Abstract
In this project, we introduced new heterogeneous nanocatalysts featuring amine branches and decorated with inexpensive copper metal (Fe3O4@SiO2@M-D-Cu and Fe3O4@SiO2@M-D). These nanocatalysts were characterized using several analytical techniques, including SEM, FT-IR, EDX, XRD, TGA, and ICP-OES. We investigated their catalytic activity in traditional pot reactions, focusing on the three-component synthesis of xanthene and chromene compounds. The properties and future potential of these nanocatalysts include high yield, recyclability, ease of operation, and the use of environmentally friendly solvents. Additionally, the catalysts can be easily removed from the reaction solution using an external magnet and reused up to eight times without a decrease in catalytic activity.
Similar content being viewed by others
Introduction
Nowadays, green chemistry with its 12 principles has grown a lot in the pharmaceutical and chemical industries. The majority of catalytic reaction solvents are toxic, corrosive and inflammable, therefore, they creates pollution for humans and the environment. Hence, the green approach to the removal of hazardous and toxic materials is highly developed. According to the principles of green chemistry, as much as possible, the process of converting the reactant to the product should be done with excellent green solvents such as water and the minimum required reaction time. Among the important approaches of green chemistry is the use of catalysts and decreasing of by-products in chemical processes1,2,3,4,5.
Catalysts are among the most effective techniques for environmentally friendly chemical transformations and are usually performed under mild conditions .In catalytic processes, activity and high selectivity are considered two important issues for carrying out a reaction6. On the other hand, separating and reusing the catalyst is a challenging issue. Despite the very desirable activity and selectivity of homogeneous catalysts, the difficulty of separation and reduction of their catalytic performance during the reaction time has caused these catalysts to be rarely used in industry. In order to avoid these problems, the construction of heterogeneous catalysts was given much attention owing to their thermal and mechanical stability and high structural diversity. One of the most attractive properties of magnetically synthesized nanoparticles is their separation method. In heterogeneous catalytic systems, a smoothing or centrifugation step is required. However, due to their paramagnetic nature, catalysts fixed on magnetic nanoparticles can be readily isolated from the reaction medium by using a simple external magnet, and there is no need for smoothing steps7,8,9.
Recently, magnetic nanoparticles (MNPs) have gained widespread application in a range of fields, because of their unique characteristics. Hence, the synthesis of them has a special position10 .Iron oxide NPs, have notable advantages such as high surface area, non-toxicity, proper thermal stability, high reactivity, convenience, cost-effectiveness, and good biocompatibility. Therefore, magnetic nanoparticles have attracted much attention as a suitable candidate for catalyst support11. Modifying the surface with coating materials is one of the ways to solve this problem. Today, among the various coatings the use of silica is developed owing to its easy connection with ligands and diverse linkers7,11,12,13,14,15,16,17.
Over the past years, copper has been introduced as an effective transition metal in catalytic systems. This metal is inexpensive, abundant, and less toxic than the other transition metals. Also, copper-based catalysts can enhance catalytic activity. Another advantage of using copper catalysts is that they decrease the costs of organic reactions. Hence, many efforts have been made to replace palladium with cheap metals like copper18,19,20.
Heterocyclic compounds because of their various applications in agriculture and pharmaceutical or biological activities are an important class of organic compounds. In this respect, multicomponent reactions (MCRs) in organic synthesis are an important and effective tool to access heterocyclic compounds. Multicomponent reactions (MCRs) allow the simultaneous integration of multistep synthesis of organic compounds into a single reaction using a one-pot method21. This significantly reduces the consumption of solvents and reagents and saves overall reaction time. Additionally, they have provided better yield than conventional stepwise synthesis methods. By forming and breaking multiple carbon–carbon and carbon-heteroatom bonds in one step, MCRs have emerged as a highly efficient method for making complex and diverse organic molecules. The goal of green chemistry is to design effective and sustainable chemical processes to reduce environmental impacts. These reactions by reducing waste generation through atom economy, align with the principles of green chemistry22,23,24,25,26,27,28,29.
4H-chromene and its derivatives form an important category of O-heterocyclic compounds. Moreover, the synthesis and development of these compounds, due to their wide utilization in pharmaceuticals and varied biological activities such as anticancer, antitumor, anticoagulant, anti-inflammatory, antiallergenic, anti-HIV, anti-rheumatic, antihypertensive, and antioxidant properties, have received attention throughout the globe30,31,32,33,34.
Xanthenes are another type of heterocyclic compound. This class of heterocyclic compounds due to the presence of oxygen in their structure, has a diverse collection of beneficial properties, such as antimicrobial, antiviral, antibacterial, and antiproliferative. On the other hand, playing a basic and crucial role in laser technologies, fluorescent sensors and also preparation of dyes. The production of xanthenes and their derivatives is accomplished via multicomponent reactions conducted under catalytic conditions14,35,36.
Based on our earlier research into catalyst synthesis and their applications, herein we presented a new scheme aimed at synthesizing a recyclable magnetic nanocatalyst with branches containing amine groups and then doped with inexpensive Cu metal. To demonstrate its capabilities, we utilized the nanocatalyst mentioned above in the preparation of 4H-chromene and xanthene derivatives.
Experimental
The materials used in this work were purchased from Merck or Sigma, as detailed in the experimental section. These materials are common in the field of chemistry, and their characteristics are described in the text and on their respective sites. Philips PW-1830 was used to obtain the X-ray difraction (XRD) pattern. Melting points were determined using an electro thermal 9100 apparatus. The FT-IR spectra were detected using a Shimadzu IR-470 spectrophotometer. VSM (Value stream mapping) model MDKB from Danesh Pajohan Kavir Co. Kashan, Iran. was used to record magnetic analysis curves. Using a Bruker DRX 400-Avance spectrometer, the results of the 1H and 13C spectra of the products were obtained. A SEM VEGA3 was used to record the SEM images. The EDS analysis was performed using MAP and LINE SCAN techniques. The TGA STA6000 was used to record the Termogravimetric analysis (TGA).
Preparation of Fe3O4@SiO2@Pr-Cl
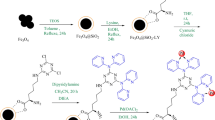
The synthetic steps of Fe3O4@SiO2 were performed as described in previously published literature37. 1 g of Fe3O4@SiO2 mixed in 30 ml of dry toluene was exposed to ultrasonic waves. The reaction mixture after adding 10 mmol (2 ml) 3-chloropropyltrimethoxysilane (CPTMS) was stirred for 24 h at reflux temperature under argon atmosphere. Eventually, the material was gathered using an external magnetic field, then washed with toluene and thoroughly dried.
Preparation of Fe3O4@SiO2@M
1 g of Fe3O4@SiO2@Pr-Cl was dispersed in EtOH (40 mL). In the following step, 2 mmol (0.25 g) of melamine was added to the reaction mixture and it was refluxed for 24 h. The synthesized nanoparticles were washed with EtOH/H2O and dried37.
Preparation of Fe3O4@SiO2@M-Pr
In 30 ml of dry toluene, 1 g of Fe3O4@SiO2@M was suspended, after that 1,3-dibromopropane (20 mmol, 2.2 ml) and KI (2 mmol, 0.33 g) were stirred under N2 gas at reflux temperature.
Preparation of Fe3O4@SiO2@M-D
15 mmol of diethylenetriamine and 15 mmol (2 ml) of triethylamine were added to 1 g of Fe3O4@SiO2@M-Pr nanoparticles dispersed in dry dichloromethane solvent and stirred for 18 h. EtOH solvent was used to wash the synthesized nanocatalyst after separation by the magnetic field (Fig. 1).
Preparation steps of nanomagntic catalyst.
Preparation of Fe3O4@SiO2@M-D-Cu
Fe3O4@SiO2@M-D nanocatalyst (1 g) was dispersed in EtOH solvent. Then 2 mmol (0.3 g) Cu(OAc)2 was added and stirred at reflux temperature for 24 h. The magnetic field was used to separate the synthesized nanocatalyst, which was then washed with EtOH solvent (Fig. 1).
General procedure for the synthesis of chromene derivatives
1 mmol of different OH-acids, 1 mmol of aromatic aldehydes, and 1 mmol of malononitrile were stirred in the presence of 0.03 g of nanocatalyst in 5 mL of EtOH- H2O at 50 °C. At the end of the reaction, the catalyst was separated by magnetic field. The resulting precipitate was recrystallized in EtOH (Fig. 2).
General procedure for the preparation of chromene derivatives.
General procedure for the Synthesis of xanthene derivatives
1 mmol of 2-naphthol, 1 mmol of aryl aldehyde and 1 mmol of dimedone were stirred in the presence of 0.03 g of nanocatalyst in 5 mL of EtOH at 80 °C. The Fe3O4@SiO2@M-D-Cu was extracted from the reaction mixture with the aid of an external magnetic field and the remaining precipitate was recrystallized in EtOH (Fig. 3) (Spectral data for products are reported in supplementary data).
General procedure for the preparation of xanthene derivatives.
Results and discussion
A new design aimed at synthesizing a recyclable magnetic nanocatalyst with branches containing amine groups and then doped with inexpensive copper metal has been presented. Initially involved the synthesis of magnetic nanoparticles using the co-precipitation method. Next, a silica coating was applied to protect the nanoparticles and create a surface that can be modified. This coating also gave the nanoparticles a spherical morphology. Then, the synthetic nanocatalysts were used in appropriate conditions to perform the reactions of chromene and xanthene derivatives.
FT-IR spectrum to affirm the synthesis of Fe3O4@SiO2@Pr-Cl, Fe3O4@SiO2@M, Fe3O4@SiO2@M-Pr, Fe3O4@SiO2@M-D and Fe3O4@SiO2@M-D-Cu are shown in Fig. 4. The Fe–O bond vibration was characterized by a strong peak at 596 cm−1. Moreover, the sharp peak at 1101 cm−1 confirms the existence of Si–O groups. The peaks at 2852 and 2922 cm−1 correspond to the (C–H sp3) stretching vibrations. The peak at 1652 cm⁻1 is related to the stretching vibration mode of the C=N bond. In addition to the mentioned peaks for Fe3O4@SiO2@M, the peaks at 3332–3468 cm−1 are ascribed with NH and NH2 groups, which shows the presence of a melamine ring in the nanocatalyst. After adding 1,3-dibromopropane to the previous layer, an increase in the height of the peak at 2855–2926 cm−1 was observed, which is related to the stretching vibration of alkyl halides. The peaks at 3421–3464 cm−1 correspond to the N–H stretching vibrations of amino groups, which confirms that diethylenetriamine is well attached to the previous layer. As expected in the spectra of the final product Fe3O4@SiO2@M-D-Cu, after loading Cu on the nanocatalyst the change in peaks from 3421 to 3410 cm−1 and 3464 to 3437 cm−1 was observed.
FT-IR spectra of (A) Fe3O4@SiO2@Pr-Cl, (B) melamine, (C) Fe3O4@SiO2@M, (D) Fe3O4@SiO2@M-Pr, (E) Fe3O4@SiO2@M-D and (F) Fe3O4@SiO2@M-D-Cu.
The thermal stability of the Fe3O4@SiO2@M-D-Cu nanocatalyst was investigated through thermal gravimetric analysis (Fig. 5). The weight loss of 3% up to 150 °C is due to the removal of solvents that are physically adsorbed on the surface. The 5% weight loss observed between temperatures of 150 °C and 800 °C is likely caused by the evaporation of both free water and bound water in the SiO2 layer (Fe3O4@SiO2). A weight loss of 20% is observed in the Fe3O4@SiO2@M diagram between temperatures of 250 and 300 °C. This weight loss can be attributed to the decomposition of melamine and alkene groups that are present in the nanocatalyst substrate. The catalyst surface in the Fe3O4@SiO2@M-D-Cu diagram experienced a 30% weight loss, which can be attributed to the separation of organic ligands., including diethylenetriamine and other organic components.
TGA result of Fe3O4@SiO2, Fe3O4@SiO2@M, Fe3O4@SiO2@M-D-Cu.
The XRD analysis was used to investigate the crystallinity of the Fe3O4@SiO2@M-D-Cu synthesis. Compared with Fe3O4, the synthesized catalyst exhibits similar peaks, indicating that they are well-crystallized and remain in their crystalline phase throughout the synthesis process. Specifically, the peaks observed at 2θ = 30.5, 36.1, 43.7, 53.4, 57.5, and 62.8 can be assigned to the (220), (311), (400), (422), (511), and (440) reflections, respectively (Fig. 6). As a result of XRD pattern analysis and Debye–Scherrer equation, nanoparticles have a crystallite size of about 50 nm, which is in line with SEM measurement.
Magnetization curves of Fe3O4, Fe3O4@SiO2@M-D, and Fe3O4@SiO2@M-D-Cu.
EDX analysis of Fe3O4@SiO2@M-D-Cu clearly shows the presence of related support atoms including C, Si, Fe, N, O, and Cu which proves the successful performance of modification with organic ligands and placement of Cu metal (Fig. 7). Elemental mapping analysis distribution reveals that the elements on the surface of the Fe3O4@SiO2@M-D-Cu are uniformly distributed (Fig. 8).
EDX analysis of Fe3O4@SiO2@M-D-Cu.
Elemental mapping analysis distribution of nanocatalyst.
VSM analysis was used to measure the magnetic behavior of Fe3O4@SiO2@M-D-Cu at ambient temperature. synthesized Fe3O4 nanoparticles have magnetic properties of 48.40 emu/g, while Fe3O4@SiO2@M-D-Cu has a saturation magnetization of 22.5 emu/g, which indicates that during the modification process, the saturation magnetization decreased which is due to the formation of a coating on the surface of the possible magnetic particles. Which can affect their magnetic ability. However, the catalyst retained its magnetic behavior (Fig. 9).
VSM analysis of Fe3O4 and Fe3O4@SiO2@M-D-Cu.
The morphology, particle size, and state of aggregation of the Fe3O4@SiO2@M-D-Cu nanoparticles were investigated using FE-SEM images (Fig. 10). By immobilizing the ligands on the Fe3O4@SiO2 and placing a layer of silica on top, nanoparticles were obtained. They had an average particle size of 48 nm and a spherical structure.
FE-SEM images of Fe3O4@SiO2@M-D-Cu.
Several factors can influence the synthesis of chromene derivatives, including the type of temperature, catalyst amount, and solvent used. The reaction between dimedone, malononitrile, and 4-chlorobenzaldehyde was chosen as a model reaction to optimize these parameters. At first, the model reaction was carried out to find a acceptable solvent in the presence of solvents such as H2O, MeOH, n-Hexane, Toluene, EtOH-H2O, EtOH, CH3CN, and THF (Table 1, entries 1–8). According to the yield obtained, the best solvent was an equal mixture of EtOH- H2O. In general, the use of polar protic solvents is more advantageous than the use of polar aprotic solvents when carrying out reactions for the synthesis of chromium derivatives, probably because polar protic solvents provide better stabilization of reactants or intermediates and participate in hydrogen bonding in these reactions. Based on the results, among different temperature values (25–70 °C), the reaction efficiency at 50 and 70 °C are almost the same, so 50 °C was chosen as the optimal temperature (Table 1, entries 8–10). Finally, based on the yields obtained, the most effective catalyst was 3 mg (Table 1, entries 10).
In order to demonstrate the effectiveness of this nanocatalyst, we conducted reactions between various aldehydes, OH-acids, and malononitrile under optimized conditions. The outcomes can be found in Table 2. The results show that aldehydes with substituents that either donate or withdraw electrons produced related products with satisfactory yields.
Figure 11 illustrates a plausible mechanism for synthesizing chromene derivatives. Malononitrile is initially converted into cyano olefin A through a Knoevenagel condensation with an aldehyde in the presence of a nanocatalyst. Next, the enolate of B is added to A via a Michael addition, resulting in the formation of intermediate C. Finally, the target product is obtained through a series of steps, which include the removal of acidic hydrogen by the catalyst, intramolecular cyclization, and tautomerization44.
A plausible mechanism for synthesizing chromene derivatives.
To show the greater effectiveness of the prepared catalyst, in another part, the synthetic process of xanthene compounds was evaluated. To optimize the reaction conditions, including the solvent, catalyst amount, and temperature, benzaldehyde has been chosen as the model compound for the reactions (using 1 mmol of 4-chlorobenzaldehyde and 2 mmol of dimedone). The findings will be presented in Table 3. According to Table 3, EtOH (Among the solvents CH2Cl2, CH3CN, MeOH, DMF, Toluene, n-Hexane, and EtOH) was determined to be an appropriated solvent in the presence of the catalyst (Table 3, entries 1–8). Also, the reaction of the model was studied by examining different temperature values (involving 25, 50, and 80 °C). The reaction was conducted at 80 °C with the best efficiency compared to other temperatures (Table 3, entries 8–10). Finally, in the presence of different amounts of catalyst, the best amount was reported to be 30 mg, and further increase did not affect the yield of the product.
Afterward, we analyzed the reaction’s scope by utilizing different aromatic aldehydes, dimedone, and 2-naphthol. The outcomes of this examination are presented in Table 4. Unlike electron-donating groups, with the placement of electron-withdrawing groups, efficiency increases and reaction time decreases.
The synthesis of xanthene derivatives occurs through a two-step process. First, the catalyst activates the carbonyl group, making aldehydes more susceptible to nucleophilic attack by dimedone, leading to the formation of intermediate (I). Next, another dimedone molecule is added via a Michael reaction, yielding intermediate (II). The desired product (Fig. 12) is obtained by eliminating H2O and undergoing intramolecular cyclization6.
A plausible mechanism for synthesizing xanthene derivatives.
In the final part, the efficiency of Fe3O4@SiO2@M-D and Fe3O4@SiO2@M-D-Cu were compared with other previously reported catalysts to evaluate its activity relative to others. It is evident from Table 5 that the current system is more efficient, simpler, and yields higher results for synthesizing xanthene and chromene derivatives compared to the other methods.
Reusability of catalysts is a significant challenge for chemists involved in catalyst synthesis. Therefore, it is essential to conduct research on this important issue. In this experiment, the catalysts for reuse were separated, washed with EtOH, and then dried. Research has demonstrated that this catalyst can perform at least 8 steps without remarkable impact on its performance (Fig. 13). Furthermore, as part of the additional study, FT-IR spectra, FE-SEM images, and XRD patterns were examined after 8 recycling cycles (Fig. 14). These analyses revealed no significant differences. In addition, copper nanoparticles were examined for possible leaching from the Fe3O4@SiO2@M-D-Cu during the model reactions. The ICP-OES analysis revealed that after 8 recycling steps, the catalyst’s Cu leaching amount is only 3.8% by weight. This value is quite insignificant when compared to the initial loading of Cu in the fresh catalyst, which is 4.2% by weight.
Diagram of catalyst reusability for model reactions.
(A) IR spectrum, (B) XRD and (C) FE-SEM patterns of Fe3O4@SiO2@M-D-Cu after 8 cycles of reuse.
Conclusion
We have successfully synthesized a novel heterogeneous catalytic system with amine branches, designated as Fe3O4@SiO2@M-D. Subsequently, we prepared the nanocatalyst Fe3O4@SiO2@M-D-Cu by decorating cost-effective copper metal ions onto the surface of magnetic nanoparticles. This synthesis was confirmed through various analyses, including SEM, FT-IR, TGA, EDX, XRD, and ICP-OES. Under optimal reaction conditions, we synthesized a wide range of chromene derivatives (yielding 85–98% at 50 °C) and xanthene derivatives (yielding 85–98% at 80 °C) using the prepared nanocatalysts in the presence of ethanol and ethanol–water as green solvents. This high yield can be attributed to the catalyst’s excellent dispersion in water (PI = 0.379), the significant solubility of the reactants in ethanol, and the stabilization of ions. These nanocatalysts’ properties and future applications include high stability, high activity, reusability, and easy recovery. Their nature eliminates the need for filtration and centrifugation of the reaction mixture and enables separation of the catalyst from the reaction medium using a magnetic field. Ultimately, we conclude that the catalyst can be recycled and reused for at least eight consecutive cycles while maintaining good stability for this class of reactions.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Kalantari, F., Rezayati, S., Moghadam, M. M. & Ramazani, A. A novel Cu (II) nanoparticles anchored on cyanuric chloride-attached magnetic nanocatalyst: A highly stable, recyclable and efficient nanocatalyst for the synthesis of chromene derivatives. Inorg. Chem. Commun. 164, 112371 (2024).
Patil, S., Tandon, R. & Tandon, N. A current research on silica coated ferrite nanoparticle and their application. Curr. Res. Green Suatain. Chem. 4, 100063 (2021).
Patil, S., Tandon, R. & Tandon, N. Synthesis and characterization of Fe3O4@SiO2@K10 NPs applicable for N-ter-butyloxycarbonylation using solvent-free conditions. J. Phys. Conf. Ser. 2267, 012107 (2021).
Tandon, R., Patil, S., Tandon, N. & Kumar, P. Magnetically recyclable silica-coated magnetite-molybdate nanocatalyst and its applications in N-formylation reactions under solvent-free conditions. Lett. Org. Chem. 19, 616–626 (2022).
Pise, A., Patil, S. M. & Ingale, A. P. Malic acid as a green catalyst for the N-boc protection under solvent-free condition. Lett. Org. Chem. 21, 620–629 (2024).
Naderi, S., Sandaroos, R., Peiman, S. & Maleki, B. Synthesis and characterization of a novel crowned Schiff Base ligand linked to ionic liquid and application of its Mn(III) complex in the epoxidation of olefins. Chem. Methodol. 7, 392–404. https://doi.org/10.22034/CHEMM.2023.385248.1651 (2023).
Peiman, S., Maleki, B. & Ghani, M. Dendrimer templated ionic liquid nanomagnetic for efficient coupling reactions. Sci. Rep. 14, 25082 (2024).
Tandon, R., Tandon, N. & Patil, S. M. Overview on magnetically recyclable ferrite nanoparticles: Synthesis and their applications in coupling and multicomponent reactions. RSC Adv. 11, 29333–29353 (2021).
Tandon, N., Patil, S. M., Tandon, R. & Kumar, P. Magnetically recyclable silica-coated ferrite magnetite-K 10 montmorillonite nanocatalyst and its applications in O, N, and S-acylation reaction under solvent-free conditions. RSC Adv. 11, 21291–21300 (2021).
Patil, S. M. Magnetically recoverable molybdate supported silica decorated ferrite nanocat (Fe–Si–Mo) and their utilization for reductive amination of carbonyl compound under benign condition. J. Indian Chem. Soc. 101, 101261 (2024).
Patil, S. M. Novel silica-coated magnetic nanoparticles and their synthetic applications. Iran. J. Catal. 13 (2023).
Ebrahimzadeh, P., Maleki, B., Ghani, M. & Peiman, S. High-performance Fe₃O₄@SiO₂@Mel@DABCO Catalyst for Synthesis of Chromene Derivatives and Solid Phase Microextraction of Fipronil and Prometryn in Food Samples Followed by HPLC-UV Determination. Chem. Methodol. 8, 833–855. https://doi.org/10.48309/chemm.2024.480851.1832 (2024).
Yao, M.-J., Guan, Z. & He, Y.-H. Simple, catalyst-free, one-pot procedure for the synthesis of 2-amino-3-cyano-1, 4, 5, 6-tetrahydropyrano [3, 2-c] quinolin-5-one derivatives. Synth. Commun. 43, 2073–2078 (2013).
Peiman, S. & Maleki, B. Fe3O4@ SiO2@ NTMPThio-Cu: A sustainable and eco-friendly approach for the synthesis of heterocycle derivatives using a novel dendrimer template nanocatalyst. Sci. Rep. 14, 17401 (2024).
Patil, S. M. R. & Bedre, A. V. Recent progress in Fe3O4 nanoparticles and their green applications in organic transformations. Iran. J. Catal. 13, 235–270 (2023).
Patil, S. M., Tandon, R. & Tandon, N. Magnetically recoverable silica-decorated ferromagnetic-nanoceria nanocatalysts and their Use with O-and N-butyloxycarbonylation reaction via solvent-free condition. ACS Omega 7, 24190–24201 (2022).
Patil, S. M., Ingale, A. P., Pise, A. S. & Bhondave, R. S. Novel cobalt-supported silica-coated ferrite nanoparticles applicable for acylation of amine, phenol, and thiols derivatives under solvent-free condition. ChemistrySelect 7, e202201590 (2022).
Peiman, S., Baharfar, R. & Hosseinzadeh, R. CuI NPs immobilized on a ternary hybrid system of magnetic nanosilica, PAMAM dendrimer and trypsin, as an efficient catalyst for A3-coupling reaction. Res. Chem. Intermed. 48, 1365–1382 (2022).
El-Ghamry, H. A., Alharbi, B. K., Takroni, K. M. & Khedr, A. M. A series of nanosized Cu (II) complexes based on sulfonamide azo dye ligands: An insight into complexes molecular structures, antimicrobial, antitumor and catalytic performance for oxidative dimerization of 2-aminophenol. Appl. Organomet. Chem. 37, e6978 (2023).
Beiraghi Toosi, R. & Hakimi, M. Synthesis nickel (II) and copper (II) complexes with a new oxazoline ligand, bis 2-(2-phenyl-2-(pyridin-2-yl) oxazolin-3-yl)ethanol. Chem. Methodol. 8, 745–752. https://doi.org/10.48309/chemm.2024.479911.1829 (2024).
Jopale, M. K. et al. Synthesis and characterisation of mixed oxides of Co–Ni catalyst and its application in microwave mediated synthesis of Benzo [b] Pyrans. J. Indian Chem. Soc. 101, 101265 (2024).
Mohlala, R. L. & Coyanis, E. M. The vital use of isocyanide-based multicomponent reactions (MCR) in chemical synthesis. Phys. Sci. Rev. 9, 995–1032 (2024).
Peiman, S., Maleki, B. & Ghani, M. Fe3O4@gC3N4@ Thiamine: A novel heterogeneous catalyst for the synthesis of heterocyclic compounds and microextraction of tebuconazole in food samples. Sci. Rep. 14, 21488 (2024).
Yazdan Kushkoo, F., Khabazzadeh, H. & Khaleghi, M. Synthesis of a new series of 1,3,4-oxadizole-based azo derivatives and In Vitro evaluation of their antibacterial activities. Chem. Methodol. 8, 645–661. https://doi.org/10.48309/chemm.2024.474180.1819 (2024).
Ramezanpour-Touchahi, M., Mazloumi, M., Taherpour Nahzomi, H., Shirini, F. & Tajik, H. Introduction of a new ionic liquid based on tropine for the acceleration of the synthesis of several hetero-aromatic compounds and investigation of its physicochemical property and antibacterial activity. Chem. Methodol. 8, 401–438. https://doi.org/10.48309/chemm.2024.452735.1789 (2024).
Ingale, A. P., Patil, S. M. & Shinde, S. V. Catalyst-free, efficient and one pot protocol for synthesis of nitriles from aldehydes using glycerol as green solvent. Tetrahedron Lett. 58, 4845–4848 (2017).
Pise, A. S., Ingale, A. P. & Patil, S. M. An efficient synthesis of 1, 3-oxazine derivatives catalyzed under ceric ammonium nitrate in an aqueous medium at ambient temperature. Polycycl. Aromat. Compd. https://doi.org/10.1080/10406638.2023.2259569 (2023).
Patil, S. et al. One-pot protocol for the reductive amination of aldehydes using thiamine hydrochloride as a green catalyst under solvent-free condition. Synth. Commun. 53, 1545–1558 (2023).
Patil, S. M. et al. Magnetite-supported montmorillonite (K 10)(nanocat-Fe-Si-K 10): An efficient green catalyst for multicomponent synthesis of amidoalkyl naphthol. RSC Adv. 13, 17051–17061 (2023).
Danehchin, M. & Esmaeili, A. A. Synthesis of Fe3O4@SiO2@Pr-NH2@DAP as a magnetic recyclable nano-catalyst for efficient synthesis of pyranothiazolopyrimidines and 4 H-pyrans under solvent-free condition. Sci. Rep. 13, 14937 (2023).
Maleki, B. et al. A novel recyclable hydrolyzed nanomagnetic copolymer catalyst for green, and one-pot synthesis of tetrahydrobenzo [b] pyrans. Sci. Rep. 14, 30940 (2024).
Sajjadifar, S., Abakhsh, F. & Arzehgar, Z. Design and characterization of Fe₃O₄@n-pr-NH₂@Zn₃(BTC)₂ magnetic MOF: A catalyst for dihydropyrimidine and 2-amino-4H-chromene synthesis. Chem. Methodol. 8, 550–568. https://doi.org/10.48309/chemm.2024.472969.1817 (2024).
Azizi, Z. et al. Benzimidazolium dihydrogen phosphate: A novel green catalyst for the preparation of pyrano[2,3–d]pyrimidines and pyrano[3,2- c]chromenes in aqueous media. Chem. Methodol. 8, 626–644. https://doi.org/10.48309/chemm.2024.457715.1796 (2024).
Zare, A., Oraki, M., Rostami, E., Jazinizadeh, E. & Abshirini, Z. Efficacious fabrication of 2-amino-4H-cromenes using a new inorganic-organic hybrid nanomaterial. Chem. Methodol. 8, 478–491. https://doi.org/10.48309/chemm.2024.458403.1797 (2024).
Mirhasani, M., Sandaroos, R., Maleki, B. & Peiman, S. Synthesis, identification and application of a new ionic liquid based on 1-vinylimidazole, immobilized on graphene oxide nanosheets. Case Stud. Chem. Environ. Eng. 11, 101063 (2024).
Zare, A. & Mostaghar, F. Construction of a novel Mmagnetic nanomaterial, and its utility as an effectual catalyst for the fabrication of 1,8-dioxo-octahydroxanthenes. Chem. Methodol. 8, 23–36. https://doi.org/10.48309/chemm.2024.422175.1733 (2023).
Peiman, S., Maleki, B. & Ghani, M. Fe3O4@SiO2@Mel-Rh-Cu: A high-performance, green catalyst for efficient xanthene synthesis and its application for magnetic solid phase extraction of diazinon followed by its determination through HPLC-UV. Chem. Methodol. 8, 257–278. https://doi.org/10.48309/chemm.2024.442693.1767 (2024).
Boroumand, H., Alinezhad, H., Maleki, B. & Peiman, S. Triethylenetetramine-grafted magnetic graphene oxide (Fe3O4@GO-NH2) as a reusable heterogeneous catalyst for the one-pot synthesis of 2-amino-4 H-benzopyran derivatives. Polycycl. Aromat. Compd. 43, 7853–7869 (2023).
Sharghi, H., Razavi, S. F., Aberi, M., Sabzalizadeh, F. & Karbalaei-Heidari, H. R. Nanostructured coumarin-based cobalt complex as an efficient, heterogeneous and recyclable catalyst for the three-component synthesis of benzo [b] pyran and 3, 4-dihydropyrano [c] chromene derivatives. J. Iran. Chem. Soc. 18, 1641–1655 (2021).
Parthiban, D. Bi-tailed N-cationic surfactant promoted green synthesis of 2-amino-5-oxo-4-alkyl-4, 5-dihydropyrano [3, 2-c] chromene-3-carbonitrile derivatives via multicomponent reaction. J. Heterocycl. Chem. 60, 1210–1222 (2023).
Patil, U., Patil, R. C. & Patil, S. S. Biowaste-derived heterogeneous catalyst for the one-pot multicomponent synthesis of diverse and densely functionalized 2-amino-4H-chromenes. Org. Prep. Proced. Int. 53, 190–199 (2021).
Ghorbani-Vaghei, R., Rahmatpour, F., Sarmast, N., Mahmoudi, J. & Shahriari, A. DABCO as a green catalyst for the synthesis of pyranoquinoline derivatives. Can. J. Chem. 95, 601–604 (2017).
Kumar, K., Sahani, R. K., Garai, S. & Bhattacharya, S. Synthesis and structural features of indium (III) furan-2-thiocarboxylates showing efficient catalytic activity toward multicomponent reactions via Knoevenagel condensation. Dalton Trans. 52, 17499–17513 (2023).
Singh, P., Yadav, P., Mishra, A. & Awasthi, S. K. Green and mechanochemical one-pot multicomponent synthesis of bioactive 2-amino-4 H-benzo [b] pyrans via highly efficient amine-functionalized SiO2@Fe3O4 nanoparticles. ACS Omega 5, 4223–4232 (2020).
Mousavifar, S. M., Kefayati, H. & Shariati, S. Fe3O4@ Propylsilane@ Histidine [HSO4-] magnetic nanocatalysts: Synthesis, characterization and catalytic application for highly efficient synthesis of xanthene derivatives. Appl. Organomet. Chem. 32, e4242 (2018).
Merroun, Y. et al. Synthesis, characterization, and catalytic application of SnP2O7 for the highly efficient synthesis of xanthene derivatives. Poly. Aromat. Compd. 44, 4349–4363 (2024).
Merroun, Y. et al. Triple superphosphate modified by tin (II) chloride: As a reusable and efficient catalyst for the one-pot synthesis of xanthene and xanthenone derivatives under green conditions. J. Mol. Struct. 1294, 136383 (2023).
Ghassamipour, S. & Ghashghaei, R. Zirconium dodecylphosphonate promoted synthesis of xanthene derivatives by condensation reaction of aldehydes and β-naphthol or dimedone in green media. Monatsh Chem. 146, 159–163 (2015).
Maghsoodlou, M. T., Habibi-Khorassani, S. M., Shahkarami, Z., Maleki, N. & Rostamizadeh, M. An efficient synthesis of 2, 2′-arylmethylene bis (3-hydroxy-5, 5-dimethyl-2-cyclohexene-1-one) and 1, 8-dioxooctahydroxanthenes using ZnO and ZnO–acetyl chloride. Chin. Chem. Lett. 21, 686–689 (2010).
Niknam, K., Panahi, F., Saberi, D. & Mohagheghnejad, M. Silica-bonded S-sulfonic acid as recyclable catalyst for the synthesis of 1, 8-dioxo-decahydroacridines and 1,8-dioxo-octahydroxanthenes. J. Heterocycl. Chem. 47, 292–300 (2010).
Moradi, L. & Mirzaei, M. Immobilization of lewis acidic ionic liquid on perlite nanoparticle surfaces as a highly efficient solid acid catalyst for the solvent-free synthesis of xanthene derivatives. RSC Adv. 9, 19940–19948 (2019).
Oskooie, H. A., Tahershamsi, L., Heravi, M. M. & Baghernejad, B. Cellulose sulfonic acid: An efficient heterogeneous catalyst for the synthesis of 1,8-dioxo-octahydroxanthenes. J. Chem. 7, 717–720 (2010).
Amini, A., Fallah, A., Cheng, C. & Tajbakhsh, M. Natural phosphate-supported Cu (II), an efficient and recyclable catalyst for the synthesis of xanthene and 1, 4-disubstituted-1, 2, 3-triazole derivatives. RSC Adv. 8, 41536–41547 (2018).
Azath, I. A., Puthiaraj, P. & Pitchumani, K. One-pot multicomponent solvent-free synthesis of 2-amino-4 H-benzo [b] pyrans catalyzed by per-6-amino-β-cyclodextrin. ACS Sustain. Chem. Eng 1, 174–179 (2013).
Sayyahi, S., Fallah-Mehrjardi, M. & Saghanezhad, S. J. Synthesis of heterocyclic compounds by catalysts supported on nano-magnetite (Fe3O4)-an update. Mini-Rev. Org. Chem. 18, 11–26 (2021).
Hosseini Mohtasham, N. & Gholizadeh, M. Horsetail plant (Equisetum arvense) and horsetail plant ash: Application and comparison of their catalytic activities as novel and natural porous lewis acid catalysts for the one-pot green synthesis of 2-amino-4 H-chromene derivatives under solvent-free conditions. J. Iran. Chem. Soc. 17, 397–409 (2020).
Maleki, A., Jafari, A. A. & Yousefi, S. Green cellulose-based nanocomposite catalyst: Design and facile performance in aqueous synthesis of pyranopyrimidines and pyrazolopyranopyrimidines. Carbohydr. Polym. 175, 409–416 (2017).
Aghaei-Hashjin, M., Yahyazadeh, A. & Abbaspour-Gilandeh, E. Zr@IL-Fe3O4 MNPs as an efficient and green heterogeneous magnetic nanocatalyst for the one-pot three-component synthesis of highly substituted pyran derivatives under solvent-free conditions. RSC Adv. 11, 23491–23505 (2021).
Acknowledgements
The authors acknowledge the Research Council of the University of Mazandaran.
Funding
None.
Author information
Authors and Affiliations
Contributions
Writing: V.D.;S.P.; B.M.; M.Gh Conceptualization: V.D.; S.P.; B.M.; M. Gh. Data curation: V.D.; S.P.; B.M.; M.Gh. Formal analysis: V.D.; S.P.; B.M.; M.Gh. Project administration: V.D.;S.P.; B.M.; M.Gh. Methodology: V.D.; S.P.; B.M.; M.Gh. Validation: V.D.;S.P.; B.M.; M.Gh. Review and editing: V.D.;S.P.; B.M.; M.Gh. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This work does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dorostian, V., Maleki, B., Peiman, S. et al. High-performance Fe3O4@SiO2@M-D and Fe3O4@SiO2@M-D-Cu with amine branches and decorated with copper metal for one-pot synthesis of chromene and xanthene derivatives. Sci Rep 15, 10571 (2025). https://doi.org/10.1038/s41598-025-94548-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94548-3
Keywords
This article is cited by
-
One-pot synthesis of 1,8-dioxo-decahydroacridines and 1,8-dioxo- octahydroxanthene derivatives using novel biopolymer xanthan perchloric acid (XPA) catalyst
Research on Chemical Intermediates (2026)
-
Enhancement of photocatalytic and antibacterial functions through synergistic effects of Cu/CeO2 nanocomposite supported on activated carbon
Ionics (2026)
-
Cu(II)-Functionalized Magnetic Fe₃O₄@SiO₂–TCCP Catalyst for the Synthesis of Imidazo[1,2-a]pyridines in Deep Eutectic Solvents
Journal of Inorganic and Organometallic Polymers and Materials (2026)
-
Design and catalytic performance of a novel two-dimensional copper metal-organic framework for green synthesis of tetrahydrobenzo[b]pyrans
Scientific Reports (2025)
-
Deep eutectic solvent-promoted sustainable synthesis of 4H-pyrimido [2,1-b] benzothiazoles via multicomponent reactions
Research on Chemical Intermediates (2025)