Abstract
Gas explosion is a recurrent event in coal mining that cause severe spleen damage. This study aimed to investigate the role and mechanism of oxidative stress in gas explosion-induced spleen injury. 120 male Sprague–Dawley (SD) rats were randomly divided into a control group (NC), a gas explosion-induced spleen injury model group (Model), an Nrf2 inhibitor group (Model + ATRA), and an Nrf2 induction group (Model + TBHQ). After explosion, the rats of the inhibitor group and induction group were immediately given intraperitoneal injection of all-trans-retinoicacid (ATRA, 5 mg/kg) or tertiary butylhydro-quinone (TBHQ, 1 mg/kg) once. Then, the rats were anesthetized with blood taken from the abdominal aorta at 24 h, 72 h and 7 days. The results showed that gas explosion reduced the spleen index. The expression of oxidative stress-related genes and proteins Nrf2, HO-1, COX2 and GPX4 were increased significantly (P < 0.05) after gas explosion. Compared with the model group, TBHQ improved the spleen index, and reduced inflammation. Moreover, the expression of inflammatory factor IL-6 and ROS was decreased (P < 0.05), HMOX1 and the expression of oxidative stress-related genes and proteins were increased (P < 0.05), but the opposite results were observed in the inhibitor group. Taken together, we firstly found that TBHQ may regulate the degree of oxidative stress in spleen injury induced by gas explosion through the Nrf2/HO-1 signaling pathway.
Similar content being viewed by others
Introduction
Gas explosions are a prevalent hazard during coal mining. When the concentration of gas in the atmosphere reaches a certain level and comes into contact with an open flame, it can result in an explosion. The incidence of gas explosions can cause casualties and considerable economic losses and even restrict the development of the coal industry1. Gas explosions can generate shock waves, high-temperature flames, and toxic gases2, which can worsen multiple injuries. Previous studies have demonstrated that gas explosions can lead to multiorgan injuries, affecting the nervous system, lungs, kidneys, and spleen3,4. The spleen is the largest immune organ in the human body, and its fragile texture increases susceptibility to injury during gas explosions. In recent decades, significant changes have occurred in the treatment of splenic trauma due to concerns about late complications such as impaired immune function and postoperative infections after splenectomy. Nonsurgical treatments are currently preferred, with the aim of preserving the spleen and its functionality5. Recent clinical studies indicate that trauma patients with traumatic brain injury (TBI) and concomitant spleen injury who undergo splenectomy exhibit a more severe injury profile, a more compromised hemodynamic status, and higher in-hospital mortality rates compared to patients without splenectomy6. Indeed, one study found that the spleen contributes to chronic restraint stress-induced lung injury through splenic CD11b + cells7. The timely implementation of spleen-preserving treatment is crucial for the prognosis of patients with gas explosion-induced spleen injury. Furthermore, understanding the molecular mechanisms underlying the pathogenesis of the early stage of spleen injury is a key research direction in the search for early treatment methods for spleen preservation.
Oxidative stress is a physiological response that occurs when the body is exposed to harmful external and internal environments, leading to the production of reactive oxygen species (ROS) and reactive nitrogen species. The accumulation of ROS in cells can result in an imbalance that severely damages proteins, lipids, and DNA8. Previous studies have shown that oxidative stress is mainly centered around the superoxide, and scholars have consequently focused on studying the pathological sources of this oxygen-derived free radical and the types of molecular damage it may cause, as well as the protection provided by antioxidant enzymes.
In recent years, oxidative stress has been recognized as a pathological response resulting from imbalances in genetic factors or dysregulation of gene expression. The center of this new focus is the E2-related factor 2 (Nrf2), a nuclear factor widely recognized as the pivotal regulator of the antioxidant response9. Nrf2 regulates hundreds of genes and activates antioxidant responsive element (ARE)-dependent gene expression by binding with antioxidant AREs to form the Nrf2/ARE complex. This complex plays a crucial physiological role in anti-inflammation, antioxidation, detoxification, autophagy, and proteasome functions10. Tertiary-butylhydroquinone (TBHQ) is an approved food-grade phenolic antioxidant with antioxidant activity in a variety of cells and tissues, many studies have shown that TBHQ reduces oxidative stress levels in mammals, which can be used as an activator of Nrf211. TBHQ, an Nrf2 agonist, primarily mitigates oxidative stress through the following mechanisms: First, Nrf2 is a key transcription factor responsible for activating gene expression in response to oxidative stress. Upon exposure to a multitude of stimuli, Nrf2 translocates from the cytoplasm to the nucleus, where it binds to antioxidant response elements (AREs). Thus, TBHQ exerts its antioxidative effects by activating Nrf2 and promoting ARE-mediated transcription. Second, TBHQ directly activates AREs/electrophile response elements, and the resulting increase in gene expression further protects against excessive oxidative stress12. In vitro, Nrf2 activated by TBHQ can inhibit the expression of cytokine-induced pro-inflammatory genes and oxidative stress genes, change macrophage phenotype, and promote autophagy activity13. Our team successfully established an animal model of gas explosion injury in a real tunnel environment. In this study, we explored from a new perspective whether the Nrf2/HO1 signaling pathway plays an antioxidant and protective role in gas explosion-induced spleen tissue injury. The aim of this study was to provide a therapeutic basis for the early clinical treatment of spleen injury caused by gas explosion.
Results
Effects of oxidative stress on the spleen index in rats exposed to gas explosion
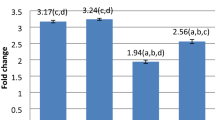
We quantified the changes in the rats spleen indices to directly determine the extent of spleen atrophy and indirectly indicate the status of the immune system. As shown in Fig. 1, the spleen indices of the rats at 24 h, 72 h, and 7 d post-explosion were 46.3%, 52.9%, and 61.9%, respectively, of those in the NC group. Following treatment with the ATRA inhibitor, the corresponding values were reduced to 40.3%, 37.5%, and 40.6% relative to those of the NC group at equivalent time points post-explosion. Conversely, administration of the agonist TBHQ resulted in spleen indices of 49.3%, 60.3%, and 68.9% compared to those of the NC group at the indicated time intervals post-explosion. This finding suggests that severe spleen atrophy occurs in rats after gas explosion, but the TBHQ activator can mitigate this effect by indirectly reducing immune function damage.
Effects of oxidative stress on spleen index after gas explosion in rats. The change of spleen index after gas explosion at the 24 h, 72 h and 7 day.
Changes in Splenic tissue injury in rats after gas explosion
No fatalities were observed in the rat specimens 220 m away from the explosion. According to the histological analysis based on HE staining, the spleen structure in the NC group was normal and intact, with neatly arranged cells and plump nuclei. A clear demarcation was observed between the red pulp (RP) and white pulp (WP), and the WP contained abundant lymphocytes with a dense arrangement. Inflammatory bleeding, decreased lymphocytes, loose arrangement, blurred cell structure, disappearance of some cell nuclei, and diffuse necrosis were observed in the spleen tissue of the rats in the Model group. Compared with the Model group, the inhibitor group showed increased damage to the spleen and infiltration of inflammatory cells in multiple locations and a large number of vacuoles were formed (As indicated by the arrow), while the activator group displayed milder spleen injury with reduced congestion and hemorrhage in the RP (Fig. 2A). Quantitative results show that the proportion of WP in NC group at 24 h, 72 h, and 7 days was 28.7%, 31% and 29%, respectively. And decreased to 10.3%, 11.7% and 13.6% at 24 h, 72 h, and 7 days after gas explosion. When treated with TBHQ, the proportion of WP was 15.8%, 22% and 25%, respectively (Fig. 2B). Taken together, these data indicated that TBHQ can alleviate spleen injury caused by gas explosion.
Effects of gas explosion on spleen damage in rats. (A) HE staining results on spleen structure in each group (HE×100). The yellow arrow indicated vacuoles formed by gas explosion; (B) The quantitative analysis of white pulp. *P < 0.05, **P < 0.01.
Changes in inflammatory factors and injury markers in the spleens of rats
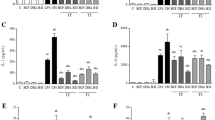
The expression levels of inflammatory factors are shown in Fig. 3. The levels of IL-6 and IL-10 in the spleens of rats in the Model group were significantly greater (P < 0.01) than those in the NC group. Meanwhile, in contrast to the Model group, the inhibitor group exhibited a notable increase in the expression level of IL-6 (P < 0.05). The expression level of IL-10 did not significantly change at 24 h, but it significantly decreased at 72 h and 7 d. The agonist TBHQ significantly downregulated the expression of IL-6 (P < 0.05) and upregulated the expression of the anti-inflammatory factor IL-10 (P < 0.01). The levels of the injury marker ROS and the oxidative stress regulatory factor HMOX1 in the spleens of the Model group were significantly greater (P < 0.05) than those in the spleens of the NC group. In contrast to the findings for the Model group, the ROS level in the inhibitor group significantly increased (P < 0.05), while that in the HMOX1 group significantly decreased (P < 0.05). The level of ROS in the agonist group significantly decreased compared with that in the Model group, while that in the HMOX1 group significantly increased (P < 0.05).
Effects of gas explosion on spleen inflammatory and injury factors in rats. The levels of the inflammatory factors IL-6 and IL-10 and the injury factors ROS and HMOX1 in the rat spleen were determined by ELISA. * P < 0.05, **P < 0.01.
Nrf2/HO-1 signaling pathway resists oxidative stress injury in gas explosion
Nrf2 plays a crucial role in cellular defense against oxidative stress. Western blotting and RT‒qPCR revealed significant upregulation of Nrf2 and HO-1 protein and mRNA expression in the Model group (P < 0.05) (Fig. 4). However, compared to those in the activator group, the Nrf2 and HO-1 protein and mRNA levels in the inhibitor group were substantially lower (P < 0.05) (Fig. 4).
Expression changes of genes and proteins related to oxidative stress pathways. (A, B) The expression changes of genes by qPCR; (C, D) The expression changes of proteins by Western blot. * P < 0.05, **P < 0.01.
Discussion
Gas explosion-induced injury represents a complex multiorgan trauma primarily affecting the nervous system, lungs, and spleen, among other organs14,15. The etiology of this condition remains poorly understood, and specific treatments are currently lacking. Furthermore, due to the inherent difficulties in acquiring clinical data on human gas explosion injuries, research involving relevant animal models is limited, and the underlying molecular mechanisms remain largely unelucidated16,17.
In recent years, research on nonapoptotic programmed cell death has gradually garnered widespread attention. The excessive release of ROS from damaged mitochondria, leading to oxidative stress, may be a central mechanism in tissue and cell injury18,19,20. Oxidative stress is a cellular state characterized by an imbalance between the production of reactive oxygen species and the capacity of antioxidant defense systems, often triggered by exposure to harmful stimuli. It is considered a pathological condition resulting from dysregulation of gene expression21. As a key activator of the antioxidant response element, Nrf2 regulates the expression of numerous genes, mediating physiological processes such as anti-inflammation and antioxidation. Our team successfully established spleen injury model of rat simulating gas explosions in a real tunnel environment. This study aimed to explore whether the Nrf2/HO1 signaling pathway plays a role in antioxidant protection against spleen tissue injury caused by gas explosion from a novel perspective. The aim of this study was to provide a basis for the early clinical treatment of gas explosion-induced splenic injury.
Recent studies have demonstrated that upon stimulation by external factors, Nrf2 dissociates from Keap1 and enters the nucleus, where it binds to the sMaf protein, activating ARE and enhancing Nrf2-mediated regulation of HO-1 and NQO1 transcription22. Within the antioxidant defense system, Nrf2 can bind to AREs to modulate antioxidant responses in nerve cells, safeguarding against oxidative damage. Moreover, HO-1 and NQO-1 contribute to cellular antioxidant responses while promoting axonal regeneration23 and suppressing the expression of tumor necrosis factor α and IL-1β, thus inhibiting inflammatory reactions24. Wang et al.25 found that treatment with sagebrush phenol significantly downregulated the expression of NF-κB and COX-2 in rats with spinal cord injury, and upregulated the expression of Nrf2 to regulate key proteins involved in inflammation and redox status, which effectively inhibited oxidative stress and the inflammatory response induced by spinal cord injury. In our study, severe inflammatory reactions were observed in the spleens of rats in the Model group. This phenomenon was accompanied by the upregulation of inflammatory factors such as IL-6, ROS (markers of tissue injury) and HMOX1 (an oxidative stress regulator). However, the expression levels of the inflammatory factor IL-6 and injury marker ROS were significantly downregulated in the TBHQ-treated group, while significant upregulation was detected in the expression of the anti-inflammatory factor IL-10, the oxidative stress regulator HMOX1, and the pathway-related genes Nrf2, HO-1, COX2, and GPX4.
This study investigates the regulatory effects of TBHQ on oxidative stress via the Nrf2/HO-1 signaling pathway, thereby mitigating spleen injury induced by gas explosions in a rat model. While several mechanisms have been elucidated, explosion incidents elicit a complex interplay of factors beyond oxidative stress, and the present study did not comprehensively explore these additional mechanisms. Future research should aim to address this limitation through a more in-depth investigation.
Conclusions
In summary, TBHQ may modulate oxidative stress-induced injury in the rat spleen following gas explosion via the Nrf2/HO-1 signaling pathway, thereby attenuating the inflammatory response and alleviating the severity of spleen injury. Future investigations will focus on elucidating the molecular mechanisms underlying the amelioration of gas explosion-induced oxidative stress in the rat spleen, with the ultimate aim of identifying novel therapeutic targets for gas explosion-related spleen injuries.
Methods
Instruments and equipment
CIT-2MK infrared temperature sensor (Institute of Automation, Chinese Academy of Sciences): temperature measurement range: 400 ~ 2000℃, response time: 67 ms, application range: 0 ~ 60℃; The CYG series solid-state piezoresistive pressure sensors (Baoji Sensor Research Institute of Shaanxi Qinming Electronics Group Co., Ltd.) have ranges of 0.2 MPa and 0.1 MPa, applicable range: -40 ~ 80℃, output signal range: 0 ~ 5 V; Flame sensor (Baoji Sensor Research Institute of Shaanxi Qinming Electronics Group Co., Ltd.); CS20000 dynamic testing and analysis system (Chengdu Taist Electronic Information Co., Ltd.); BX60 fluorescence microscope (Olympus, Japan); 120 A electronic analytical balance (Drecisa, Switzerland); LS-O410 constant temperature oven (Fisher Scientific, USA); OFT-100 Rat Opening Activity Experimental System (Chengdu Taimeng Software Co., Ltd.); The gas explosion large-scale tunnel test system (Chongqing Research Institute Co., Ltd. of China Coal Science and Industry Group) is shown in Fig. 5.
Layout of large-scale real tunnel test system. (1) Tunnel entrance; (2) Auxiliary section; (3) Protective section; (4) Hydraulic explosion-proof door; (5) Detonating chamber; (6) Valve circulation system; (7) Ignition system; (8) Flat tunnel; (9) Comprehensive explosion test system; (10) Ceiling box; 11. Inclined tunnel; 12. Tunnel exit.
Establishment of the rat gas explosion model and grouping
A total of 120 healthy male Sprague‒Dawley (SD) rats were purchased from the Experimental Animal Center of Daping Hospital, Army Medical University (Chongqing, China). The rats were randomly divided into four groups (10/group, total 30 in three time points) as follows: the control group (NC), the gas explosion model group (Model), the Nrf2 inhibitor group (Model + ATRA), and the Nrf2 activator group (Model + TBHQ). The rats in the model and intervention groups were placed in the tunnel at a distance of 220 m from the ignition point for the gas explosion test, following the specific procedures described in reference. In the Model + ATRA group, the rats received a daily intraperitoneal injection of ATRA at a dose of 5 mg/kg26 after gas explosion. In the Model + TBHQ group, the rats received a daily intraperitoneal injection of 12.5 mg/kg TBHQ27 after the gas explosion. The rats in the NC and Model groups were administered an equivalent volume of PBS. At 24 h, 72 h, and 7 days after the intervention, 10 rats from each group were deeply anesthetized using 1% pentobarbital sodium28, and blood samples were collected from the abdominal aorta. Animals were humanely euthanized by intraperitoneal injection of 2.5% Avertin (50 ml/kg). The spleen tissues were then harvested, a timeline of the specific experimental design is shown in Fig. 6. This study was approved by the Ethics Committee of Xinxiang Medical University (Approval No.: XYLL-2020007). All animal procedures were performed in accordance with the ARRIVE guidelines for animal research.
A timeline of specific experimental design for the animals.
Detection of the Splenic tissue index in rats
The test rats were weighed at the designated time points and sacrificed under sodium pentobarbital anesthesia at the 48 h after gas explosion29, after which the intact spleen was removed for weighing. The splenic index was calculated as the spleen mass (mg) divided by the body weight (g).
Hematoxylin–eosin (HE) staining
After the mice were euthanized by abdominal aortic anesthesia, the spleen tissue was removed and fixed with 4% paraformaldehyde in liquid nitrogen bottles for pathological sectioning. The spleen tissue was embedded in paraffin, cut into 4 μm thick slices, and sliced at 60 °C for 60 min. After gradient dewaxing with xylene and gradient hydration with alcohol, the slices were stained with HE and sealed. The degree of spleen tissue injury was observed under a 100× optical microscope.
Enzyme-linked immunosorbent assay (ELISA)
The levels of the inflammatory factors IL-6 (Cat. No: RK00020, ABclonal, Wuhan, China) and IL-10 (Cat. No: RK00050, ABclonal) and the injury factors ROS (Cat. No: RK15283, ABclonal) and HMOX1 (Cat. No: RK03723, ABclonal) in the rat spleen were determined by ELISA. Spleen tissues from each group were collected and homogenized with lysate. After centrifugation at 4 °C and 12,000 rpm for 10 min, the supernatant was collected. All procedures were performed strictly following the kit instructions. The optical densities of standard substances at different concentrations were measured at a wavelength of 450 nm using an ELISA instrument to generate a standard curve for calculating the levels of related factors in the samples.
qRT‒PCR analysis
Two hundred milligrams of spleen tissue was weighed and rapidly pulverized in liquid nitrogen, followed by total RNA extraction using TRIzol (Invitrogen, Carlsbad, USA). Subsequently, the RNA concentration was determined, and cDNA synthesis was performed by reverse transcription following the protocol of the PrimeScript™ RT Master Mix kit. Real-time fluorescence quantitative PCR was performed according to the protocol provided by the Step One Plus Real Time PCR System kit. The fluorescence signals of the gene amplification products were detected, and relative gene expression was calculated using β-actin as the internal reference. The primers were synthesized by Sangon Biotech Co., Shanghai, China (Table 1). The qRT‒PCR conditions were as follows: 95 °C for 30 min, followed by 40 cycles of denaturation at 95 °C for 5 s and annealing/extension at 60 °C for 30 s. The obtained data were analyzed quantitatively using the 2−ΔΔCt method. The data for each sample were processed three times.
Western blot analysis
Two hundred milligrams of spleen tissue was combined with 1 mL of RIPA protein lysate. The tissue suspension was homogenized using an ultrasonic homogenizer and kept in an ice bath for 30 min, followed by centrifugation at 4 °C and 12,000 rpm for 20 min. The resulting supernatant was collected and quantified using a BCA kit. Subsequently, the target proteins were transferred to a PVDF membrane after SDS‒PAGE. After blocking with 5% non-fat milk powder, the membrane was washed with TBST and incubated overnight at 4 °C with primary antibodies β-actin (dilution of 1:3000, Abbkine, Wuhan, China, Cat. No: A01010), Nrf2 (dilution of 1:1000, Abclonal, Cat. No: A11159), HO-1 (dilution of 1:1000, Proteintech, Wuhan, China, Cat. No: 66743), COX2 (dilution of 1:1000, Abclonal, Cat. No: A1253), and GPX4 (dilution of 1:1000, Abclonal, Cat. No: A11243). Then, secondary antibodies (1:5000, Abbkine, Cat. No: A21020) were added and incubated at room temperature for 1 h. Finally, BCIP/NBT chromogenic plates (Sungyo, Shanghai, China) were developed and exposed using an automatic exposure machine. β-actin served as the internal reference. A computer software was utilized to calculate the gray values of both the target bands and the internal reference bands. All experiments were repeated three times.
Statistical analysis
The experimental data were analyzed and graphed using GraphPad Prism X7 software. The experimental results were expressed as mean ± standard error (mean ± SEM), and the t-test was employed for comparison between two groups. One-way ANOVA analysis was utilized for comparison among multiple groups. Dunnett T3’s post hoc test (data with unequal variance) or by Tukey’s post hoc test (data with equal variance) were performed when necessary. An alpha level of 0.05 was considered statistically significant for all analyses. *P < 0.05, **P < 0.01.
Data availability
The data that support the findings of this study are available on request from the corresponding author, Junhe Zhang, upon reasonable request.
References
Tian, L. Q. et al. The abnormalities of coagulation and fibrinolysis in acute lung injury caused by gas explosion. Kaohsiung J. Med. Sci. 36, 929–936 (2020).
Song, Y. & Zhang, Q. The quantitative studies on gas explosion suppression by an inert rock dust deposit. J. Hazard. Mater. 353, 62–69 (2018).
Zhang, Y. et al. Abnormality of the corpus callosum in coalmine gas explosion-related posttraumatic stress disorder. PLoS One. 10, e0121095 (2015).
Dong, X. et al. Short-term effect of gas explosion in real roadway environment on rats’ brain neural behavior. Wei Sheng Yan Jiu. 49, 889–894 (2020).
Coccolini, F. et al. Splenic trauma: WSES classification and guidelines for adult and pediatric patients. World J. Emerg. Surg. 12, 40 (2017).
Mader, M. M. et al. Traumatic brain injury with concomitant injury to the spleen: characteristics and mortality of a high-risk trauma cohort from the traumaregister DGU®. Eur. J. Trauma. Emerg. Surg. 48, 4451–4459 (2022).
Li, Y. et al. Spleen contributes to chronic restraint stress-induced lung injury through Splenic CD11b + cells. Int. Immunopharmacol. 126, 111258 (2024).
Costa, L. G. et al. Mechanisms of Neuroprotection by Quercetin: Counteracting Oxidative Stress and More. Oxid. Med. Cell. Longev. 2986796 (2016). (2016).
Zhu, L. et al. GPX4-Regulated Ferroptosis Mediates S100-Induced Experimental Autoimmune Hepatitis Associated with the Nrf2/HO-1 Signaling Pathway. Oxid. Med. Cell. Longev. 6551069 (2021). (2021).
Tu, W. et al. The Anti-Inflammatory and Anti-Oxidant mechanisms of the Keap1/Nrf2/ARE signaling pathway in chronic diseases. Aging Dis. 10, 637–651 (2019).
Song, H. et al. Tertiary butylhydroquinone alleviates gestational diabetes mellitus in C57BL/KsJ-Lep Db/++ mice by suppression of oxidative stress. J. Cell. Biochem. 120, 15310–15319 (2019).
Zhao, Y. L. et al. TBHQ-Overview of Multiple Mechanisms against Oxidative Stress for Attenuating Methamphetamine-Induced Neurotoxicity. Oxid. Med. Cell. Longev. 8874304 (2020). (2020).
Deng, S. et al. TBHQ attenuates ferroptosis against 5-fluorouracil-induced intestinal epithelial cell injury and intestinal mucositis via activation of Nrf2. Cell. Mol. Biol. Lett. 26, 48 (2021).
Svetlov, S. I. et al. Biomarkers of blast-induced neurotrauma: profiling molecular and cellular mechanisms of blast brain injury. J. Neurotrauma. 26, 913–921 (2009).
Hazell, G. A. et al. Injury scoring systems for blast injuries: a narrative review. Br. J. Anaesth. 128, 127–134 (2022).
Zhang, J. et al. Causation analysis of risk coupling of gas explosion accident in Chinese underground coal mines. Risk Anal. 39, 1634–1646 (2019).
Shao, F. et al. Burn wound bacteriological profiles, patient outcomes, and tangential excision timing: A prospective, observational study. Ostomy Wound Manage. 64, 28–36 (2018).
Sutradhar, S., Deb, A. & Singh, S. S. Melatonin attenuates diabetes-induced oxidative stress in spleen and suppression of splenocyte proliferation in laboratory mice. Arch. Physiol. Biochem. 128, 1401–1412 (2022).
Liu, F. et al. Difenoconazole causes spleen tissue damage and immune dysfunction of carp through oxidative stress and apoptosis. Ecotoxicol. Environ. Saf. 237, 113563 (2022).
Zhang, X. et al. N-acetyl-L-cysteine alleviated the oxidative stress-induced inflammation and necroptosis caused by excessive NiCl2 in primary spleen lymphocytes. Front. Immunol. 14, 1146645 (2023).
Li, D. L. et al. Quercetin protects retina external barrier from oxidative stress injury by promoting autophagy. Cutan. Ocul Toxicol. 40, 7–13 (2021).
Loboda, A. et al. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell. Mol. Life Sci. 73, 3221–3247 (2016).
Piermarini, E. et al. Frataxin Silencing alters microtubule stability in motor neurons: implications for Friedreich’s ataxia. Hum. Mol. Genet. 25, 4288–4301 (2016).
Li, D. et al. Zinc promotes functional recovery after spinal cord injury by activating Nrf2/HO-1 defense pathway and inhibiting inflammation of NLRP3 in nerve cells. Life Sci. 245, 117351 (2020).
Wang, Z. H. et al. Carnosol protects against spinal cord injury through Nrf-2 upregulation. J. Recept Signal. Transduct. Res. 36, 72–78 (2016).
Li, H. X. et al. Role of Keap1-Nrf2/ARE signal transduction pathway in protection of Dexmedetomidine preconditioning against myocardial ischemia/reperfusion injury. Biosci. Rep. 42, BSR20221306 (2022).
Wang, Z. et al. Tert-butylhydroquinone alleviates early brain injury and cognitive dysfunction after experimental subarachnoid hemorrhage: role of Keap1/Nrf2/ARE pathway. PLoS One. 9, e97685 (2014).
Chang, Y. et al. Palmitoylation regulates myelination by modulating the ZDHHC3-Cadm4 axis in the central nervous system. Signal. Transduct. Target. Ther. 9, 254 (2024).
Dong, X. et al. Evaluation of Gas Explosion Injury Based on Analysis of Rat Serum Profile by Ultra-Performance Liquid Chromatography/Mass Spectrometry-Based Metabonomics Techniques. Biomed. Res. Int. 8645869 (2020). (2020).
Acknowledgements
This work was supported by the Open Project Program of the Third Affiliated Hospital of Xinxiang Medical University (No. KFKTYB202108).
Funding
This work was supported by the Open Project Program of the Third Affiliated Hospital of Xinxiang Medical University (No. KFKTYB202108).
Author information
Authors and Affiliations
Contributions
MJ, ZJ and RW drafted the manuscript and completed the figures and tables; XL, QJ, MS, YS and LJ performed the data analysis; ZJ and RW revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was performed in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (Bethesda, MD, USA). Eighth Edition, 2010. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Xinxiang Medical University (XYLL-20220077).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ma, J., Zhang, J., Xi, L. et al. Tertiary butylhydroquinone regulates oxidative stress in spleen injury induced by gas explosion via the Nrf2/HO-1 signaling pathway. Sci Rep 15, 11987 (2025). https://doi.org/10.1038/s41598-025-97096-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-97096-y