Abstract
The non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio (NHHR) is a novel lipid index. Prior research has established a connection between lipid irregularities and chronic kidney disease (CKD). This study aims to establish a possible link between NHHR and CKD. Data from the National Health and Nutrition Examination Survey (NHANES) spanning from 2003 to 2016 was used to examine the relationship between NHHR and CKD among the elderly population. This research utilized weighted logistic regression, smoothed curve fitting and subgroup analyses along with interaction tests to evaluate the association between NHHR and CKD. The findings reveal a positive correlation between NHHR and CKD in fully adjusted Model 3. Besides, NHHR had a J-curve relationship with CKD. Subgroup analysis indicated that compared with those with lower body mass index (BMI), individuals with higher BMI are more prone to CKD. Research has shown that increased NHHR levels are associated with a higher likelihood of developing CKD in individuals aged above 60 in the United States. NHHR’s role in lipid metabolism suggests it might be an effective marker for tracking CKD’s progression.
Similar content being viewed by others
Introduction
CKD is marked by a decline in the glomerular filtration rate, heightened urinary albumin excretion, or a combination of these factors1. Patients with CKD often exhibit no obvious clinical symptoms in the early stages, but as the condition progresses, they may experience a range of symptoms, including edema, hypertension, increased risk of infections, and disturbances in lipid and electrolyte metabolism2,3,4. CKD, marked by diminished glomerular filtration rates and increased urinary albumin excretion, poses a significant public health issue, affecting around 10% of the worldwide population, notably among the elderly5. Thus, early and precise identification, intervention, and management of CKD are crucial to alleviate the healthcare burden on both patients and society. The causes of CKD are multifaceted and remain incompletely defined; however, risk factors such as inflammation, obesity, diabetes, hypertension, and cardiovascular disease (CVD) have been recognized6,7.
The relationship between lipid metabolism and CKD is complex, with various studies having delved into the same. In the renal parenchyma, lipid deposits may induce inflammation and fibrosis. Renal tubulointerstitial fibrosis, a major histopathological manifestation, is closely associated with CKD advancement and eventual renal failure8. Commonly, CKD patients show raised levels of triglyceride-rich lipoprotein particles (TRLs), higher low-density lipoprotein cholesterol (LDL-C), and lower HDL-C. The NHHR, integrating the properties of both HDL-C and non-HDL-C, serves as a new composite index for evaluating atherosclerosis risk. Previous studies have confirmed that it is superior to traditional blood lipid indexes with respect to their assessment of atherosclerosis and metabolic syndrome risk9,10. Moreover, it has been reported that NHHR is related to type 2 diabetes, hypertension, and osteoporosis in previous research. So, this may be one of the reasons why physicians can consider the index of NHHR when evaluating disease risk in clinical practice11,12,13. Because of the superiority of NHHR in metabolic disease prediction, combining the relationship between CKD and lipid metabolism, the hypothesis was raised that NHHR might relate to CKD.
While NHHR has been studied in various aspects, the relationship between NHHR and elderly CKD patients has not been fully expounded. In this regard, the study leveraged data from NHANES from 2003 to 2016 to conduct the research on the potential link that may exist between NHHR and elderly CKD patients. It is anticipated to address the need and establish a foundation for monitoring and preventing CKD patients from the standpoint of lipid metabolism.
Methods
Survey description and study population
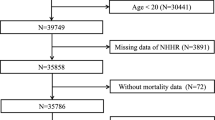
The NHANES is a recurring population-based database that is compiled in 2-year intervals by the National Center for Health Statistics (NCHS), which is a part of the Centers for Disease Control and Prevention (CDC) in the United States. The NHANES uses a complex, multistage, stratified, clustered probability design to create a dataset representative of noninstitutionalized U.S. residents14. All participants are asked to provide their informed permission once they have been fully explained the study’s goals. The study utilized data from the NHANES, covering the years 2003 to 2016, starting with a base of 71,058 participants. As shown in Fig. 1 exclusion criteria were systematically applied to refine the sample: (1) exclusion of individuals under 60 years old, numbering 58,639; (2) removal of participants without complete NHHR data, totaling 1,378; (3) exclusion of subjects lacking comprehensive CKD data, amounting to 371; (4) patients with incomplete data on covariates such as poverty-to-income ratio (PIR), alcohol consumption, diabetes mellitus (DM), BMI, educational attainment, smoking status and hypertension (n = 6,955). Ultimately, 3,715 participants were deemed eligible for the study.
Flow diagram of study cohort selection.
Definition of NHHR and CKD
All participants in this study had venous blood samples drawn in a fasting state. NHHR represents the quotient of non-HDL-C and HDL-C, with non-HDL-C calculated by deducting HDL-C from total cholesterol (TC)15,16. CKD was defined as an eGFR < 60 mL/min/1.73m2 or the presence of albuminuria14. In order to determine eGFR, the CKD-Epidemiology Collaboration (CKD-EPI) formula is used17. The main sign of albuminuria is an albumin-to-creatinine ratio (UACR) of 30 mg/g or above18.
Covariates
For this study, we investigated various factors that could influence the connection between NHHR and CKD. We took into account demographic information like gender, age, race, education level, marital status, and PIR. Laboratory and questionnaire data included triglycerides (TG), TC, HDL-C, glomerular filtration rate, urea, BMI, diabetes, smoking habits, alcohol intake, blood pressure status, and CVD. The levels of PIR were classified as low (PIR ≤ 1), middle (1 < PIR < 4), and high (PIR ≥ 4). The smoking status was classified as never smoked, past smoker, or current smoker. The levels of alcohol intake were likewise classified as none, light, moderate, or heavy. Detailed definitions of these variables are available in supplementary table S1, with complete data accessible on the NHANES website.
Statistical analysis
Considering the intricacies of the NHANES sampling process, this research adhered to CDC guidelines to select appropriate sampling weights for statistical analysis. Specific sub-weight for TC and HDL cholesterol (WTSAF2YR) were utilized. Continuous variables had their means and standard errors computed, whereas categorical variables had their percentages computed. In order to compare groups based on categorical data, we used weighted chi-square tests, and for continuous variables, we used weighted Student’s t-tests. To increase the validity of the findings, confounders that might affect the relationship between NHHR and CKD were controlled for. Three weighted logistic regression models were used to further analyze the connection between NHHR and CKD. Basic demographic factors in Model 2 included: age, gender, race, education level, and marital status; Model 3 corrected for all study variables comprehensively; and Model 1 was left unadjusted. Moreover, NHHR levels were divided into quartiles to conduct a more nuanced analysis.
The individuals were categorized according to their gender, race, education level, marital status, BMI, PIR, smoking status, hypertension, and DM. Our objective was to analyze the correlation between NHHR and CKD across various clinical and demographic subgroups. To examine how this association varied among the subgroups, interaction tests were used. Employing techniques such as threshold effect analysis, smoothed curve fitting, the study investigated the correlation between NHHR and CKD. In the case of missing data, we opted to perform deletion of the missing values. Statistical analyses were executed using R (version 4.2.0) and Empower software, applying two-tailed tests with a significance threshold set at P < 0.05.
Results
Baseline characteristics
Table 1 details the baseline characteristics of the study’s participants. The cohort comprised 3,715 individuals, with 49.48% male and 50.52% female. Non-Hispanic whites formed the largest racial group, representing 58.59% of the sample. CKD was prevalent in 35.99% of the participants, with an average NHHR of 2.70 ± 1.13. As detailed in Table 1, differences in covariates such as sex, age, race, PIR, alcohol consumption and BMI were significant across NHHR quartiles (P < 0.05). Conversely, no significant disparities were found in smoking, chronic diseases like hypertension, cardiovascular disease and diabetes.
The association between NHHR and CKD
Table 2 displays the outcomes of a weighted multifactor logistic regression analysis that examined the correlation between NHHR and CKD. In total, three models were used for the analysis: one that was unadjusted, one that was adjusted for demographic characteristics such as age, sex, race, education level, and marital status, and one that was fully adjusted for all covariates. Model 2 showed a positive correlation with odds ratios (ORs) of 1.14 (95% CI: 1.04, 1.25) and Model 3 showed an odds ratio of 1.18 (95% CI: 1.07, 1.31), respectively, when NHHR was considered as a continuous variable. Further analysis dividing NHHR into quartiles showed a 41% increase in CKD likelihood in the highest quartile compared to the lowest in Model 2 (OR = 1.41; 95% CI: 1.08,1.86) and a 50% increase in Model 3 (OR = 1.50; 95% CI: 1.08,2.09), with both models exhibiting statistically significant trends (p for trend 0.015 and 0.019, respectively). Conversely, Model 1 did not exhibit any significant associations.
Smoothed curve fitting indicated a nonlinear relationship between NHHR and CKD (Fig. 2), manifesting as a J-shaped relationship. Threshold effect analysis revealed a statistically significant difference between linear and segmented linear regression models (P = 0.002). Table 3 identifies an inflection point of 1.8, with OR as 0.65 (95% CI: 0.44, 0.96) when the OR was below this inflection point, suggesting a negative correlation between NHHR and CKD. Conversely, a significant positive correlation existed between NHHR and CKD with an OR of 1.27 (95% CI: 1.16 ,1.39) above this inflection point, indicating a rise of 27% in CKD prevalence for every unit increase in NHHR.
Smooth curve fitting for NHHR and CKD. Subjects who did and did not develop CKD are identified as 1 and 0 on the y-axis, respectively. NHHR was a continuous variable. The solid red line represents the smooth curve fit between variables. Blue dotted lines represent the 95% CI from the fit. Adjustment factors included age, sex, race, education attainment, marital status, BMI, PIR, smoking status, drinking status, hypertension, DM, CVD, dietary cholesterol intake and total cholesterol.
Subgroup analysis
Subgroup analysis and interaction tests assessed whether the correlation between NHHR and CKD varied across different subgroups. Table 4 demonstrates that the correlation remained consistent across subgroups stratified by sex, race, educational attainment, smoking status, PIR, BMI, hypertension, and DM (P values for all interactions > 0.05, except for BMI). Within the subgroup of individuals with BMI ≥ 30, there was a noTable 24% increase in the prevalence of CKD for every unit increase in NHHR. There was a clear difference observed in the BMI subgroup as shown in Fig. 3, figure S1 and figure S2, which is consistent with previous research.
Association between NHHR and CKD in different BMI group. Adjustment factors included age, sex, race, education attainment, marital status, PIR, smoking status, drinking status, hypertension, DM, CVD, dietary cholesterol intake and total cholesterol.
Discussion
This study focused on exploring the correlation between the NHHR and CKD among elderly Americans. Analyzing data from 3,715 subjects, the study found that elevated NHHR levels correlated significantly with heightened CKD risk, persisting even after adjustments for confounding variables. Employing smoothed curve fitting and threshold effect analysis, a J-shaped curve was identified in the relationship between NHHR and CKD, with a notable inflection point occurring at 1.8. Further analyses, including subgroup assessments and interaction tests, upheld the consistency of this relationship across various demographics, with BMI being the sole exception.
Over 10% of people worldwide suffer with CKD, making it a public health concern19,20. The incidence of CKD is progressively increasing year by year, with a higher prevalence seen among individuals aged over 601. By 2050, the percentage of the population that is elderly is predicted to rise from 10% in 2024 to 16%21. Kidney function declines with age, and aging is a major risk factor for CKD22,23. The incidence of chronic conditions like diabetes, hypertension, and cardiovascular diseases has increased with population aging, which has aided in the development of CKD24,25,26. Thus, the link between NHHR and CKD in the senior population is the main focus of this study. It is especially crucial to focus on the assessment, diagnosis, and treatment of CKD in elderly.
One important risk factor for chronic kidney disease (CKD) is dyslipidemia, which is characterized by elevated TG, LDL-C, TC, or decreased HDL-C27,28,29. A cohort study involving 1,891 adults in China found a substantial link between a heightened NHHR and increased CKD risk, even after adjusting for confounders like uric acid. This relationship was especially strong among women, individuals who are overweight, and those with hypertension30. A similar association was noted in research by Wen et al., which further supported a positive relationship between NHHR and CKD28. These results are supported by Korean research that found a nonlinear U-shaped curve between non-HDL-c levels and CKD31. This highlights even more how crucial NHHR is for clinical CKD assessment and treatment, as well as its use as a prognostic marker.
Extensive research indicates that disturbances in lipid metabolism contribute to the development and progression of CKD. Cholesterol metabolism is an important component of lipid metabolism32. Compared with healthy individuals, lipid characteristics of CKD patients usually show low levels of HDL-C33. HDL-C, known for its role in the reverse transport of cholesterol and its antioxidant capabilities, helps decrease the occurrence of atherosclerosis and CVD risk34,35. In a prospective U.S. study, lower levels of HDL-C were linked with a 40% and 77% increased risk of stroke and myocardial infarction, respectively, among CKD patients36. Conversely, elevated LDL-C levels and subsequent foam cell formation enhance the release of proinflammatory cytokines, which intensify inflammation. This process can impair renal function by altering lipid metabolism and promoting oxidative stress. Furthermore, LDL-C has been found to be a distinct determinant of renal illness, which may be associated with reduced plasma levels of the enzyme lecithin cholesterol acyltransferase (LCAT), which is critical for the elimination of cholesterol from the bloodstream. In CKD, one manifestation of abnormal lipid metabolism is the ectopic deposition of lipids within the kidney37. Such fat deposits are commonly found in the perirenal space, renal sinus, and renal parenchyma. Adipocytes located in these areas release proinflammatory adipokines, leading to renal inflammation, fibrosis, and dysfunction. These pathological conditions are linked with renal-cell damage, glomerulosclerosis, interstitial fibrosis, and proteinuria38. A cross-sectional study by Krievina et al. demonstrated that serum levels of kidney injury molecule-1 (sKIM-1) and fibroblast growth factor-21 (FGF-21) are associated with ectopic fat deposition in the kidney. sKIM-1 and FGF-21 are markers of renal injury associated with abnormal lipid metabolism39.
Subgroup analysis indicated an elevated prevalence of CKD among individuals possessing a BMI exceeding 30, underscoring the substantial influence of obesity on CKD progression. Research supports that both obesity and metabolic syndrome independently increase the risks associated with CKD and end-stage renal disease (ESRD). A comprehensive cohort investigation conducted by Hsu et al. confirmed that individuals classified as obese face a heightened risk of progressing to ESRD relative to their normal-weight counterparts, identifying high BMI as a potent risk factor40. Research conducted by Iseki et al. in Japan found a significant relationship between BMI levels and the prevalence of CKD among older adults, suggesting that maintaining a healthy body weight could lower the risk of this disease41. The link between obesity and CKD is affected by factors such as insulin resistance, lipotoxicity, adipocytokine dysregulation, and high blood pressure, making weight control and a healthy lifestyle crucial for CKD patients42,43,44.
The study has several strengths. Firstly, the use of the NHANES database provides a large, representative sample collected through standardized procedures, significantly reducing error. Thus, study results can be reliably extrapolated to the entire U.S. population using appropriate sampling weights. Secondly, the inclusion of extensive covariates such as demographic data, laboratory tests, and questionnaire responses enhanced the credibility of the results. Additionally, subgroup analysis and interaction tests were undertaken to explore variability among diverse demographic groups. Nevertheless, this study faces several limitations. Primarily, its cross-sectional design necessitates subsequent longitudinal studies to confirm a causal link between NHHR and CKD. Secondly, although adjustments were made for numerous critical covariates, residual confounding factors remain unaccounted for. Lastly, the reliance on NHANES data, which focuses on the U.S. population, may limit the applicability of the findings to other global populations or ethnic cohorts.
The J-shaped relationship between NHHR and CKD is an important finding of this study. Our research indicates that both excessively high and low levels of NHHR are associated with an increased prevalence of CKD, particularly in obese patients (BMI > 30). At the public health policy level, regular monitoring of NHHR may help identify high-risk populations in a timely manner to protect their kidney health. At the same time, patients should be encouraged to exercise actively, control their weight, and strive to maintain NHHR at optimal levels, which may help reduce the incidence of CKD.
Conclusion
This study indicates a link between elevated NHHR and a heightened risk of CKD among U.S. residents aged over 60, particularly among obese individuals (BMI > 30). It suggests that lipid management could be crucial for preventing CKD and highlights the need for increased focus on renal health in those with elevated NHHR levels. Further large-scale, prospective studies are needed to confirm these findings.
Data availability
All data for this cross-sectional study were obtained from the official NHANES website, and readers can access the data free of charge from the website (https:// www. cdc. gov/ nchs /nhanes / index. htm).
References
Jha, V. et al. Chronic kidney disease: global dimension and perspectives. Lancet 382, 260–272. https://doi.org/10.1016/s0140-6736(13)60687-x (2013).
Evans, M. et al. A narrative review of chronic kidney disease in clinical practice: current challenges and future perspectives. Adv. Ther. 39, 33–43. https://doi.org/10.1007/s12325-021-01927-z (2022).
Kurniawan, A. L. et al. Association between metabolic parameters and risks of anemia and electrolyte disturbances among stages 3–5 chronic kidney disease patients in Taiwan. BMC Nephrol. 22, 385. https://doi.org/10.1186/s12882-021-02590-w (2021).
Levey, A. S. et al. Chronic kidney disease as a global public health problem: approaches and initiatives - a position statement from kidney disease improving global outcomes. Kidney Int. 72, 247–259. https://doi.org/10.1038/sj.ki.5002343 (2007).
Global & national burden of chronic kidney disease. 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet 395, 709–733. https://doi.org/10.1016/s0140-6736(20)30045-3 (2020).
Ejerblad, E. et al. Obesity and risk for chronic renal failure. J. Am. Soc. Nephrol. 17, 1695–1702. https://doi.org/10.1681/asn.2005060638 (2006).
Brennan, E., Kantharidis, P., Cooper, M. E. & Godson, C. Pro-resolving lipid mediators: regulators of inflammation, metabolism and kidney function. Nat. Rev. Nephrol. 17, 725–739. https://doi.org/10.1038/s41581-021-00454-y (2021).
Mitrofanova, A., Merscher, S. & Fornoni, A. Kidney lipid dysmetabolism and lipid droplet accumulation in chronic kidney disease. Nat. Rev. Nephrol. 19, 629–645. https://doi.org/10.1038/s41581-023-00741-w (2023).
Kim, S. W. et al. Non-HDL-cholesterol/HDL-cholesterol is a better predictor of metabolic syndrome and insulin resistance than apolipoprotein B/apolipoprotein A1. Int. J. Cardiol. 168, 2678–2683. https://doi.org/10.1016/j.ijcard.2013.03.027 (2013).
Fan, Y. Q. et al. The non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio as a predictive indicator of CKD risk in NAFLD patients: NHANES 2017–2020. Front. Nutr. 11, 1501494. https://doi.org/10.3389/fnut.2024.1501494 (2024).
Wang, J., Li, S., Pu, H. & He, J. The association between the non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio and the risk of osteoporosis among U.S. Adults: analysis of NHANES data. Lipids Health Dis. 23, 161. https://doi.org/10.1186/s12944-024-02152-7 (2024).
Wu, J. & Guo, J. Non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio (NHHR) and hypertension in American adults: a NHANES cross-sectional study. Front. Physiol. 15, 1398793. https://doi.org/10.3389/fphys.2024.1398793 (2024).
Tan, M. Y. et al. The association between non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio with type 2 diabetes mellitus: recent findings from NHANES 2007–2018. Lipids Health Dis. 23, 151. https://doi.org/10.1186/s12944-024-02143-8 (2024).
Lu, J. D. et al. Association between serum neurofilament light chain levels and chronic kidney disease: a cross-sectional population-based study from the National health and nutrition examination survey (2013–2014 cycle). Ren. Fail. 46, 2427178. https://doi.org/10.1080/0886022x.2024.2427178 (2024).
Hu, H. et al. Non-High-Density lipoprotein cholesterol and risk of cardiovascular disease: the Japan epidemiology collaboration on occupational health study. J. Atheroscler Thromb. 29, 1295–1306. https://doi.org/10.5551/jat.63118 (2022).
Cheng, Q. et al. Association of non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio (NHHR) and gallstones among US adults aged ≤ 50 years: a cross-sectional study from NHANES 2017–2020. Lipids Health Dis. 23, 265. https://doi.org/10.1186/s12944-024-02262-2 (2024).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150, 604–612. https://doi.org/10.7326/0003-4819-150-9-200905050-00006 (2009).
KDIGO 2021 Clinical practice guideline for the management of glomerular diseases. Kidney Int. 100, S1–s276, doi:https://doi.org/10.1016/j.kint.2021.05.021 (2021).
Hill, N. R. et al. Global prevalence of chronic kidney disease - A systematic review and meta-analysis. PLoS One. 11, e0158765. https://doi.org/10.1371/journal.pone.0158765 (2016).
[Guidelines for the early evaluation. And management of chronic kidney disease in China. Zhonghua Nei Ke Za Zhi. 62, 902–930. https://doi.org/10.3760/cma.j.cn112138-20221013-00755 (2023).
Chesnaye, N. C., Ortiz, A., Zoccali, C., Stel, V. S. & Jager, K. J. The impact of population ageing on the burden of chronic kidney disease. Nat. Rev. Nephrol. 20, 569–585. https://doi.org/10.1038/s41581-024-00863-9 (2024).
Denic, A. et al. The substantial loss of nephrons in healthy human kidneys with aging. J. Am. Soc. Nephrol. 28, 313–320. https://doi.org/10.1681/asn.2016020154 (2017).
Denic, A., Rule, A. D. & Glassock, R. J. Healthy and unhealthy aging on kidney structure and function: human studies. Curr. Opin. Nephrol. Hypertens. 31, 228–234. https://doi.org/10.1097/mnh.0000000000000780 (2022).
Alicic, R. Z., Rooney, M. T. & Tuttle, K. R. Diabetic kidney disease: challenges, progress, and possibilities. Clin. J. Am. Soc. Nephrol. 12, 2032–2045. https://doi.org/10.2215/cjn.11491116 (2017).
Ku, E., Lee, B. J., Wei, J. & Weir, M. R. Hypertension in CKD: core curriculum 2019. Am. J. Kidney Dis. 74, 120–131. https://doi.org/10.1053/j.ajkd.2018.12.044 (2019).
Zoccali, C. & Mallamaci, F. The cardiovascular-renal link and the health burden of kidney failure. Eur. Heart J. 44, 1167–1169. https://doi.org/10.1093/eurheartj/ehad039 (2023).
Baragetti, A. et al. High density lipoprotein cholesterol levels are an independent predictor of the progression of chronic kidney disease. J. Intern. Med. 274, 252–262. https://doi.org/10.1111/joim.12081 (2013).
Wen, J. et al. Association of the TG/HDL-C and Non-HDL-C/HDL-C ratios with chronic kidney disease in an adult Chinese population. Kidney Blood Press. Res. 42, 1141–1154. https://doi.org/10.1159/000485861 (2017).
Lyu, K. et al. The effect of blood lipid profiles on chronic kidney disease in a prospective cohort: based on a regression discontinuity design. Biomed. Environ. Sci. 37, 1158–1172. https://doi.org/10.3967/bes2024.113 (2024).
Zuo, P. Y. et al. Non-HDL-cholesterol to HDL-cholesterol ratio as an independent risk factor for the development of chronic kidney disease. Nutr. Metab. Cardiovasc. Dis. 25, 582–587. https://doi.org/10.1016/j.numecd.2015.03.003 (2015).
Suh, S. H. et al. Non-High-Density lipoprotein cholesterol and progression of chronic kidney disease: results from the KNOW-CKD study. Nutrients 14 https://doi.org/10.3390/nu14214704 (2022).
Noels, H., Lehrke, M., Vanholder, R. & Jankowski, J. Lipoproteins and fatty acids in chronic kidney disease: molecular and metabolic alterations. Nat. Rev. Nephrol. 17, 528–542. https://doi.org/10.1038/s41581-021-00423-5 (2021).
Lamprea-Montealegre, J. A. et al. Chronic kidney disease, lipids and apolipoproteins, and coronary heart disease: the ARIC study. Atherosclerosis 234, 42–46. https://doi.org/10.1016/j.atherosclerosis.2014.02.006 (2014).
Soppert, J., Lehrke, M., Marx, N., Jankowski, J. & Noels, H. Lipoproteins and lipids in cardiovascular disease: from mechanistic insights to therapeutic targeting. Adv. Drug Deliv Rev. 159, 4–33. https://doi.org/10.1016/j.addr.2020.07.019 (2020).
Brewer, H. B. Jr., Remaley, A. T., Neufeld, E. B., Basso, F. & Joyce, C. Regulation of plasma high-density lipoprotein levels by the ABCA1 transporter and the emerging role of high-density lipoprotein in the treatment of cardiovascular disease. Arterioscler. Thromb. Vasc Biol. 24, 1755–1760. https://doi.org/10.1161/01.ATV.0000142804.27420.5b (2004).
Bajaj, A. et al. Lipids, apolipoproteins, and risk of atherosclerotic cardiovascular disease in persons with CKD. Am. J. Kidney Dis. 73, 827–836. https://doi.org/10.1053/j.ajkd.2018.11.010 (2019).
Mende, C. & Einhorn, D. Fatty kidney disease: the importance of ectopic fat deposition and the potential value of imaging. J. Diabetes. 14, 73–78. https://doi.org/10.1111/1753-0407.13232 (2022).
Jiang, Z. et al. Obesity and chronic kidney disease. Am. J. Physiol. Endocrinol. Metab. 324, E24–e41. https://doi.org/10.1152/ajpendo.00179.2022 (2023).
Krievina, G. et al. Ectopic adipose tissue storage in the left and the right renal sinus is asymmetric and associated with serum kidney injury Molecule-1 and fibroblast growth Factor-21 levels increase. EBioMedicine 13, 274–283. https://doi.org/10.1016/j.ebiom.2016.10.020 (2016).
Hsu, C. Y., McCulloch, C. E., Iribarren, C., Darbinian, J. & Go, A. S. Body mass index and risk for end-stage renal disease. Ann. Intern. Med. 144, 21–28. https://doi.org/10.7326/0003-4819-144-1-200601030-00006 (2006).
Iseki, K. et al. Body mass index and the risk of development of end-stage renal disease in a screened cohort. Kidney Int. 65, 1870–1876. https://doi.org/10.1111/j.1523-1755.2004.00582.x (2004).
Rutkowski, P., Klassen, A., Sebekova, K., Bahner, U. & Heidland, A. Renal disease in obesity: the need for greater attention. J. Ren. Nutr. 16, 216–223. https://doi.org/10.1053/j.jrn.2006.04.017 (2006).
Arabi, T. et al. Obesity-related kidney disease: beyond hypertension and insulin-resistance. Front. Endocrinol. (Lausanne). 13, 1095211. https://doi.org/10.3389/fendo.2022.1095211 (2022).
Nakashima, A., Kato, K., Ohkido, I. & Yokoo, T. Role and treatment of insulin resistance in patients with chronic kidney disease: A review. Nutrients 13 https://doi.org/10.3390/nu13124349 (2021).
Acknowledgements
We thank all the staff who worked hard on the NHANES project, and all the volunteers who participated in the survey.
Funding
This study was supported by the National Natural Science Foundation of China (Item No: 82370777), Major Project of Jiangsu Commission of Health: Basic research and clinical application of biomarkers for early diagnosis of prostate cancer (Item No: ZD2021002), Wuxi Taihu Talent Plan, Team in Medical and Health High-level Talents Project: Basic and clinical study of urothelial carcinoma of upper urinary tract (Item No: THRCJH20200406).
Author information
Authors and Affiliations
Contributions
Ninghan Feng designed this study and provided guidance. Zhou Li and Yifan Sun conducted data collection, data analysis, data interpretation for this study. Yifan Sun drafted the initial manuscript. All authors reviewed the manuscript, provided comments and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The NHANES protocols received approval from the National Center for Health Statistics Ethics Review Board of the U.S. CDC, and all participants provided written informed consent during the survey.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, Y., Li, Z. & Feng, N. Association of non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio and chronic kidney disease in elderly insights from NHANES. Sci Rep 15, 12611 (2025). https://doi.org/10.1038/s41598-025-96299-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96299-7
Keywords
This article is cited by
-
Exposure to ambient air pollutants and incident chronic kidney disease in patients with hypertension: a CHARLS-based study
International Urology and Nephrology (2026)