Abstract
Previous studies on gait changes in mild cognitive impairment (MCI) are inconsistent. Alzheimer’s disease (AD) plasma biomarkers, amyloid beta (Aβ) and phosphorylated-tau (p-tau), are relevant to gait disorders. This study explores gait changes in MCI and the relationship between gait performance and AD plasma biomarkers. 231 participants were recruited and stratified based on p-tau181 levels into: low p-tau181 with normal cognition (lT-NC), low p-tau181 with MCI (lT-MCI), and high p-tau181 with MCI (hT-MCI). The same cohort was subsequently stratified by Aβ42/Aβ40 levels into: high Aβ42/Aβ40 with normal cognition (hA-NC), high Aβ42/Aβ40 with MCI (hA-MCI), and low Aβ42/Aβ40 with MCI (lA-MCI). Demographic, cognitive and gait data were compared across groups. The hT-MCI and lA-MCI groups were older than the other groups. Significant differences in stride length were found between lT-NC and hT-MCI, lT-MCI and hT-MCI, but not between lT-NC and lT-MCI. Neuropsychological assessments revealed poorer performance in hT-MCI and lT-MCI groups relative to lT-NC, while global cognitive function was comparable between hT-MCI and lT-MCI groups. No such associations were observed between stride length and Aβ42/Aβ40 levels. Decreased stride length, which is generally considered to be indicative of poorer gait, was significantly associated with elevated p-tau181 levels and independent of global cognitive status. These findings highlight the potential of p-tau181 as a biomarker for tau-related motor dysfunction in MCI.
Similar content being viewed by others
Introduction
Mild cognitive impairment (MCI) is recognized as an intermediate stage between intact cognitive function and dementia1, with progression to dementia occurring in 4–15% of cases within one year2. It has been acknowledged that MCI can lead to alterations in gait and balance3,4. Gait disorders and cognitive impairments emerge concomitantly, affecting approximately 50% of elderly individuals aged over 855,6,7, which seriously affect the quality of life and increase the risk of death of patients. It is worth noting that not only does gait disorder usually precede cognitive impairment in the aging process8,9,10, but gait function declined faster than cognitive ability11. Thus, the gait disorder may not be directly caused by cognitive impairment, but may serve as a sensitive indicator of early tiny changes in brain function. Some scholars have proposed that gait variability serves as the most sensitive indicator of cognitive decline12, while others have suggested that reduced gait speed indicates early cognitive deterioration13. Conversely, certain studies have argued against the existence of gait changes in MCI14. The conflicting findings in the existing literature necessitate an exploration of the underlying reasons.
From the biochemical perspective, biomarkers such as amyloid beta (Aβ) and phosphorylated-tau181 (p-tau181) was believed to be closely related to the cognitive-related gait disorders15, but most studies have focused on idiopathic normal pressure hydrocephalus patients16. Besides, biomarkers were primarily obtained through invasive procedures like lumbar puncture or costly techniques like positron emission tomography (PET). As a result, blood-based biomarkers are increasingly being promoted as a more viable option17. Single molecule array (Simoa) is a highly sensitive method for detecting dementia plasma biomarkers, achieving detection sensitivities at the atomic level18. It exhibits high sensitivity to p-tau18119, Aβ40 and Aβ4220.
Previous studies examining the relationship between gait and Alzheimer’s disease (AD) biomarkers have yielded inconsistent results. In transgenic mice, tau pathology showed a marked delay in motor initiation21. In a Figure-of-eight test to assess dynamic balance, prolonged completion times was correlate with Aβ42/Aβ40 ratios22. Interestingly, under dual-task conditions, the Timed Up and Go (TUG) test showed a negative correlation with cerebrospinal fluid p-tau levels in cognitive impairment patients23.
Earlier researches on cognitive-related gait disorders have primarily focused on Aβ and gait speed, with gait measurements often being simplistic and subjective, unable to detect subtle changes. Additionally, some studies have had small sample sizes, contributing to the variability in findings24,25. Hence, our study aims to explore the forms of gait changes in early cognitive impairment and analyze the influence of AD plasma biomarkers therein, through a quantitative motion system that combines high-precision vision sensor and artificial intelligence learning algorithm. By clarifying the potential correlation, we can pave the way for the subsequent exploration of causality, and provide a certain theoretical basis for early intervention and prevention of age-related diseases in the future.
Materials and methods
Participants
In this study, 231 subjects (aged 41 to 88 years) were enrolled in the Nanjing Drum Tower Hospital from 2020 to 2023. All subjects underwent background information collection, neuropsychological assessment, AD plasma biomarker testing and gait testing which were collected at intervals of no more than 2 weeks. This study has been approved by the Ethics Committee of the Nanjing Drum Tower Hospital (Ethics number: 2022-472-02), and the study conforms with World Medical Association Declaration of Helsinki published on the website of the Journal of American Medical Association.
The inclusion criteria were as follows: (1) complete demographic information, cognitive assessment information, blood test results and gait information; (2) normal cognitive or MCI; (3) cooperate with the completion of this study; (4) right handed.
The exclusion criteria were as follows: (1) cognitive grading to dementia; (2) any other conditions affecting gait, such as joint disease, extremity fracture, and so on; (3) history of ischemic stroke with infarct size more than 1.5 cm in diameter; (4) occlusion or stenosis > 50% of intracranial or extracranial large arteries; (5) cerebral hemorrhage; (6) other neurological diseases, such as Parkinson’s disease, epilepsy, and so on; (7) serious systemic diseases; (8) limited cooperation with the study.
Background information collection
Background information collection covered gender, age, education, body mass index, and personal history. In personal history, we concentrated on vascular risk factors, involving hypertension, diabetes, hyperlipidemia, smoking and drinking.
Neuropsychological assessment
The neuropsychological assessment was conducted by a trained psychiatrist. The detailed tests included the following assessments: global cognitive evaluation (Mini Mental State Examination, MMSE; Montreal Cognitive Assessment, MoCA), psychiatric assessment (Hamilton Depression Rating Scale, HAMD; Hamilton Anxiety Rating Scale, HAMA), short-term memory assessment (Digit Span Forward, DSF), working memory assessment (Digit Span Backward, DSB), visual learning and memory function evaluation (Visual Reproduction Immediate Recall, VRIR; Visual Reproduction Delayed Recall, VRDR; Visual Reproduction Recognition, VRR; Visual Reproduction Copy, VRC), cognitive control test (Stroop Color Word Test- Dot, SCWT- Dot; Stroop Color Word Test- World, SCWT- Word; Stroop Color Word Test- Interference, SCWT- Interference), psychomotor speed and visual search assessment (Trail Making test-A, TMT-A), divided attention assessment (Trail Making test-B, TMT-B), encoding and retrieval verbal memory assessment (Auditory Verbal Learning Test Immediate Recall, AVLTIR; Auditory Verbal Learning Test Short Time Delay Recall, AVLTSTDR; Auditory Verbal Learning Test Long Time Delay Recall, AVLTLTDR; Auditory Verbal Learning Test Recognition, AVLTR), visuospatial function assessment (Visual Objective and Space Perception battery- Silhouettes, VOSP—Silhouettes).
In this study, participants’ cognitive grading was defined by the scores of MMSE and MoCA. The education-adjusted cut-off values for the MMSE were > 17 for illiteracy, > 20 for 1‒6 years of education, and > 24 for > 6 years of education26. The education-adjusted cut-off values for the MoCA were > 13 for illiteracy, > 19 for 1‒6 years of education, > 24 for > 6 years of education27. The normal cognition was defined as both the MoCA and MMSE scores above the cut-off values. The MCI was defined as (1) cognitive impairment detected by patients, informants, or clinical physicians; (2) having a MMSE score above the cut-off value and a MoCA score below its cut-off value; (3) normal activity of daily living.
AD plasma biomarker testing
Peripheral blood samples of each subject were obtained by venipuncture. The plasma samples were centrifuged at room temperature for 2500 rpm × 15 min within 2 h, and divided into 0.5 ml parts in a polypropylene storage tube (Sarstedt, Germany) and then stored at -80 °C for further use. AD plasma biomarker testing was performed by using an ultra-sensitive single-molecule array (SIMOA/qPCR) (Quanterix, MA, USA), a technique that can detect 10−16 molar concentration in plasma28. The biomarkers include Aβ42, Aβ40 and p-tau181. The Quanterix Accelerator Laboratory utilized the Neurology 3-Plex A Kit (101,995) to measure Aβ40 and Aβ42, and the P-Tau 181 Advantage V2 Kit (103,714) to quantify p-tau181 levels.
Since there was no consistent cut-off value, some studies determined the cut-off value by calculating the median. Considering our small sample size, and the subjects data sourced for single center hospital, we chose to adopt the cut-off values obtained from published studies conducted on the Chinese population using the same Simoa method. Specifically, p-tau181 > 2.41 pg/ml was defined as high level, and Aβ42/Aβ40 < 0.056 was defined as low level29.
Gait testing
Gait testing was conducted by the ReadyGo quantitative motor function assessment system. Subjects were asked to wear everyday clothing, without skirts and high heels, and continuously walk three straight back and forth at the daily walking speed between two squares. The two squares are 3 m apart. The ReadyGo quantitative motor function assessment system could identify 32 bone points in the whole body in real time through high-precision vision sensor and artificial intelligence learning algorithm, and the real-time position of bone points in the process of movement was electronically restored in three-dimensional through the Time-of-Flight depth sensor. At the same time, the key actions such as lifting, landing, turning, standing, and sitting, were semantically analyzed and automatically annotated by artificial intelligence. Gait indicators include gait velocity, stride length, step speed, swing velocity, step height, step width, stride frequency, stance phase, swing phase, and double-support phase, with their average, median, maximum, minimum presented. The gait indicators were automatically calculated by the system and corrected manually before group comparison.
The definition of gait indicators were as follows: (1) gait velocity: the distance traveled in unit time, unit m/s; (2) stride length: longitudinal straight distance between two points of heel or toe on the same side, unit m; (3) step speed: the stride length in each stride cycle time, unit m/s; (4) swing velocity: the distance within swing time, unit m/s; (5) step height: the vertical distance between the midpoint of the foot and the ground, unit m; (6) step width: the transverse distance between the midpoint of the two feet, unit m. (7) stride frequency: the number of steps taken per unit time, unit steps/min; (8) stance phase: the time between the foot touching the ground and leaving the ground again, that is, the stage of foot on the ground, accounts for about 60% of the walking cycle; (9) swing phase: the time between the foot leaving the ground and landing again, that is, the stage of foot in the air, accounting for about 40% of the walking cycle; (10) double-support phase: the stage of both feet land at the same time, accounting for about 20% of a walking cycle.
Statistical analysis
SPSS (Statistical Product and Service Solutions) 26.0 was used for statistical analysis in this study. The counting data were analyzed by Pearson’s chi-squared test, presented as number of cases (percentage). Measurement data which conformed to a normal distribution were tested using ANOVA or independent samples t-test, and were expressed as mean ± standard deviation. Measurement data which conformed to a abnormal distribution were analyzed by Kruskal–Wallis test or Mann–Whitney U test, and shown as median (interquartile range). Additionally, demographics data showing remarkable differences was added as covariate in general linear model (GLM) to compare the gait differences between groups. Covariate used in the GLM and partial correlation that showed non-normal distribution was logarithm-transformed. Bonferroni correction was applied in GLM for multiple comparison corrections. Partial correlation analysis was conducted to analysis the correlation between gait indicators and each cognitive domains. p < 0.05 was considered statistically significant.
Results
Background data and neuropsychological data
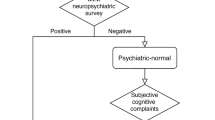
According to the inclusion criteria, 231 subjects were eventually enrolled in the study. Based on p-tau181 levels and neuropsychological assessment, subjects were divided into the following groups: low p-tau181 level with normal cognition (lT-NC), low p-tau181 level with MCI (lT-MCI), and high p-tau181 level with MCI (hT-MCI) groups. Then the same cohort of 231 subjects was grouped again by Aβ42/Aβ40 levels: high Aβ42/Aβ40 level with normal cognition (hA-NC), high Aβ42/Aβ40 level with MCI (hAβ-MCI), low Aβ42/Aβ40 level with MCI (lA-MCI) groups. Detailed information is presented in Fig. 1.
The enrollment flowchart of this study illustrates the participant inclusion process, which ultimately resulted in the inclusion of 231 subjects based on the defined inclusion and exclusion criteria. Note: MRI: magnetic resonance imaging; NC: normal cognition; MCI: mild cognitive impairment.
The study included 77 lT-NC subjects, 90 lT-MCI subjects, and 43 hT-MCI subjects. The hT-MCI group was significantly older than the other two groups. There were no significant differences in gender, years of education, and body mass index (BMI) among the three groups (p > 0.05). Similarly, there were no significant differences in vascular risk factors such as hypertension, diabetes, hyperlipidemia, smoking and drinking (p > 0.05). Neuropsychologically, the hT-MCI and lT-MCI groups performed worse than the lT-NC group. While global cognitive function (MMSE and MoCA) did not differ between hT-MCI and lT-MCI groups (MMSE: p = 0.429; MoCA: p = 0.554), hT-MCI participants exhibited significantly worse performance on psychiatric assessment (HAMA: p = 0.09), cognitive control test (SCWT-Dot: p = 0.031; SCWT-Word: p = 0.003; SCWT-Interference: p = 0.004) and memory assessment (AVLTIR: p = 0.01; AVLTLTDR: p = 0.018; AVLTR: p = 0.002). Detailed information is presented in Table 1 and S1.
Grouped according to Aβ42/Aβ40, we included 76 hA-NC subjects, 113 hA-MCI subjects, and 20 lA-MCI subjects. Demographically, there were no significant differences in gender, years of education and BMI (p > 0.05). The lA-MCI group was also significantly older than the other two groups. There were no significant differences in vascular risk factors (p > 0.05). Notably, p-tau181 levels showed significant differences among the groups, and the hA-MCI and lA-MCI groups had worse cognitive performance than the hA-NC group (p < 0.05). Detailed information is presented in Table 2 and S2.
Additionally, we observed that 21.43% of subjects (n = 21) in the normal cognition group were p-tau181 high level, and 22.45% subjects (n = 22) in the normal cognition group were Aβ42/Aβ40 low level. These subjects were not followed up for further analysis.
Gait analysis
Gait differences among the three groups were compared using GLM with logarithm-transformed age as a covariate, corrected for multiple comparisons by Bonferroni. The results indicated significant differences in four indicators of stride length (including average and median, left and right) between the lT-NC and hT-MCI groups, as well as between the lT-MCI and hT-MCI groups. However, there was no significant difference in stride length between the lT-NC and lT-MCI groups. Thus, we conclude that in patients with MCI, those who are high p-tau181 level exhibit worse gait performance, with stride length being a more sensitive indicator of gait changes associated with high p-tau181 level MCI.
There was also a significant difference in step speed between the lT-NC and hT-MCI groups. In addition, swing velocity showed difference between the lT-MCI and hT-MCI groups. However, these differences were limited to one or two indicators. No significant differences were observed in other gait indicators among the three groups. Detailed information is presented in Table 3, with additional details in supplementary information (Table S3).
In the groups categorized by Aβ42/Aβ40 level (hA-NC, hA-MCI and lA-MCI), with age and p-tau181 value logarithm-transformed as covariates, the GLM analysis of gait indicators was carried out. The results revealed differences in swing velocity between the hA-NC and lA-MCI groups, as well as between the hA-MCI and lA-MCI groups. However, these differences were limited to a single indicator, the minimum-right. In addition, significant differences in stance phase and swing phase were observed between the hA-MCI and lA-MCI groups; step height differed between the hA-NC and hA-MCI groups, though these differences were also confined to a single indicator. Although gait changes were present in MCI with low Aβ42/Aβ40 level, the gait indicators were scattered and singular, indicating a weak association. Detailed information is presented in Table S4.
Correlation analysis
Partial correlation analysis between gait indicators and each cognitive domains, with logarithm-transformed age as a covariate, revealed that stride length was significantly correlated with MoCA, DSB and TMT-B. In addition, there were also correlations between stride length and DFS, VRR, TMT-A and AVLTLTDR, though these correlations were not strong. Detailed information is presented in Fig. 2.
A partial correlation analysis was conducted between gait parameters and cognitive domains, with age as a covariate. Blue circles indicate positive correlations, while red circles represent negative correlations. The size of the circle reflects the strength of the correlation, with larger circles indicating stronger relationships. Asterisks within the circles denote statistical significance: *** for p < 0.001, ** for p < 0.01, and * for p < 0.05. Note: MMSE: Mini Mental State Examination; MoCA: Montreal Cognitive Assessment; DSF, Digit Span Forward; DSB: Digit Span Backward; HAMD: Hamilton Depression Rating Scale; HAMA: Hamilton Anxiety Rating Scale; VRIR: Visual Reproduction Immediate Recall; VRDR: Visual Reproduction Delayed Recall; VRR: Visual Reproduction Recognition; VRC: Visual Reproduction Copy; SCWT- Dot: Stroop Color Word Test- Dot; SCWT- Word: Stroop Color Word Test- World; SCWT- Interference: Stroop Color Word Test- Interference; TMT-A: Trail Making test- A; TMT-B: Trail Making test- B; AVLTIR: Auditory Verbal Learning Test Immediate Recall; AVLTSTDR: Auditory Verbal Learning Test Short Time Delay Recall; AVLTLTDR: Auditory Verbal Learning Test Long Time Delay Recall; AVLTLTDR: Auditory Verbal Learning Test Recognition; VOSP—Silhouettes: Visual objective and space perception battery- Silhouettes.
Discussion
This study explored the forms of gait alterations in MCI. Previous research on gait associated with MCI have yielded inconsistent results. For the first time, we identified the decreased stride length, which is generally considered to be the most sensitive indicator of poorer gait, was significantly associated with elevated level of p-tau181 and were independent of the cognitive status. This observation was not seen in the Aβ42/Aβ40 group. Moreover, correlation analysis revealed that stride length was closely correlated with MoCA, DSB and TMT-B scores.
The National Institute on Aging (NIA) and the Alzheimer’s Association (AA) introduced the concept of biochemically defined Alzheimer’s disease, suggesting that AD plasma biomarkers may be more indicative of biochemically defined AD rather than dementia30. In our study, we observed a mismatch between clinical assessments and biomarker results, with some patients showing positive biomarkers but negative clinical assessments. Previous research had also reported such discrepancies. For instance, 29.5% of cognitively unimpaired individuals over 65 years old tested positive for Aβ PET31. Similarly, autopsy study had found high levels of Aβ deposition in elderly individuals without any evidence of cognitive impairment32. Frank et al. found that the positivity rate for cerebrospinal fluid Aβ in cognitively normal subjects was 27.2%, and for p-tau181, it was 15.6%, which were comparable to our findings33. This population may progress more rapidly to subjective cognitive decline or MCI and could benefit the most from early interventions34. In our study, we did not conduct a detailed analysis of this subgroup, nor were they included in the normal cognition cohort. Instead, our focus was primarily on exploring the relationship between MCI and hematological biomarkers. Subsequent investigations should involve comparative analyses of this specific subgroup.
The abnormal folding of Aβ40 and Aβ42 peptides make up amyloid plaques, while hyperphosphorylated tau proteins make up neurofibrillary tangles. These pathophysiological processes occur approximately a decade or more before the clinical syndrome of dementia, progressing over time to synaptic loss, decreased brain metabolism, and structural or functional brain changes35. Aβ42/Aβ40 and p-tau181 have been recognized as critical biomarkers for predicting future cognitive decline36. Cross-sectional study showed that the Aβ42/Aβ40 was positively correlated with gait variability in mild AD patients37. Longitudinal assessment had linked lower plasma Aβ42/Aβ40 ratios to declining cognition and gait speed over time38.
P-tau181 is a highly specific pathological biomarker for AD39, with concentrations beginning to rise as early as 16 years prior to estimated symptom onset in presymptomatic and symptomatic familial AD40. Gait changes can occur as early as 12 years before the onset of cognitive impairment41, highlighting the importance of focusing on the gait changes in the early stages of cognitive impairment, such as MCI. Animal study had shown that excessive p-tau accumulation in the hippocampus and cortex can lead to subtle gait abnormalities and spatial memory impairments42. Consistent with this, tau-PET study in MCI patients had shown a significant correlation between increased tau burden in Braak I-IV stages and increased gait speed variability43. Interestingly, previous study also showed p-tau levels was not correlated with gait scale scores23.
Previous study had demonstrated that abnormalities in mental state may influence gait performance by increasing gait variability44. Montero-Odasso et al. reported an association between declines in executive function and impaired gait control, particularly under dual-task conditions45. Additionally, Allali et al. provided evidence that memory dysfunction may indirectly affect gait, potentially through mechanisms such as attentional deficits or impaired task-switching abilities46. These findings collectively highlight the complex interplay between cognitive and emotional factors and motor function in neurodegenerative diseases. Our results were consistent with these findings, and the potential synergistic effects of tau burden and these factors on gait control warrant further investigation.
MCI patients may exhibit slower gait speed, lower cadence, shorter stride length, and greater stride variability, compared to normal individuals47. The presence of abnormal gait was a significant predictor of dementia development48, accelerating the conversion of amnestic MCI to AD49. In a study involving middle-aged and elderly individuals, poor baseline cognition and slower gait speed respectively predicted faster declines in gait speed and cognition50. In a large Chinese community study that also using high-precision gait detection, significant gait differences were observed between cognitive impairment patients and normal controls. Turn time was notably correlated with plasma p-tau, and a combination of dual-task gait, dual-task eye-tracking assessment, and plasma p-tau181 effectively identified cognitive impairment patients51. While gait is associated with cerebrospinal fluid tau levels, it appears unrelated to longitudinal cognitive decline, we considered these findings may be due to insufficient follow-up duration47.
In our study, both the hT-MCI and lA-MCI groups were older than the other two groups, which is similar to previous studies30, suggesting that biomarkers indicate the beginning of the pathological process of cognitive impairment. When age was controlled as a covariate, correlation analysis suggested that impaired gait was closely related to global cognitive function (MoCA), working memory (DSB) and divided attention (TMT-B). This may be related to the location of p-tau181 deposition. It has been shown that the accumulation of p-tau181 usually begins in the entorhinal cortex, gradually reaches marginal areas such as the hippocampus, and eventually spreads to the extensive neocortex52. From a neuroimaging perspective, some scholars suggest that individuals at risk of cognitive-motor decline exhibit more than a 50% greater atrophy in the frontal and parietal lobes compared to those with purely cognitive impairment53. White matter fibers serve as important channels connecting cortical and subordinate neurons, and their lesions, white matter hyperintensities, was thought to be closely associated with cognitive and gait impairment in older adults54.
As an important indicator of fibrinous tangle damage, we proposed that p-tau181 may be cutting off the cortical signal to control gait, while Aβ, which is deposited in the cortex as amyloid plaques, is more likely to be involved in cognitive function than gait function. Besides, Aβ pathology plateaus early in AD, exhibiting smaller dynamic changes, whereas plasma p-tau181 rises during preclinical stages and progresses with disease severity23. These may explain why we did not find gait differences according to Aβ42/Aβ40 grouping. Our study found that p-tau181 may be a biomarker of the earliest interference between cognition and motor control.
Additionally, we found that most of the gait differences between groups were observed in the left side, which may be related to the lateralization of brain activity. The human brain exhibits structural, functional and behavioral asymmetries, a phenomenon known as brain lateralization55. In terms of motor control, the left hemisphere dominates in motor control, exhibiting a left hemisphere motor advantage56. Moreover, activation patterns differ between right-handed and left-handed individuals, left-handed individuals activate larger brain areas and greater brain volume during continuous movement, with a significantly lower degree of brain lateralization57. This suggests that non-dominant limb movement activates both hemispheres more equally58. In Parkinson’s disease, patients with onset in the dominant side exhibit fewer motor deficits, indicating greater neural reserve, which enables better adaptation to disease-related changes59. In our study, all participants were right-handed, and as the left side has less neural reserve, it may more readily show differences when examining early cognitive and gait impairments. This may explain why the majority of group differences were observed on the left side. In terms of cognition, Wu et al. found that in middle-aged individuals, greater left-lateralization of the prefrontal cortex was associated with better cognitive performance, whereas in older adults, lower left-lateralization was linked to better cognitive outcomes60. In our study, both left- and right-stride length were associated with cognitive domains, such as MoCA, DSB and TMT-B. However, there were slight differences in the strength of these correlations, with right-stride length showing a stronger association with MoCA compared to left-stride length. This may reflect lateralized cognitive functions, but as our sample includes both middle-aged and older adults, the specific degree of lateralization remains unclear and warrants further investigation through functional magnetic resonance imaging.
Although plasma p-tau181 has been shown to discriminate AD dementia from non-AD neurodegenerative disorders (AUC = 0.93, sensitivity = 92%, specificity = 87%)23, our findings did not exclude primary age-related tauopathy. Nevertheless, we highlight the pragmatic value of plasma p-tau181 as a screening marker for gait decline in resource-limited settings, even without Aβ confirmation. Furthermore, our gait differences did not meet the criteria for pathological gait disorders, they might represent alterations in preclinical motor function. This is consistent with the preclinical AD framework, where biomarker abnormalities precede the emergence of overt symptoms. Future longitudinal studies should validate whether these subclinical gait changes foreshadow subsequent motor or cognitive decline.
This study proposes that decreased stride length was significantly associated with elevated p-tau181 levels and independent of global cognitive status. These findings highlight the potential of p-tau181 as a biomarker for tau-related motor dysfunction in MCI. At the same time, this study also has some shortcomings, we still need further research to explore the detailed mechanism. In addition, we need a larger sample size and longitudinal studies to complete and enrich our research.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kuan, Y. C. et al. Balance and gait performance in older adults with early-stage cognitive impairment. Eur J Phys Rehabil Med 57, 560–567. https://doi.org/10.23736/S1973-9087.20.06550-8 (2021).
Manly, J. J. et al. Estimating the prevalence of dementia and mild cognitive impairment in the US: The 2016 health and retirement study harmonized cognitive assessment protocol project. JAMA Neurol 79, 1242–1249. https://doi.org/10.1001/jamaneurol.2022.3543 (2022).
Doi, T. et al. Mild cognitive impairment, slow gait, and risk of disability: A prospective study. J Am Med Dir Assoc 16, 1082–1086. https://doi.org/10.1016/j.jamda.2015.07.007 (2015).
Yu, L. et al. Incident mobility disability, mild cognitive impairment, and mortality in community-dwelling older adults. Neuroepidemiology 53, 55–62. https://doi.org/10.1159/000499334 (2019).
Beauchet, O. et al. Poor gait performance and prediction of dementia: Results from a meta-analysis. J Am Med Dir Assoc 17, 482–490. https://doi.org/10.1016/j.jamda.2015.12.092 (2016).
Verghese, J. et al. Abnormality of gait as a predictor of non-Alzheimer’s dementia. N Engl J Med 347, 1761–1768. https://doi.org/10.1056/NEJMoa020441 (2002).
Nutt, J. G. Classification of gait and balance disorders. Adv Neurol 87, 135–141 (2001).
Quan, M. et al. Walking pace and the risk of cognitive decline and dementia in elderly populations: A meta-analysis of prospective cohort studies. J Gerontol A Biol Sci Med Sci 72, 266–270. https://doi.org/10.1093/gerona/glw121 (2017).
Mielke, M. M. et al. Assessing the temporal relationship between cognition and gait: Slow gait predicts cognitive decline in the Mayo Clinic Study of Aging. J Gerontol A Biol Sci Med Sci 68, 929–937. https://doi.org/10.1093/gerona/gls256 (2013).
Skillback, T. et al. Slowing gait speed precedes cognitive decline by several years. Alzheimers Dement 18, 1667–1676. https://doi.org/10.1002/alz.12537 (2022).
Jayakody, O. et al. Relative trajectories of gait and cognitive decline in aging. J Gerontol A Biol Sci Med Sci 77, 1230–1238. https://doi.org/10.1093/gerona/glab346 (2022).
Byun, S. et al. Gait variability can predict the risk of cognitive decline in cognitively normal older people. Dement Geriatr Cognit Disord 45, 251–261. https://doi.org/10.1159/000489927 (2018).
Jayakody, O., Breslin, M., Srikanth, V. K. & Callisaya, M. L. Gait characteristics and cognitive decline: A longitudinal population-based study. J Alzheimers Dis 71, S5–S14. https://doi.org/10.3233/JAD-181157 (2019).
Bovonsunthonchai, S. et al. Quantitative gait analysis in mild cognitive impairment, dementia, and cognitively intact individuals: A cross-sectional case-control study. BMC Geriatr 22, 767. https://doi.org/10.1186/s12877-022-03405-9 (2022).
Chen, P. H., Lin, S. I., Liao, Y. Y., Hsu, W. L. & Cheng, F. Y. Associations between blood-based biomarkers of Alzheimer’s disease with cognition in motoric cognitive risk syndrome: A pilot study using plasma Abeta42 and total tau. Front Aging Neurosci 14, 981632. https://doi.org/10.3389/fnagi.2022.981632 (2022).
Gronning, R. et al. Association between ventricular CSF biomarkers and outcome after shunt surgery in idiopathic normal pressure hydrocephalus. Fluids Barriers CNS 20, 77. https://doi.org/10.1186/s12987-023-00475-8 (2023).
Jack, C. R. Jr. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimers Dement https://doi.org/10.1002/alz.13859 (2024).
Dong, R., Yi, N. & Jiang, D. Advances in single molecule arrays (SIMOA) for ultra-sensitive detection of biomolecules. Talanta 270, 125529. https://doi.org/10.1016/j.talanta.2023.125529 (2024).
Bayoumy, S. et al. Clinical and analytical comparison of six Simoa assays for plasma P-tau isoforms P-tau181, P-tau217, and P-tau231. Alzheimers Res Ther 13, 198. https://doi.org/10.1186/s13195-021-00939-9 (2021).
Chatterjee, P. et al. Plasma Abeta42/40 ratio, p-tau181, GFAP, and NfL across the Alzheimer’s disease continuum: A cross-sectional and longitudinal study in the AIBL cohort. Alzheimers Dement 19, 1117–1134. https://doi.org/10.1002/alz.12724 (2023).
Jang, H. et al. Gait ignition failure in JNPL3 human tau-mutant mice. Exp Neurobiol 28, 404–413. https://doi.org/10.5607/en.2019.28.3.404 (2019).
Nilsson, M. H. et al. The effects of tau, amyloid, and white matter lesions on mobility, dual tasking, and balance in older people. J Gerontol A Biol Sci Med Sci 76, 683–691. https://doi.org/10.1093/gerona/glaa143 (2021).
Ahman, H. B. et al. Dual-task performance and neurodegeneration: Correlations between timed up-and-go dual-task test outcomes and Alzheimer’s disease cerebrospinal fluid biomarkers. J Alzheimers Dis 71, S75–S83. https://doi.org/10.3233/JAD-181265 (2019).
Tangen, G. G. et al. Mobility and associations with levels of cerebrospinal fluid amyloid beta and tau in a memory clinic cohort. Front Aging Neurosci 15, 1101306. https://doi.org/10.3389/fnagi.2023.1101306 (2023).
Shaaban, C. E. et al. Brain health correlates of mobility-related confidence. Exp Gerontol 163, 111776. https://doi.org/10.1016/j.exger.2022.111776 (2022).
Katzman, R. et al. A Chinese version of the Mini-Mental State Examination; impact of illiteracy in a Shanghai dementia survey. J Clin Epidemiol 41, 971–978. https://doi.org/10.1016/0895-4356(88)90034-0 (1988).
Lu, J. et al. Montreal cognitive assessment in detecting cognitive impairment in Chinese elderly individuals: a population-based study. J Geriatr Psychiatry Neurol 24, 184–190. https://doi.org/10.1177/0891988711422528 (2011).
Rissin, D. M. et al. Single-molecule enzyme-linked immunosorbent assay detects serum proteins at subfemtomolar concentrations. Nat Biotechnol 28, 595–599. https://doi.org/10.1038/nbt.1641 (2010).
Xiao, Z. et al. Plasma Abeta42/Abeta40 and p-tau181 predict long-term clinical progression in a cohort with amnestic mild cognitive impairment. Clin Chem 68, 1552–1563. https://doi.org/10.1093/clinchem/hvac149 (2022).
Stevenson-Hoare, J. et al. Plasma biomarkers and genetics in the diagnosis and prediction of Alzheimer’s disease. Brain 146, 690–699. https://doi.org/10.1093/brain/awac128 (2023).
Sperling, R. A. et al. Association of factors with elevated amyloid burden in clinically normal older individuals. JAMA Neurol 77, 735–745. https://doi.org/10.1001/jamaneurol.2020.0387 (2020).
Morris, J. C. et al. Cerebral amyloid deposition and diffuse plaques in “normal” aging: Evidence for presymptomatic and very mild Alzheimer’s disease. Neurology 46, 707–719. https://doi.org/10.1212/wnl.46.3.707 (1996).
Jessen, F. et al. Subjective cognitive decline and stage 2 of Alzheimer disease in patients from memory centers. Alzheimers Dement 19, 487–497. https://doi.org/10.1002/alz.12674 (2023).
Jack, C. R. Jr. et al. Associations of Amyloid, Tau, and neurodegeneration biomarker profiles with rates of memory decline among individuals without dementia. JAMA 321, 2316–2325. https://doi.org/10.1001/jama.2019.7437 (2019).
Sperling, R. A. et al. Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7, 280–292. https://doi.org/10.1016/j.jalz.2011.03.003 (2011).
Smirnov, D. S. et al. Plasma biomarkers for Alzheimer’s disease in relation to neuropathology and cognitive change. Acta Neuropathol 143, 487–503. https://doi.org/10.1007/s00401-022-02408-5 (2022).
Koychev, I. et al. Abeta42/Abeta40 and Abeta42/Abeta38 Ratios are associated with measures of gait variability and activities of daily living in mild Alzheimer’s disease: A pilot study. J Alzheimers Dis 65, 1377–1383. https://doi.org/10.3233/JAD-180622 (2018).
He, L. et al. Plasma Abeta and neurofilament light chain are associated with cognitive and physical function decline in non-dementia older adults. Alzheimers Res Ther 12, 128. https://doi.org/10.1186/s13195-020-00697-0 (2020).
Skillback, T. et al. Cerebrospinal fluid tau and amyloid-beta1-42 in patients with dementia. Brain 138, 2716–2731. https://doi.org/10.1093/brain/awv181 (2015).
O’Connor, A. et al. Plasma phospho-tau181 in presymptomatic and symptomatic familial Alzheimer’s disease: a longitudinal cohort study. Mol Psychiatry 26, 5967–5976. https://doi.org/10.1038/s41380-020-0838-x (2021).
Buracchio, T., Dodge, H. H., Howieson, D., Wasserman, D. & Kaye, J. The trajectory of gait speed preceding mild cognitive impairment. Arch Neurol 67, 980–986. https://doi.org/10.1001/archneurol.2010.159 (2010).
Yan, D. et al. Tau hyperphosphorylation and P-CREB reduction are involved in acrylamide-induced spatial memory impairment: Suppression by curcumin. Brain Behav Immun 71, 66–80. https://doi.org/10.1016/j.bbi.2018.04.014 (2018).
Lindh-Rengifo, M. et al. Effects of brain pathologies on spatiotemporal gait parameters in patients with mild cognitive impairment. J Alzheimers Dis 96, 161–171. https://doi.org/10.3233/JAD-221303 (2023).
Hausdorff, J. M., Balash, J. & Giladi, N. Effects of cognitive challenge on gait variability in patients with Parkinson’s disease. J Geriatr Psychiatry Neurol 16, 53–58. https://doi.org/10.1177/0891988702250580 (2003).
Montero-Odasso, M., Verghese, J., Beauchet, O. & Hausdorff, J. M. Gait and cognition: A complementary approach to understanding brain function and the risk of falling. J Am Geriatr Soc 60, 2127–2136. https://doi.org/10.1111/j.1532-5415.2012.04209.x (2012).
Allali, G., Garibotto, V. & Assal, F. Parkinsonism differentiates idiopathic normal pressure hydrocephalus from its mimics. J Alzheimers Dis 54, 123–127. https://doi.org/10.3233/JAD-160428 (2016).
Muurling, M. et al. Gait disturbances are associated with increased cognitive impairment and cerebrospinal fluid tau levels in a memory clinic cohort. J Alzheimers Dis 76, 1061–1070. https://doi.org/10.3233/JAD-200225 (2020).
Verghese, J., Wang, C., Lipton, R. B. & Holtzer, R. Motoric cognitive risk syndrome and the risk of dementia. J Gerontol A Biol Sci Med Sci 68, 412–418. https://doi.org/10.1093/gerona/gls191 (2013).
Tuena, C. et al. Prognostic relevance of gait-related cognitive functions for dementia conversion in amnestic mild cognitive impairment. BMC Geriatr 23, 462. https://doi.org/10.1186/s12877-023-04175-8 (2023).
Li, H. et al. The Bidirectional Association Between Cognitive Function and Gait Speed in Chinese Older Adults: Longitudinal Observational Study. JMIR Public Health Surveill 9, e44274. https://doi.org/10.2196/44274 (2023).
Lin, J. et al. A detection model of cognitive impairment via the integrated gait and eye movement analysis from a large Chinese community cohort. Alzheimers Dement 20, 1089–1101. https://doi.org/10.1002/alz.13517 (2024).
Braak, H., Alafuzoff, I., Arzberger, T., Kretzschmar, H. & Del Tredici, K. Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112, 389–404. https://doi.org/10.1007/s00401-006-0127-z (2006).
Gomez, G. T. et al. The association of motoric cognitive risk with incident dementia and neuroimaging characteristics: The atherosclerosis risk in communities study. Alzheimers Dement 18, 434–444. https://doi.org/10.1002/alz.12412 (2022).
Zhao, H., Wei, W., Xie, H. & Huang, Y. Motoric cognitive risk syndrome among Chinese older adults with white matter lesions: A cross-sectional observational study. J Alzheimers Dis 91, 925–931. https://doi.org/10.3233/JAD-220712 (2023).
Larkin, D. Movement laterality and its relationship to hemispheric specialization. Am J Occup Ther 43, 308–312. https://doi.org/10.5014/ajot.43.5.308 (1989).
Geschwind, N. The apraxias: Neural mechanisms of disorders of learned movement. Am Sci 63, 188–195 (1975).
Solodkin, A., Hlustik, P., Noll, D. C. & Small, S. L. Lateralization of motor circuits and handedness during finger movements. Eur J Neurol 8, 425–434. https://doi.org/10.1046/j.1468-1331.2001.00242.x (2001).
Kapreli, E. et al. Lateralization of brain activity during lower limb joints movement. An fMRI study. Neuroimage 32, 1709–1721. https://doi.org/10.1016/j.neuroimage.2006.05.043 (2006).
Wu, T., Hou, Y., Hallett, M., Zhang, J. & Chan, P. Lateralization of brain activity pattern during unilateral movement in Parkinson’s disease. Hum Brain Mapp 36, 1878–1891. https://doi.org/10.1002/hbm.22743 (2015).
Hennessee, J. P. et al. Relationship of prefrontal brain lateralization to optimal cognitive function differs with age. Neuroimage 264, 119736. https://doi.org/10.1016/j.neuroimage.2022.119736 (2022).
Acknowledgements
We would like to thank Department of Neurology of Drum Tower Hospital for the technical support.
Funding
This research was supported by the National Natural Science Foundation of China (82130036, 82471454), the STI2030-Major Projects (2022ZD0211800), Jiangsu Province Key Medical Discipline (ZDXK202216).
Author information
Authors and Affiliations
Contributions
Conceptualization, Chenglu Mao and Yuting Mo; methodology, Chenglu Mao; validation, Chenglu Mao; formal analysis, Yuting Mo; investigation, Yuting Mo; resources, Yun Xu; data curation, Chenglu Mao, Jialiu Jiang and Shuang Fang, Zhihong Ke, Zheqi Hu; writing-original draft preparation, Chenglu Mao and Yuting Mo; writing—review and editing, Chenglu Mao, Hui Zhao and Yun Xu; visualization, Chenglu Mao; supervision, Yuting Mo, Hui Zhao and Yun Xu; project administration, Hui Zhao and Yun Xu; funding acquisition, Yun Xu and Hui Zhao. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study have been approved by the Ethics Committee of the Nanjing Drum Tower Hospital (Ethics number: 2022-472-02) and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Written informed consent has been obtained.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, C., Mo, Y., Jiang, J. et al. Association between high plasma p-tau181 level and gait changes in patients with mild cognitive impairment. Sci Rep 15, 14679 (2025). https://doi.org/10.1038/s41598-025-94472-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94472-6