Abstract
In this study, fly ash and metakaolin are examined as replacements in cementitious materials to reduce environmental pollution. An orthogonal test was conducted with nine groups of geopolymer mortars prepared by varying the ratio of the cementitious materials and the amount of composite alkali activator (NaOH-Na2SiO3). The optimal ratio of the geopolymer mortar and its feasibility as a non-vibrated pipe trench backfill mortar were examined using fluidity and mechanical property tests. Combined with the microscopic tests, the pore structure and hydrate phase of the geopolymer mortar were analysed. The results showed that the geopolymer mortar with 30% fly ash (F30), 10% metakaolin (M10), and 4% alkali activator (A4) had the best performance and achieved compressive and flexural strengths of 17.2 MPa and 5.55 MPa, respectively, with a fluidity of 157 mm; these results meet the requirements for backfill materials. In addition, according to the strength comparison of the different groups, metakaolin had a better effect on the mechanical properties of the material than fly ash. Microscopic analysis revealed that the hydratable substances such as potassium feldspar and montmorillonite decomposed under the action of OH− and formed C-(A)-S-H and N-A-S-H gels, along with potassium A-type zeolite crystals; these substances improved the pore structure and density of the mortar. This study provides valuable insights into the potential use of alkali-activated geopolymer mortars as vibration-free backfill materials for pipeline trenches.
Similar content being viewed by others
Introduction
In recent years, with the accelerated construction of China’s infrastructure, pipeline construction has become an important part of infrastructure construction, and the selection of pipeline trench backfill materials has gradually become a key issue in the engineering field1. Traditional cement mortar materials are widely used for trench backfilling2 because of their high strength and ease of construction. However, the high energy consumption and environmental pollution problems associated with cement production have become a research focus. With the intensification of global climate change, the construction industry, which is a major carbon emitter, has become an important target for promoting sustainable development. The carbon footprint needs to be reduced by preparing more environmentally friendly building materials3.
Geopolymers are environmentally friendly building materials prepared as auxiliary cementitious materials and have received widespread attention because of their low carbon emission, high durability and good mechanical properties4. Geopolymer mortars made of metakaolin5, coal gangue6 and fly ash7 are widely used in road construction, building structure and restoration projects. In particular, under harsh conditions such as high temperatures and extreme pH environments, geopolymer mortars exhibit excellent mechanical properties8,9. Studies have shown that geopolymers contain a large amount of high-bond energy SiO2 with significant pozzolanic activity. After excitation, high-bond energy SiO2 generates silicate anions (SiO44−); these anions interact with the calcium hydroxide generated during the hydration of cement to produce calcium silicate gel. The gel-like products formed during this reaction process can increase the strength and density of cementitious materials. C-S-H gel is the most critical structural material in cementitious materials and can effectively improve the microstructure, reduce harmful pores, and improve mechanical properties10.
Metakaolin and fly ash are used as the main raw materials of geopolymers. After alkali excitation treatment, a dense three-dimensional network structure is formed and enhances the mechanical properties of cement-based materials11. Some scholars have reported12 that metakaolin is a material with high pozzolanic activity, but when it is directly mixed into mortar, its ability to improve strength is limited; additionally, its strength needs to be improved by appropriate alkali activation or compounding with other admixtures. The aluminosilicate network in metakaolin dissolves under alkali activation conditions to form a three-dimensional crosslinked sodium alumino-silicate hydrate. This gel has a good filling effect, which can reduce the porosity of the material and increase its compactness, thereby improving the compressive and flexural strengths13,14. Fly ash is rich in SiO2, Al2O3 and other components, and the alkali activator interacts with the material components to produce silicate and aluminosilicate gels15,16. Moreover, the spherical particle shape of fly ash improves the porosity of the geopolymer material, increasing its compressive and flexural properties17,18. Studies have shown19 that the use of Na2SiO3 and NaOH as base activators has a significant effect on the initial curing time and crushing strength of geopolymers. These findings indicate that using sodium silicate as an alkali activator results in the shortest initial setting time and the best mechanical performance of the materials. Some scholars have used fly ash and metakaolin as the main materials and water glass (Na2SiO3) and sodium hydroxide (NaOH) as alkaline solutions to prepare geopolymer mortar. When no alkali activator is added, the raw material fails to adequately react, causing a decline in mortar strength20. In summary, to protect the environment, metakaolin and fly ash were used to replace part of the cement to make a new cement-based mortar, and water glass was used as an activator to enhance the hydration properties of the polymers.
To investigate the potential application of ternary binder materials consisting of metakaolin, fly ash, and cement for pipeline trench backfilling, alkali-activated geopolymer cement mortars were made by replacing cement with different proportions of fly ash and metakaolin after activation. Nine groups of mix proportions were designed by orthogonal tests, and the effects of these variables on the key parameters of geopolymer materials, such as the initial curing time, flowability, crushing strength, and bending strength, were comprehensively analysed. The mechanical properties of the nine groups of mortar were compared using compression and flexural tests to obtain the optimal replacement amounts of fly ash and metakaolin and the amount of alkali activator to be added. The mechanism of their mechanical properties was also explored using microscopic tests. This study provides a scientific basis and technical support for the practical application of environmentally friendly building materials in the backfilling of pipeline trenches.
Test materials and methods
Test materials
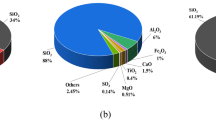
(1) Cement: Jidong Cement P.O 42.5, with a fineness of 1.4%. The initial setting time lasts for 180 min. The final setting period is 385 min. The volume stability meets the required standards. Table 1 lists the physical properties and chemical composition; (2) Fly ash (FA) is a Class II, Type F, low calcium fly ash supplied by the Jinchuan Power Plant in Hohhot, Inner Mongolia, and appears as a grey powder; metakaolin (MK) appears as a white powder. Figs 1 and 2 show the microstructure and phase composition of FA and MK, respectively. The micrographs of fly ash mainly show spherical particles and irregular blocky particles, while metakaolin is composed of various irregular blocky and flaky particles. Figure 2a-b presents the results of the XRD analysis. The main phase components of FA and MK were revealed. The FA sample mainly contains the crystalline phases of quartz, albite, potassium feldspar and calcite, while the MK sample is mainly composed of quartz and potassium feldspar. Table 2 lists chemical analysis of FA and MK. The particle size distribution was determined by laser particle size analyzer. Figure 3a-b shows the distribution of particle size. The median particle size of FA is 15.67 μm and its apparent density is 587.33 m2/kg. The median particle size of MK is 14.16 μm and the apparent density is 1060.92 m2/kg.
(3) Fine aggregate: conventional river sand; (4) Water: ordinary tap water; (5) Activator: a composite alkali activator (sodium hydroxide and water glass) with a modulus of 1.4 was selected.
SEM image analysis of FA and MK.
XRD patterns of FA and MK.
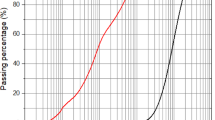
Particle size distribution of FA and MK.
Orthogonal test design
The method of analysis of the extreme difference using orthogonal experiments was used. The replacement rate of fly ash and metakaolin (replacing cement by equal mass) and alkali equivalent (calculated based on Na2O content) were used as the control factors. An L9 (33) orthogonal experiment was designed. Factor levels are listed in Table 3.
Mix proportion design
The M25 mortar is designed following ASTM C109/C109M-20b, “Standard Test Method for Compressive Strength of Hydraulic Cement Mortar.” 30%, 50%, 70% fly ash and metakaolin (0%, 10%, 20%) and other mass substitutions for cement, and alkaline activator (0%, 4%, 8%) with alkali equivalent (Na2O in the activator as a percentage of the fly ash mass) mixed externally to prepare alkali activated fly ash-metakaolin-cement mortar test blocks. Mix details are in Table 4.
Test methods for determining the mechanical properties and microstructure
Mechanical properties and flow properties test
The cement, fly ash, metakaolin, river sand, alkali activator and water were poured into the mixing pan in that order. Cubic specimens with 70.7 mm sides were prepared. After 24 h, the specimens were cured in a standard curing box. In accordance with the Standard for Mechanical Testing of Ordinary Mortars (GB/T50081-2002), the unconfined compressive strength and flexural strength of the specimens were measured at curing periods of 3 d, 7 d, 14 d, and 28 d. Mortar fluidity tests followed GB/T 2419 − 2005, which is the standard for cement mortar flow measurement. The cement, fly ash, metakaolin, river sand, alkali activator and water were mixed in proportion, poured into a mixing pan and stirred until a uniform mortar was formed; the mixed mortar was poured into a cement mortar flow tester, and the flow test was performed following the “cement mortar flow test method” (GB/T2419-2005). Before the test, the table top was moistened with a damp cloth to prevent the mortar from sticking to the table. The mortar was loaded into the test mould in two batches. The first batch was loaded to approximately 2/3 of the height of the test mould and then tamped 15 times with a tamping rod. The mortar was then loaded until it was approximately 20 mm higher than the test mould. After tamping for 10 times, the formwork was removed, the mortar that was higher than the truncated cone mould was cut off, the truncated cone mould was lifted upwards, and the jumping table was activated and jumped 25 times; after jumping was complete, a calliper was used to measure the diameters in two perpendicular directions, and the average (round up to the nearest integer), which represented the flowability of the mortar, was taken.
Microscopic characterization test of each mortar group
First, the microstructure of the mortar pores in each group was examined using NMR after mechanical testing. Variations in the pore characteristics of the polymer mortar were observed for different amounts of cementitious materials and alkali activator contents. The 28 d mortar samples were placed in a -0.8 MPa vacuum water saturation device for 24 h to saturate them with water, and the porosity of the mortar was then measured using nuclear magnetic resonance (OMR Neumark MES-23-060v-i).
Next, the mass changes in the hydration products of each group were analysed using TG-DTG. After 28 days of curing, the mortar samples were placed in an aluminium crucible and then placed in a thermal analyser. The temperature was increased at 10 °C/min. The test temperature ranged from room temperature to 995 °C. TG‒DTG curves were employed to examine the influence of varying replacement levels on water, carbonates, and other hydration products in the mortar during hydration. X-ray diffraction (XRD) and infrared spectroscopy (FTIR) were used for analysis. The phase transformations of the hydration products in alkali-activated fly ash-metakaolin-cement blends with various replacement ratios were analysed. The XRD test was conducted using a Mini-Flex 600 instrument to analyse the phase composition of the mortar samples. With a Cu target, the scanning rate was 2°/min. The scanning range was 5° to 90°.
Finally, the analysis was conducted using SEM (Tescan-Mira-Lms). The microstructures of the samples in each group were observed. The results were combined with Xplore30-point energy dispersive spectroscopy (EDS). The influence of varying fly ash–metakaolin replacement ratios and composite alkali activator dosages on the elemental composition of the hydration products in geopolymer mortar was analysed.
Results and discussion
Fluidity
In this experiment, by studying the influence of different metakaolin contents, fly ash replacement amounts and alkali activator amounts on slurry fluidity, the influence of each factor on fluidity differs. As shown in Fig. 4, the replacement amount of fly ash is the primary factor influencing the fluidity of the mortar. From F30M00A0, F50M00A4 and F70M00A8, as the replacement amount of fly ash increases from 30 to 50% and the dosage of activator increases from 0 to 4%, the fluidity of the mortar significantly increases to 145 mm, 156 mm and 144 mm, respectively, with a trend an initial increase and then a decrease. This occurs because the spherical structure of fly ash particles improves the lubricity of mortar21, whereas an appropriate amount of alkali activator can help accelerate the dissolution of active ingredients in metakaolin and fly ash and generate N-A-S-H and C-S-H gels with good fluidity, thus improving fluidity. However, when the replacement amount of fly ash exceeds 50% and the amount of alkali activator exceeds 4%, the fluidity begins to decline. This occurs because fly ash has a certain degree of water absorption, resulting in a reduction in free water, an increase in paste viscosity and a decrease in fluidity22. Moreover, excessive alkali activator causes the gel formation speed to occur too quickly, and the paste becomes viscous, thus reducing fluidity23.
Polymer mortar fluidity.
For F30M00A0, F30M10A4 and F30M20A8, the content of fly ash was fixed at 30%, the content of metakaolin increased from 0 to 20%, and the dosage of activator increased from 0 to 8%. The fluidity of the samples in each group was 145 mm, 157 mm and 150 mm, respectively, with a trend of an initial increase and then a decrease. These results indicated that when the fly ash content was 30%, metakaolin improved the fluidity of mortar samples under the action of activators because the gel substances generated by metakaolin under alkali excitation helped reduce the viscosity of the slurry, especially under low alkali excitation conditions, and the effect was more evident24. For groups F50M00A4, F50M10A8 and F50M20A0, when the content of metakaolin was 0%, the highest mobility of F50M00A4 was 156 mm, and when the content of metakaolin was 10%, the lowest mobility of F50M10A8 was 153 mm, with a difference of 3 mm. The fluidity of F50M20A0 was between the two above samples, and its fluidity was 154 mm; this value was 1 mm and 2 mm different from those of F50M10A8 and F50M00A4, respectively. Thus, when the fly ash content exceeded 30%, the influence of partial kaolin on the fluidity of mortar was minimal.
However, for F30M00A0, F50M00A4 and F70M00A8, with increasing activator amount, the slurry fluidity initially increased and then decreased. When the activator parameter was 4%, the fluidity of F50M00A4 reached 156 mm; this value was 11 mm greater than that of F30M00A0. According to the above analysis, fly ash had the greatest influence on the fluidity of mortar, followed by the activator, and metakaolin had the least influence.
Mechanical property
As shown in Fig. 5a-b, the compressive strength of the F30M10A4 group is optimal at all ages, and the compressive strength reaches 11.6 MPa at 3 days and 17.2 MPa at 28 days. These results occur because an appropriate amount of complex alkali activator (4%) can promote the reaction of the active ingredients in metakaolin and fly ash with Ca(OH)2 in the cement, causing more highly cohesive gels and improving the compactness of the mortar25. Compared with the F30M00A0 group lacking a composite activator, the 28 d compressive strength increased by 10.26% with 10% less cement, which showed that the composite activator could effectively activate the properties of solid waste and cause better hydration properties than cement properties. However, the F30M20A8 group exhibited a compressive strength that was 13.95% lower with 8% composite alkali activator than the F30M10A4 group, which was caused by the high dosage of the composite alkali activator. These results indicate that the absence of, or an excess of, the composite alkali activator may hinder the interaction of reactive constituents in metakaolin, fly ash, and Ca(OH)2 in the cement, leading to a reduced formation of highly cohesive gels and negatively impacting the mechanical properties of the mortar.
Furthermore, the compressive strengths of the groups with 50% replacement of geopolymer materials (F30M20A8 and F50M00A4) at 28 d greatly differed, at 14.8 MPa and 8.4 MPa, respectively. This is caused by the different replacement ratios of fly ash and metakaolin. The reactive components in metakaolin underwent a more complete reaction with Ca(OH)2 during the alkali activation process, resulting in the formation of a greater quantity of highly cohesive gels26; these gels efficiently filled the pores, increased the material’s density, and increased its compressive strength, whereas the F50M00A4 group lacked the synergistic effect of metakaolin, resulting in fewer gel products and lower strength.
The flexural strength of the F30M00A0 group at 28 d was 4.94 MPa; this value was lower than that of the F30M10A4 group. These findings indicated that the composite alkali activator significantly enhanced the flexural strength. Additionally, metakaolin showed a synergistic effect with fly ash under alkaline activation, leading to the formation of more cohesive gels27, thereby further improving the flexural performance of the mortar. The F70M10A0 group and the F70M20A4 group had the worst flexural strength performance, at 1.94 MPa and 1.32 MPa, respectively. This was caused by the low cement content in the mortar, which hindered the adequate reaction of active components in the geopolymer material, leading to a limited production of highly cohesive gels. This situation reduced the density and strength of the material. Moreover, high fly ash replacement increased the viscosity and porosity of the slurry, making the microstructure of the material loose, which ultimately resulted in a notable reduction in flexural performance.
Overall, the F30M10A4 group exhibited compressive and flexural strengths superior to those of the other groups. These findings indicated that an optimal combination of a composite alkali activator and an appropriate metakaolin–fly ash replacement ratio effectively promoted gel formation, resulting in enhanced material performance. In addition, groups with high fly ash substitution (e.g., F70 series groups) and low alkali activator contents had a negative impact on the mechanical properties. In particular, groups F70M10A0 and F70M20A4 had poor strength performance, which was likely caused by insufficient reactions, resulting in insufficient highly cohesive gel formation. To further investigate the mechanisms underlying the enhancement of the mechanical properties of the geopolymer mortar, microstructural analysis of the mortars in groups F30M00A0, F30M10A4, F30M20A8 and F50M00A4 were carried out to determine the intrinsic reasons for their strength improvement.
Mechanical properties of geopolymer mortar.
NMR results
To further explore the reasons for the excellent mechanical properties of the F30M10A4 group, the pore structure of each group of mortar samples was analysed using NMR. Density plays a crucial role in determining mortar performance, and pore composition (including pore volume and distribution) has a direct effect on mortar strength28. Therefore, the T2 spectrum from NMR is strongly correlated with the pore distribution in the geopolymer mortar. The T2 spectrum area reflects the porosity of the geopolymer mortar29. The transverse relaxation time is correlated with the pore size. A smaller transverse relaxation time corresponds to a lower porosity30.
As shown in Fig. 6a, the T2 spectrum of each group of samples at 28 d is mainly composed of three spectral intervals, which represent the pore water relaxation of micropores, small pores and large pores. Here, the influence of varying replacement levels of fly ash and metakaolin on the pore structure of geopolymer mortar were revealed. The effects of different dosages of alkali activators on the pore structure of the mortar were also examined. Overall, the use of an optimal amount of composite alkali activator can effectively refine the pore structure of the mortar, decrease the presence of large pores, and increase the proportion of small pores, thus increasing the compactness of the mortar. As shown in Fig. 6a, the T2 spectral area of the F30M10A4 group in each spectral interval is significantly smaller than those of the other groups. These results indicate that this group has the optimal ratio in the geopolymer mortar system. At this ratio, the number of pores is minimal, and the density of the mortar is highest. The T2 spectral area of the F30M20A8 group in each interval is significantly smaller than that of the F50M00A4 group; these results indicate that the density of the F30M20A8 group is better. Thus, metakaolin has a more significant effect on the pore structure than fly ash.
T2 spectrum and pore proportion of geopolymer mortar.
According to the relevant literature31, the pore size of mortar can be classified into harmless pores (0–0.01 μm), less harmful pores (0.01–0.1 μm), harmful pores (0.1–1 μm), and highly harmful pores (1–100 μm). The ratio of harmless and less harmful pores is crucial in determining the mechanical properties of the mortar. As shown in Fig. 6b, the F30M10A4 group has the smallest proportion of highly harmful and harmful pores, whereas the proportion of harmless and less harmful pores is the highest. Compared with those of the F30M00A0, F30M20A8 and F50M00A4 groups, the total porosity is 20.86, 24.13 and 31.12% lower, respectively. Thus, using the F30M10A4 ratio, the highly harmful and harmful pores in the geopolymer mortar are filled with hydration products, which convert them into harmless and less harmful pores. The pores are filled, the porosity is reduced, and the pore size decreases. These changes in pore composition directly improve the material’s microscopic load-bearing structure. Therefore, F30M10A4 has the highest strength performance. This result is consistent with the deductions in Sect. 2.1.2. The porosity of F30M20A8 is slightly lower than that of F50M00A4, with a 0.11% decrease in the harmful pore volume, whereas the volume of less harmful pores increases by 0.09%. These changes in pore composition also verify that the impact of metakaolin on the pores of mortar is greater than that of fly ash.
In the F30M20A8 and F50M00A4 groups, the composite cementitious material prepared with metakaolin and fly ash replaced more than 40% of the cement. The amount of CaO in the cementitious material that can undergo hydration was significantly reduced, and the decrease in the amount of hydration products generated resulted in insufficient filling of the highly harmful and harmful pores in the mortar. Therefore, the interval between multiple harmful pores and harmful pores in the T2 spectrum has a large spectral area. In addition, the composite alkali activator contains a large amount of Na+ and a small amount of Ca2+, which cannot replace Na+ to form a high-strength hydration gel. A large amount of low-strength, low-viscosity, and weakly stable sodium-activated aluminosilicate-hydrate gel cannot effectively play a bonding role and improve the pore structure of the mortar system. Therefore, the material density and mechanical properties are diminished.
TG-DTG results
The NMR test results indicate that the F30M10A4 geopolymer mortar has the optimal pore structure, with a denser internal structure. The pore composition of the material is closely related to the type and content of hydration products in the mortar system, and the gel content with a filling effect is the key to determining the compactness of the geopolymer mortar system. Therefore, to further verify the content and type of hydration products in each group of geopolymer mortar samples, their hydration products at the age of 28 d were analysed using TG-DTG.
Figure 7a-d shows the TG-DTG test results. Throughout the heating process, nonchemically bound water evaporates, leading to the dehydration of key hydration phases (C-(A)-S-H, gypsum, AFt, and AFm) between 70 °C and 200 °C. The temperature range of 400 ℃ to 500℃ corresponds to the dehydroxylation of Ca(OH)2. The 650 ℃ to 830 ℃ range corresponds to the decarbonation of CaCO3 and volatile components32. As illustrated in Fig. 7a-d, in the range of 70 ℃ to 200 ℃, all groups had sustained weight loss, in descending order: F30M10A4, F30M00A0, F30M20A8, and F50M00A4. A comparison of F30M20A8 and F50M00A4 revealed a slightly greater weight loss of 14.31% for F30M20A8 than for F50M00A4 (12.95%). These results indicate that incorporating metakaolin facilitates the formation of additional C-(A)-S-H gel, improving the strength of the geopolymer mortar and supporting the initial hypothesis. Ca(OH)2 decomposes into calcium oxide and water in the range of 400 °C to 500 °C. The weight loss rates in descending order are as follows: F30M00A0, F30M20A8, F30M10A4, and F50M00A4. Compared with the F50M00A4 group, the F30M20A8 group presented a weight loss rate of 2.93%. These results indicate that metakaolin incorporation enhances the formation and hydration of CaO. In the range of 650 °C to 830 °C, the formation of CaCO3 reduces the calcium content in hydrated tricalcium silicate and reduces the calcium‒silicon ratio in cementitious material. The weight loss rates in the decomposition temperature range from largest to smallest are F50M00A4, F30M00A0, F30M20A8, and F30M10A4; thus, the F30M10A4 geopolymer mortar has the highest content of C-(A)-S-H gel and Ca(OH)2. The weight loss rate of the F30M20A8 group was 9.81%, whereas that of the F50M00A4 group was 16.35%. Therefore, a higher CaCO3 content is found in the F50M00A4 group, and metakaolin has a more significant effect on gel formation than fly ash.
TG-DTG diagram of geopolymer mortar.
These findings also support those from the previously discussed mechanical property and NMR tests; F30M10A4 has the optimal mix ratio for geopolymer mortar. The Ca(OH)2 content and gel substance content of the remaining groups are lower than those of F30M10A4; these results verify that when the amount of composite cementitious material exceeds 40%, the hydration reaction of the geopolymer in the mortar system is incomplete. At this time, the inclusion of a composite alkali activator does not effectively enhance the hydration reaction of the geopolymer system. As a result, the unreacted Ca(OH)₂ undergoes carbonation, leading to the formation of CaCO₃ and an increase in the porosity of the internal structure of the mortar. The weakening of the pore structure subsequently results in a decrease in the mechanical properties of the material. A comparison of the TG‒DTG test results of the two groups of geopolymer mortars, F30M20A8 and F50M00A4, reveals that the F30M20A8 group has slightly greater Ca(OH)2 and gel contents; these results indicate that metakaolin reacts more completely with the Ca(OH)2 in the cement during the alkali activation process than fly ash, leading to a more pronounced effect on the gel.
XRD and FTIR results
Based on the results of the aforementioned mechanical property tests, NMR tests, and TG-DTG tests, the F30M10A4 group geopolymer mortar generates more gelling substances. These substances fill the pores, producing a denser mortar. As a result, it has the best mechanical properties. The aim of this study is to explore the differences in the types of gel material produced by the hydration reactions of the optimal F30M10A4 geopolymer mortar and other geopolymer mortars. XRD and FTIR tests are used to analyse the gel products. The focuses of this analysis are on phase and physical property groups.
Figures 8a-d show the XRD phase identification maps and infrared spectrum analysis diagrams of the samples in each group, respectively. Figure 8a-b reveals significant differences in the hydration products of the F30M10A4 samples compared with those of the other groups. The diffraction peaks of quartz, montmorillonite (noted for their high absorbency and expansiveness), albite, and C-(A)-S-H were identified in the samples of the F30M00A0, F30M20A8, and F50M00A4 groups. In the F50M00A4 group of samples, the diffraction peaks of quartz and montmorillonite were the sharpest, and the C-(A)-S-H diffraction peaks nearly disappeared. In the F30M10A4 group of samples, the diffraction peaks of montmorillonite disappeared, whereas the peaks for albite and some quartz were weakened, and the diffraction peaks of KA (potassium A-type zeolite) appeared. In addition, sharper characteristic diffraction peaks of C-S-H appeared at 2θ = 19.12°, 17.4° and 31.04°, whereas the characteristic diffraction peaks of C-A-S-H appeared at 2θ = 8.4° with groups F30M00A0 and F30M20A8. Notably, the F30M10A4 group also showed a diffuse peak with N-A-S-H characteristics at 2θ = 29°; these results indicated significant phase changes in the hydration products within the F30M10A4 geopolymer mortar. OH− ions promote the decomposition of the composite cementitious material. They react with Ca(OH)2 formed during cement hydration, leading to the generation of N-A-S-H gel and KA, as well as C-S-H and C-A-S-H gels. In the F50M00A4 group, although the use of composite activators can still promote the reaction of the active ingredients in the geopolymer material with the hydration products of the cement, the diffraction peaks of the inert components such as quartz and albite in F30M20A8 may still be more evident. This occurs because metakaolin has a stronger influence on the formation of C-(A)-S-H gels in geopolymer mortar than does fly ash.
XRD and FTIR spectra of geopolymer mortar.
The infrared spectrum analysis in Fig. 8c-d shows that the characteristic spectral bands of the hydration products mainly appear between 900 and 1300 cm− 1 at the physical group level. This range is attributed mainly to the asymmetric vibration of the silicon-oxygen-tetrahedral (T: silicon/aluminium) in the gel product. These structural features confirm the role of the composite activator. This significantly enhances the mechanical properties of the geopolymer mortar. This results occur because the gel formation is promoted, and the internal structure of the mortar is optimized.
The F30M10A4 geopolymer mortar exhibits an infrared absorption band at 453 cm− 1. This band is attributed to the bending vibration of the silicon‒oxygen bond in the SiO4 group. Compared with those of the other groups, the absorption peak of F30M10A4 at the bending vibration of the silicon–oxygen linkage shifts to a lower wavenumber, with a maximum shift of 13 cm− 1. This result indicates that the F30M10A4 geopolymer mortar has the lowest degree of SiO2 polymerization. A significant amount of SiO2 dissolves and reacts with Ca(OH)2 from cement hydration. This leads to the formation of additional silicate and aluminosilicate gel products. Moreover, the maximum absorption wavenumber of the asymmetric vibration of the silicon‒oxygen tetrahedral bond in F30M10A4 increased from 998 cm− 1 to 1009 cm− 1 compared with those of the other groups; these results indicated that more highly polymerized aluminosilicate gel products were generated inside the F30M10A4 mortar. Four peaks were obtained in the main absorption peaks of each group of samples distributed between 800 and 1300 cm− 1, which were located at 800–900 cm− 1, 900–980 cm− 1, 980–1050 cm− 1 and 1050–1300 cm− 1. As illustrated in Fig. 9a-d, the F30M10A4 samples had the smallest area for the non-polymerized silicate deconvolution peak in the 800–900 cm− 1 range. The area of the calcium silicate deconvolution peak in the 900–980 cm− 1 range was the largest. The deconvolution peak area of calcium aluminate hydrate in the 980–1050 cm− 1 range was also the largest. Moreover, the relative concentration of the silicon‒oxygen tetrahedra in the F30M10A4 group of geopolymer mortars in the 980–1050 cm− 1 wavenumber range was significantly greater than that in the other groups, with a maximum increase of 54.53% these results indicated the formation of new crystalline phase substances in the F30M10A4 group. For the F30M00A0, F30M20A8 and F50M00A4 samples, the peak areas in the 900–980 cm− 1 and 980–1050 cm− 1 wavenumber ranges are small, indicating insufficient gel formation and incomplete hydration. This occurs because the amount of composite gelling material is too high, resulting in a low cement content in the polymer mortar system. Although a certain amount of composite alkali activator is added, the CaO content in the mortar system is too low to effectively carry out the hydration reaction to form Ca(OH)2, which in turn leads to the generation of N-A-S-H gel and C-(A)-S-H gel. The gel content in the F30M20A8 group is notably lower than those in the other groups. In the 900–1300 cm⁻¹ range, the asymmetric vibration peak of the silicon‒oxygen tetrahedral bond in F30M20A8 is much greater than that in F50M00A4. In particular, the deconvolution peak area between 980 and 1050 cm− 1 is significantly larger in F30M20A8 than in F50M00A4. The results revealed that more silicate and aluminosilicate gels with high polymerization degrees were generated in the F30M20A8 samples than in the other samples; these results further verified that metakaolin had a more significant effect on gel formation than fly ash.
FTIR peak fitting diagram and element analysis diagram.
The analysis revealed that a significant number of N-A-S-H gels with high polymerization and three-dimensional network structures formed in the F30M10A4 group. Compared with fly ash, metakaolin promotes gel formation more effectively. Figure 9e shows the three-dimensional network of N-A-S-H gels, whereas Fig. 9f shows the replacement of Na+ with Ca2+ during cement hydration, resulting in the formation of lower-polymerized C-A-S-H gels. Compared with the other groups, the F30M10A4 group generated more C-(A)-S-H gels, N-A-S-H gels, and KA, enhancing the filling effect. These findings further support the deduction that the gel content was greater in the F30M10A4 group than in the other groups, as observed via TG-DTG analysis.
SEM-EDS results
Based on the results of the above tests, the hydration products in the F30M10A4 mortar sample mainly include C-(A)-S-H gel, N-A-S-H gel, and KA. The combined effect of these hydration products effectively improves the mechanical properties of the mortar. The 4% alkali activator helps the aluminium silicates in metakaolin completely dissolve, forming a denser three-dimensional network structure. Concurrently, the silicate provided by the fly ash combines with calcium ions, promoting the formation of gel products. This improved the overall strength of the mortar.
Four groups of mortar samples were used for SEM examination to analyse the microstructure. The test results are shown in Fig. 10a-d. Compared with F30M00A0 and F30M20A8, F30M10A4 clearly has the highest amount of internal hydration products, and the microstructure is more compact. F50M00A4 has the most compact microstructure, followed by F30M00A0, and F30M20A8 has the loosest microstructure. These results show that when the amount of cement replacement is large, the inclusion of 4% alkali activator effectively refines the internal structure of the mortar, increasing its density.
SEM-EDS image analysis.
F50M00A4 has more Ca(OH)2 and CaCO3 than F30M20A4, and relatively few amorphous gels are observed. However, the F30M20A8 mortar has amorphous gels distributed throughout its structure, and these gels effectively fill the pores. Based on the above tests, metakaolin has a greater effect on the mechanical properties of geopolymer mortar than fly ash. Although the 4% compound alkali activator promotes cement hydration to some extent, excess OH− ions cannot effectively promote the decomposition of the cementitious material. This prevents sufficient reaction with Ca(OH)2 from cement hydration, leading to carbonation in the F50M00A4 group and the formation of additional CaCO3. This influences the formation of gelling substances, and as a result, the microscopic pores cannot be effectively filled. The Ca(OH)2 generated from cement hydration in F30M20A8 reacts fully with the Al2O3 in metakaolin, forming highly polymerized C-A-S-H gel that fills the pores. As a result, the mechanical properties of the F30M20A8 group polymer mortar are slightly superior to those of the F50M00A4 group. As shown in Fig. 10e-f, the main elements of the hydration products of F30M20A8 and F30M10A4 are composed of Ca, Si, Al, Na, and K, with a small amount of Mg. F30M20A8 contains C-S-H, C-A-S-H, N-A-S-H, and Ca(OH)2, and F30M10A4 also contains potassium A zeolite crystals with high hardness. These products promote closer connections between the internal gels and improve the internal microstructure of the polymer mortar. Thus, its mechanical properties are improved.
To summarize the above analysis, the geopolymer mortar in the F30M10A4 group has the best mechanical properties, and its microscopic mechanism is shown in Fig. 11. Under the action of OH−, the silica, alumina, and montmorillonite in the composite cementitious material generate structures such as silicon‒oxygen tetrahedrons and aluminium‒oxygen tetrahedrons; these tetrahedrons combine with elements such as Ca and Al produced by the hydration of cement to generate more C-(A)-S‒H gel, N‒A‒S‒H gel and KA. The various gels and zeolite crystals form an interwoven three-dimensional structure that fills the pores in the mortar, significantly improving its internal microstructure and producing denser appearance; thus, the mechanical properties of the geopolymer mortar are significantly enhanced.
Mechanism diagram of the effect of composite cementitious materials on the mechanical properties of geopolymer mortar after excitation.
To avoid too many test samples, the material mix ratio was designed according to the orthogonal test, and samples with similar mechanical properties were selected in a later study to provide a more detailed division of the material incorporation amount. Moreover, water glass is selected as the alkali activator in this study, and other alkali activators are selected in a later study to explore their influence on the mechanical properties of cement-based mortar.
Conclusions
In this study, the effects of different fly ash and metakaolin replacement amounts and alkali activator dosages on the performance of geopolymer mortars and their mechanisms of action are examined. Therefore, a systematic analysis of fluidity, mechanical properties (compressive strength and splitting tensile strength) and microscopic mechanisms (phase characterization, physical property groups, gel content and microscopic morphology) was carried out, and the key conclusions are as follows:
(1) Spherical particles of fly ash can improve fluidity; however, when the replacement amount exceeds 50%, the fluidity decreases due to the increase in water absorption of fly ash. Metakaolin has little influence on fluidity and can moderately improve fluidity when the replacement amount of metakaolin is between 0% and 20%.
(2) When the replacement amount of fly ash is 30%, the replacement amount of metakaolin is 10% and the amount of compound alkali activator is 4%, the mechanical properties of the geopolymer mortar are significantly improved. The compressive strength of the geopolymer mortar in the F30M10A4 group reaches 17.2 MPa at 28 d of age, and the flexural strength reaches 5.55 MPa; these values meet the basic performance requirements of the material for backfilling pipe trenches.
(3) In the F30M20A8 and F50M00A4 groups, the mixing ratio of fly ash to metakaolin was changed from 3:2 to 5:0. Compared with F50M00A4, F30M20A8 exhibited notably superior mechanical properties; these results indicated that metakaolin had a better effect on the mechanical properties of the material than did fly ash.
(4) According to the results from the microscopic tests (NMR, TG-DTG, XRD and FTIR), the F30M10A4 group of geopolymer mortars has a large number of gels with a filling effect (C-(A)-S-H, N-A-S-H and KA not found in the remaining groups); these gels coexist, fill the pores. Thus, the density of the mortar is improved, and its mechanical properties are optimal. The F30M20A8 group geopolymer mortar produced more C-S-H and C-A-S-H gels than the F50M00A4 group geopolymer mortar; these results indicated that the F30M20A8 group geopolymer mortar has a better pore structure than the F50M00A4 group geopolymer mortar.
(5) According to the SEM analysis, the F30M10A4 mortar not only produced hydration products similar to those of the other groups but also produced other clusters of slender prismatic hydration products that did not appear in the other groups. Further analysis using the EDS results revealed that these slender prismatic hydration products are mainly composed of Ca, Si, Al, Na, K, and O, with a small amount of Mg.
(6) Group F30M20A8 not only has amorphous gel products with a wide distribution inside the mortar but also has a microscopic representation of more amorphous gel material filling the pores. The pores are more effectively filled than those in group F50M00A4; thus, the compactness of the F30M20A8 group is improved. These results also verify that metakaolin reacts more fully with cement hydration than fly ash in the alkali-induced process.
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Xia, Z. et al. Multi-objective optimization of geothermal heating systems based on thermal economy and environmental impact evaluation. Renew. Energy. 237, 121858 (2024).
Jin, Q., Qi, Y. H., Li, Z. F., Li, H. Y. & Chen, J. P. Research and application of modified cement grouting materials for gushing water disaster in karst conduit. J. Geotech. Geol. Eng. 40, 5855–5868 (2022).
Ahmadi, M., Piadeh, F., Hosseini, M. R., Zuo, J. & Kocaturk, T. Unraveling Building sector carbon mechanisms: critique and solutions. Renew. Sustainable Energy Rev. 205, 114873 (2024).
Bui, Q. B., Nguyen, T. P. & Schwede, D. Manually compressed soil blocks stabilised by fly Ash based geopolymer: a promising approach for sustainable buildings. Sci. Rep. 13, 22905 (2023).
Rashad, A. M., Essa, G. M. F., Morsi, W. M. & Fahmy, E. A. Calcium nitrate as a modifier agent for metakaolin-based geopolymer mortar. Constr. Build. Mater. 456, 139199 (2024).
Liu, B., Zhang, Y. & Yang, S. Influence of coal gangue-aeolian sand aggregate gradation on rheological properties and pipeline transportation characteristics of filling slurry. Sci. Rep. 15, 789 (2025).
Pu, S. et al. Investigation on the behavior of fly Ash phosphate-based geopolymer stabilized acidic lead contaminated soil. J. Environ. Chem. Eng. 12, 114739 (2024).
Tchakounte, A. et al. RSM-CCD design of volcanic Ash/rice husk Ash based phosphate geopolymer for crystal Violet adsorption: kinetics and isotherms. Sci. Rep. 14, 29422 (2024).
Yuan, B. et al. Eco-efficient recycling of engineering muck for manufacturing low-carbon geopolymers assessed through LCA: exploring the impact of synthesis conditions on performance. Acta Geotech., 1–21 (2025).
Zarzuela, R. et al. Producing C-S-H gel by reaction between silica oligomers and Portlandite: A promising approach to repair cementitious materials. Cem. Concrete Res. 130, 106008 (2020).
Liu, L. et al. Explicable AI-based modeling for the compressive strength of metakaolin-derived geopolymers. Case Stud. Constr. Mater. 21, e03849 (2024).
Zhang, L., Zhang, Y., Wang, Q., Zhang, W. & Li, Z. Sustainable alkali-activated materials: leveraging spontaneous combustion coal gangue for enhanced cementitious performance. Mater. Today Commun. 41, 111044 (2024).
Dhanalakshmi, K., Sudarvizhi, M. S., Jose, P. A. & Dhanasekaran, M. Exploring the chemistry of metakaolin-based geopolymers. J. Polym. Res. 31, 349 (2024).
Zhang, B. et al. Harnessing iron tailings as supplementary cementitious materials in limestone calcined clay cement (LC3): an innovative approach towards sustainable construction. Constr. Build. Mater. 453, 139111 (2024).
Zhang, B. et al. Effects of pretreated recycled powder substitution on mechanical properties and microstructures of alkali-activated cement. Constr. Build. Mater. 406, 133360 (2023).
Chen, Y., Tang, L., Sun, Y., Cheng, Z. & Gong, W. Physical–mechanical properties and microstructure degradation of acid–alkali contaminated granite residual soil. Geomech. Energy Environ. 36, 100501 (2023).
Jia, X. Construction of coal fly ash-based spherical grain adsorbents and their adsorption characteristics on phenolic compounds. Colloids Surf. C Environ. Aspects. 210, 100042 (2024).
Anand, A., Tiwari, S. K. & Prakash, V. Experimental investigation and optimization of morphological characteristics in mechanically agitated aluminium 6061-fly Ash cenosphere composite. Mater. Wiss Werkstofftech. 54, 1598–1610 (2023).
Yadav, M. et al. Impact of the fly Ash/alkaline activator ratio on the microstructure and dielectric properties of fly Ash KOH-based geopolymer. CivilEng 5, 537–548 (2024).
Dong, Y., Naggar, E. R. M., Gao, R., Li, Y. F. & Zhao, Y. X. Impact of microwave Pre-Curing on pore structure and environmental performance of Metakaolin- and fly Ash-Based geopolymers. Buildings 14, 3918–3918 (2024).
Xia, D. et al. Understanding the synergetic effect of SAP and nano-silica on the mechanical properties, drying shrinkage and microstructures of alkali-activated slag/fly ash-based concrete. Constr. Build. Mater. 455, 139223 (2024).
Lekhya, A. & Kumar, N. S. A study on the effective utilization of ultrafine fly Ash and silica fume content in high-performance concrete through an experimental approach. Heliyon 10, e39678 (2024).
Mahamaya, M., Alam, S. & Das, S. K. Development and characterization of alkali activated controlled low strength material using mining waste. Constr. Build. Mater. 452, 138928 (2024).
Akbulut, Z. F., Guler, S., Osmanoglu, F., Kıvanç, M. R. & Ashour, A. Exploring the flowability, physical, and mechanical properties of eco-friendly colored cement mortars with Metakaolin under sulfuric (H2SO4) and nitric acid (HNO3) attacks. J. Build. Eng. 109, 463 (2024).
Li, Z., Du, P., Zhou, Y., Wang, J. & Cheng, X. Synchronous hot-pressed metakaolin-fly Ash based geopolymer: compressive strength and hydration products. J. Build. Eng. 97, 110997 (2024).
Viola, V., Allah, P., Perumal, P. & Catauro, M. Alkali activation of Metakaolin and Wollastonite: reducing sodium hydroxide use and enhancing gel formation through carbonation. Materials 17, 4910 (2024).
Qiao, J. et al. Performance and characterization of fly ash-metakaolin-based geopolymer pastes. Mater. Today Commun. 41, 110293 (2024).
Jiang, Z. et al. A recycled crushed rock sand mortar based on Talbot grading theory: correlation of pore structure and mechanical properties. Constr. Build. Mater. 44, 137980 (2024).
Wang, T., Fan, X. & Gao, C. Strength, pore characteristics, and characterization of fly ash-slag-based geopolymer mortar modified with silica fume. Structures 69, 107525 (2024).
Zhu, J., Cao, Y. & Chen, J. Transversal inertial effect on relaxation/retardation time of cement mortar under harmonic wave. Acta Mech. Solida Sin. 21, 44–50 (2008).
Li, P. et al. Experimental study and analytical model for the pore structure of epoxy latex-modified mortar. Sci. Rep. 12, 5822 (2022).
Ma, H. Q. et al. Effects and mechanisms of slag reinforced coal gangue geopolymers. J. IOP Conf. Ser. : Mater. Sci. Eng. 474, 012040 (2019).
Acknowledgements
The study was carried out with the major science and technology project of ordos city (Grant No. 2022EEDSKJZDZX014), the Natural Science Foundation of Inner Mongolia Autonomous Region (Grant No. 2021MS05045), and the Natural Science Foundation of Inner Mongolia Autonomous Region (Grant No. 2024MS059002).
Author information
Authors and Affiliations
Contributions
Investigation, L.S.W.; Methodology, Y.L.; Supervision, H.L.G and H.L.; Writing – original draft, H.Z.; Writing – review & editing, H.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, H., Li, H., Guo, H. et al. Mechanical properties of alkali activated geopolymer cement mortar for non vibratory compacted trench backfilling. Sci Rep 15, 12347 (2025). https://doi.org/10.1038/s41598-025-96291-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96291-1
Keywords
This article is cited by
-
Experimental and GEP-based evaluation of compressive strength in eco-friendly mortars with waste foundry sand and varying cement grades
Scientific Reports (2025)
-
Impact of alkaline activator concentration on mechanical properties and microstructure of a ternary blended one-part geopolymer cement
Scientific Reports (2025)
-
Ultra-High Strength Geopolymer Concrete from Industrial Waste: Reducing Anhydrous Sodium Silicate and Steel Fiber Effects
Iranian Journal of Science and Technology, Transactions of Civil Engineering (2025)
-
Performance based evaluation of black cotton soil geopolymer mortar on fresh properties, mechanical strength, durability, and molecular dynamics correlation
Multiscale and Multidisciplinary Modeling, Experiments and Design (2025)