Abstract
The goal of this investigation was to assess the adverse impacts of polystyrene microplastics (PS-MPs) on Labeo rohita with a comprehensive assessment of growth performance, hematological changes, and histopathological effects. Six test diets were established with different MPs levels using canola meal as the basal diet: control 0.0%, 0.5%, 1%, 1.5%, 2%, and 2.5% MPs. For 90 days, 315 fish (15 fish per tank with three replicates) were fed experimental diets at a rate of 5% of their live, twice a day. L. rohita fingerlings treated with 2.5% MPs leading to a notable decline in growth and feed consumption (P < 0.05). As PS-MPs increased, carcass, mineral and digestibility content declined. Additionally, a histology of the gut revealed significant abnormalities in intestine, including villi structure disruption and increased mucus cell proliferation, at a 2.5% PS-MPs concentration. Hematological indices such as RBCs, Hb, PLT, MCHC, and PCV decreased significantly when exposed to MPs, whereas WBCs, MCH, and MCV dramatically increased. Conclusively, this study demonstrated that the PS-MPs exert detrimental effects on growth performance, and induce histopathological and hematological changes in L. rohita.
Similar content being viewed by others
Introduction
Microplastics (MPs) may be found in large quantities in freshwater environments and can be discharged from various routes, such as urban dust, wastewater and agricultural runoff from cities. The ingestion of MPs derived from commonly used petroleum-based polymers such as polyvinyl chloride (PVC), polystyrene (PS) and polyethylene terephthalate (PET) have been extensively studied, revealing detrimental effects on the digestive system of aquatic organisms1,2.
Research has shown that MPs can harm fish and other aquatic life in multiple ways3,4. Upon consumption, MPs can cause intestinal inflammation, immune responses, and oxidative stress, ultimately leading to reduced growth, metabolism, and reproductive toxicity5,6,7,8. The gastrointestinal tract is particularly vulnerable to MP absorption, with significant quantities of MPs absorbed into the blood through the intestinal lining9.
After entering the intestines, MPs may be transported to other parts of the body via the bloodstream10. This can lead to a range of negative effects, particularly in stressful environments where organisms often experience decreased energy availability. In such environments, fish may exhibit dysfunctions in feeding, digestion, and energy metabolism. Furthermore, nutrient absorption can be directly impacted by changes in elemental composition and growth performance11,12.
The freshwater fish Rohu (Labeo rohita), known for its high price in Asia, is one of the most well-known species. Global production reached a notable milestone of almost 1.5 million tons in 201213, Rohu is among the most commercially valuable fish species, boasting high market demand and consumer acceptance. The nutritional quality of Rohu, however, is influenced by various external factors, including feed composition and environmental conditions14. The negative effects observed in the study were due to PS-MPs, not the canola meal. This shows that the canola meal did not affect the results and the harm was caused directly by the PS-MPs. The basal diet consisted of canola meal, a nutritionally balanced and commonly used feed for fish15. To ensure that the observed effects were solely attributed to PS-MPs, we verified that the canola meal itself did not influence the results. Our findings confirm that the negative effects observed in this study were directly caused by the PS-MPs, rather than the canola meal16. The current study determines the impacts of PS-MPs on L. rohita with a comprehensive assessment focusing on growth performance, histopathological effects and hematological changes.
Materials and methods
This research was executed at the GC University Faisalabad, Pakistan.
Ethical considerations
The research design was authorized by the Ethics Review Committee of the GC University Faisalabad, Faculty of life Science (Ref. No. GCUF/ERC/ 378). All experimental methods complied with relevant guidelines and regulations, ensuring the highest standards of ethical conduct.
MPs evaluation
The GCUF Department of Environmental Sciences supplied the polystyrene microplastics (PS-MPs). The mean size of these whitish, scrubby particles was 69 μm. Table 1 presents the physical and chemical characteristics of the particles.
Fish adaptability and experimental setup
The L. rohita were procured from a local hatchery and were progressively acclimated to 70-liter V-shaped tanks in the laboratory setting over a 90-day period. The fish received a 5 g/L sodium chloride treatment to control external parasites and minimize the risk of subsequent infections17. To minimize stress, water quality indicators were routinely checked, including temperature (25–28 °C), pH levels (7.5–8.5) and dissolved oxygen (DO, 6.0–7.5 mg L–1)18. After every feeding session, the tanks were thoroughly cleaned, and fresh water was added twice a day.
Feed and feed component formulations
Six isocaloric and isonitrogenous diets based on canola meal were formulated. The test diets (II–VI) were formulated with increasing concentrations of PS-MPs, ranging from 0.5 to 2.5%, while the control diet (TD-I) was prepared without any PS-MPs (0%), serving as a reference (Table 2). The feed components were powdered to pass through a 0.5 mm mesh sieve. Fish oil was added gradually to the feed components after they had been combined for five minutes. A fine dough was created by adding approximately 12% water. The dough was then processed into feed pellets using a pelleting machine. PS-MPs were added to test diets in different ratios. No animals were euthanized after completion of the study.
Growth parameters
The standard formulae were used in order to evaluate growth indices (Feed conversion ratio, feed intake, specific growth rate) by following Hussain et al.18. The growth rate of L. rohita was measured by calculating the difference between their initial weight (IW) and final weight (FW).
Chemical analysis of feed and feces
Homogenized samples of muscle, feed components, and feces were all tested using the standard methods outlined in AOAC19. Crude fat (CF) content was determined by petroleum ether extraction using the Soxtec HT2 1045 apparatus. To ascertain the moisture content of fish body, feed samples and excrement they were continually dried at 105 °C in an oven for 12 h. Moreover, the percentages of ash and crude protein (CP) were determined using an electric furnace and a micro Kjeldahl, respectively. Gross energy (GE) content was analyzed using a bomb calorimeter (model GD-ISO1716, GDY-1 A).
Calculation nutrients digestibility
The digestibility coefficient (ADC %) of several diets was assessed using the established methodology described by NRC15.
Histopathology analysis
The gut was segmented, labeled, and fixed in 10% formalin for preservation. Following fixation, the samples were embedded in paraffin wax to provide support and stability. Thin sections, measuring 5 μm in thickness, were then obtained using a microtome (Accu-Cut® SRM™, USA). These sections were subjected to hematoxylin and eosin (H&E) stain, a conventional histological staining technique, to enhance tissue morphology and facilitate microscopic analysis. Finally, a microscope was employed to capture images of various histological alterations.
Blood parameters
For hematological analysis, blood samples (1–2 mL) were collected from three fish per tank (n = 9), using sterile syringes and EDTA anticoagulant tubes. Prior to sampling, fish were anesthetized with a 50 mg/L tricane methanesulfonate solution (MS-222, Syndel, Canada), and blood was drawn from the caudal vein. Leukocytes (WBCs) and erythrocytes (RBCS) counts were performed using a hemocytometer20. Hemoglobin (Hb) concentrations and packed cell volume (PCV) were also measured. Additionally, mean corpuscular hemoglobin concentration (MCHC), MC volume (MCV) and MC hemoglobin (MCH) were calculated using conventional equations.
Calculating the concentration of minerals
To assess the minerals of the body samples, fingerlings underwent a wet digestion process. The samples were immersed in a 2:1 (v/v) boiling mixture of HClO4 and HNO3. This acid mixture, obtained from Sigma-Aldrich (USA), effectively oxidized the organic matrix, releasing the mineral constituents into solution. The resulting digest was then analyzed using atomic absorption spectrometry (AAS). This technique enabled the quantitative determination of essential mineral elements present in the body composition samples19.
Statistic analysis
The data presented are expressed as mean values of triplicate replicates (± SD). For every set of data, a one-way analysis of variance (ANOVA) was performed21. Using an ANOVA, the research determined the mean variations amongst the six test diets. The p-value of less than 0.05 was used to evaluate the statistical significance. The Co-Stat programme was used to ascertain the data analyses.
Contamination control
To minimize the risk of contamination, all equipment was cleansed with dish detergent and tap water and subsequently rinsed with ethanol and deionized water. Throughout the experiment, gloves and cotton lab coats were worn to minimize the possibility of clothing contamination from the air. Muslined cloths were used to cover aquariums to keep airborne contaminants out of the water.
Results
Growth parameters
The parameters of growth of L. rohita fish declined significantly with increasing levels of PS-MPs in their diet (Table 3). Fingerlings given a 0% PS-MPs diet exhibited the maximum FW (32.83 g) and WG (19.23 g), whereas when fed 2.5% PS-MPs showed the minimum FW (21.55 g) and WG (8.33 g). Intermediate inclusion levels (0.5–2%) showed a progressive decline in FW and WG, with a corresponding increase in FCR and decrease in SGR. At the 2.5% PS-MPs level, there was a statistically significant negative impact on growth performance, with the highest FCR (2.45%) and lowest SGR (0.54%) observed (p < 0.05).
Body composition
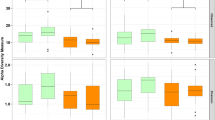
Figure 1 displays the fish’s initial and ultimate whole-body composition. The L. rohita fish fed the 0% PS-MPs diet had the greatest CP content (17.43%) and the lowest CF content (4.16%). Conversely, fish receiving a 2.5% PS-MPs diet exhibited the lowest CP (14.55%) and the highest CF (5.86%). The 0% PS-MPs diet had the highest ash content (4.91%), while the least value was observed in 2.5% PS-MPs (3.14%). Moreover, 2.5% PS-MPs revealed the highest moisture content (75.96%), significantly different from other diets (p < 0.05), with the 0% PS-MPs diet having the lowest moisture content (72.74%).
Body composition of L. rohita fish fed the canola meal supplemented polystyrene microplastics (PS-MPs): A—crude protein (CP), B—crude fat (CF), C—ash, D—moisture.
Nutrient digestibility
The nutritional composition of different test diets for L. rohita, including CP, CF, and GE, is displayed in Table 4 for feed and feces, and apparent digestibility (ADC%) is illustrated in Fig. 2. Among all the treatment groups, the 2.5% PS-MPs diet had the highest levels of CP (17.31%), CF (3.75%), and GE (2.04%) in the feces. In contrast, the 0% PS-MPs diet revealed the maximum digestibility values for CP (72.28%), CF (76.25%), and GE (68.04%), as well as the lowest nutrient values in feces.
Digestibility coefficient (ADC, %) of L. rohita fed polystyrene microplastic (PS-MPs) supplemented diets.
Histopathology
Histopathological analysis of L. rohita gut samples revealed abnormalities in gut tissue that increased with higher PS-MP exposure. These abnormalities included villi breaks, degeneration, and structural disturbances (Table 5). Variations in damage and deformation were seen in the intestinal villi of each experimental group. The most severe effects were observed at the 2.5% PS-MP level, while the control group (0% MPs) showed no anomalies (Fig. 3). In contrast to the control, exposed fingerlings with PS-MP showed histological abrasions in their guts. The abrasions exhibited a dose-and size-dependent pattern, becoming worse as PS-MP size and concentration increased.
Histopathology of intestine of L. rohita fed non-biodegradable polystyrene microplastics (PS-MPs) (Magnification: 400×). Green arrow: normal structure of villi of gut, Blue arrows: serosa (S), Yellow arrows: breakage of villi structure (BV), red arrows: increase of mucus cell (IMC) and black arrows: joining of villi structure. The highest abnormalities were seen in diet containing 2.5% PS-MPS. As the microplastic concentration increased, the severity of damage also increased, indicating a dose-dependent relationship between MP exposure and intestinal abnormalities. These outcomes indicate that prolonged exposure to MPs could lead to significant disruptions in intestinal integrity and overall health.
Hematology
The results indicated a significantly increasing trend in WBC count with a higher PS-MPs level relative to the control. The Table 6 demonstrates that L. rohita fed a 2.5% PS-MPs had the reduced PCV (28.33%), MCHC (25.79%), Hb (7.31 g/100 ml), RBC (1.24 × 106 mm–3) and highest WBC (2.87 × 103 mm–3) count. In contrast, the control diet (0% PS-MPs) resulted in the highest values for PLT (310.75), Hb (8.91 g/100 ml), PCV (32.88%), and MCHC (27.11%).
Minerals analysis
Fishes fed biodegradable canola meal based diets, the minerals concentration dramatically (p < 0.05) dropped with increasing level of PS-MPs. The maximum value of minerals was found in test diet-I, which did not contain PS-MPs (Table 7).
Discussion
The findings of research demonstrated that ingesting PS-MPs significantly (p < 0.05) reduced fish growth. Consistent results were obtained by Lai et al.22, who found that survival rates and SGR decreased sharply with increasing dietary PS-nanoplastic levels. When organisms consume MPs, their gastrointestinal tracts may get blocked which can also result in larval death and growth reduction23. Putrajab et al.24 found a significant difference in specific WG, FCR, and length with MPs at 1 mg per 0.75 g dose of feed than without MPs. According to Wen et al.25, long-term exposure to PE-MP altered the enzyme activity linked to energy consumption and digestion but had no effect on the growth and survival of juvenile Amazonian cichlids. Some evidence suggests that using MPs instead of food may hinder growth26. Upon ingestion, MPs cause physical damage to the gastrointestinal tract, leading to the production of reactive oxygen species (ROS) and activation of stress signaling pathways, including the JNK and p38 MAPK pathways. This stress response inhibits the insulin-like growth factor-1 (IGF-1) signaling pathway, which plays a crucial role in regulating growth and development. Specifically, the inhibition of IGF-1 signaling reduces the activity of the mechanistic target of rapamycin (mTOR) pathway, leading to decreased protein synthesis and, ultimately, impaired growth27.
Present findings showed that the CP, CF ash, and moisture levels in the MP diets differed significantly from those in the control. The supplementation of PS-MPs altered the nutrient profile of fish, as environmental factors and diet are known to significantly influence the nutritional composition of fish22. Diets supplemented with 8% HDPE resulted in significantly decreased ash and protein levels in fish compared to those receiving diets with 0–2% HDPE28. Our results suggested that the fish’s poor nutritional absorption and digestion may be the cause of the treatments’ lower protein and ash levels in contrast to the control. These alterations may be due to PS-MPs-induced gut damage, which reduces nutrient absorption and protein metabolism. The increased moisture content could result from inflammation and water retention (edema), while the elevated CF content might indicate a stress-induced metabolic shift towards lipid storage29.
Present findings showed that digestibility decreased as PS-MP levels increased. Physically, MPs can clog the digestive tract, leading to reduced nutritional absorption and impaired energy allocation30. Furthermore, MPs can cause genotoxicity, compromise immunological function, and alter energy metabolism31. At the microbial level, MPs can substantially alter the gut microbiota, leading to dysbiosis and impaired host health. This disruption can contribute to metabolic disorders, including impaired nutrient absorption, altered energy metabolism, and compromised immune function. Ultimately, changes in the gut microbiome’s functional potential can further impair metabolic processes, increasing disease susceptibility32.
The present investigation revealed a significant histopathological change associated with the highest PS-MP concentration. Our results align with existing research, which have reported alterations in intestinal histology following MP exposure. These changes include mucosal epithelial separation, hyperplasia, villous swelling, enterocyte shortening, and vacuolation, as well as increased infiltration of white blood cells33,34. Similar effects on intestinal tissues have been documented in other studies35,36, further corroborating our results. Notably, Pitt et al.37 demonstrated that despite a negligible rate of clearance, there was still a significant accumulation of plastic occurred in the stomach.
Major blood homeostasis abnormalities may arise from MPs entering the fish circulatory system straight via the gills. In our research, fingerlings had considerably reduced blood indices including as PCV, RBCs, Hb, PLT, and MCHC. Hematological attributes and plasma components may also function as markers for MP toxicity10. MP exposure can trigger an inflammatory response and induce tissue hypersensitivity reactions. Exposure to PS-MPs can disrupt hematopoiesis, leading to decreased erythrocyte count, hematocrit, and hemoglobin concentrations, which increases the risk of anemia. The cytotoxic effects of MPs on erythrocytes cause hemolysis, compromising oxygen delivery to tissues. Furthermore, MP-induced physical and chemical stress can damage erythrocyte membranes, leading to hemolysis and altered hematological parameters10. Similarly, according to Pastorino et al.38, plasma constituents and blood parameters are considered to be the most significant physical indicators of health and stress problems in fish39. MPs can disrupt haemopoietic organ function and induce endocrine abnormalities, potentially changing fish blood profiles when they enter their circulatory system10.
The greatest concentration of PS-MP exposure affected the fingerlings’ mineral content, with increasing concentrations leading to decreased mineral levels. Sayed et al.40 found that fish body fluid electrolyte concentrations are essential for the dynamic control of ion outflow and water input. Fish health is assessed generally by the electrolyte imbalance, which also acts as a bio-indicator of environmental stressors. Moreover, contaminants can toxically affect the structure and permeability of fish gills, disrupting the osmoregulatory system’s function41. The present findings are consistent with earlier research, which also reported alterations in serum electrolyte levels in fish following exposure to MPs However, the underlying mechanisms of MP toxicity require further investigation to be fully elucidated.
Conclusions
The study revealed that PS-MPs had a significant detrimental impact on the growth and overall health of L. rohita. This investigation also showed that after ingesting PS-MPs for 90 days, L. rohita accumulated PS-MPs in their bodies. PS-MPs exposure caused substantial alterations in growth, digestibility, intestinal histology, hematological profile and mineral composition. The outcomes indicate that long-term PS-MPs exposure may compromise fish well-being and nutritional uptake.
Data availability
Data will be available on demand from corresponding author.
References
Cheng, H. et al. Toxicities of microplastic fibers and granules on the development of zebrafish embryos and their combined effects with cadmium. Chemosphere 269, 128677. https://doi.org/10.1016/j.chemosphere.2020.128677 (2021).
Qin, L. et al. Size-dependent impact of polystyrene microplastics on the toxicity of cadmium through altering neutrophil expression and metabolic regulation in zebrafish larvae. Environ. Pollut. 291, 118169. https://doi.org/10.1016/j.envpol.2021.118169 (2021).
Banaee, M., Faraji, J., Amini, M., Multisanti, C. R. & Faggio, C. Rainbow trout (Oncorhynchus mykiss) physiological response to microplastics and Enrofloxacin: novel pathways to investigate microplastic synergistic effects on pharmaceuticals. Aquat. Toxicol. 2023, 261–272. https://doi.org/10.1016/j.aquatox.2023.106627 (2023).
Rakib, M. R. J. et al. Microplastic toxicity in aquatic organisms and aquatic ecosystems: a review. Water Air Soil. Poll. 234(1), 52. https://doi.org/10.1007/s11270-023-06062-9 (2023).
Zitouni, N. et al. Uptake, tissue distribution and toxicological effects of environmental MPs in early juvenile fish Dicentrarchus labrax. J. Hazard. Mater. 403, 124055–124067. https://doi.org/10.1016/j.jhazmat.2020.124055 (2021).
Ding, J., Zhang, S., Razanajatovo, R. M., Zou, H. & Zhu, W. Accumulation, tissue distribution, and biochemical effects of polystyrene MPs in the freshwater fish red tilapia (Oreochromis niloticus). Environ. Pollut. 238, 1–9. https://doi.org/10.1016/j.envpol.2018.03.001 (2018).
Wu, H., Hou, J. & Wang, X. A review of microplastic pollution in aquaculture: sources, effects, removal strategies and prospects. Ecotoxicol. Environ. Saf. 252, 114567–114583. https://doi.org/10.1016/j.ecoenv.2023.114567 (2023).
Zicarelli, G. et al. Diet and plastic ingestion in the Blackmouth catshark Galeus melastomus, Rafinesque 1810, in Italian waters. Animals 13(6), 1039–1046. https://doi.org/10.3390/ani13061039 (2023).
Garcia, A. G., Suárez, D. C., Li, J. & Rotchell, J. M. A comparison of microplastic contamination in freshwater fish from natural and farmed sources. Environ. Sci. Pollut Res. 28(12), 14488–14497. https://doi.org/10.1007/s11356-020-11605-2 (2021).
Kim, J. H., Yu, Y. B. & Choi, J. H. Toxic effects on bioaccumulation, hematological parameters, oxidative stress, immune responses and neurotoxicity in fish exposed to mps: a review. J. Hazard. Mater. 413, 125423–125441. https://doi.org/10.1016/j.jhazmat.2021.125423 (2021).
Hochachka, P. W. & Somero, G. N. Biochemical Adaptation: Mechanism and Process in Physiological Evolution (Oxford University Press, 2002).
Wilder, S. M. & Jeyasingh, P. D. Merging elemental and macronutrient approaches for a comprehensive study of energy and nutrient flows. J. Anim. Ecol. 85(6), 1427–1430. https://doi.org/10.1111/1365-2656.12573 (2016).
Fao, F. A. O. S. T. A. T. Food and agriculture organization of the United Nations. Rome. http://faostat.fao.org 403–403 (2018).
Xu, H., Dong, X., Zuo, R., Mai, K. & Ai, Q. Response of juvenile Japanese Seabass (Lateolabrax japonicus) to different dietary fatty acid profiles: growth performance, tissue lipid accumulation, liver histology and flesh texture. Aquaculture 461, 40–47. https://doi.org/10.1016/j.aquaculture.2016.04.023 (2016).
NRC (National Research Council). Nutrient Requirements of Fish114 (National Academy, 2011).
Gatlin, I. I. I. et al. Expanding the utilization of sustainable plant products in Aquafeeds: a review. Aquac Res. 38(6), 551–579. https://doi.org/10.1111/j.1365-2109.2007.01704.x (2007).
Rowland, S. J. Diseases of Australian Native Freshwater Fishes with Particular Emphasis on the Ectoparasitic and Fungal Diseases of Murray Cod (Maccullochella peeli), golden perch (Macquaria ambigua) and silver perch (Bidyanus bidyanus) (Dept. of Agriculture, 1991).
Hussain, S. M. et al. Evaluation of growth, nutrient absorption, body composition and blood indices under dietary exposure of iron oxide nanoparticles in common carp (Cyprinus carpio). J. Anim. Physiol. Anim. Nutr. 108(2), 366–373. https://doi.org/10.1111/jpn.13898 (2024).
AOAC (Association of Official Analytical Chemists). Official methods of analysis. In 15th Ed. Association of Official Analytical Chemists, Washington, D.C. USA. 1094. https://doi.org/10.1002/0471740039.vec0284 (2005).
Blaxhall, P. C. & Daisley, K. W. Routine haematological methods for use with fish blood. J. Fish. Biol. 5, 771–781. https://doi.org/10.1111/j.1095-8649.1973.tb04510.x (1973).
Steel, R. G. D. & Torrie, J. H. Principles and Procedures of Statistics: A Biometrical Approach (McGraw-Hill, 1960).
Lai, W. et al. Dietary polystyrene nanoplastics exposure alters liver lipid metabolism and muscle nutritional quality in carnivorous marine fish large yellow croaker (Larimichthys crocea). J. Hazard. Mater. 419, 126454. https://doi.org/10.1016/j.jhazmat.2021.12645 (2021).
White, E. M. et al. Ingested micronizing plastic particle compositions and size distributions within stranded post-hatchling sea turtles. Environ. Sci. Technol. 52(18), 10307-1 (2018).
Putrajab, E. W., Setyono, B. D. H. & Alim, S. The effect of exposure to microplastic Polyvinyl chloride (PVC) in feed on the growth and survival of tilapia (Oreochromis niloticus). J. Fish. Health. 4(1), 42–51. https://doi.org/10.29303/jfh.v4i1.4940 (2024).
Wen, B. et al. Microplastics have a more profound impact than elevated temperatures on the predatory performance, digestion and energy metabolism of an Amazonian cichlid. Aquat. Toxicol. 195, 67–76. https://doi.org/10.1016/j.aquatox.2017.12.010 (2018).
Qiao, R. et al. Accumulation of different shapes of microplastics initiates intestinal injury and gut microbiota dysbiosis in the gut of zebrafish. Chemosphere 236, 124334–124346. https://doi.org/10.1016/j.chemosphere.2019.07.065 (2019).
Kadac-Czapska, K., Ośko, J., Knez, E. & Grembecka, M. Microplastics and oxidative stress—current problems and prospects. Antioxidants 13(5), 579. https://doi.org/10.3390/antiox13050579 (2024).
Lu, X. et al. Chronic exposure to high-density polyethylene microplastic through feeding alters the nutrient metabolism of juvenile yellow perch (Perca flavescens). Anim. Nutr. 9, 143–158. https://doi.org/10.1016/j.aninu.2022.01.007 (2022).
Von Moos, N., Burkhardt-Holm, P. & Köhler, A. Uptake and effects of microplastics on cells and tissue of the blue mussel Mytilus Edulis L. after an experimental exposure. Environ. Sci. Technol. 46(20), 11327–11335. https://doi.org/10.1021/es302332w (2012).
Prata, J. C., da Costa, J. P., Duarte, A. C. & Rocha-Santos, T. Suspected MPs in Atlantic horse mackerel fish (Trachurus trachurus) captured in Portugal. Mar. Pollut. Bull. 174, 113249–113256. https://doi.org/10.1016/j.marpolbul.2021.113249 (2022).
Rochman, C. M. et al. Classify plastic waste as hazardous. Nature 494, 169–171. https://doi.org/10.1038/494169a (2013).
Feng, S. et al. Polystyrene microplastics alter the intestinal microbiota function and the hepatic metabolism status in marine Medaka (Oryzias melastigma). Sci. Total Environ. 759, 143558. https://doi.org/10.1016/j.scitotenv.2020.143558 (2021).
Jovanović, B. Ingestion of microplastics by fish and its potential consequences from a physical perspective. Integr. Environ. Assess. Manag. 13(3), 510–515. https://doi.org/10.1002/ieam.1913 (2017).
Ahrendt, C. et al. Microplastic ingestion cause intestinal lesions in the intertidal fish Girella laevifrons. Mar. Pollut. Bull. 151, 110795. https://doi.org/10.1016/j.marpolbul.2019.110795 (2020).
Jabeen, K. et al. Effects of Virgin microplastics on goldfish (Carassius auratus). Chemosphere 213, 323–332. https://doi.org/10.1016/j.chemosphere.2018.09.031 (2018).
Zhu, M., Chernick, M., Rittschof, D. & Hinton, D. E. Chronic dietary exposure to polystyrene microplastics in maturing Japanese Medaka (Oryzias latipes). Aquat. Toxicol. 220, 105396–1053105. https://doi.org/10.1016/j.aquatox.2019.105396 (2020).
Pitt, J. A. et al. Uptake, tissue distribution, and toxicity of polystyrene nanoparticles in developing zebrafish (Danio rerio). Aquat. Toxicol. 194, 185–194. https://doi.org/10.1016/j.aquatox.2017.11.017 (2018).
Pastorino, P. et al. Changes in serum blood parameters in farmed rainbow trout (Oncorhynchus mykiss) fed with diets supplemented with waste derived from supercritical fluid extraction of sweet Basil (Ocimum Basilicum). Fishes 7(2), 89–102. https://doi.org/10.3390/fishes7020089 (2022).
Thummabancha, K., Onparn, N. & Srisapoome, P. Analysis of hematologic alterations, immune responses and Metallothionein gene expression in nile tilapia (Oreochromis niloticus) exposed to silver nanoparticles. J. Immunotoxicol. 13, 909–917. https://doi.org/10.1080/1547691X.2016.1242673 (2016).
Sayed, A. E. D. H., Hamed, M., Badrey, A. E. & Soliman, H. A. Bioremediation of hemotoxic and oxidative stress induced by polyethylene microplastic in Clarias Gariepinus using lycopene, citric acid, and Chlorella. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 250, 109189. https://doi.org/10.1016/j.cbpc.2021.109189 (2021).
Jasim, S. A. et al. Effects of short-term exposure to the heavy metal, nickel chloride (Nicl2) on gill histology and osmoregulation components of the Gray mullet, Mugil cephalus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 258, 109361. https://doi.org/10.1016/j.cbpc.2022.109361 (2022).
Acknowledgements
The study was funded by HEC Pakistan Projects No. 20-4892/NRPU/R&D/HEC/14/1145, for which the authors are grateful. The authors extend their sincere appreciation to the Researchers Supporting Project Number (RSP2025R48), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
E.R.: Original draft and process in writing. S.M.H.: Software, Formal Analysis, Methodology, Investigation, Supervisor, and Writing-Review & Editing. S.A.: Conceptualization, Software, Writing-review & editing. D.K. and J.N.: Conceptualization and Funding acquisition. K.A.A.G.: Investigation and Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rashid, E., Hussain, S.M., Ali, S. et al. Polystyrene microplastics exposure in freshwater fish, Labeo rohita: evaluation of physiology and histopathology. Sci Rep 15, 12888 (2025). https://doi.org/10.1038/s41598-025-95811-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95811-3
Keywords
This article is cited by
-
Polyethylene nanoplastics intensify toxicity of potassium clavulanate in African catfish (Clarias gariepinus)
Scientific Reports (2025)
-
Adsorption of Polystyrene (PS) Microplastic from Aqueous Solution by Black Tea Magnetic Nanoparticles (BTMNPs): Adsorbability, Mechanism, and Reusability
Water, Air, & Soil Pollution (2025)