Abstract
This study explored the kinetics of Cr(VI) conversion into Cr(III) by sodium formate on a nitrogen-doped magnetic mesoporous carbon material infused with Pd and Fe nanoparticles. This study provides the mechanism, kinetic parameters, and optimal conditions for Cr(VI) reduction by employing kinetic Monte Carlo simulations and response surface methodology. It has been proposed that Cr(OH)6 is an intermediate in this reaction. The influences of various factors, such as the initial concentrations of sodium formate and dichromate, pH, and reaction time, on the reaction efficiency were also investigated. These results suggest that the ideal conditions for Cr(III) synthesis involve a synergy of low pH and dichromate levels, a heightened sodium formate concentration, and prolonged reaction periods. The optimal conditions for Cr(III) production on Pd/Fe-NMC surface are pH = 1.37, Reaction time = 161.30 min, Cr2O72− initial concentration = 8.3 × 10−4 M, and HCOONa initial concentration = 1.02 M. Under the proposed optimal conditions, the Cr(VI) reduction efficiency of the Pd/Fe-NMC surface was 99.82%.
Similar content being viewed by others
Introduction
Water is one of the essential requirements of all life-supporting processes. Access to clean and safe water is one of the world’s main problems. The textile industry’s waste effluents usually contain high concentrations of pigments, dyes, and toxic materials, which must be removed1,2,3,4,5,6. Hexavalent chromium (Cr(VI)) is recognized as a significant environmental pollutant and a potent carcinogen in humans. The World Health Organization recommends maintaining Cr(VI) levels in drinking water below 0.05 mg/L to mitigate health hazards7. Cr(VI), a more pernicious form compared to its trivalent counterpart, is associated with a range of health issues, such as allergic reactions, anemia, skin burns, and reproductive harm. Long-term exposure and accumulation of Cr can result in various pathophysiological conditions7,8.
Various strategies have been developed to remove heavy metals from aquatic environments due to their pronounced toxicity and pollution from industrial activities. These approaches include reverse osmosis and nanofiltration9, adsorption10,11, ion exchange, chemical reduction, and coagulation processes12. Reverse osmosis and nanofiltration, which are membrane-based methods, demonstrate remarkable effectiveness in removing Cr(VI). The employment of various adsorbents in adsorption processes has shown significant advantages, especially with recent advancements in composite materials enhancing their adsorption capacity. Also, the agricultural wastes have been modified to be used as the adsorbent4,13,14. Resin-based ion exchange methods have effectively reduced contaminant levels to minimal amounts. The process of chemical reduction, succeeded by coagulation and filtration, has been established as a feasible approach through scientific verification.
Current research is directed towards creating new techniques to improve the sustainability and effectiveness of these removal processes9,12,15. The catalytic reduction method proficiently converts the highly toxic Cr(VI) to the less dangerous Cr(III), diminishing its adverse environmental effects. This approach stands out for its efficiency and eco-friendliness relative to alternative Cr(VI) elimination methods, presenting an enhanced solution for cleansing environments tainted with Cr(VI). Mesoporous carbon catalyzes the reduction of Cr(VI), possessing key characteristics such as a well-organized structure with evenly distributed pores of the same size16, increased reactivity facilitated by mesoporous that allows access to active sites16, and the ability to enhance interaction with reactants through surface functionalization17. Palladium and iron bimetallic nanoparticles have demonstrated high efficacy in converting Cr(VI) to Cr(III), rendering them highly promising for eco-friendly remediation purposes18.
Design of Experiments (DOE) is a methodical strategy employed to investigate the relationship between process variables and results, aiming to pinpoint causal relationships and enhance processes by modifying variables. Response Surface Methodology (RSM) is a DOE technique that seeks to improve processes by creating models that show the connection between many inputs and a specific output to maximize the output19,20,21. A central composite design (CCD) is utilized to build a second-order polynomial for the response variables in RSM. CCD is used to consider the importance of variables in the response.
Kinetic Monte Carlo (KMC) simulation can study the time-dependence progression of phenomena such as chemical reactions and particle diffusion22,23. This method is a variant specifically designed to investigate kinetic information, mechanisms, and fundamental processes to model the temporal evolution of a chemical system. KMC method is a simple, powerful, and flexible tool for exercising the mechanisms to gain a model of the phenomena. Combining KMC with DOE methodologies could significantly improve simulation research. This method aims to enhance the predicted accuracy and operational efficiency of the reactions24,25. Although RSM is usually used to design empirical studies, this method has been applied to simulate the catalytic reactions24,25,26,27. Recently, this method was used to theoretically examine the Cr(VI) reduction rate to Cr(III) on Pd-CNTs nanocatalyst, with HCOOH as a reducing agent28. Using KMC and DOE together, a reaction condition was proposed to give the Cr(VI) reduction efficiency as 100%.
In this study, we employed KMC simulations alongside RSM to delve into the formation of trivalent Cr(III) from the reduction of Cr(VI) using magnetic mesoporous carbon material doped with nitrogen loaded by Pd and Fe nanoparticles (Pd/Fe-NMC). Our primary aim is to develop a proposed mechanism for this reaction using the experimental work of Li et al.29. Furthermore, we will explore how different independent variables, including the concentration of sodium formate (used as a reducing agent), the concentration of dichromate (Cr2O72−), pH levels, and the duration of the reaction, influence the efficiency of Cr(III) production. Also, we will determine the most favorable conditions for this reaction.
Methods
Simulation method
Kinetic Monte Carlo methods are an advanced computational approach for simulating the temporal dynamics of atomic or molecular systems by using reaction rates to illustrate state changes. These methods are complemented by the chemical master equation, which provides an in-depth probabilistic examination of the system’s dynamic state transitions over time. In KMC simulations, the system’s condition is represented by a statistical distribution. The term Pα signifies the probability that the system is in a specific state α at time t. The effectiveness of these methods in predicting the system’s temporal behavior heavily depends on this probabilistic framework. Transition rates between states are critical for the simulation’s precision. The notation Wαβ denotes the rate at which the system transitions from state β to state α, reflecting the likelihood of this change happening within a given timeframe. Gillespie’s algorithm, integral to KMC methods, is particularly adept at simulating the stochastic development of chemical systems. It precisely determines the order and timing of reactions, such as the reversible A ↔ B, by leveraging the probabilities and rates of reactions30,31:
This approach enables detailed and precise modeling of chemical kinetics, capturing the discrete and random nature of molecular interactions.
The simulation algorithm is structured around a set of defined reactions, denoted as R = (R1, R2,…, RM), where each Ri corresponds to a specific chemical conversion; for example, R1 is associated with the transformation from A to B (A → B), and R2 with the reverse, from B to A (B → A). The rate constant, symbolized by k, quantifies the likelihood per unit of time a reaction occurs, which varies with the concentration of the chemical species. The stoichiometric coefficients indicated as ai and bi, denote the number of molecules of reactants and products participating in each reaction, respectively. This method sequentially evaluates individual reactions to depict the entire reaction system accurately. The probability of each reaction occurring is calculated using the formula ai = ki· Ci, where Ci represents the concentration of the reacting species for the ith reaction, and ki is its respective rate constant. The cumulative reaction probability, represented by "a," is computed by summing up these individual probabilities (a1, a2,…, aм), effectively integrating the likelihoods of all reactions within the system.
The rate constant, denoted as k, is affected by the concentrations of reactants and products, resulting in a nonlinear behavior of the chemical rate constant, as seen in molecular dynamics (MD) simulations. This complexity suggests that the conventional rate equations fall short, necessitating a more elaborate theoretical framework to depict even straightforward processes accurately. Two random numbers are generated in the formulas applied, each ranging from zero to one, following a uniform distribution. The first of these numbers referred to as r1, is used to calculate the duration of each step in the process, where τ symbolizes the time factor.
The symbol r2 represents the specific reaction occurring. The KMC model relies heavily on an algorithm that consistently updates the list of possible events, maintaining the alignment of the simulation’s temporal progression with real-time dynamics. This continuous updating process is a core component of the model, essential for ensuring that the simulation duration corresponds accurately to actual temporal developments32.
This research employed Chemical kinetic simulator (CKS) version 1.01 software, incorporating Gillespie’s algorithm, to perform KMC simulations. Developed at IBM’s Almaden Research Center, the CKS software package is programmed in C and C++ and has been grounded in Gillespie’s algorithm since 2001. The software has established a substantial reputation across various chemical research fields as a dependable tool for accurately simulating chemical reactions. Its widespread recognition is reflected in the significant number of citations, underscoring its efficiency and utility in computational chemistry. The software’s ability to precisely mimic complex chemical phenomena has made it an indispensable resource in the industry33,34,35.
Response surface methodology
Applying RSM involves a systematic approach, from conducting two-factor experiments to deeply exploring and evaluating essential input factors. These initial experiments are critical for identifying the main elements influencing the desired outcome. RSM employs regression analysis to explore the relationship between input variables and the resulting response, providing insights into how input changes affect the outcome. This approach improves process efficiency by narrowing the focus to the most critical variables, reducing experimental time and costs36. Optimization methods are then applied to ascertain the optimal settings for the input variables to enhance the result’s quality or efficiency33,37. Factorial designs, particularly beneficial under resource constraints, enable concurrently examining multiple variables’ effects on the result, thus offering comprehensive insights into the system under study38. A two-factorial experimental design specifically assesses how variables at different levels, high and low, affect the response variable, employing statistical methods for this analysis. This enables researchers to identify significant variables and quantify their effects on the outcome. In scenarios with k variables, two-factorial designs require 2k experiments. Fractional factorial designs are adopted for cases involving numerous variables, conducting only a portion of the 2k experiments, thereby streamlining the research process and reducing costs39.
Central Composite Design (CCD) is a prominent technique in RSM utilized for constructing second-order response surface models and executing factorial experiments. CCD excels in evaluating how input factors influence the response variable, thereby deepening the understanding of the fundamental relationships and interactions. The design strategically selects the midpoint values of critical variables as the central point, facilitating the assessment of variations and effects of other inputs40. In this study, version 7.0.0 of a renowned test design software, well-regarded in the DOE domain, was employed to identify the primary factors affecting a particular quality attribute in the process. The systematic approach of CCD is crucial for thoroughly exploring and improving the response surface, playing a vital role in process optimization. Through this method, researchers are equipped to ascertain the optimal conditions that significantly enhance the desired quality attribute41. The test design software streamlines the experimental workflow, enabling the precise and reliable determination of key factors.
Results and discussion
We have used fitting between the experimental data and simulation results to find the best mechanism and kinetic data. By using the experimental data and fitting them to the simulation results, we can get the best mechanism and kinetic data for a process. After finding the mechanism and kinetic data and confirming them with experimental data, we have used KMC simulation to suggest an optimal condition. Using RSM and the proposed mechanism and kinetic data, we can recommend the optimal condition for this process without going to the laboratory, spending money, buying materials, and wasting time.
Mechanism
Mechanism
The research presented herein draws upon the foundational experimental efforts of Li et al. (2016), who 2016 successfully synthesized a novel nitrogen-doped magnetic mesoporous carbon material loaded by palladium and iron nanoparticles (Pd/Fe-NMC). The research investigates the catalytic characteristics of Pd/Fe-NMC, focusing on its effectiveness in promoting the reduction of Cr(VI) within industrial effluents, which augments the catalytic performance of the process. It was noted that the capability of Pd-Fe/NMC to decrease Cr(VI) levels declines with an increase in the initial concentration of chromium. Sodium formate (HCOONa) was the reducing agent within this experimental framework. The experimental setup and kinetic analysis required measuring various parameters, such as the reaction temperature, its time, the initial concentrations of the reactants, the initial concentration of active sites on the catalyst, and the rate constants for different reaction stages. Specifically, the initial concentrations of Cr2O72− and HCOONa were adjusted at 0.008824 M and 0.00017 M, respectively. The reaction medium’s pH was consistently held at 2. To enhance the catalytic activity, the initial amount of active sites on the surface of the nanocatalyst was meticulously adjusted through repeated simulation iterations. The objective was to improve the correlation coefficient between the simulated outcomes and the experimental findings, which was accomplished when the concentration reached 50 M. The reaction took place at a temperature of 30 °C for 60 min. The suggested mechanism for the conversion of Cr(VI) to Cr (III) involves the interaction between HCOONa and the Pd/Fe-NMC catalyst, comprising several distinct steps:
Step 1:
Step 2:
Step 3:
Step 4:
Step 5:
Step 6:
Step 7:
Step 8:
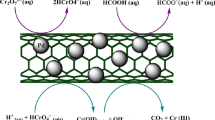
Figure 1 illustrates a comprehensive diagram of this procedure.
Schematic representation of the Cr(VI) removal process utilizing Pd-Fe/NMC.
The described stages outline the catalytic process for converting Cr(VI) to Cr(III) utilizing the Pd/Fe-NMC catalyst. This sequence includes the adsorption of reactants, the creation of intermediate compounds, and the release of end products, with the catalyst sites refreshed for future cycles. The rate constant for the initial step (k1, k-1) was lower than that of subsequent stages, designating it as the rate-limiting step within this mechanism. The rate constants for each reaction stage were established as modifiable variables and refined through repeated simulation iterations and subsequent comparisons between theoretical predictions and experimental outcomes. Table 1 outlines the rate constants determined for each specific reaction stage.
Figure 2 illustrates the alignment between the theoretical model and empirical data. The significant concordance, evidenced by the changes in Cr2O72− concentration over time, validate the proposed reaction pathway and the established rate constants, underscored by an R2 value of 0.9872. This exceptional degree of match bolsters confidence in the accuracy of the adopted reaction mechanism and kinetic parameters. In the research conducted by Li et al. (2016), the results and data affirm a reduction efficiency of 99.82% for Cr2O72−.
Comparison of experimental (circles) and predicted Cr(III) production (solid line) curve on the Pd/Fe-NMCTs surface.
Central composition design
After analyzing the study paper, we have discovered four crucial criteria that have the most significant impact on creating Cr(III). Concurrently, we determined the upper and lower limits for each variable. The variables encompass:
-
Reaction time (variable A): The selected variable for the study is the duration of the reaction, which influences the efficiency of the Cr(VI) reduction process.
-
The concentration of hydrogen ions (variable B): Based on experimental research on dichromate reduction, it was shown that pH is one of the parameters that affect the effectiveness of the process. A suitable pH range was chosen after carefully analyzing the specifics of multiple articles and experimental designs.
-
Concentration of Cr2O72− (denoted as variable C): The efficiency of the Cr2O72− reduction test is influenced by the initial concentration of Cr2O72−. Thus, one of the factors considered for this test plan is the concentration of Cr2O72−, which is established by examining relevant papers on the subject. The selection of maximum and lowest values (ranges) should be done to allow the test design program to provide valid analyses.
-
Variation in the concentration of the HCOONa (denoted as variable D): In the present study, HCOONa was used as the reducing agent. To assess the effect of the concentration of the reducing agent, an appropriate range was determined based on the research conducted by Li et al.29. The specific ranges of factors that impact the reaction for all three systems are documented in Table 2.
Enhanced results were obtained by examining factors affecting the process and adjusting the reaction’s time and pH conditions. The optimal variable was identified by thoroughly assessing the distinct features and attributes of the method. The yield percentage of Cr(III) production was calculated using the subsequent correlation:
In this equation, the production yield percentage acts as the dependent variable. The initial concentration of Cr2O72− for each experiment is indicated by a distinct symbol. The equation’s coefficient, 2, illustrates the stoichiometric relationship between the moles of one substance utilized and the moles of another substance generated. After identifying the most pivotal factors and determining their values, an appropriate experimental framework was chosen to examine the key elements’ varied magnitudes and interconnected effects. For this purpose, a central composite design was employed. In this specific framework, each variable was characterized by five distinct levels. The experimental design software facilitated the input of the highest and lowest values for the variables, thus establishing five respective levels for each. The numerical value of α was also established to be 2 (Table 2).
After entering the factors affecting the Cr(III) production process and their respective ranges of variation into the experimental design software, the program generated a series of 30 distinct experiments. Following this, 30 simulations were carried out, with the necessary adjustments applied to the variables in each case. The catalytic formation of Cr(III) in a solution containing sodium formate was emulated using kinetic simulation software, adhering to the experimental parameters defined by the design software. The yield percentage of Cr(III) production for each experiment was calculated using Eq. (12), taking into account the produced Cr(III) concentration and the initial dichromate concentration. Table 3 presents the results for the response variable along with the detailed experimental conditions of the system.
The Design Expert software suggests a model for the relationship between the variables and the response variable, which is described using the quadratic polynomial equation (Eq. 13).
The variable A indicates reaction time, variable B corresponds to pH, variable C signifies the concentration of Cr2O72−, and variable D represents the concentration of HCOO−. The equation provided designates the response variable as R1. In contrast, the primary components are represented by the variables A through D. The relationships among these elements are defined by AB through CD. In contrast, the secondary impacts of the variables are shown by A2 through D2. This statistical significance extends to all variables within the model, including reaction time, pH, sodium acetate concentration, and dichromate concentration, as well as their interactions, each showing p-values less than 0.05, underlining their relevance and significance within the framework. The factors most significantly impacting the yield percentage of Cr(VI) reduction are pH (variable B) and then dichromate concentration (variable C). The pH and dichromate concentration variables negatively affect the reduction of Cr(VI). Therefore, Cr(VI) reduction on Pd-Fe/NMC has high efficiency at acidic conditions. The factors with low effects on the yield percentage of Cr(VI) reduction are reaction time (variable D) and sodium acetate concentration (variable A). The reaction time and sodium acetate concentration variables positively affect the reduction of Cr(VI).
Sodium formate is a reducing agent in this reaction, a factor that causes Cr(VI) to be converted to Cr(III). As the presented mechanism shows, sodium formate is first converted to formate, which is then adsorbed to the catalyst’s surface. The adsorbed formate reduces Cr(VI) to Cr(III). Therefore, it is effective in the reaction yield, and its effect should be investigated. As observed in Eq. (13), the impact of sodium formate concentration on the reaction efficiency is negligible when considering its changes from 0.1 to 1.8 M, but these changes cannot be ignored. The results showed that increasing the concentration of sodium formate increases the response, although this effect is smaller than pH or Cr2O72− concentration or reaction time.
Table 4 presents an analysis of variance (ANOVA), which is employed to assess the significance and accuracy of the model put forward and to determine the influence of the variables on the dependent variable. The selected model demonstrates a high level of appropriateness, as evidenced by P-value less than 0.0001, a high F-value of 55.00, and high R2 of 0.9809. The model’s correlation coefficient, R2 = 0.9809, indicates a robust capacity to account for variations in the response variable, demonstrating how well changes in the response are explained by the model’s factors and their interactions. This high correlation coefficient suggests that the model effectively captures the variations in the dependent variable. The adjusted correlation coefficient (Adj R2 = 0.9631) aligns well with the predicted correlation coefficient (Pred R2 = 0.8899), showing good predictive accuracy. Additionally, the coefficient of variation (C.V.% = 56.89) confirms the precision and thoroughness of the experiments. The system’s signal-to-noise ratio, at 30.224, is deemed excellent. The strong concordance between observed and predicted values affirms the model’s accuracy and reliability.
Assessing diagnostic and perturbation plots
Figure 3 compares the predicted data from the program and the actual experimental results. The values are uniformly distributed around the 45° diagonal, indicating the model’s high predictive accuracy for estimating the efficiency percentage of Cr(III) production. Consequently, the model exhibits commendable precision in mirroring the experimental outcomes.
Plot comparing the predicted and actual Cr(III) production efficiency on the Pd/Fe-NMC surface.
Figure 4 demonstrates the impact of varying one parameter at a time while keeping the others fixed on the response variable. The constants are set as follows: time at 200.5, pH at 2.57, Cr2O72− concentration at 0.00505 M, and HCOONa concentration at 0.95 M. The variable causing the most significant deviation from the baseline significantly influences the outcome. The graph highlights that pH (Variable B) is the critical factor affecting Cr(III) production efficiency. Conversely, HCOONa concentration (Variable D) shows minimal deviation, suggesting a negligible impact. The plot indicates a negative correlation between the dependent variable and both dichromate concentration and pH, signifying a reduction in the dependent variable. In contrast, positive slopes for time and HCOONa concentration imply an increase in the dependent variable. pH is noted as a crucial control parameter in the adsorption of metal ions. At a pH of 0.5, Cr predominantly exists as HCrO4− and Cr2O72− ions in the solution. Additionally, this pH level increases the concentration of H+ ions on the catalyst’s surface, leading to an electrostatic attraction between Cr states and the adsorbent surface due to the charge disparity, thus enhancing adsorption. At elevated pH levels, adsorption efficiency significantly drops due to the competition between HCrO4− and OH− ions on the adsorbent’s surface. The study suggests that Cr(VI) adsorption peaks at a pH of 0.5, greatly enhancing reduction efficiency. Furthermore, the data concerning Cr2O72− concentration (Variable C) indicate a decline in removal efficiency with increasing Cr concentrations, attributed to the saturation of adsorbents’ active sites at higher concentrations.
Perturbation plot of Cr(III) production efficiency on the Pd/Fe-NMC surface.
An extended duration enhances the yield of Cr(III), providing additional time for transforming Cr2O72− ions into Cr(III). As shown in Fig. 4, the consistent and linear trend associated with HCOONa concentration indicates a negligible impact on the production efficiency of Cr(III) within the scope of the program’s explored design space.
Assessing the interaction of variables on Cr(III) production efficiency
This section utilizes Pareto graphical analysis and three-dimensional visualizations to examine the interactions between variables. Pareto analysis helps eliminate less critical factors from the process, focusing instead on the more influential ones42,43. The impact percentage of each factor is determined using Eq. (13) and the coefficients outlined in the model presented in Eq. (14).
In this context, the term Pi denotes the relative impact of each factor, while αi symbolizes the respective coefficient within the proposed model. The results obtained from this analytical approach are depicted in Fig. 5. In this framework, the pH level (Variable B) significantly influences the efficiency of Cr(III) production. Subsequently, the concentrations of Cr2O72− (Variable C) and time (Variable A) are identified as having considerable effects on the outcome variable.
Pareto graphical analysis for Cr(III) production efficiency on the Pd/Fe-NMC surface.
Optimization of effective variables on Cr(III) production efficiency
The research further investigates the interactions among essential variables and their effect on Cr(III) production efficiency in a three-stage chrome manufacturing process, using three-dimensional graphics for analysis. As described earlier, each graph is constructed based on the developed model, altering two variables simultaneously while maintaining the others constant. The set values for the variables are as follows: time = 200.5, pH = 2.75, [Cr2O72−] = 0.00505 M, and [HCOONa] = 0.95 M. Figure 6a illustrates the simultaneous effects of time (Variable A) and pH (Variable D) on Cr(III) production efficiency. Maximum efficiency in Cr(III) production is observed with the most extended duration and lowest acidity. The interplay between time and pH does not significantly alter the yield percentage of Cr(III) production. Figure 6b shows the joint effect of time (Variable A) and Cr2O72− concentration (Variable C) on Cr(III) synthesis efficiency. The graph indicates increased efficiency correlates with more extended periods and lower Cr2O72− concentrations. This suggests the most effective Cr(III) generation occurs with prolonged durations and minimal Cr2O72− concentrations, highlighting that an extended timeframe enhances Cr(III) output. Nonetheless, the efficiency diminishes with higher dichromate levels, underlining a decreased significance of time in enhancing Cr(III) yield at elevated concentrations. Figure 6c illustrates the combined effects of time (Variable A) and HCOONa concentration (Variable D) on Cr(III) production efficiency. The optimal conditions for maximizing Cr(III) production efficiency are longer time frames and higher HCOONa concentrations, indicating a significant interaction between these factors.
Plots illustrating the concurrent interaction effects on the response surface of (a) Time (A) and pH (B), (b) Time (A) and [HCOONa] (D), c Time (A) and [Cr2O72−] (C) on the Cr(III) production.
Figure 7a depicts the synergistic effects of pH (Variable B) and Cr2O72− concentration (Variable C) on Cr(III) production efficiency. The highest efficiency is observed at the minimum values of both pH and Cr2O72− concentration variables. As seen in this figure, a simultaneous increase in the concentration of Cr2O72−and pH causes a decrease in Cr(VI) reduction. This negative result is also shown in Eq. (13). pH has a negative effect on the reaction efficiency. According to step 6 of the mechanism, H+is necessary to reduce Cr(VI) to Cr(III), and the reaction proceeds in acidic conditions. Similarly, increasing the initial concentration of Cr2O72− causes a decrease in the efficiency. In the conditions we considered for the experimental design, both of these parameters, pH and Cr2O72− concentration, cause a reduction in the response. The surface plot (Fig. 7a) and Eq. (13) also showed that a simultaneous increase in these two variables causes an increase in the response, and a concurrent rise in these two variables causes a decrease in the response.
Plots illustrating the concurrent interaction effects on the response surface (a) pH (B) and [Cr2O72−] (C), (b) pH (B) and [HCOONa] (D), (c) [Cr2O72−] (C) and [HCOONa] (D) on the Cr(III) production.
Figure 7b shows the impact of combining pH (Variable B) and HCOONa concentration (Variable D) on Cr(III) synthesis efficiency, measured in percentage. The relationship between pH and HCOONa concentration reveals that HCOONa concentration does not vary significantly at higher pH levels. Hence, the most favorable conditions for Cr(III) production efficiency are lower pH values and higher HCOONa concentrations. In Fig. 7c, the interaction between Cr2O72− concentration (Variable C) and HCOONa concentration (Variable D) on Cr(III) synthesis efficiency is demonstrated. The efficiency of Cr(III) production diminishes with increased Cr2O72− concentration but improves with a higher HCOONa concentration. Consequently, optimal efficiency in Cr(III) synthesis is achieved by combining low Cr2O72− concentration with high HCOONa levels.
The software specifies recommended values and ranges for the variables, as outlined in Table 5. The model’s predictive accuracy was validated through simulations of the catalytic process for Cr(III) production using the optimal variable settings. The agreement between the computed values and the predicted outcomes affirms the model’s reliability, which was established through the response surface methodology. The simulation results demonstrate a higher Cr(III) yield than the experimental values, suggesting enhanced production efficiency under the proposed optimal conditions. The findings from the simulations in this study are contrasted with empirical results from existing literature, detailed in Table 6. This study aimed at improving the conversion from Cr(VI) to Cr(III) utilizing a nitrogen-doped magnetic mesoporous carbon framework embedded with palladium and iron nanoparticles (Pd/Fe-NMC), as reported by Li et al.29. The derived optimal conditions, featuring a Cr2O72− concentration of 0.00083 M and an HCOONa concentration of 1.02 M, resulted in a 99.82% reduction of Cr2O72−, which closely matches the 100% efficiency achieved in laboratory conditions, as indicated in Table 6.
Additionally, the efficiency of the Pd/Fe-NMC system in reducing Cr2O72− was compared with other systems, including nitrogen-doped porous carbon with magnetic nanoparticles (Fe₃O₄ and Fe) known as RHC-mag-CN44, magnetic mesoporous carbon hybrids infused with Pd nanoparticles (Pd/Fe-NMC)29, Fe3O4/Pd nanoparticles within a nitrogen-doped carbon shell (Fe₃O₄/Pd@NC) magnetic iron nanoparticles within ordered mesoporous carbon (Fe/CMK-3), and magnetic mesoporous carbon with polyaniline (PANI–Fe/OMC)47.The present study exhibits superior Cr₂O₇2− reduction efficiency compared to most cited works. Nevertheless, it should be noted that the Fe₃O₄/Pd@N–C catalyst described in45 achieved a 100% reduction efficiency, outperforming our catalyst in this specific criterion.
Conclusion
This study explored a simulation examination of the Cr(VI) reduction rate on Pd/Fe-NMC using the reducing agent HCOONa. We utilized KMC simulation paired with RSM to understand the reaction mechanisms and identify the optimal conditions for Cr(VI) reduction. The mechanism and kinetics parameters of Cr(VI) reduction on Pd/Fe-NMC were studied using experimental data. The Cr(OH)6 was proposed as an intermediate of this reaction. The effects of variables such as [HCOONa], [Cr2O72−], pH, and reaction time were studied on Cr(VI) reduction. The results showed that pH is the most effective parameter for Cr(IV) reduction. Decreasing pH and [Cr2O72−] increases the reaction yield, while [HCOONa] and Reaction time positively influence it. The optimum condition was obtained, which gives the Cr(IV) reduction efficiency of Pd/Fe-NMC 99.82%. The successful application of these optimal conditions demonstrates the potential as an effective catalyst and emphasizes the necessity of precise parameter control for achieving the best results. This work contributes to developing a clean and sustainable method for chromium reduction, aiding in reducing environmental pollution and advancing sustainability in chemical manufacturing.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Batool, M., Javed, T., Wasim, M., Zafar, S. & Din, M. I. Exploring the usability of cedrus deodara sawdust for decontamination of wastewater containing crystal violet dye. Desalin. Water Treat. 224, 433–448 (2021).
Imran, M. S., Javed, T., Areej, I. & Haider, M. N. Sequestration of crystal violet dye from wastewater using low-cost coconut husk as a potential adsorbent. Water Sci. Technol. 85(8), 2295–2317 (2022).
Majeed, H. J. et al. Synthesis and application of novel sodium carboxy methyl cellulose-g-poly acrylic acid carbon dots hydrogel nanocomposite (NaCMC-g-PAAc/ CDs) for adsorptive removal of malachite green dye. Desalin. Water Treat. 320, 100822 (2024).
Bashiri, H., Nesari, S. & Sarabadan, M. Microwave assistant production of a high performance adsorbent from rice husk. Korean J. Chem. Eng. 37(2), 240–248 (2020).
Sarabadan, M., Bashiri, H. & Mousavi, S. M. Removal of crystal violet dye by an efficient and low cost adsorbent: Modeling, kinetic, equilibrium and thermodynamic studies. Korean J. Chem. Eng. 36(10), 1575–1586 (2019).
Sarabadan, M., Bashiri, H. & Mousavi, S. M. Adsorption of crystal violet dye by zeolite-montmorillonite: modeling, kinetic and equilibrium studies. Clay Miner. 54(4), 357–368 (2019).
Gözeten, İ & Tunç, M. Palladium nanoparticles supported on activated carbon (C) for the catalytic hexavalent chromium reduction. Water Air Soil Pollut. 233, 1–14 (2022).
Hossini, H. et al. A comprehensive review on human health effects of chromium: Insights on induced toxicity. Environ. Sci. Pollut. Res. 29(47), 70686–70705 (2022).
Mnif, A.; Bejaoui, I.; Mouelhi, M.; Hamrouni, B., Hexavalent chromium removal from model water and car shock absorber factory effluent by nanofiltration and reverse osmosis membrane. International journal of analytical chemistry 2017, 2017.
Shah, A. et al. Adsorptive removal of arsenic from drinking water using KOH-modified sewage sludge-derived biochar. Cleaner Water 2, 100022 (2024).
Shah, A. et al. Removal of cadmium and zinc from water using sewage sludge-derived biochar. Sustainable Chemistry for the Environment 6, 100118 (2024).
Bahowick, S.; Dobie, D.; Kumamoto, G., Ion-exchange resin for removing hexavalent chromium from ground water at treatment facility C: Data on removal capacity, regeneration efficiency, and operation. Lawrence Livermore National Laboratory 1993.
Sabando-Fraile, C., Corral-Bobadilla, M., Lostado-Lorza, R. & Somovilla-Gomez, F. Eco-efficient cadmium(II) removal from water using alcohol distillate waste: A study of life cycle assessment. Environ. Technol. Innov. 37, 103916 (2025).
Falaki, Z. & Bashiri, H. Preparing an adsorbent from the unused solid waste of Rosewater extraction for high efficient removal of Crystal Violet. J. Iran. Chem. Soc. 18(10), 2689–2702 (2021).
Pakade, V. E., Tavengwa, N. T. & Madikizela, L. M. Recent advances in hexavalent chromium removal from aqueous solutions by adsorptive methods. RSC Adv. 9(45), 26142–26164 (2019).
Cheng, Y., Zhou, W. & Zhu, L. Enhanced reactivity and mechanisms of mesoporous carbon supported zero-valent iron composite for trichloroethylene removal in batch studies. Sci. Total Environ. 718, 137256 (2020).
Kokorina, A. A., Prikhozhdenko, E. S., Sukhorukov, G. B., Sapelkin, A. V. & Goryacheva, I. Y. Luminescent carbon nanoparticles: synthesis, methods of investigation, applications. Russ. Chem. Rev. 86(11), 1157 (2017).
Shi, D., Ouyang, Z., Zhao, Y., Xiong, J. & Shi, X. Catalytic reduction of hexavalent chromium using iron/palladium bimetallic nanoparticle-assembled filter paper. Nanomaterials 9(8), 1183 (2019).
Kim, D. et al. Polymer-supported Zn-containing imidazolium salt ionic liquids as sustainable catalysts for the cycloaddition of CO2: a kinetic study and response surface methodology. ACS sustainable chemistry & engineering 6(11), 14743–14750 (2018).
Bagheri, S.; Aghaei, H.; Monajjemi, M.; Ghaedi, M.; Zare, K., Novel Au–Fe3O4 NPs loaded on activated carbon as a green and high efficient adsorbent for removal of dyes from aqueous solutions: Application of ultrasound wave and optimization. Eurasian J Analytical Chem 2018, 13 (3).
Dil, E. A. et al. Modeling and optimization of Hg 2+ ion biosorption by live yeast Yarrowia lipolytica 70562 from aqueous solutions under artificial neural network-genetic algorithm and response surface methodology: kinetic and equilibrium study. RSC Adv. 6(59), 54149–54161 (2016).
Mohamadi, S. & Bashiri, H. Kinetic study of hydrogen sulfide decomposition on Pt(111) surface. Int. J. Chem. Kinet. 52(1), 16–22 (2020).
Bashiri, H. & Mohamadi, S. Hydrogen sulfide decomposition on Ni surface: A kinetic Monte Carlo study. Appl. Catal. A 509, 105–110 (2016).
Pahlevanpour, G. & Bashiri, H. Kinetic Monte Carlo simulation of hydrogen production from photocatalytic water splitting in the presence of methanol by 1 wt% Au/TiO2. Int. J. Hydrogen Energy 47(26), 12975–12987 (2022).
Pahlevanpour, G.; Bashiri, H., Photocatalytic hydrogen production by Ni/TiO2 (0.5 wt%): Kinetic Monte Carlo simulation. Journal of the Taiwan Institute of Chemical Engineers 2023, 152, 105159.
Rafiee, M. & Bashiri, H. Catalytic decomposition of formic acid on Cu (100): optimization and dynamic Monte Carlo simulation. Catal. Commun. 137, 105942 (2020).
Ghamsari, Z. S. & Bashiri, H. Hydrogen production through photoreforming of methanol by Cu (s)/TiO2 nanocatalyst: optimization and simulation. Surfaces and Interfaces 21, 100709 (2020).
Hajloo, H. & Bashiri, H. Optimizing Cr(VI) reduction to Cr(III) using Pd-CNTs nanocatalyst: kinetic Monte Carlo simulation and experimental design insights. Sci. Rep. 14(1), 19192 (2024).
Li, S. et al. Catalytic reduction of hexavalent chromium by a novel nitrogen-functionalized magnetic ordered mesoporous carbon doped with Pd nanoparticles. Environ. Sci. Pollut. Res. 23, 22027–22036 (2016).
Wittkop, M., Sommer, J.-U., Kreitmeier, S. & Göritz, D. Monte Carlo simulations of a single polymer chain under an external force in two and three dimensions. Phys. Rev. E 49(6), 5472 (1994).
Gillespie, D. T. A general method for numerically simulating the stochastic time evolution of coupled chemical reactions. J. Comput. Phys. 22(4), 403–434 (1976).
Jalali, H. M., Bashiri, H. & Rasa, H. Study of photo-oxidative reactivity of sunscreening agents based on photo-oxidation of uric acid by kinetic Monte Carlo simulation. Mater. Sci. Eng., C 50, 59–63 (2015).
Wu, Y., Zhang, S., Guo, X. & Huang, H. Adsorption of chromium (III) on lignin. Biores. Technol. 99(16), 7709–7715 (2008).
Chand, R., Watari, T., Inoue, K., Torikai, T. & Yada, M. Evaluation of wheat straw and barley straw carbon for Cr(VI) adsorption. Sep. Purif. Technol. 65(3), 331–336 (2009).
Hespanhol, I. & Prost, A. WHO guidelines and national standards for reuse and water quality. Water Res. 28(1), 119–124 (1994).
Sabando-Fraile, C., Corral-Bobadilla, M., Lostado-Lorza, R. & Cabredo-Pinillos, S. A synergistic integration of life cycle assessment and response surface methodology to optimize grape stalk waste-based biosorption for effective Cd(II) removal. J. Clean. Prod. 450, 141938 (2024).
Junyapoon, S. & Weerapong, S. Removal of hexavalent chromium from aqueous solutions by scrap iron fillings. KMITL Science and Technology Journal 6, 1–12 (2006).
Shao-feng, N., Yong, L., Xin-Hua, X. & Zhang-hua, L. Removal of hexavalent chromium from aqueous solution by iron nanoparticles. J. Zhejiang Univ. Sci. B 6, 1022–1027 (2005).
Rafati, L.; Asgari, A. R.; Mahvi, A. H., Evaluating the performance of Iron Nano Particle Resin (Liwatit FO36) in Removing Cr(VI) from Aqueous Solutions. 2009.
Rahmani, A.; Nourouzi, R.; Samadi, M.; Afkhami, A., Hexavalent chromium removal from aqueous solution by produced iron nanoparticles. 2009.
Shirzad Siboni, M. et al. Photocatalytic removal of hexavalet chromium and divalent nickel fromaqueous solution by UV irradiation in the presence of titanium dioxide vanoparticles. Iranian Journal of Health and Environment 3(3), 261–270 (2010).
Nöh, K., Niedenführ, S., Beyß, M. & Wiechert, W. A Pareto approach to resolve the conflict between information gain and experimental costs: multiple-criteria design of carbon labeling experiments. PLoS Comput. Biol. 14(10), e1006533 (2018).
Irawan, D., Naujoks, B., Bäck, T. & Emmerich, M. Dominance-based variable analysis for large-scale multi-objective problems. Nat. Comput. 22(2), 243–257 (2023).
Li, Y. et al. N-doped porous carbon with magnetic particles formed in situ for enhanced Cr(VI) removal. Water Res. 47(12), 4188–4197 (2013).
Tian, X., Liu, M., Iqbal, K., Ye, W. & Chang, Y. Facile synthesis of nitrogen-doped carbon coated Fe3O4/Pd nanoparticles as a high-performance catalyst for Cr(VI) reduction. J. Alloy. Compd. 826, 154059 (2020).
Tang, L. et al. Synergistic effect of iron doped ordered mesoporous carbon on adsorption-coupled reduction of hexavalent chromium and the relative mechanism study. Chem. Eng. J. 239, 114–122 (2014).
Yang, G. et al. Effective removal of Cr(VI) through adsorption and reduction by magnetic mesoporous carbon incorporated with polyaniline. RSC Adv. 4(102), 58362–58371 (2014).
Acknowledgements
Our thanks to the University of Kashan for Grant No. (1143438/6).
Author information
Authors and Affiliations
Contributions
Conception and design of study: H. Bashiri; H. Hajloo. Collected the data, analysis and/or interpretation of data: H. Hajloo. Wrote the manuscript: H. Hajloo; H. Bashiri. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hajloo, H., Bashiri, H. Theoretical insights in catalytic reduction of Cr(VI) using Pd/Fe nitrogen doped mesoporous carbon. Sci Rep 15, 14746 (2025). https://doi.org/10.1038/s41598-025-98082-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-98082-0