp(a) levels and NAFLD in specific populations. Among individuals aged ≥ 50 years, higher Lp(a) levels were significantly associated with a reduced risk of NAFLD. Compared to individuals in the lowest quartile (Q1) of Lp(a), those in the second (Q2), third (Q3), and highest (Q4) quartiles had ORs of 0.607 (95% CI 0.369, 0.997; P < 0.05), 0.447 (95% CI 0.271, 0.737; P < 0.01), and 0.403 (95% CI 0.240, 0.678; P < 0.01), respectively, showing a significant decreasing trend (P for trend = 0.002). Additionally, in individuals with BMI ≥ 30 kg/m2, the risk of NAFLD was significantly reduced in the Q4 group, with an OR of 0.424 (95% CI 0.229, 0.784; P < 0.01), with P for trend = 0.048. In the non-diabetic population, the risk of NAFLD was significantly lower in the Q2 group, with an OR of 0.678 (95% CI 0.478, 0.961; P < 0.05). Among individuals with hypertension, the risk of NAFLD was significantly lower in the Q3 and Q4 groups, with ORs of 0.504 (95% CI 0.275, 0.925; P < 0.05) and 0.512 (95% CI 0.269, 0.974; P < 0.05), respectively. In contrast, the association between Lp(a) and NAFLD did not reach significance among individuals aged < 50 years, those with BMI < 30 kg/m², those with diabetes, or those without hypertension. Similarly, in the gender subgroup analysis, the association between Lp(a) and NAFLD was not statistically significant in either males or females across all quartiles. For males, the ORs (95% CI) for Q2, Q3, and Q4 compared to Q1 were 0.835 (0.515–1.354), 0.710 (0.432–1.168), and 0.700 (0.400-1.226), respectively, with a P for trend of 0.552. For females, the ORs (95% CI) for Q2, Q3, and Q4 compared to Q1 were 0.707 (0.449–1.115), 0.738 (0.461–1.180), and 0.646 (0.396–1.054), respectively, with a P for trend of 0.312. Additionally, the RCS plot indicated a linear negative association between Log10Lp(a) and the risk of NAFLD (P = 0.029, P nonlinearity = 0.888) (Fig. 2).
Restricted cubic spline plot of the association between Lp(a) and NAFLD. Lp(a), lipoprotein(a); NAFLD, non-alcoholic fatty liver disease.
Discussion
This study, based on a cross-sectional analysis of the NHANES III database, explored the association between Lp(a) levels and NAFLD. The results indicated that higher Lp(a) levels were significantly associated with a reduced risk of NAFLD, and this association varied across specific populations. The following discussion will delve into the significance of the findings and the possible mechanisms underlying them.
Firstly, the baseline characteristics show that participants in the NAFLD group were older than those in the non-NAFLD group and had higher levels of BMI, SBP, DBP, TG, TC, UA, FPG, and HbA1c, while levels of HDL-C, Lp(a), and ALB were significantly lower. These findings are consistent with existing literature, as the development of NAFLD is often accompanied by metabolic disorders such as obesity, hypertension, and insulin resistance20,21,22. Notably, the reduced Lp(a) levels in the NAFLD group suggest a potential association with a reduced risk of NAFLD. Multivariate logistic regression analysis further confirmed the inverse relationship between Lp(a) levels and the risk of NAFLD. Interestingly, the subgroup analysis in this study revealed that the association between Lp(a) and NAFLD varies across different populations. Among individuals aged ≥ 50 years, higher Lp(a) levels significantly reduced the risk of NAFLD, with a clear decreasing trend in NAFLD risk as Lp(a) levels increased. This may be related to age-associated metabolic changes, as older individuals often experience a gradual decline in metabolic function23. In the population with a BMI ≥ 30 kg/m², the risk of NAFLD was significantly reduced in the Q4 group, suggesting that higher Lp(a) levels may have a stronger protective effect against NAFLD in obese individuals. This finding may be related to lipid metabolism disorders associated with obesity24. Obesity is often accompanied by insulin resistance and lipid metabolism dysfunction, and Lp(a) may reduce liver fat accumulation by modulating these metabolic pathways25. Furthermore, in individuals without diabetes, higher Lp(a) levels were significantly associated with a lower risk of NAFLD, whereas no significant association was observed in individuals with diabetes. This result may be related to the altered metabolic environment in diabetic patients. Diabetic individuals typically have higher levels of insulin resistance and chronic inflammation, which may weaken the protective effect of Lp(a) against NAFLD26. It is also noteworthy that higher Lp(a) levels may exert a protective effect in individuals with hypertension. Hypertension is often accompanied by vascular dysfunction and inflammation, and the unique structure of Lp(a) may influence NAFLD by modulating these pathological processes27,28. However, the association between Lp(a) and NAFLD did not reach statistical significance in individuals without hypertension, suggesting that the impact of Lp(a) on NAFLD may differ under different blood pressure conditions. RCS analysis further confirmed the linear inverse correlation between Log10Lp(a) and the risk of NAFLD, and this result remained significant in the adjusted model. This suggests that the protective effect of Lp(a) against NAFLD may be exerted through a stable linear mechanism, rather than through nonlinear or complex interactions. Based on this, Lp(a) levels could potentially serve as a biomarker for NAFLD risk assessment.
Similar to our findings, other studies have also confirmed the inverse correlation between Lp(a) and NAFLD29,30,31. For example, Nam et al., in a cross-sectional study involving 2242 non-diabetic Korean adults, found that Lp(a) concentrations decreased with the increasing severity of NAFLD and the prevalence of NAFLD decreased across higher tertiles of Lp(a) concentrations, with Lp(a) concentrations showing a significant inverse association with the presence of NAFLD after adjusting for multiple risk factors29. Additionally, Mehta et al., in a study involving 151 patients with biopsy-proven NAFLD, found that Lp(a) levels were significantly higher in patients with nonalcoholic fatty liver (NAFL) compared to those with nonalcoholic steatohepatitis (NASH), with Lp(a) levels being 50% lower in NASH patients than in NAFL patients, and Lp(a) concentrations were inversely associated with hepatocyte ballooning, lobular inflammation, and fibrosis30. Besides, Meroni et al., in a study involving 600 patients with biopsy-proven NAFLD, found that patients with lower levels of Lp(a) had a higher risk of developing severe hepatic fibrosis and cirrhosis, while Lp(a) levels were consistent with the hepatic expression of the LPA gene and decreased with the progression of NAFLD, and the study also revealed that combining Lp(a) with transaminases could enhance the predictive capability for advanced hepatic fibrosis31. Beyond the association between Lp(a) and NAFLD, a growing body of research has also revealed that Lp(a) is closely related to metabolic diseases and as well as the risk, severity, and mortality of CVD32,33,34. For example, in a cross-sectional study involving 410 patients with new-onset acute myocardial infarction, Lp(a) levels were shown to have a nonlinear correlation with the prevalence of aortic valve calcification, with a positive association observed when Lp(a) levels were below 840 mg/L, while this association disappeared when Lp(a) levels reached or exceeded 840 mg/L32. Furthermore, Ulloque-Badaracco et al., conducted a systematic review and meta-analysis including 50 studies with a total of 150,519 participants to evaluate the association between Lp(a) and metabolic syndrome, finding that lower Lp(a) levels were associated with an increased risk of metabolic syndrome, suggesting that Lp(a) could serve as a potential biomarker for identifying individuals at risk of metabolic syndrome, although further research is needed to elucidate the underlying mechanisms of this association33. These data suggest that Lp(a) may not only serve as a potential protective factor for NAFLD but also play a significant role in modulating the pathological processes related to metabolic syndrome and the occurrence of cardiovascular events. Therefore, further exploration of Lp(a)’s role in metabolic regulation and its underlying mechanisms could not only deepen our understanding of NAFLD pathogenesis but also provide new insights for the prevention and treatment of various metabolic-related diseases.
The results of this study suggest that higher levels of Lp(a) are significantly associated with a reduced risk of NAFLD, indicating that Lp(a) may have a protective role in the pathophysiology of NAFLD. Firstly, due to its structural similarity to LDL, Lp(a) may be involved in lipid transport and clearance, reducing fat accumulation in the liver and thereby lowering the risk of NAFLD34. Additionally, apolipoprotein(a), a component of Lp(a), possesses anti-inflammatory and antioxidant properties, which can suppress the release of inflammatory factors and reduce lipid oxidative stress35. These effects may alleviate chronic liver inflammation and cell damage, thereby slowing the progression of NAFLD. Furthermore, Lp(a) may enhance insulin sensitivity, regulating fat deposition in the liver and mitigating the negative effects of insulin resistance36. These mechanisms suggest that Lp(a) could play a crucial role in the prevention and treatment of NAFLD. However, while these mechanisms provide a plausible explanation, further experimental studies are needed to validate the precise role of Lp(a) in the pathogenesis of NAFLD.
Despite uncovering a significant negative correlation between Lp(a) levels and the risk of NAFLD, several limitations must be considered. Firstly, as a cross-sectional study, it is unable to establish a causal relationship between Lp(a) and NAFLD, limiting our understanding of the temporal dynamics between these variables. Secondly, although this study adjusted for multiple confounding factors, there may still be unmeasured confounders, such as genetic background, dietary habits, and environmental factors, that could influence the results. Additionally, since the study is based on the NHANES database in the United States, the findings may only be applicable to specific populations and may need validation in different racial and geographic groups. The diagnosis of NAFLD primarily relied on ultrasound, rather than more precise tools such as biopsy or MRI, which could lead to misdiagnosis or underdiagnosis, potentially affecting the accuracy of the study. Furthermore, this study uses the term NAFLD rather than metabolic dysfunction-associated steatotic liver disease (MASLD), which has been proposed as the updated terminology emphasizing the metabolic dysfunction underlying liver fat accumulation. While NAFLD was the prevailing term during the study design and remains widely recognized, adopting MASLD may offer a more comprehensive perspective on the metabolic context of the disease. Future studies should consider aligning with updated terminology to ensure consistency and enhance the understanding of metabolic-associated steatotic liver conditions. Finally, variations in the methods used to measure Lp(a) may impact the comparability of the results. Therefore, future research should adopt standardized measurement protocols and further validate these findings across different populations. These limitations provide avenues for future exploration to better understand the role of Lp(a) in NAFLD.
Conclusion
In conclusion, this study systematically evaluated the association between Lp(a) levels and the risk of NAFLD. The results showed that higher Lp(a) levels were significantly associated with a reduced risk of NAFLD, particularly in older adults, individuals with obesity, and those with hypertension. This suggests that Lp(a) could be a potential biomarker associated with the risk of NAFLD. In clinical practice, measuring Lp(a) levels in high-risk populations could facilitate early screening and stratified risk assessment of NAFLD. Moreover, integrating Lp(a) levels into personalized management strategies, including lifestyle interventions and metabolic control, may help reduce the risk of NAFLD development and progression. Future longitudinal studies and mechanistic investigations are needed to further validate this association and help elucidate the underlying pathophysiological mechanisms.
Data availability
The data used in this study were obtained from the publicly available NHANES database. The research data can be accessed through the NHANES website in accordance with their data usage policies.
References
Cotter, T.G. & Rinella, M. Nonalcoholic fatty liver disease 2020: The state of the disease. Gastroenterology 158(7), 1851–1864. https://doi.org/10.1053/j.gastro.2020.01.052 (2020).
Stefan, N., Häring, H. U. & Cusi, K. Non-alcoholic fatty liver disease: Causes, diagnosis, cardiometabolic consequences, and treatment strategies. Lancet Diabetes Endocrinol. 7(4), 313–324. https://doi.org/10.1016/S2213-8587(18)30154-2 (2019).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64(1), 73–84. https://doi.org/10.1002/hep.28431 (2016).
Polyzos, S. A., Kechagias, S. & Tsochatzis, E. A. Review article: Non-alcoholic fatty liver disease and cardiovascular diseases: Associations and treatment considerations. Aliment. Pharmacol. Ther. 54(8), 1013–1025. https://doi.org/10.1111/apt.16575 (2021).
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M. & Sanyal, A. J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 24(7), 908–922. https://doi.org/10.1038/s41591-018-0104-9 (2018).
Bhatia, H. S. et al. Lipoprotein(a), platelet function and cardiovascular disease. Nat. Rev. Cardiol. 21(5), 299–311. https://doi.org/10.1038/s41569-023-00947-2 (2024).
Wang, Z. W., Li, M., Li, J. J. & Liu, N. F. Association of lipoprotein(a) with all-cause and cause-specific mortality: A prospective cohort study. Eur. J. Intern. Med. 106, 63–70. https://doi.org/10.1016/j.ejim.2022.09.010 (2022).
Duarte Lau, F. & Giugliano, R. P. Lipoprotein(a) and its Significance in Cardiovascular Disease: A Review [published correction appears in JAMA Cardiol. ;7(7):776. (2022). https://doi.org/10.1001/jamacardio.2022.2074]. JAMA Cardiol. 7(7), 760–769 https://doi.org/10.1001/jamacardio.2022.0987 (2022).
Wang, Z., Yan, X., Fang, L., Tang, J. & Zhang, J. Association between lipoprotein(a), fibrinogen and their combination with all-cause, cardiovascular disease and cancer-related mortality: Findings from the NHANES. BMC Public. Health. 24(1), 1927. https://doi.org/10.1186/s12889-024-19443-4 (2024). Published 2024 Jul 18.
Tsimikas, S. A test in context: Lipoprotein(a): Diagnosis, prognosis, controversies, and emerging therapies. J. Am. Coll. Cardiol. 69(6), 692–711. https://doi.org/10.1016/j.jacc.2016.11.042 (2017).
Schmidt, K., Noureen, A., Kronenberg, F. & Utermann, G. Structure, function, and genetics of lipoprotein (a). J. Lipid Res. 57(8), 1339–1359. https://doi.org/10.1194/jlr.R067314 (2016).
Fan, C. et al. Nobiletin ameliorates hepatic lipid deposition, oxidative stress, and inflammation by mechanisms that involve the Nrf2/NF-κB Axis in nonalcoholic fatty liver disease. J. Agric. Food Chem. 71(50), 20105–20117. https://doi.org/10.1021/acs.jafc.3c06498 (2023).
Pawlak, M., Lefebvre, P. & Staels, B. Molecular mechanism of PPARα action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 62(3), 720–733. https://doi.org/10.1016/j.jhep.2014.10.039 (2015).
Peng, H. et al. Prediction of MAFLD and NAFLD using different screening indexes: A cross-sectional study in U.S. Adults. Front. Endocrinol. (Lausanne). 14, 1083032. https://doi.org/10.3389/fendo.2023.1083032 (2023).
Bull, F. C. et al. World health organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 54(24), 1451–1462. https://doi.org/10.1136/bjsports-2020-102955 (2020).
Chobanian, A. V. et al. The seventh report of the joint National committee on prevention, detection, evaluation, and treatment of high blood pressure: The JNC 7 report [published correction appears in JAMA. 2003;290(2):197]. JAMA 289(19), 2560–2572. https://doi.org/10.1001/jama.289.19.2560 (2003).
American Diabetes Association Professional Practice Committee. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetes-2022. Diabetes Care 45(Suppl 1), S17–S38 https://doi.org/10.2337/dc22-S002 (2022).
Authors/Task Force Members; ESC Committee for Practice Guidelines (CPG); ESC National Cardiac Societies. ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk [published correction appears in Atherosclerosis. 2020;292:160–162. doi: 10.1016/j.atherosclerosis.2019.11.020] [published correction appears in Atherosclerosis. 2020;294:80–82. doi: 10.1016/j.atherosclerosis.2019.12.004]. Atherosclerosis. 2019;290:140–205. (2019). https://doi.org/10.1016/j.atherosclerosis.2019.08.014
Mikolasevic, I. et al. Transient elastography (FibroScan(®)) with controlled Attenuation parameter in the assessment of liver steatosis and fibrosis in patients with nonalcoholic fatty liver disease - Where do we stand? World J. Gastroenterol. 22(32), 7236–7251. https://doi.org/10.3748/wjg.v22.i32.7236 (2016).
Milić, S., Lulić, D. & Štimac, D. Non-alcoholic fatty liver disease and obesity: Biochemical, metabolic and clinical presentations. World J. Gastroenterol. 20(28), 9330–9337. https://doi.org/10.3748/wjg.v20.i28.9330 (2014).
Yuan, M. et al. Hypertension and NAFLD risk: Insights from the NHANES 2017–2018 and Mendelian randomization analyses. Chin. Med. J. (Engl). 137(4), 457–464. https://doi.org/10.1097/CM9.0000000000002753 (2024).
Fujii, H. (ed Kawada, N.) Japan Study Group Of Nafld Jsg-Nafld The role of insulin resistance and diabetes in nonalcoholic fatty liver disease. Int. J. Mol. Sci. 21 11 3863 https://doi.org/10.3390/ijms21113863 (2020). Published 2020 May 29.
Zhou, Q., Yu, L., Cook, J. R., Qiang, L. & Sun, L. Deciphering the decline of metabolic elasticity in aging and obesity. Cell. Metab. 35(9), 1661–1671e6. https://doi.org/10.1016/j.cmet.2023.08.001 (2023).
Vekic, J., Zeljkovic, A., Stefanovic, A., Jelic-Ivanovic, Z. & Spasojevic-Kalimanovska, V. Obesity and dyslipidemia. Metabolism 92, 71–81. https://doi.org/10.1016/j.metabol.2018.11.005 (2019).
Martyn, J. A., Kaneki, M. & Yasuhara, S. Obesity-induced insulin resistance and hyperglycemia: Etiologic factors and molecular mechanisms. Anesthesiology 109(1), 137–148. https://doi.org/10.1097/ALN.0b013e3181799d45 (2008).
Rohm, T. V., Meier, D. T., Olefsky, J. M. & Donath, M. Y. Inflammation in obesity, diabetes, and related disorders. Immunity 55(1), 31–55. https://doi.org/10.1016/j.immuni.2021.12.013 (2022).
Aboukhater, D. et al. Inflammation and hypertension: Underlying mechanisms and emerging Understandings. J. Cell. Physiol. 238(6), 1148–1159. https://doi.org/10.1002/jcp.31019 (2023).
Guzik, T. J. & Touyz, R. M. Oxidative stress, inflammation, and vascular aging in hypertension. Hypertension 70(4), 660–667. https://doi.org/10.1161/HYPERTENSIONAHA.117.07802 (2017).
Nam, J. S. et al. Association between lipoprotein(a) and nonalcoholic fatty liver disease among Korean adults. Clin. Chim. Acta. 461, 14–18. https://doi.org/10.1016/j.cca.2016.07.003 (2016).
Mehta, A. et al. Discordant association of nonalcoholic fatty liver disease with lipoprotein(a) and markers of atherogenic dyslipidemia. J. Clin. Lipidol. 17(6), 828–833. https://doi.org/10.1016/j.jacl.2023.09.003 (2023).
Meroni, M. et al. Low Lipoprotein(a) levels predict hepatic fibrosis in patients with nonalcoholic fatty liver disease. Hepatol. Commun. 6(3), 535–549. https://doi.org/10.1002/hep4.1830 (2022).
Wang, Z., Li, M. & Liu, N. The nonlinear correlation between lipoprotein (a) and the prevalence of aortic valve calcification in patients with new-onset acute myocardial infarction. Acta Cardiol. 77(10), 950–959. https://doi.org/10.1080/00015385.2022.2129183 (2022).
Ulloque-Badaracco, J. R. et al. Association of apolipoproteins and lipoprotein(a) with metabolic syndrome: A systematic review and meta-analysis. Lipids Health Dis. 22(1), 98. https://doi.org/10.1186/s12944-023-01860-w (2023).
Chemello, K., Chan, D. C., Lambert, G. & Watts, G. F. Recent advances in demystifying the metabolism of lipoprotein(a). Atherosclerosis 349, 82–91. https://doi.org/10.1016/j.atherosclerosis.2022.04.002 (2022).
Kostner, K. M. & Kostner, G. M. Lp(a) and the risk for cardiovascular disease: Focus on the Lp(a) paradox in diabetes mellitus. Int. J. Mol. Sci. 23(7), 3584. https://doi.org/10.3390/ijms23073584 (2022). Published 2022 Mar 25.
Haffner, S. M. et al. Insulin sensitivity and Lp(a) concentrations in normoglycemic men. Diabetes Care. 18(2), 193–199. https://doi.org/10.2337/diacare.18.2.193 (1995).
Acknowledgements
The authors would like to thank all the researchers and technical support teams involved in this study, particularly the NHANES team for their data collection and management. Special thanks are extended to the study participants whose contributions made this research possible.
Funding
None.
Author information
Authors and Affiliations
Contributions
CBL: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Visualization, Writing - original draft, Writing - review & editing. MCL: Data curation, Formal analysis, Investigation, Visualization, Writing - original draft, Writing - review & editing. ZWW: Conceptualization, Funding acquisition, Project administration, Supervision, Validation, Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was conducted using publicly available data from the NHANES, which is collected and managed by the U.S. Centers for Disease Control and Prevention (CDC). NHANES data are de-identified and made publicly accessible, ensuring participant confidentiality. Therefore, this study did not require additional ethical approval or informed consent. The research followed the principles of the Declaration of Helsinki.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, C., Li, M. & Wang, Z. There is a linear negative correlation between lipoprotein(a) and non-alcoholic fatty liver disease. Sci Rep 15, 8538 (2025). https://doi.org/10.1038/s41598-025-93518-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93518-z