Abstract
Printed electronics is gaining increasing research attention and a growing market share due to it’s lower production cost and easy scalability. Here, we demonstrate the use of Electro-Static Inkjet (ESJET) printing technology to deposit silver nanoparticle (Ag nps) inks as grid electrodes for non-vacuum processed Cu(In,Ga)S2 (CIGS) solar cells, bridging the gap between high viscosity screen printed materials and low viscosity inkjet processes. We investigated the effect of composition of Ag inks compositions and the corresponding sintering condition of ESJET printed Ag grids on the photovoltaic performance of CIGS solar cells. High sintering temperatures (220 °C) of the printed Ag electrodes caused a significant performance loss in the CIGS cells. However, sintering at 160 °C produced cells with efficiency comparable to reference cells using thermal evaporated Ag grids. Results from stability test (ISOS-D-1) shows the device efficiency of devices with ESJET printed Ag grids decreased from 11.14 to 10.44% after 3 months in air without encapsulation, corresponding to efficiency loss of approximate 0.6%. Our work demonstrates that ESJET printing is a viable method for deposition of photovoltaic metal grids, enabling finer electrode patterns, minimizing material waste, and significantly reducing the production cost of thin film solar cells.
Similar content being viewed by others
Introduction
With advancements in solar cell research, solar cell efficiency has greatly improved over the last decades. Crystalline silicon (Si) modules are the most common commercial photovoltaic (PV) modules, representing more than 85% of world PV cell market due to their low cost and high conversion efficiency. However, crystalline Si solar cells are bulky and rigid, requiring high installation cost and limiting their applications in flexible devices. In contrast, thin film solar cells offer advantages such as lightweight, flexibility, and the ability to be deposited on various substrates, including glass, plastic, and metal. Common types include cadmium telluride (CdTe), copper indium gallium selenide (CIGS), and amorphous silicon (a-Si) solar cells. Their lower material consumption and potential for large-area production make them attractive for applications in building-integrated photovoltaics (BIPV) and portable solar technologies. Compared to a-Si solar cells, CIGS offers superior light absorption and higher efficiency, with laboratory-scale devices achieving power conversion efficiencies of 23.6% on rigid glass1, 20.8% on polymer2 and 19.4% on stainless steel3. Additionally, unlike CdTe, which contains toxic tellurium, CIGS presents fewer environmental concerns. Vacuum-based processes, such as co-evaporation and sputtering, have been used to fabricate high-efficiency CIGS solar cells but come with high production costs and complex manufacturing requirements. As a cost-effective alternative, non-vacuum deposition techniques are gaining attention. Methods such as hydrazine-based deposition4, electrodeposition5, nanocrystals-based depostion6, and spin coating7,8 have been widely studied for depositing CIGS absorbers. While non-vacuum CIGS solar cells still lag behind vacuum-processed counterparts in efficiency, recent advancements have pushed their performance above 15%9,10, making them increasingly viable for commercial applications. Some non-vacuum processes, however, involve the use of toxic or harmful chemicals. To address environmental concerns associated with non-vacuum processes, our previous work demonstrated a non-vacuum processed, CIGS absorber with an efficiency of 11.7%, utilizing green solvents and the electrostatic spray-assisted vapor deposition (ESAVD) method 11,12. However, in laboratory settings, high efficiency cells still require metal finger grids/electrodes to be deposited via high-vacuum thermal evaporation-one of the major cost-determining and efficiency-limiting steps in solar cell manufacturing. Since thermal evaporation is too expensive for low-cost production and scale-up, the most common metallization technique applied in the thin film solar cell industry is thick-film silver(Ag) contacts via screen-printing13. Screen-printing, however, is a contact printing method that can risk damaging fragile substrates due to applied contact forces. In contrast, inkjet printing is a non-contact, additive digital lithography method that allows for the precise deposition of metal electrodes while reducing material waste compared to screen printing. Compared to screen-printing, inkjet printing reduces material wastage. However, its limitations include the need for low-viscosity inks with limited metal content and relatively low resolution, with feature sizes typically between 50 and 100 µm. Meanwhile, aerosol printing faces scalability challenges due to limitations in multi-nozzle systems. Electrostatic Inkjet (ESJET) is an alternative additive and non-contact digital lithography printing technology in which an applied potential generates an electrostatic force on the surface of the liquid ink. When the electrostatic force overcomes the surface tension, a Taylor cone forms, and a liquid jet is emitted from the apex. The applied voltage can be adjusted to control both the electric field and the flow rate, making the printing system more compact and easier to control14,15. ESJET printing has demonstrated great potential for 2D/3D printing varied materials in electronics and biotechnology. Compared with widely used electrohydrodynamic(EHD) inkjet printing technology16,17,18, ESJET printing operates in a full voltage- controlled manner without requiring additional pressure from pump or carrier gas. When operating in nanoelectrospray (nES) mode, the fluid emission rate can be exclusively adjusted by the applied voltage, eliminating the need for external fluid pumping. Another key advantage is that very low flow rates can be achieved by selecting an appropriate nozzle and liquid combination, enabling the controlled ejection of minimum liquid volumes- as low as sub-femtoliter level- allowing for precise surface patterning. ESJET is capable of achieving high-resolution(down to ≤ 1 µm resolution) printing for a range of conductive, semi-conductive, insulating, and other functional inks with varied viscosities (1 to 10,000cP) and frequencies -capabilities not available with classical piezo-based ink jet technologies. The low cost, fine resolution, and high print speed of ESJET makes it a promising alternative for depositing finger electrodes in solar cells. The ability of ESJET to print fine lines could reduce the shadowing effect of finger electrodes, increasing the amount of transmitted light reaching the absorber and thereby enhancing solar cell efficiency.
Regarding metallic inks for conductive features, these inks are typically composed of nanoparticle suspensions of gold (Au), Ag or copper (Cu). Au has high conductivity and excellent thermal/chemical stability but is prohibitively expensive. Cu is more affordable but easily oxidizes in air, reducing its conductivity. Silver, by contrast, offers a balance of cost, chemical stability in air, and high conductivity, making it the most widely studied material for nanoparticles (nps) inks. Additionally, Ag provides a suitable work function for electron extraction in CIGS solar cells, and silver inks are widely available in the printed electronics market.
In our work, we demonstrate the application of ESJET printing technology for fabricating top metallic grid electrodes in CIGS solar cells for the first time. We investigate the effect of sintering temperature on the performance of printed Ag electrodes and examine cell stability under ISOS-D-1 and ISOS-D-3b standard tests. The ISOS-D-1 (dark storage) test evaluates a device’s tolerance to atmospheric components such as oxygen and moisture at ambient temperature and relative humidity (RH). Our device was stored at approximately 20 °C, and its photovoltaic performance was measured after three months. The ISOS-D-3b (damp heat) test assesses the impact of humidity (set at 85% RH) under elevated temperatures (85 °C), providing insights into long-term device stability.
Experiment and characterization
Preparation of CIGS solar cells
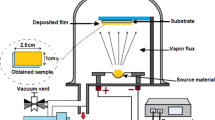
The CIGS absorber was deposited by ESAVD method as reported in the reference. In brief, during the ESAVD process, the precursor mixture of copper(II) chloride (CuCl2), indium(III) chloride (InCl3), gallium(III) nitrate (Ga(NO3)3), and thiourea (SC(NH2)2) in aqueous solution is atomized to form an aerosol, which is then charged (5–15 kV) and directed towards a heated substrate where it undergoes decomposition and a chemical reaction , forming a stable solid film of CIGS onto the Mo substrate. The as-deposited CIGS films were selenised at 550 °C for 30 min under Se vapor to allow grain growth into highly crystalline and pure photovoltaic grade CIGSSe films. After selenisation, a thin layer (50–70 nm) of CdS was deposited onto the CIGS absorber using chemical bath method to form a p–n junction. Subsequently, an approximately 50 nm thick intrinsic zinc oxide (i-ZnO) followed by 700 nm of Aluminum-doped ZnO (AZO) film was deposited using HHV sputtering system13. Finally, Ag electrodes were deposited using either ESJET printing or thermal evaporation for charge collection.
Structure, optical and electrical characterization
Crystal structure of the CIGS films was characterized by a grazing angle incidence X-ray diffraction (XRD) technique with a Bruker D8 discovery diffractometer using Cu Kα1 radiation (λ = 0.15406 nm). Raman spectroscopy measurement was carried out with a micro-Raman spectrometer (Renishaw inVia Raman Microscope) using a diode-pumped solid-state laser with excitation wavelength of 514 nm. The microstructure of the CIGSSe films was characterized using scanning electron microscope (SEM, JEOL JSM-6480LV). Solar cells performance were measured under AM1.5 simulated solar light with an intensity of 100 mW/cm2 using an Oriel Solar 1A.
ESJET printing of Ag electrode
Ink preparation
Silver ink was supplied by Dycotec Materials Ltd and further reformulated by Precision Varionic International Ltd to achieve a viscosity of 500cP and a solid content of 45wt%.
Single nozzle ESJET printing procedure
The Ag ink was loaded into the fluid reservoir, ensuring that the tip of the gold ink electrode was submerged. ESJET printing was controlled using a waveform synchronized with the stage movement to deposit the required pattern, as shown in Fig. 1a and b.
(a) ESJET single-nozzle setup, consisting of an FCA (Front Contact Assembly), HVS (High Voltage Supply), HVPS (High Voltage Power Supply), nozzle, emitter, and an X–Y-Z stage. (b) ESJET print system (500 mm × 500 mm × 50 mm) with an X–Y–Z nano-precision stage, designed and built by PVI.
During idle periods, when printing was not desired, a voltage Vi (idle voltage) was applied to the ink electrode. Vi is a sub-threshold voltage, normally in the range of 0.5–1.0 kV, deliberately set below the onset voltage for electrospray to prevent jet formation and maintain the system in a standby state. When the voltage was increased to Vp (print voltage, Vp is typically higher than Vi and is adjusted based on the desired print mode, generally in the range of 1.5–3.0 kV) which exceeds the onset threshold, the system initially exhibited a pulsating mode. In this phase, rather than forming discrete droplets, the system produced a pulsating nano-electrospray with intermittent, short-lived, and highly unstable jets. This phase is sometimes mistakenly referred to as “drop formation” in the literature; however, in this context, no actual drops are formed. Instead, tiny jets are ejected sporadically from the liquid meniscus. As the applied voltage was further increased from Vp to V2 (operating voltage for stable cone-Jet mode, typically around 2.0–2.5 kV, optimized for each specific ink and application), the system transitioned into a stable cone-jet mode. In this mode, a continuous and stable jet was formed, enabling consistent printing. For depositing metallic grid contacts, V2 was carefully selected to maintain stable cone-jet mode throughout the print cycle. In this regime, the fluid throughput (Q) is directly proportional to the square of the applied voltage (Vp2), ensuring a steady and reliable ink flow. Furthermore, scaling laws for pulsed electrohydrodynamic drop formation allowed control over line width and thickness as a function of table speed. The substrate speed was varied between 50 to 500 mm/s to achieve the required line geometry. To further enhance the print quality, the distance between the print nozzle and substrate was set to 50% of jet length14.
Thermal treatment conditions of Ag electrodes
Organic solvent and polymer additives are normally used in the Ag inks to improve its viscosity and printability. The existence of these organic residues deteriorates the conductivity of the Ag electrode. To remove these residues and enhance the direct contact between Ag nano/micro particles. Sintering of the ESJET Ag-Ink deposited CIGS cell was box oven cured in an air environment at various temperatures (220 °C, 160 °C, and 120 °C) for 30 min. The choice of sintering conditions was crucial to removing organic residues from the Ag ink and achieving good electrical conductivity while avoiding damage to the CIGS/CdS interface. The higher the sintering temperature, the less organic residues stay in the Ag electrode. However, too high temperature (> 250 °C) can destroy the CIGS/CdS interface, ultimately damaging the solar cell.
Results and discussion
Characterization of CIGS absorber
Non-vacuum deposited CIGS absorber films were selenization at 550 °C for 30 min. Figure 2 demonstrated the surface morphology and crystalline phase purity characterization of the CIGS absorber. As shown in the SEM images in Fig. 2a) and b, the CIGS thin film absorber reveals a polycrystalline structure with grain sizes of several micrometers. The grain morphology plays a crucial role in charge carrier transport and recombination processes, influencing overall solar cell efficiency. Larger, well-connected grains generally reduce grain boundary recombination, enhancing device performance. XRD and Raman spectroscopy were used to characterize the properties of the deposited CIGS absorber, with the results presented in Fig. 2c and d. The primary XRD peaks observed at 2θ values of 28.3°, 46.9°, and 53.0° in the as-deposited absorbers correspond to the (112), (204)/(220), and (116)/(312) crystal planes of the polycrystalline chalcopyrite CIGS structure. This confirms the successful formation of polycrystalline CIGS films. Additionally, the deposited absorber exhibits a predominant (112) orientation. Raman characterization revealed two strong characteristic peaks of CIGSSe. The peak at 178 cm−1 is associated with the “A1” mode of lattice vibration for chalcopyrite structures, while the peak at 218 cm−1 corresponds to the “B2/E” mode of lattice vibration. The peak observed at 295 cm−1 corresponds to the “A1” vibrational mode of CIGS in the absorber, likely resulting from a small residual amount of sulfur retained during the selenization process. The absence of additional Raman peaks suggests that the CIGS films are phase-pure. Both XRD and Raman results demonstrate that the deposited CIGS absorber is well-crystallized, exhibiting clear characteristic peaks of CIGS, with no detectable binary or ternary impurity phases.
Characterization of CIGS absorbers: (a) surface morphology (b) cross section SEM, (c) XRD and (d) Raman spectra of selenization CIGS absorber.
Design and characterization of ESJET printed Ag
For CIGS modules, the transparent conductive metal oxide window layer leads to high resistive losses. Depositing highly conductive but nontransparent metal grids on top of the AZO layer can efficiently improve current collection. These metal grids help reduce resistive losses caused by the low conductivity of the AZO layer, as well as mitigate long-term degradation. However, these nontransparent grids on top of the AZO layer could also create a shadowing effect, decreasing the amount of light transmitted to the absorber layer, thereby reducing the photocurrent generated in the device. The shadowing losses from metal grids are calculated as the total grid area divided by the total cell area. For a standard structured device with a simple grid pattern, as shown in Fig. 3a, the power lost due to shadowing (Pshadow) from the front contact grids is given by:
where PL is the intensity of the incident light and η is the photovoltaic efficiency of the solar cell. Wbus, Lbus, Wfinger and Lfinger are the width and length of bus and finger electrode respectively. D and L are the width and length of the entire device, d is the distance between two finger electrodes, and n is the number of finger electrodes. Resistance in CIGS solar cells arises from multiple sources, as shown in Fig. 3b, including resistance of Mo (R1), bulk resistance in the CIGS absorber (R2), resistance of AZO layer(R3) and contact resistance at the AZO/Ag interface (R4), as well as the resistance of the silver finger electrode (R5) and bus bars(R6), all of these factors influence charge transport and collection efficiency in the device. The dimensions of the busbars and contact fingers play a crucial role in balancing series resistance and power losses caused by the shadowing effect. Narrower contact fingers reduce shadowing losses, increasing light absorption. However, narrower contact fingers also increase series resistance due to the higher resistance of the finger electrode (R5) and the contact resistance (R4) at the AZO/Ag interface. Thus, the front electrode parameters should be optimized to achieve a balance between low resistance and minimal shadowing losses. The bus bars collect charge carriers from the fingers and enable a serial/ parallel connection to adjacent cells. Ideally, the grid lines should be tall but narrow to minimize shadow losses while maintaining efficient charge collection19,20.
(a) Finger and bus electrodes design of CIGS solar cells; (b) resistance in CIGS solar cells.
At the lab scale, vacuum-deposited Ni/Al is normally used as metal grids for CIGS solar cells, and the pixel cells are separated into small area of 20 mm2 (as shown in Fig. 4a) by mechanical scribing. To match the size of thermally evaporated metal grids (500 µm width Ag in this work), ESJET-printed finger electrodes with the same size and pattern were fabricated for small-sized pixel cells. Commercially CIGS solar modules are produced by screen printing metal inks, followed by high temperature sintering. The module can be manufactured using either stringing a series of individual standard cells together, or monolithically interconnecting a series of cells. In this work, we adopted the standard cell method for scale-up samples due to its ease of processing. To achieve an optimal trade-off between optical transparency and electrical performance, the reported optimized width of screen-printed finger electrodes in CIGS solar cells typically falls in the range of 50–200 µm, with spacing between electrodes in the range of 1–2 mm. In this study, the metal grids consist of fine parallel lines with an average height of 500 nm and a width ranging from 50 to 500 µm, intersected by two thicker bus bars (2 mm wide), as shown in Fig. 4b. The sheet resistance and thickness of ESJET-printed Ag grids were characterized using a four-point probe and profilometer, respectively., The related results were summarized in Table1. Compared to screen-printed Ag grids, which have a thickness of 1.94um and a sheet resistance of 0.121Ω/□, ESJET printed Ag grids exhibit a higher average thickness of 4.64um and lower sheet resistance of 0.082 Ω/□. While this value is slightly higher than the sheet resistance of vacuum-deposited silver electrode (≤ 1–10 mΩ/□), it is comparable to the reported sheet resistance of 10–100 mΩ/□ for other printed silver electrode widely used in thin film solar cells. Compared with ESJET printed electrodes composed of Ag flakes, ESJET printed Ag grids based on Ag nps exhibit a similar height but improved conductivity. Figure 4c shows the structure and morphology of the ESJET printed Ag grids under a microscope. The Ag ink demonstrates good wetting properties and is compatible with the top AZO layer of CIGS solar cells, allowing the fine lines of ESJET deposited grids to form with clear and well-defined edges. It is demonstrated that very fine lines with widths as small as 50 μm can be achieved using the ESJET printing method. From high magnification microscope images in Fig. 4d, it is clearly seen that the Ag grids are composed of Ag nps, which agglomerate into clusters of hundreds of nanometers in size.
Designed metal grid patterns for (a) small-area and (b) module-structured CIGS solar cells. Microscope images of ESJET-printed electrodes on FTO glass (c) optical microscope images (low magnification) and (d) SEM image (high magnification).
Photovoltaic performance of CIGS solar cells
After the Ag grids were ESJET-printed on top of the CIGS devices, the photovoltaic performance of the devices was characterized under simulated sunlight. The photovoltaic parameters of solar cells with ESJET-printed Ag grids are shown in Fig. 5 and summarized in Table 2. From Fig. 5a, it is clear that the solar cells show very poor performance when the Ag grids are annealed at 220 °C for 30 min, as the J–V curve appears almost linear. As the sintering temperature of the Ag grids decreases from 160 to 120 °C, the short circuit current (Jsc), open circuit voltage (Voc), and the resulting fill factor (FF) show a slight decline due to increased series resistance within the device. This increase in resistance is attributed to the incomplete removal of organic residues at 120 °C, which affects the conductivity of the ESJET-printed Ag nps. The efficiency variations among the devices were primarily influenced by the annealing temperature. As previously discussed in the section on sintering Ag grids, CIGS solar cells are highly sensitive to the sintering temperatures. Heating above 250 °C leads to irreversible degradation of the CIGS/buffer p‐n junction and element diffusion within the device21. Therefore, all post-deposition sintering processes of after the CdS layer deposition should be conducted at low temperatures to minimize cell degradation at the p–n junction interface. A high sintering temperature for an extended duration (220 °C for 30 min) also results in a lower fill factor and, consequently, reduced efficiency due to the increased defect density. Jsc is related to the charge carrier collection, and the slightly higher Jsc at 160 °C indicates that the Ag nps inks can be sufficiently cured at this temperature to form a good ohmic contact with the AZO layer. From Figure 5b, the best efficiency achieved by the device with ESJET-printed Ag grids is 12.13%, which is comparable with the 12.42% efficiency of the reference cell with thermally evaporated Ag grids. However, in comparison with the device using thermal evaporated Ag grids, solar cells with ESJET-printed Ag girds exhibit lower Voc and FF, likely due to the decomposition or diffusion of the CdS layer during the thermal treatment of the printed Ag ink. ESJET is a digital printing process that enables high-speed, large-area patterning. Figure 5c presents the J-V curve of CIGS devices with Ag finger and bus electrode patterns, which are typically used in commercial thin-film solar modules. The scaled-up cell, with a size of 1.6 cm2, achieves an efficiency of approximately 11.96%.
J-V curve of CIGS solar cells with ESJET printed finger electrodes (a) under different sintering conditions and the best efficiency solar cell at (b) small scale and (c) large scale devices.
Stability test of CIGS solar cells with ESJET printed finger electrodes
Apart from solar cell efficiency, the long- term stability of printed contacts is also a critical factor in evaluating solar cells22. Based on stability and lifetime tests conducted in accordance with ISOS procedures, ISOS-D-1 and ISOS-D-3b stability tests were carried out for small area CIGS solar cells with ESJET-printed grids. For the ISOS-D-1 stability test, the prepared CIGS solar cells with ESJET-printed Ag grids were stored in air for 3 months. The efficiency of the CIGS device decreased from 11.14 to 10.44%, corresponding to an efficiency loss of approximately 0.6%, as shown in Fig. 6a.
Stability test (a) ISOS-D-1 and (b) and (c) ISOS-D-3b results for CIGS solar cells with ESJET printed finger electrodes.
The ISOS-D-3b stability test was conducted to estimate the lifetime (for 80% of the original efficiency) of CIGS solar cells with ESJET-printed Ag grids, without encapsulation. A humidity chamber (Binder KMF115, temperature range: − 10 °C to 100 °C; humidity range: 10% to 98% RH) was used for the Damp Heat (DH) test under conditions of 85 °C and 85% relative humidity (RH). After 30 h of DH testing, the efficiency of CIGS solar cells without encapsulation decreased from 11.91 to 9.43%, as shown in Fig. 6b and c. The acceleration factor (AF) under DH testing conditions was calculated based on the Arrhenius equation and Peck’s relationship16:
In the equation, RHu and RHt denote relative humidity at use condition and test condition respectively, Ea is the activation energy (0.569 eV based on reference)23,24, k is the Boltzman’s constant (8.617385 × 10–5 eV/k),Tu and Tt are the temperature(Kelvin) for the use and test condition respectively. Using Eq. (2), the estimated lifetime of CIGS solar cells with ESJET-printed grids was approximately 32,638 h (3.73 years) when operated at 25 °C and 25% humidity without encapsulation. During the DH test, the Voc of the solar cells, which serves as an indicator of the CIGS/CdS junction quality, remained unchanged. However, the device performance of CIGS solar cells with ESJET-printed Ag grids was mainly limited by a decreasing Jsc and FF, caused by an increase in series resistance. This increase was attributed to the rising resistance of the AZO layer. Additionally, during the DH test, the surface color of AZO became less uniform, with visible stains (translucent milky color) appearing on top of the layer. These color changes led to a significant increase (tens to hundreds of times) of sheet resistance of AZO. However, no notable increase in the sheet resistance of Ag grids was observed after 30 h of DH testing. The stability tests of CIGS solar cells with ESJET-printed Ag grids demonstrated promising results. Encapsulation with an improved humidity barrier layer could further enhance stability, making the prepared CIGS solar cells more suitable for commercial applications.
Conclusions
In this work, we successfully ESJET-printed Ag grids for CIGS solar cells, achieving a best power conversion efficiency of 12.13%, which is comparable with efficiency (12.42%) of the reference cell with vacuum deposited Ag grids. ESJET printing offers significant advantages over conventional metallization techniques due to its high-resolution digital printing capability, which enables rapid prototyping of different contact geometries and patterns without the need for masks. The ESJET printing reported in this work is focused on small size lab scale devices (< 30 mm × 30 mm). Throughput is an important consideration since ESJET printing deposits grid fingers individually. However, this limitation can be mitigated by its high-speed capability and scalability. The ESJET method used in this study can achieve print speeds of 50–1200 mm/s, which is relatively fast for fine-line deposition. PVI and Oliveris are developing various multi-nozzle ESJET systems that can significantly increase throughput, making the process competitive with traditional methods. Current work provides a simple and feasible ESJET printing technique for the preparation of fine metal electrode for CIGS and other type of solar cells, enabling material-efficient and roll-to-roll processing while reducing material waste. These benefits can effectively lower production cost and promote the widely commercial adoption of solar cells. Beyond solar cells, ESJET also has great potential for printing fine lines/meshes in light emitting diodes, biosensors, displayers, and the other optoelectronics.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Keller, J. et al. High-concentration silver alloying and steep back-contact gallium grading enabling copper indium gallium selenide solar cell with 23.6% efficiency. Nat. Energy https://doi.org/10.1038/s41560-024-01472-3 (2024).
Carron, R. et al. Advanced alkali treatments for high-efficiency Cu(In, Ga)Se solar cells on flexible substrates. Adv. Energy Mater. 9(24), 1900408. https://doi.org/10.1002/aenm.201900408 (2019).
Ramanujam, J. et al. Flexible CIGS, CdTe and a-Si: H based thin film solar cells: A review. Prog. Mater. Sci. 110, 100619. https://doi.org/10.1016/j.pmatsci.2019.100619 (2020).
Mitzi, D. B. et al. Hydrazine-based deposition route for device-quality CIGS films. Thin Solid Films 517(7), 2158–2162. https://doi.org/10.1016/j.tsf.2008.10.079 (2009).
Bhattacharya, R. N. et al. 15.4% CuInGaSe-based photovoltaic cells from solution-based precursor films. Thin Solid Films 361, 396–399. https://doi.org/10.1016/S0040-6090(99)00809-3 (2000).
McLeod, S. M., Hages, C. J., Carter, N. J. & Agrawal, R. Synthesis and characterization of 15% efficient CIGSSe solar cells from nanoparticle inks. Prog. Photovolt. 23(11), 1550–1556. https://doi.org/10.1002/pip.2588 (2015).
Uhl, A. R. et al. Non-vacuum deposition of Cu(In, Ga)Se2 absorber layers from binder free, alcohol solutions. Prog. Photovolt. 20(5), 526–533. https://doi.org/10.1002/pip.1246 (2012).
Hao, S. S. et al. Effect of K doping on the performance of aqueous solution-processed Cu(In, Ga)Se solar cell. Adv. Energ. Sust. Res. 3(7), 2200006. https://doi.org/10.1002/aesr.202200006 (2022).
Zhang, T. et al. High efficiency solution-processed thin-film Cu(In, Ga)(Se, S) solar cells. Energ. Environ. Sci. 9(12), 3674–3681. https://doi.org/10.1039/c6ee02352e (2016).
Zhao, Y. H. et al. Controllable formation of ordered vacancy compound for high efficiency solution processed Cu(In, Ga)Se solar cells. Adv. Funct. Mater. 31(10), 2007928. https://doi.org/10.1002/adfm.202007928 (2021).
Wang, M. Q. et al. An alternative non-vacuum and low cost ESAVD method for the deposition of Cu(In, Ga)Se absorber layers. Phys. Status Solidi A 212(1), 72–75. https://doi.org/10.1002/pssa.201431295 (2015).
Wang, M. Q., Hossain, M. A. & Choy, K. L. Effect of sodium treatment on the performance of electrostatic spray assisted vapour deposited copper-poor Cu(In, Ga)(S, Se) solar cells. Sci. Rep. 7, 6788. https://doi.org/10.1038/s41598-017-07027-9 (2017).
Advanced Screen Printable Thin Film PV Front Side Silver Conductor Compositions. Ieee Phot Spec Conf 2009, 40–42.
Lin, Y. F., Van Kerschaver, E., Cabanas-Holmen, K. Laser Sintering of screen-printed silver paste for silicon solar cells. In 2013 IEEE 39th Photovoltaic Specialists Conference (Pvsc) 3445–3447 (2013).
Wang, K. & Stark, J. Dispersion of nano-Au suspension using novel pulsed jet nanoelectrospraying approach. Mater Lett 123, 120–123. https://doi.org/10.1016/j.matlet.2014.03.016 (2014).
Lee, Y. G. & Choi, W. S. Electrohydrodynamic Jet-printed zinc-tin oxide TFTs and their bias stability. ACS. Appl. Mater. Interfaces 6(14), 11167–11172. https://doi.org/10.1021/am5009826 (2014).
Li, X. et al. Direct writing of silver nanowire electrodes via dragging mode electrohydrodynamic jet printing for organic thin film transistors. Org. Electron. 62, 357–365. https://doi.org/10.1016/j.orgel.2018.07.027 (2018).
Song, Y. H. et al. Electrostatic jet engineering of flexible composite pressure sensors for physical applications. ACS Appl. Polym. Mater. 4(2), 868–878. https://doi.org/10.1021/acsapm.1c01357 (2022).
Rowell, M. W. & McGehee, M. D. Transparent electrode requirements for thin film solar cell modules. Energ. Environ. Sci. 4(1), 131–134. https://doi.org/10.1039/c0ee00373e (2011).
Kessler, J., Wennerberg, J., Bodegård, M. & Stolt, L. Highly efficient Cu(In, Ga)Se mini-modules. Sol. Energ. Mat. Sol. C 75(1–2), 35–46. https://doi.org/10.1016/S0927-0248(02)00102-2 (2003).
Koprek, A. et al. Effect of Cd diffusion on the electrical properties of the Cu(In, Ga)Se2 thin-film solar cell. Sol. Energ. Mat. Sol. C 224, 110989. https://doi.org/10.1016/j.solmat.2021.110989 (2021).
Guillemoles, J. F. et al. Stability issues of Cu(In, Ga)Se-based solar cells. J. Phys. Chem. B 104(20), 4849–4862. https://doi.org/10.1021/jp993143k (2000).
Ott, T., Walter, T., Hariskos, D., Kiowski, O., Schäffler, R. Accelerated aging and contact degradation of CIGS solar cells. In 2012 IEEE 38th Photovoltaic Specialists Conference (Pvsc) vol. 2 (2013). https://doi.org/10.1109/Jphotov.2012.2226141.
Sanaie, G., Schenkelberg, F. Using reliability modeling and accelerated life testing to estimate solar inverter useful life. In 59th Annual Reliability and Maintainability Symposium (Rams) (2013).
Acknowledgements
The authors would like to acknowledge the financial support from, EPSRC & Innovate UK Project: Hi-Prospects (102470), “High resolution Printing of Solar Photovoltaic Electrode Structures” EU Commission H2020 project titled “Innovative High Resolution Electro-Static printing of Multifunctional Materials”, Hi-Response (646296). The European Commission supported this work under the H2020 HI-ACCURACY project (Grant Agreement ID: 862410).
Author information
Authors and Affiliations
Contributions
M.W prepared and did characterisation of CIGS solar cells. M.W also wrote the draft. M.Q, M.H, and R.S did the ESJET printing and related characterisation. P.O did the writing on ESJET printing. P.O, K.C and M.W initiated the concept. All authors contributed to the proofreading of the draft.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, M., Obene, P., Questianx, M. et al. Electrostatic InkJet printed silver grids for non-vacuum processed CIGS solar cells. Sci Rep 15, 11048 (2025). https://doi.org/10.1038/s41598-025-94348-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94348-9