Abstract
Delayed wound healing in diabetic foot ulcer (DFU) is a major cause of amputations, with ferroptosis impeding recovery. Dracorhodin (DP), a flavonoid from Dragon’s Blood, has shown anti-inflammatory and wound-healing properties, though its molecular mechanisms is unclear. This study investigates DP’s role in DFU treatment through bioinformatics and experimental approaches. A rat model of DFU was created with a high-fat/high-glucose diet and streptozotocin (STZ) induction, and wound healing was monitored after applying varying DP doses. Histopathological analysis and ELISA assessed tissue changes, inflammatory markers, and growth factors. Network pharmacology and molecular docking were used to identify core targets and pathways, while human umbilical vein endothelial cells (HUVECs) were used for in vitro testing. The results demonstrated that DP accelerated wound healing in DFU rats in a dose-dependent manner by enhancing collagen synthesis, angiogenesis, and growth factor levels, while simultaneously reducing inflammation and ROS levels. Network pharmacology and molecular docking analyses identified the Nrf2-mediated ferroptosis pathway as a potential key mechanism underlying DP’s therapeutic effects in DFU. In vitro experiments further revealed that DP improved cell viability and migration, while decreasing ROS and lipid peroxidation levels, effects attributed to Nrf2 pathway activation. These outcomes were significantly attenuated by the Nrf2 inhibitor ML385. In conclusion, DP promotes DFU healing via activation of the Nrf2 pathway and inhibition of ferroptosis.
Similar content being viewed by others
Introduction
Diabetic foot ulcer (DFU) is a serious complication of diabetes, characterized by chronic, non-healing wounds prone to infection, and is a leading cause of lower-limb amputations1. Epidemiological studies report that among 537 million individuals with diabetes mellitus globally2, 19–34% develop DFU, with approximately 20% of DFU cases necessitating lower extremity amputations3. Over 85% of foot-related amputations are directly attributed to DFU4. Mortality rates among DFU patients escalate sharply, reaching 13.1% at 1 year, 49.1% at 5 years, and 76.9% at 10 years post-diagnosis5. Notably, post-amputation mortality within 5 years remains alarmingly high, ranging from 53–91.7%6. Furthermore, DFU recurrence within 5 years of initial wound healing affects 65% of survivors7, reflecting the difficult nature of this disease. The healing disorders of DFU is due to multiple complex mechanisms: peripheral neuropathy drives insidious tissue injury progression through sensory deficits, motor impairment-driven deformities, and autonomic dysregulation; ischemic injury from combined macro-/microvascular insufficiency impedes reparative processes by limiting oxygen/nutrient delivery; while immune incompetence perpetuates infection susceptibility through compromised microbial clearance and sustains a pro-inflammatory state that disrupts regenerative cascades – collectively underpinning DFU chronicity and therapeutic recalcitrance8,9. Delayed wound healing in DFU heightens the risk of severe infections and amputation, highlighting the importance of accelerating healing to reduce these outcomes. Current standard therapy for DFU integrates glycemic/metabolic optimization, targeted infection control, vascular revascularization, advanced debridement with pressure offloading, and evidence-based bioengineered interventions within a multidisciplinary framework to promote healing and reduce amputation risks10,11. Despite advances in diabetic foot care, effective therapies specifically targeting DFU wound healing remain limited, emphasizing the urgent need for innovative treatment strategies12.
The delayed healing of DFU is driven by various pathological mechanisms, including peripheral neurovascular damage and an amplified inflammatory response. Recent research highlights ferroptosis, triggered by excessive oxidative stress in hyperglycemic conditions, as a critical factor impairing DFU healing13. In diabetes, prolonged hyperglycemia induces oxidative stress in ulcerated tissues, leading to the depletion of glutathione (GSH), a key precursor for GPX4 synthesis. This depletion leads to the accumulation of membrane lipid ROS, which, in excess, damages biomolecules like DNA, lipids, and proteins, initiating lipid peroxidation and ultimately causing ferroptosis. This process reduces the proliferative and migratory capacities of fibroblasts and keratinocytes, damages nerves and microvasculature, inhibits granulation tissue formation and epithelial cell growth, and ultimately hampers wound tissue repair and regeneration14,15.
Recent studies have revealed that phytomedicines possess the ability to prevent or repair various forms of cellular damage, including that observed in chronic wounds. Phytochemicals such as flavonoids, polyphenols, alkaloids, and terpenoids display multifunctional effects, including stimulating fibroblast proliferation, promoting collagen synthesis, enhancing cell migration and re-epithelialization, as well as displaying antimicrobial, antioxidant, anti-inflammatory, and immunomodulatory properties. These activities play a crucial role in chronic wound healing16. Dragon’s blood, a red resin obtained from the palm species Daemonorops draco, is a traditional Chinese medicinal plant. Research indicates that it is one of the most frequently referenced herbal remedies for tissue regeneration in classical Chinese medical literature17. Dragon’s blood is known for its ability to invigorate blood circulation, resolve blood stasis, stop bleeding, and promote tissue regeneration. Clinically, it has shown significant efficacy in enhancing the healing of DFU18. Dracorhodin (DP), the primary active compound in dragon blood, is considered a key marker for its therapeutic properties. The quality standards for dragon blood in the Chinese Pharmacopoeia are established based on its DP content, which is recognized for its effectiveness in tissue healing, wound repair, and the management of diabetes-related complications19,20. Due to its inherent instability and susceptibility to reduction, DP is commonly used in the form of Dracorhodin Perchlorate21. In our previous studies, we demonstrated that local application of DP accelerates wound healing in DFU rats22. However, the optimal dosage and underlying mechanisms remain unclear.
In this study, we established a DFU rat model to evaluate the efficacy of different concentrations of DP intervention on DFU. Rapidly advancing network pharmacology and molecular docking were employed to predict the potential mechanisms, which were further validated through in vitro cellular experiments. The results of this research are intended to provide additional evidence supporting the preclinical application of DP in promoting DFU wound healing.
Method and materials
Reagents and materials
DP (GC35898) was purchased from GLPBIO (USA). STZ (S6089) and sodium citrate buffer (C1013) were obtained from Macklin Biochemical Co., Ltd. (China). Dimethyl sulfoxide (DMSO) (D8371) was sourced from Solarbio Science & Technology Co., Ltd. (China). Recombinant human epidermal growth factor (rhEGF) was supplied by Huanov Gene Pharmaceutical Co., Ltd. (Guilin, China). ELISA kits for TNF-α (MM-0180R2), IL-6 (MM-0190R2), VEGF (MM-0807R2), PDGF (MM-0076R2), EGF (MM-0050R2), and ROS (MM-21264R2) were acquired from Enzyme Immunoassay Biotech Co., Ltd. (Wuhan, China). HUVECs (FH-H040) were purchased from Shanghai Fuheng Biological Technology Co., Ltd. (China). Cell Counting Kit-8 (CCK-8) (CK04) and 0.25% Trypsin-EDTA solution (C0201) were obtained from DoJinDo Molecular Technologies (Japan) and Shanghai Biyuntian Biotechnology Co., Ltd. (China), respectively. Phosphate-buffered saline (PBS) (C0221A) and complete extracellular matrix (ECM) medium (1001) were sourced from Sciencell Research Laboratories (USA). Transwell® inserts (3422) with polycarbonate membranes (6.5 mm diameter, 8.0 μm pore size) and Matrigel® (356234) were purchased from Corning Costar (USA) and BD Biosciences (USA), respectively. The Nrf2 inhibitor ML385 (GC19254) and primary antibodies against Nrf2 (16396-1-AP), GPX4 (Ab125066), HO-1 (10701-1-AP), and SLC7A11 (Ab307601) were acquired from GLPBIO (USA), Proteintech Group, Inc. (USA), and Abcam plc (UK). Blood glucose levels were monitored using an Accu-Chek® glucometer (Roche Diagnostics, Switzerland). DP ointment was prepared by dissolving DP in DMSO to final concentrations of 200, 100, and 50 µg/mL, followed by mixing these solutions with Vaseline at a ratio of 1 mL solution to 16 g Vaseline. The ointment was stored at 4 °C. The preparation was carried out by the standards outlined in the 2020 edition of the Chinese Pharmacopoeia for ointment formulations.
Laboratory animals
Seventy SPF rats (200 ± 20 g) were obtained from Beijing Sibeifu Biotechnology Co., Ltd. (License No: SCXK (Beijing) 2019-0010). These rats were housed at the Experimental Animal Center of Beijing University of Chinese Medicine, in a well-ventilated environment maintained at a constant temperature of 25 °C, with ad libitum access to water and food. The animal study protocol was approved by the Animal Ethics Committee of the Beijing University of Chinese Medicine (Approval No: BUCM-2023062809-2228), and was conducted in adherence to the ARRIVE guidelines. All methods were performed in accordance with the relevant guidelines and regulations.
Development and grouping of DFU rat models
Sixty SD rats were acclimated for one week before being randomly assigned to one of the following groups: the control group, which continued on a normal diet, or one of the experimental groups: the model group, positive drug group (rhEGF), and high (DP-H), medium (DP-M), and low (DP-L) dose DP groups. All experimental groups were fed a high-sugar, high-fat diet. After one month, rats in the high-sugar, high-fat diet groups were given a single intraperitoneal injection of STZ (40 mg/kg), while the control group received an equivalent volume of sodium citrate buffer. Blood glucose levels were measured using an Accu-Chek glucometer over three consecutive days, and symptoms such as polydipsia, polyphagia, weight loss, yellowing of fur, and wet bedding were monitored. Successful diabetes induction was confirmed by fasting blood glucose levels ≥ 16.7 mmol/L and the presence of diabetes-related symptoms 72 h (h) after the STZ injection. Anesthesia was induced using isoflurane, and a circular wound (15 mm in diameter) was created on the dorsum of each rat. Following model establishment, the control group received standard wound care for natural healing, while the DFU group received the designated treatment. On days 7 and 14 post-treatment, samples were randomly collected from rats in each group under anesthesia. Skin, wound edges, granulation tissue, and serum were collected from a 2 mm area surrounding the ulcer for further analysis23. At the end of the experiment, all rats were euthanized with an overdose of isoflurane.
Wound healing rate
Wound healing rates in DFU and control rats were evaluated on days 0, 3, 7, 10, and 14. The rate of wound healing was determined using the following formula:
.
Wound areas were quantified using ImageJ, an open-source image analysis software developed by the National Institutes of Health (NIH).
Wound histopathological analysis
On days 7 and 14 post-intervention, at least three rats from each group were randomly selected for sampling. Skin specimens from the areas surrounding the ulcers were collected, dehydrated, and embedded in paraffin. The paraffin sections were then dewaxed, rehydrated, and stained with HE and Masson’s trichrome. Histological characteristics were evaluated through microscopic observation and image analysis.
Immunohistochemistry
After dewaxing and rehydrating the paraffin sections, antigen retrieval was performed, followed by blocking, circumscribing, and serum blocking. The sections were then incubated with the primary antibody (CD34, dilution 1:200) and the secondary antibody (dilution 1:500). Staining was completed using DAB for chromogenic detection and nuclear counterstaining. Under 50× magnification, the three regions with the highest vascular density were identified. Five non-overlapping regions were then selected at 200× magnification to quantify microvessel counts, with the average count calculated.
ELISA
Blood samples were collected from the abdominal aortas of rats in each group on days 7 and 14 and then stored at 4 °C for 30 min (min). The samples were centrifuged at 3000 rpm for 15 min to separate the serum. Serum levels of TNF-α, IL-6, VEGF, EGF, PDGF, and ROS were quantified using ELISA kits, following the manufacturer’s protocols.
Network pharmacology research
The SMILES format of DP was retrieved from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and uploaded to Super-PRED (https://prediction.charite.de/index.php), SwissTargetPrediction (http://swisstargetprediction.ch), and PharmMapper (www.lilab-ecust.cn/pharmmapper/) for target prediction. The corresponding gene names for the target proteins were obtained from the UniProt database. Ferroptosis-related gene targets were sourced from the FerrDb database (http://www.zhounan.org/ferrdb/) and categorized by type. Disease targets related to “diabetic ulcer,” “diabetic foot,” and “diabetic foot ulcer” were identified through keyword searches in GeneCards (https://www.genecards.org/), OMIM (https://omim.org/), TTD (http://db.idrblab.net/ttd/), and DisGeNET (http://disgenet.com). Overlapping targets between DFU, ferroptosis, and drug targets were identified and visualized using a Venn diagram. The intersecting targets were imported into the String database for Protein-Protein Interaction (PPI) analysis and visualized with Cytoscape 3.10.0. Gene Ontology (GO), Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway24, and WIKI pathway analyses of the overlapping targets were performed using the DAVID database, with histograms and bubble plots generated using Wei Shengxin (https://www.bioinformatics.com.cn).
Molecular docking
The Pymol software (using commands: remove solvent; remove organic molecules) was used to remove water and small molecules. The three-dimensional structure of DP was retrieved from the PubChem database in SDF format and optimized using ChemOffice software, then saved in PDB format. Hydrogenation of DP, ferritin, and core genes associated with DFU was performed using AutoDock 1.5.6, and docking boxes were defined. Finally, docking of these genes with DP was performed using AutoDock Vina.
Cell culture and concentration screening.
HUVECs cells were cultured in ECM complete medium under conditions of 5% CO2, 37 °C, and saturated humidity in a humidified incubator, with medium changes every 1–2 days. Cells were passaged upon reaching approximately 80% confluence. Cell viability was evaluated using the CCK-8 assay to determine the optimal working concentrations of DP for the experiment. Three effective concentrations were selected and categorized into high, medium, and low groups for further analysis. Control HUVECs were cultured in a medium containing 5 mM glucose for 24 h, while the experimental group was treated with a 30 mM glucose medium.
Cell viability assay
Based on the results from the previous cell experiments, the optimal concentration of DP was determined for use in further experiments. These were organized into the following groups: blank control group, model group, DP group (optimal concentration), DP + ML385 group, cell seeding plate, and CCK-8 assay. The experimental conditions for these groups were the same as those outlined previously.
Scratch, transwell, and tube formation assay
Scratch assay
HUVECs were plated in 6-well plates and cultured until they reached 90% confluence. A 100 µL Eppendorf tip was used to create scratches in the cell monolayer. After washing the cells 1–2 times, they were treated with glucose and varying concentrations of DP according to the experimental groups. The cells were incubated in a CO2 incubator, and changes in cell morphology were observed at 0 and 24 h using an inverted microscope.
Transwell assay
After trypsinization and cell counting, HUVECs were seeded at a concentration of 2 × 105 cells/mL. For the migration assay, 10,000 cells per well were used. The Transwell chambers were set up with serum-free medium in the upper chamber and media containing 5 mM or 30 mM glucose, along with low, medium, or high concentrations of DP, in the lower chamber. After incubating for 10–30 min at room temperature to fix the cells, the chambers were washed twice with PBS, fixed in 100% methanol for 20 min, and stained for 15–30 min. The chambers were washed three times with PBS, and air-dried, and the cells were counted and imaged using a fluorescent inverted microscope.
Tube formation assay
HUVECs were seeded onto pre-coated BD Matrigel and incubated for 6 h. Tube formation was observed using an inverted microscope, and three random fields per well were captured for analysis.
Immunofluorescence detection of ROS
DCFH-DA was diluted 1:1000 in serum-free medium to achieve a final concentration of 10 µM. The cell culture medium was removed, and an appropriate volume of the diluted DCFH-DA was added to fully cover the cells, typically 1 mL per well of a six-well plate. The cells were incubated at 37 °C for 20 min. Following incubation, the cells were washed three times with a serum-free medium to remove any unincorporated DCFH-DA. The cells were rinsed twice with PBS and imaged using a fluorescence microscope.
Lipid peroxidation assay
BODIPY™ 581/591 C11 is commonly used to assess lipid peroxidation and antioxidative activity in living cells. In the presence of ROS generated by live cells, the dye undergoes oxidation, causing a shift in its emission peak from ~ 590 nm (reduced state, red) to ~ 510 nm (oxidized state, green). Primary HUVECs are seeded in a 12-well plate pre-coated with an appropriate matrix. Following the manufacturer’s protocol, 1 mg of BODIPY™ 581/591 C11 is dissolved in 198 µL of DMSO to prepare a 10 mM stock solution, which is then diluted to 5 µM using Neurobasal-A medium. Each well is supplemented with 0.5 mL of the 5 µM working solution of BODIPY™ 581/591 C11 to ensure complete cell coverage. The cells are incubated at 37 °C for 30 min in the dark, washed gently three times with a warm basal medium, and imaged using a laser confocal microscope. Image quantification is performed using ImageJ software.
Western blotting
Each sample was homogenized in 200 µL of lysis buffer and incubated on ice for 45 min. After lysis, the samples were centrifuged at 12,000 rpm for 15 min at 4 °C. Protein concentrations were quantified using a BCA protein assay kit. The proteins were then separated by SDS-PAGE, transferred to PVDF membranes, and incubated with primary antibodies. Protein detection was performed using the ECL chemiluminescence system. The optical density of the target bands was quantified using Image Pro Plus 6.0 software for further analysis.
Statistical analysis
Quantitative data from three independent experiments were expressed as mean ± SEM and analyzed by one-way ANOVA followed by Tukey’s multiple comparisons test. Statistical plots were generated using GraphPad Prism 9.5.0, and significance was set at p < 0.05.
Results
Wound healing progression and rates in DFU rats
Figure 1A and B illustrate the changes in wound healing areas of DFU rats from day 0 to 14. On day 0, there were no significant differences in wound areas among the groups. Over time, wound areas decreased in all groups, but the model group showed a significantly slower healing rate compared to the others. By day 14, the model group still showed pronounced inflammatory responses, whereas the other groups had advanced to the remodeling phase and achieved epithelialization, with the DP-H group showing the most significant healing improvement. Analysis of healing rates at different time points (Fig. 1C) revealed no significant difference between the DP-H and positive drug groups from day 3 to 7 (p > 0.05), both of which showed higher healing rates compared to the control, DP-M, DP-L, and model groups (p < 0.05). From day 7 to 14, the DP-H group demonstrated significantly faster healing rates compared to the positive drug, control, DP-M, DP-L, and model groups (p < 0.05). These findings confirm that DP enhances DFU healing in a dose-dependent manner.
DP accelerates wound healing in DFU rats. (A) Representative images of wound area healing at various time points in DFU rats. (B) DFU rat wound healing simulation diagram. (C) Wound healing rates (%) at day 0, 3, 7, 10, and 14 across different groups.
Effect of DP on the pathological structure of DFU wounds
Hematoxylin and eosin (HE) staining results on day 7 (Fig. 2A) revealed distinct differences among the groups. The control group showed epidermal disruption, mild reduction in dermal collagen fibers, new blood vessel formation, and minimal inflammatory cell infiltration. In contrast, the model group showed severe epidermal structure loss, eschar formation, cellular necrosis, extensive inflammatory cell infiltration, and a significant reduction in dermal collagen fibers accompanied by bleeding. The rhEGF group demonstrated a relatively intact epidermal structure, thickening of the stratum spinosum, formation of new blood vessels, and minimal inflammatory cell infiltration. The DP-H group displayed a more intact epidermal structure, thickened stratum spinosum, abundant dermal collagen fibers, and new blood vessel formation compared to the model group, with no evidence of inflammatory cell infiltration. By day 14 (Fig. 2D), both the control and rhEGF groups displayed relatively complete skin structures with reduced dermal collagen fibers and ongoing new blood vessel formation, without inflammatory cell infiltration. In contrast, the model group continued to show epidermal loss, a significant reduction in dermal collagen fibers, and persistent inflammatory cell infiltration. The DP-H group maintained an intact epidermal structure, abundant dermal collagen fibers, and active new blood vessel formation, highlighting its potential to promote wound healing.
Topical application of DP significantly promotes collagen fiber deposition and neovascularization in DFU rat models, while reducing inflammatory cell infiltration and increasing MVD. (A,D) HE staining of DFU rat wounds on day 7 and 14, scale bar = 50 μm. Black arrows denote the loss of epidermal layer integrity within the field of view, yellow arrows indicate collagen fiber content in the dermis, green arrows highlight newly formed blood vessels, red arrows represent inflammatory cell infiltration, and blue arrows mark areas of bleeding. (B,E) Masson staining of DFU rat wounds on day 7 and 14, scale bar = 50 μm. (C,F) CD34 immunohistochemistry of DFU rat wounds on day 7 and 14, scale bar = 50 μm. (G) Quantitative analysis of collagen fiber occupancy by Masson staining of DFU rat wounds on day 7 and 14. (H) Quantitative analysis of MVD in DFU rat wounds on days 7 and 14. Data were expressed as mean ± SD (n = 3). *P<0.05, **P<0.01, ***P<0.001 compared with the model group.
Collagen fiber deposition was evaluated using Masson’s trichrome staining. The DP treatment groups and the rhEGF group showed extensive collagen deposition with well-organized collagen bundles in the dermis (Fig. 2B, E), whereas the Model group showed minimal collagen deposition and poorly structured collagen bundles. Quantitative analysis (Fig. 2G) indicated that the DP-H group achieved the highest percentage of collagen fibers in the dermis at both 7 and 14 days, significantly surpassing the Model group (p < 0.001). The rhEGF, DP-M, and DP-L groups also displayed significantly increased collagen fiber content compared to the Model group (p < 0.001). These findings suggest that the topical application of DP enhances collagen deposition and accelerates wound healing.
DP enhances CD34 expression and microvessel density in wounds
CD34 expression was used to assess microvessel density (MVD) and vascular distribution, reflecting tissue vascular density. Results indicated a consistent trend on both days 7 and 14, with the model group showing reduced MVD compared to the control group (Fig. 2C, F). In contrast, all DP treatment groups showed higher MVD levels than the model group. The DP-H group demonstrated a significantly higher MVD compared to the rhEGF groups (Fig. 2H). These findings suggest that DP enhances CD34 expression and increases microvessel density in wounds.
DP treatment modulates cytokines, growth factors, and ROS levels
TNF-α and IL-6 are key inflammatory cytokines implicated in DFU, with elevated levels associated with persistent ulcers and delayed wound healing. In contrast, PDGF, EGF, and VEGF are essential growth factors that promote fibroblast proliferation and migration, playing a crucial role in wound repair. ELISA assays showed that TNF-α and IL-6 levels were significantly higher in the model group compared to the control group on both day 7 and day 14 (Fig. 3A, B). In contrast, DP treatment resulted in a dose-dependent decrease in IL-6 and TNF-α levels compared to the model group. Furthermore, PDGF, EGF, and VEGF levels were significantly lower in the model group than in the control group on days 7 and 14 (Fig. 3C and E). In comparison to the model group, DP treatment increased the levels of these growth factors in a dose-dependent manner. ROS are key indicators of oxidative stress and ferroptosis in cells and tissues. Wound tissue analysis revealed a significant increase in ROS levels in the model group compared to the control group on days 7 and 14 (Fig. 3F). DP treatment led to a dose-dependent reduction in ROS levels compared to the model group. These results suggest that DP treatment decreases inflammatory factors, promotes growth factors, and alleviates oxidative stress-induced ferroptosis in DFU rat wounds, thereby enhancing wound healing.
Results of serum Elisa on day 7 and 14 in DFU rats. (A) TNF-α results. (B) IL-6 results. (C) PDGF results. (D) EGF results. (E) VEGF results. (F) ROS results. Data were expressed as mean ± SD (n = 3). *P<0.05, **P<0.01, ***P<0.001 compared with the model group.
Network pharmacological target prediction and pathway enrichment
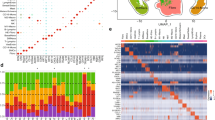
Based on data from the Super-PRED databases, a total of 778 targets of DP were identified. Furthermore, 485 ferroptosis-related genes and 3008 DFU-associated targets were obtained from various databases, resulting in the identification of 28 overlapping targets (Fig. 4A). PPI analysis highlighted the core targets (Figs. 4B, and 4C), with the top four targets ranked by degree values as PPARG, HIF1A, NFE2L2 (Nrf2), and PTGS2. GO analysis revealed that the primary biological processes involved responses to external oxidative stress, hypoxia, and inflammation. The key molecular functions identified included protein binding, ATP binding, and protein serine kinase activity (Fig. 4D). WIKI pathway analysis showed a particularly strong association with Nrf2-related pathways (Fig. 4E). KEGG pathway analysis suggested that the mechanisms through which DP may prevent and treat DFU involve the AGE-RAGE signaling pathway, lipid metabolism and atherosclerosis pathways, and ROS regulation (Fig. 4F).
Pharmacological analysis of the DP-DFU-Ferroptosis network. (A) Venny figure. (B) PPI network analysis. (C) Key components of the DP-DFU-Ferroptosis core target network. (D) GO enrichment analysis. (E) WIKI enrichment analysis. (F) KEGG enrichment analysis.
Molecular docking validates core targets
Molecular docking results indicated that the binding energies of DP with PPARG, HIF1A, NFE2L2 (Nrf2), and PTGS2 were − 8.49, -6.29, -7.07, and − 7.12 kcal/mol, respectively (Fig. 5). Generally, a binding energy lower than − 5 kcal/mol suggests a good binding affinity, while a value below − 7 kcal/mol typically indicates a strong and stable interaction. Our results suggest that DP exhibits strong affinity for PPARG, NFE2L2 (Nrf2), and PTGS2, and may exert its effects through these targets. Based on the integration of molecular docking and network pharmacology enrichment analysis, we hypothesize that DP could promote DFU wound healing by modulating the Nrf2-mediated ferroptosis signaling pathway. Subsequently, we conducted in vitro experiments to validate the therapeutic effects and underlying mechanisms.
Visualization of docking between DP and core target molecules (binding energy ≤ − 5.0 kcal/mol).
DP improves cell viability in high glucose-Induced damage
CCK-8 assays identified three final concentrations of DP: 0.05 µM, 0.1 µM, and 0.2 µM (Fig. 6A). After high glucose-induced damage, treatment with these concentrations of DP showed that cell viability was significantly lower in the HG (30 mM) group compared to the control group (p < 0.001). In contrast, cell viability was significantly higher in all DP-treated groups compared to the HG (30 mM) group, with the HG + DP groups demonstrating the highest cell viability (p < 0.001). These results suggest that DP improves cell viability in high glucose-induced damage (Fig. 6B).
Effects of DP on HUVECs with high glucose damage. (A) Screening of DP working concentrations by CCK-8 method. (B) CCK-8 assay for cell viability. (C,D,G) Scratch assay and statistical analysis. (E,H) Transwell assay and statistical analysis. (F,I) Tube formation assay and statistical analysis. Data were expressed as mean ± SD (n = 3). *P<0.05, **P<0.01, ***P<0.001 compared with the model group, #P<0.05, ##P<0.01, ###P<0.001 compared with the DP group.
DP enhances angiogenesis and promotes cell migration under high glucose-induced conditions
Scratch (Fig. 6C, D, G) and Transwell assays (Fig. 6E, H) revealed a significant reduction in migration rate in the HG (30 mM) group compared to the control group (p < 0.001). In contrast, all DP-treated groups showed significantly higher migration rates than the HG (30 mM) group, with a dose-dependent response. The tube formation assay (Fig. 6F, I) also demonstrated a significant reduction in the number of vascular structures in the HG (30 mM) group compared to the control group (p < 0.001). In contrast, all DP-treated groups displayed a significant increase in vascular formation compared to the HG (30 mM) group, also showing a dose-dependent effect. These results suggest that DP enhances angiogenesis and promotes cell migration in the context of high glucose-induced damage.
DP enhances HUVECs viability in high glucose-damaged conditions through Nrf2 pathway
Based on the experiments, the optimal concentration of DP was determined to be 0.2 µM, which was used for further studies. Viability assays performed using the CCK-8 method yielded significant results (Fig. 7A). Compared to the control group, the model group showed a significant decrease in cell viability (p < 0.001). In contrast, cell viability in the DP-treated group was significantly higher than in the model group. However, when the Nrf2 inhibitor ML385 was added, a significant reduction in cell viability was observed compared to the DP group (p < 0.001). These results suggest that DP enhances the viability of high glucose-damaged HUVECs, and this effect is diminished when Nrf2 is inhibited.
DP activates the Nrf2 pathway to inhibit ferroptosis to repair high glucose-damaged HUVECs. (A) Cell viability assay of high glucose-damaged HUVECs after treatment with DP and ML385. (B,D) Immunofluorescence and statistical analysis of ROS (scale bar = 100 μm). (C,E) Lipid peroxidation assay (scale bar = 25 μm). (F–J) WB detection of Nrf2 pathway-related protein expression and quantitative analysis. Data were expressed as mean ± SD (n = 3). *P<0.05, **P<0.01, ***P<0.001, #P<0.05, ##P<0.01, ###P<0.001 compared with the DP group. Original blots are presented in Supplementary Fig. 1–6.
DP reduces high glucose-induced ROS accumulation via the Nrf2 pathway
Immunofluorescence results (Fig. 7B, and D) showed that, compared to the control group, the model group showed a significant increase in ROS levels (p < 0.001). In contrast, the DP group displayed a significant decrease in ROS levels compared to the model group (p < 0.001). However, upon the addition of ML385, ROS levels increased again, indicating that DP effectively alleviates excessive ROS induced by high glucose injury, although this effect is diminished by ML385.
DP attenuates lipid peroxidation in high glucose-damaged HUVECs through the Nrf2 pathway
Lipid peroxidation assays demonstrated increased lipid peroxidation levels in the model group, as indicated by the intense green fluorescence of the oxidized form of C11-BODIPY. Treatment with DP significantly attenuated lipid peroxidation in high glucose-induced damage in HUVECs. However, the following addition of ML385 led to a rebound in lipid peroxidation levels (Fig. 7C, and E), suggesting that DP mitigates lipid peroxidation, an effect that is reversed by ML385.
DP activates the Nrf2-mediated ferroptosis signaling pathway to mitigate high glucose-induced damage
Western blot analysis (Fig. 7F-J) revealed that, compared to the control group, the expression levels of Nrf2, GPX4, HO-1, and SLC7A11 proteins were significantly reduced in the model group (p < 0.001). In contrast, the DP group showed significantly increased expression levels of these proteins compared to the model group (p < 0.001). However, upon the addition of ML385, the expression levels of Nrf2, GPX4, HO-1, and SLC7A11 were significantly decreased once again (p < 0.001). These results suggest that DP activates the Nrf2-mediated ferroptosis signaling pathway to mitigate high glucose-induced damage in HUVECs.
Discussion
This study reveals that DP facilitates dose-dependent wound healing in DFU rat models by enhancing angiogenesis and collagen formation while mitigating ROS levels and inflammatory responses in wound tissue. The mechanism underlying these effects involves activation of the Nrf2 pathway, which suppresses ferroptosis, as confirmed by the inhibitory effects of ML385 on this pathway.
Rapid closure of DFU wounds is essential for minimizing infection and reducing the risk of amputation25,26. In this study, a DFU rat model was established through a combination of a high-glucose, high-fat diet, STZ injections, and surgical intervention, with rhEGF serving as a positive control23. The findings demonstrate that topical application of DP significantly accelerates wound healing in DFU rats compared to both the model and rhEGF groups, with a dose-dependent improvement in healing rates. Histological analyses, including HE and Masson’s trichrome staining, showed that DP enhances collagen deposition and neovascularization while reducing inflammatory cell infiltration, aligning with previous observations27,28. CD34, a glycoprotein predominantly expressed on hematopoietic stem cells, endothelial cells, and certain stromal cells, is commonly used as a marker for endothelial cells and an indicator of angiogenesis in histological and pathological studies29,30. Immunohistochemical analysis in this study was employed to evaluate CD34 expression in DFU rat wound tissues, providing valuable insights into neovascularization. The results revealed significantly increased CD34 expression in the DP-treated group compared to both the model and positive control groups, indicating that DP significantly enhances microvascular formation in a dose-dependent manner. Effective wound healing in DFU is closely associated with the regulation of various cytokines, including inflammatory mediators (TNF-α, IL-6)31, growth factors (PDGF, EGF)32, and angiogenic factors (VEGF)33. High levels of inflammatory mediators hinder the healing process by inhibiting epidermal cell proliferation and migration, therefore delaying ulcer resolution. Similarly, a deficiency in growth factors impairs the formation and repair of blood vessels and cells, reducing vascular and cellular regeneration, which ultimately leads to delayed ulcer healing34,35. Okizaki et al. highlighted key abnormalities in diabetic animal ulcer models, including sustained production of pro-inflammatory cytokines, dysfunction in macrophages and neutrophils, impaired migration and proliferation of keratinocytes and fibroblasts, and reduced synthesis of growth factors essential for the healing process36. Present findings align with these observations. Serum ELISA analysis showed significantly higher levels of inflammatory cytokines in the DFU model group compared to the control group, accompanied by a significant decrease in growth factor levels. DP treatment effectively reduced IL-6 and TNF-α levels while increasing the concentrations of PDGF, EGF, and VEGF. These results indicate that DP suppresses inflammatory responses and stimulates growth factor production, thereby enhancing neovascularization and cellular proliferation, ultimately facilitating accelerated wound healing.
In recent years, considerable attention has been given to the role of hyperglycemia-induced free radicals and oxidative stress in cell death, contributing to delayed wound healing in diabetic ulcers37,38. Understanding the mechanisms of cell death is essential for uncovering the molecular pathways underlying disease pathogenesis. Ferroptosis, a newly recognized form of cell death, plays a crucial role in various diseases. The primary mechanism involves the inactivation of GPX4, leading to lipid peroxidation and excessive ROS accumulation, which ultimately results in ferroptosis39,40. In the context of DFU, the hyperglycemic environment exacerbates oxidative stress, elevating ROS levels. Simultaneously, the cystine-glutamate antiporter is impaired, reducing cystine uptake and hindering the synthesis of GSH, a precursor for GPX4. This disruption leads to the accumulation of lipid ROS, which significantly contributes to the progression of diabetes and its complications41. Telorack et al. demonstrated that diabetic rats with ulcerated skin show a deficiency in GSH synthetase in granulation tissue keratinocytes, resulting in reduced GPX4 activity42. This impairment negatively impacts the regeneration and repair of ulcerated skin granulation tissue. Katunga et al. further support these findings, reporting a significant reduction in GPX4 activity in the local tissues of chronic ulcers in diabetic patients43. Moreover, several important studies have demonstrated that increased local GSH levels under hyperglycemic conditions can promote granulation tissue formation and accelerate healing in diabetic ulcer models44,45. In a hyperglycemic environment, excessive oxidative stress impedes GSH synthesis, leading to reduced and less active GPX4 levels. This, in turn, results in the accumulation of ROS, causing lipid peroxidation of cell membranes. The resulting lipid peroxidation further amplifies oxidative stress, creating a harmful cycle. This mechanism represents a key molecular factor contributing to the impaired healing observed in diabetic ulcers46. Our findings indicate that ROS levels are significantly elevated in the model group compared to the control group. Treatment with DP effectively reduces ROS levels in a dose-dependent manner, a result further confirmed through in vitro validation. In vitro, assays demonstrated that DP enhances the viability of high-glucose-damaged HUVECs and promotes their migration and regenerative capacity, which is consistent with previous studies47. DP was also observed to significantly decrease lipid peroxidation levels in these cells. GPX4, a selenoprotein phospholipid hydroperoxidase, is instrumental in mitigating ferroptosis by reducing lipid peroxidation, underscoring its critical role in this process. Nrf2, a transcription factor regulating the expression of antioxidant response elements such as GPX4, exerts its antioxidant effects through nuclear translocation48. In this study, network pharmacology analysis identified Nrf2 as a core target of DP in the intervention of DFU. Molecular docking demonstrated a strong binding affinity of DP to Nrf2, and WIKI pathway enrichment analysis underscored the central role of the Nrf2 pathway in DP-mediated DFU intervention. In vitro experiments, including Western blot analysis, revealed significantly reduced Nrf2 protein levels in high-glucose-damaged HUVECs, accompanied by decreased expression of ferroptosis-related proteins GPX4, SLC7A11, and HO-1. DP treatment restored the expression of Nrf2 and its downstream proteins, including GPX4, HO-1, and SLC7A11, indicating activation of the Nrf2-mediated ferroptosis signaling pathway. However, the therapeutic effect of DP was attenuated in the presence of the Nrf2 inhibitor ML385, further confirming the pathway’s involvement (Fig. 8).
DP accelerates diabetic foot ulcer healing by alleviating ferroptosis via the Nrf2 pathway activation.
Conclusion
In conclusion, DP accelerates DFU wound healing by activating the Nrf2 signaling pathway, thereby inhibiting ferroptosis. Using a combination of network pharmacology, bioinformatics, and in vivo and in vitro experiments, the findings provide robust theoretical evidence supporting the potential of DP as a therapeutic agent for DFU intervention.
Data availability
Data is provided within the manuscript or supplementary information files.
References
GBD 2021 Diabetes Collaborators. Global, regional, and National burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the global burden of disease study 2021. Lancet 402(10397), 203–234 (2023).
International Diabetes Federation. The Diabetic Foot. Brussels, Belgium & Federation, I. D. https://www.idf.org/our-activities/care-prevention/diabetic-foot.html (Accessed 1 Aug 2022) (2020).
Yadav, J. P. et al. Phytoconstituents as modulators of NF-κB signalling: investigating therapeutic potential for diabetic wound healing. Biomed. Pharmacother. 177, 117058 (2024).
Deng, P. et al. Worldwide research trends on diabetic foot ulcer (2004–2020): Suggestions for researchers. J. Diabetes Res. 2022, 7991031. (2022).
Chen, L., Sun, S., Gao, Y. & Ran, X. Global mortality of diabetic foot ulcer: A systematic review and meta-analysis of observational studies. Diabetes Obes. Metab. 25(1), 36–45 (2023).
McDermott, K., Fang, M., Boulton, A. J. M., Selvin, E. & Hicks, C. W. Etiology, epidemiology, and disparities in the burden of diabetic foot ulcers. Diabetes Care. 46(1), 209–221 (2023).
Armstrong, D. G., Tan, T. W., Boulton, A. J. M. & Bus, S. A. Diabetic foot ulcers: A review. JAMA 330(1), 62–75 (2023).
Yadav, J. P., Patel, D. K., Verma, A. & Pathak, P. Revolutionizing diabetic wound healing: targeted therapeutic strategies based on growth factors. Obes. Med. 47, 100535. (2024).
Ramachandran, V., Mohanasundaram, T., Karunakaran, D., Gunasekaran, M. & Tiwari, R. Physiological and pathophysiological aspects of diabetic foot ulcer and its treatment strategies. Curr. Diabetes Rev. 19(8), e031122210617 (2023).
McDermott, K., Fang, M., Boulton, A. J. M., Selvin, E. & Hicks, C. W. Etiology, epidemiology, and disparities in the burden of diabetic foot ulcer. Diabetes Care. 46(1), 209–221 (2023).
Armstrong, D. G., Tan, T. W., Boulton, A. J. M. & Bus, S. A. Diabetic foot ulcer: A review. JAMA 330(1), 62–75 (2023).
Everett, E. & Mathioudakis, N. Update on management of diabetic foot ulcer. Ann. N Y Acad. Sci. 1411(1), 153–165 (2018).
Mohsin, F., Javaid, S., Tariq, M. & Mustafa, M. Molecular immunological mechanisms of impaired wound healing in diabetic foot ulcer (DFU), current therapeutic strategies and future directions. Int. Immunopharmacol. 139, 112713 (2024).
Song, J. Y. et al. Deciphering the emerging role of programmed cell death in diabetic wound Healing. Int. J. Biol. Sci. 19(15), 4989–5003 (2023).
Zhang, Y. Study on the Repairing Effect and Mechanism of Ferrostatin-1 on Ulcer Wounds in High Glucose Environment Based on iron Death Pathway (Tianjin Medical University, 2021).
Miraj, S. S. et al. Phytotherapy in diabetic foot ulcer: A promising strategy for effective wound healing. J. Am. Nutr. Assoc. 42(3), 295–310 (2023).
Yan, L. Y. Z. H. A briefanalysis ofthe external use ofancient remedies named after the efficacy of muscle growth in ancient texts. J. Guangzhou Univ. Chin. Med. 19(3), 234–235 .
Pan, X. Y. et al. Clinical study on the treatment of diabetic foot ulcer wounds with compound blood exhaust powder. Chin. J. Orthop. Traumatol. 30(07), 32–37 (2022).
Jiang, X. et al. Dracorhodin perchlorate regulates fibroblast proliferation to promote rat’s wound healing. J. Pharmacol. Sci. 136(2), 66–72 (2018).
Ching, Y. H. et al. Hypoglycemic effects of dracorhodin and dragon blood crude extract from Daemonorops draco. Bot. Stud. 65(1), 8 (2024).
Zhang, W. et al. Dracorhodin targeting CMPK2 attenuates inflammation: A novel approach to sepsis therapy. Clin. Transl. Med. 13(10), e1449 (2023).
Deng, P. et al. Combined metabolomics and network pharmacology to elucidate the mechanisms of dracorhodin perchlorate in treating diabetic foot ulcer rats. Front. Pharmacol. 13, 1038656 (2022).
Nie, X. et al. Asiaticoside nitric oxide gel accelerates diabetic cutaneous ulcers healing by activating Wnt/β-catenin signaling pathway. Int. Immunopharmacol. 79, 106109 (2020).
Kanehisa, M., Furumichi, M., Sato, Y., Matsuura, Y. & Ishiguro-Watanabe, M. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 53(D1), D672–D677 (2025).
Lavery, L. A. et al. WHS (Wound Healing Society) guidelines update: diabetic foot ulcer treatment guidelines. Wound Repair Regener. 32(1), 34–46. (2024).
Kang, Y. et al. Programmed microalgae-gel promotes chronic wound healing in diabetes. Nat. Commun. 15(1), 1042. (2024).
Xu, C. et al. Gynura divaricata (L.) DC. promotes diabetic wound healing by activating Nrf2 signaling in diabetic rats. J. Ethnopharmacol. 323, 117638 (2024).
Zhang, Z. et al. Integrating network pharmacology, molecular docking and experimental validation to explore the pharmacological mechanisms of quercetin against diabetic wound. Int. J. Med. Sci. Vol. 21, 14 2837–2850 (2024).
Díaz-Flores, L. et al. Comparison of the behavior of perivascular cells (pericytes and CD34+ stromal cell/telocytes) in sprouting and intussusceptive angiogenesis. Int. J. Mol. Sci. 23(16), 9010 (2022).
Xing, Z. et al. IDO1 inhibitor RY103 suppresses Trp-GCN2-mediated angiogenesis and counters immunosuppression in glioblastoma. Pharmaceutics. 16(7), 870 (2024).
Sun, X. et al. Paeoniflorin inhibited nod-like receptor protein-3 inflammasome and NF-κB-mediated inflammatory reactions in diabetic foot ulcer by inhibiting the chemokine receptor CXCR2. Drug Dev. Res.. 82(3), 404–411 (2021).
Liu, Y. et al. Fibroblast growth factor in diabetic foot ulcer: progress and therapeutic prospects. Front. Endocrinol. 12(14), 744868. (2021).
Schönborn, M. et al. Circulating angiogenic factors and ischemic diabetic foot syndrome advancement—A pilot study. Biomedicines 11(6), 1559 (2023).
Hajmousa, G. et al. Human adipose tissue-derived stromal cells act as functional pericytes in mice and suppress high-glucose-induced proinflammatory activation of bovine retinal endothelial cells. Diabetologia 61(11), 2371–2385 (2018).
Icli, B. et al. MicroRNA-615-5p regulates angiogenesis and tissue repair by targeting AKT/eNOS (protein kinase B/endothelial nitric oxide synthase) signaling in endothelial Cells. Arterioscler. Thromb. Vasc Biol. 39(7), 1458–1474 (2019).
Okizaki, S. et al. Suppressed recruitment of alternatively activated macrophages reduces TGF-beta1 and impairs wound healing in streptozotocin-induced diabetic mice. Biomed. Pharmacother. 70, 317–325 (2015).
Feng, J. et al. Oxidative stress and lipid peroxidation: prospective associations between ferroptosis and delayed wound healing in diabetic ulcers. Front. Cell Dev. Biol. 10(8), 898657 (2022).
Jiang, G. et al. Mitochondrial dysfunction and oxidative stress in diabetic wound. J. Biochem. Mol. Toxicol. Vol. 37, e23407 (2023).
Wei, T. et al. Janus liposozyme for the modulation of redox and immune homeostasis in infected diabetic wounds. Nat. Nanotechnol. 19(8), 1178–1189 (2024).
Vujčić, S. et al. Oxidative stress and inflammatory biomarkers in patients with diabetic foot. Med. (Kaunas). 58(12), 1866 (2022).
Qi, X. et al. An immunomodulatory hydrogel by hyperthermia-assisted self-cascade glucose depletion and ROS scavenging for diabetic foot ulcer wound therapeutics. Adv. Mater. 35(48), e2306632. (2023).
Telorack, M. et al. A glutathione-Nrf2-thioredoxin cross-talk ensures keratinocyte survival and efficient wound repair. Plos Genet. 12(1), e1005800 (2016).
Katunga, L. A. et al. Obesity in a model of gpx4 haploinsufficiency uncovers a causal role for lipid-derived aldehydes in human metabolic disease and cardiomyopathy. Mol. Metab. 4(6), 493–506 (2015).
Sakai, O. et al. Role of GPX4 in human vascular endothelial cells, and the compensatory activity of brown rice on GPX4 ablation condition. Pathophysiology 24(1), 9–15 (2017).
Arya, A. K. et al. Relationship between oxidative stress and apoptotic markers in lymphocytes of diabetic patients with chronic non healing wound. Diabetes Res. Clin. Pract. Vol. 94(3), 377–384 (2011).
Shivavedi, N. et al. Ascorbic acid therapy: A potential strategy against comorbid depression-like behavior in streptozotocin-nicotinamide-induced diabetic rats. Biomed. Pharmacotherapy = Biomedecine Pharmacotherapie. 109, 351–359 (2019).
Yu, Z. et al. Effect of dracorhodin perchlorate on angiogenesis of human umbilical vein endothelial cells . Chin. J. Aesthetic Plast. Surg. 31(06), 380–382 (2019).
Zhang, Y. et al. Dexmedetomidine alleviates ferroptosis following hepatic ischemia-reperfusion injury by upregulating Nrf2/GPx4-dependent antioxidant responses. Biomed. Pharmacotherapy = Biomedecine Pharmacotherapie. 169, 115915 (2023).
Acknowledgements
The authors extend their sincere appreciation to theexperts who dedicated their time and expertise to partici-pate in the survey rounds of this study. Their valuableinsights and active engagement significantly enriched theresearch process.
Author information
Authors and Affiliations
Contributions
G.T. drafted the manuscript. G.T. , Y.W.and P.D. performed the experiments and analyzed the data. J.W., Z.L.,R.Z, H.G, Y.Z and X.M. revised the manuscript. Z.C. designed the experiments and had primary responsibility for final content of the manuscript. All authors contributed to the article approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tang, G., Wang, Y., Deng, P. et al. Mechanism of dracorhodin in accelerating diabetic foot ulcer healing via the Nrf2 pathway, a network pharmacology, molecular docking and experimental validation. Sci Rep 15, 12492 (2025). https://doi.org/10.1038/s41598-025-97831-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-97831-5