Abstract
Various studies have reported an association between physical activity and grey matter volumes. Some studies have suggested that this relationship may be moderated by sex, yet the direction is still under debate. Focusing on hippocampus and dorsolateral prefrontal cortex (dlPFC), we tested whether the association between regional grey matter volumes and self-reported physical activity differs between women and men. We examined this interaction in five European cohorts from the Lifebrain consortium (n = 1809; age range: 18–88 years). Effect sizes were first determined by linear models run separately for each cohort, then pooled across datasets in a random-effects meta-analysis. Contrary to our hypotheses, there was no evidence of a relationship between physical activity and hippocampal or dlPFC volumes, nor was there a moderation by sex. Our null findings raise the question of whether self-report questionnaires of physical activity, which commonly feature in big datasets, are sufficiently sensitive to capture a—presumably modest—association between physical activity levels and grey matter outcomes. We conclude that the reliance on self-report questionnaires of physical activity is sub-optimal for brain-behaviour analyses.
Similar content being viewed by others
Introduction
A physically active lifestyle is associated with a higher likelihood of having a healthy brain, both in younger and older populations. In school-aged children, increased aerobic fitness has been associated with executive function1 and larger hippocampi2. In older adults, physical activity is associated with indices of brain structure3,4,5 and even delayed onset or less likely occurrence of neurological illness, such as dementia6. Attempts to translate these associations into physical activity interventions that promote healthy brain ageing have yielded promising, yet mixed, results7,8,9,10. Across observational and interventional studies, considerable individual variability in the physical activity-brain relationship can be observed, plausibly contributing to the heterogeneity of findings. One plausible, yet often overlooked, moderator of the physical activity-brain association is biological sex11.
Globally, the prevalence of insufficient physical activity differs between men and women, with women showing reduced activity levels12. In terms of cognition, physical activity may have greater beneficial effects on cognitive function in women than in men13,14,15, although the reverse has also been reported16. Given that sex differences may influence the relationship between physical activity and cognition, one would expect a similar sex difference in the efficacy of physical activity on brain structure—particularly the neural correlates of cognitive function. In terms of brain structure, no consensus has yet been reached regarding the direction of any potential interaction, even for the hippocampus, the most commonly examined region of interest in the exercise literature. Studies of the moderating effect of sex on associations between physical activity and hippocampus have also given contradictory results, including stronger associations in women17, men18, or not differing between sexes19. Accordingly, further investigation of the interaction between physical activity and sex in their effect on brain structures is needed to reveal robust and replicable associations.
Using pooled data from five independent European cohorts of the Horizon 2020 Lifebrain consortium20, we investigated the moderating effect of biological sex on the relationship between self-reported physical activity and MRI-derived measures of brain structures. In a previous publication, we found that a cumulative unhealthy lifestyle score was negatively associated with total grey matter volume21. Using a binary classification of physical inactivity, Binnewies and colleagues21 further found no evidence that sex moderated the association between being classified as physically inactive and total grey matter or hippocampal volumes. However, the use of categorical data for physical activity levels may not be sensitive enough to the full range of physical activities nor the small effects often observed in lifestyle-brain associations22. To account for the full continuum of physical activity levels, and their potential interaction with sex on brain outcomes, the current analysis used the continuous scale of derived metabolic equivalents of task (METs) minutes per week. We selected as outcome measures grey matter regions often examined in the exercise-brain literature, i.e., hippocampal volume and the dorsolateral prefrontal cortex (dlPFC). The hippocampus has a track-record of observed plasticity during physical activity interventions10,23,24,25 and may also show sex differences in its vulnerability to ageing26. Similarly, the dlPFC shows age-related changes27 and has been associated with physical activity, both in interventions28 and in observational studies29,30,31. Here, we tested if, in line with findings in the cognitive domain, volumetric indices of brain structure would show a stronger association in females than in males.
Methods
Study samples
The Lifebrain consortium aims to promote harmonised multicohort analyses across European studies on brain structure and function20. Here, we used all Lifebrain cohorts with available physical activity (in METs) and structural MRI data. Cohorts outside of Lifebrain were not included in the analyses. This resulted in a total of 1809 participants, drawn from five different Lifebrain cohorts (Table 1): the Cambridge Centre for Ageing and Neuroscience study (Cam-CAN, UK)32, Center for Lifebrain Changes in Brain and Cognition longitudinal studies (LCBC, Norway)33, Live Active Successful ageing study (LISA, Denmark)34, Whitehall-II Imaging Sub-study (UK)35 and the Netherlands Study of Depression and Anxiety (NESDA, Netherlands)36. In longitudinal studies (LISA, LCBC), only data from baseline were used. From the NESDA study, which recruited a combination of participants diagnosed with depression and anxiety and healthy controls, only the healthy controls were selected for the present analysis. We further excluded participants who had missing physical activity data or no T1-weighted MRI brain scan. A description of each study’s sample is available in Appendix A.
All studies were conducted in accordance with the guidelines and regulations set out by their relevant authorities and the Declaration of Helsinki. Informed consent was obtained from all participants, and each study received approval from their local ethical review board: the Whitehall II Imaging Sub-study from the Oxford Central University Ethics Committee (MSD‐IDREC‐C1‐2011‐71), Cam-CAN from the Cambridgeshire 2 Research Ethics Committee (reference: 10/H0308/50), LCBC from the Norwegian Regional Committee for Medical and Health Research Ethics, LISA from the Capital Region, Copenhagen (H-3-2014-017) and the Danish Data Protection Agency, and NESDA from the ethical review board of each participating research centre in Amsterdam, Leiden, and Groningen (METC Number 2003-183).
Measures
Physical activity
Physical activity questionnaires were used to estimate total physical activity in metabolic equivalent of task minutes per week (MET-min/week). The questionnaires included the international physical activity questionnaire (IPAQ) in LCBC and NESDA, the community healthy activities model program for seniors (CHAMPS) in Whitehall-II, the EPIC-EPAQ2 in Cam-CAN and the physical activity scale for the elderly (PASE) in LISA. Although the questionnaires differed between cohorts, all enabled the calculation of MET-min/week and reflected the type, frequency, and intensity of current physical activities.
MRI acquisition and processing
T1-weighted images were automatically processed in a harmonised manner across studies with the recon-all stream of FreeSurfer (version 6.0 or 5.3)37. The derived regions of interest (ROIs) included the hippocampus, dlPFC (“caudal middle frontal”) and total grey matter (GMV). Left and right hippocampal volumes were considered separately in light of previous reports that the left hippocampus may be more susceptible to the effects of exercise10. For the dlPFC, the average of the left and right volumes was used because we had no a priori hypothesis for hemispheric differences in relation to physical activity. Estimated intracranial volumes (ICV), as outputted by FreeSurfer (eTIV), were included as covariates in the model. For an overview of the scanner type, strength and MR acquisition parameters used in each cohort, see Appendix A.
Statistical analysis
Statistical analyses were carried out in RStudio version 1.4.110338, running on R version 3.6.039, with the “stats” (R Core Team, 2016) and “meta”40 packages.
For each cohort, a generalised linear model (GLM) was fitted for each volumetric brain outcome (total GM, left hippocampus, right hippocampus, dlPFC), with physical activity (MET-min/week), age, sex and ICV as the independent variables (model 1). The GLMs were then repeated to include the physical activity*sex interaction term (model 2). Sex variables were coded in the same direction across cohorts (female = 1, male = 0). As two different scanners were used in the Whitehall II data collection, “scanner” was added as an additional covariate for this study. In a supplementary analysis, we tested whether the inclusion of BMI (kg/m2) as a covariate altered results. For each brain outcome, the standardised coefficients for physical activity (from model 1) and physical activity*sex (from model 2) were then pooled across cohorts in a random-effects meta-analysis. As a measure of heterogeneity, Cochran’s Q (henceforth “Q”) was calculated for each meta-analysis. The alpha-level was Bonferroni corrected to account for the four ROIs (α = 0.0125). Finally, to compare the fit of the proposed models, model evaluation was conducted using Akaike information criteria (AIC), and Akaike weights. The AIC for each model was obtained using the R package “stats”. The AIC values were then transformed to Akaike weights, as defined in Wagenmakers and Farrell41.
Two post-hoc sensitivity analyses were conducted. First, we explored a three-way interaction term (physical activity*sex*age) in each cohort separately. In line with previous literature focusing solely on older adults, we then repeated the random-effects meta-analyses in participants aged 60 years or older. Second, to better understand which aspects of physical activity the MET values capture, we correlated METs with other measures of physical activity, physical function, and fitness in the LISA study. The LISA study was selected for this additional analysis as it offered a wide range of physical activity and function measures. To generate further hypotheses, an exploratory analysis also probed the interaction between physical activity and sex on other brain regions reported to show marked age-related changes (ref.42; Appendix B).
Results
Pooled across cohorts, physical activity levels did not significantly vary with age or sex (Appendix C), even though there was a large negative age effect in CAM-CAN, which included the widest age range, and a large positive sex effect (wherein men reported more METs than women) in Whitehall, which had the smallest proportion of females across the five studies. At the same time, there was a large heterogeneity of effects across the five cohorts, for both age (Q = 81.98, p < 0.001) and sex (Q = 35.59, p < 0.001).
No significant pooled estimates for physical activity (Figs. 1, Fig. 2), or the interaction between sex and physical activity (Fig. 3), were observed. No significant pooled estimates were observed when the analysis was stratified by sex (Appendix D), nor when BMI was included as a covariate (Appendix E).
For each brain ROI, a random effects meta-analysis was conducted to pool the associations between physical activity and grey matter volumes across cohorts, including age, sex and ICV as covariates in the model.
A scatterplot to illustrate the association between physical activity and total grey matter volume (GMV), with one linear model per cohort, using z-scores.
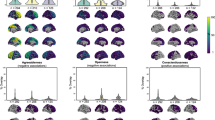
For each brain ROI, a random effects meta-analysis was conducted. Each forest plot illustrates the cohort-specific and pooled beta values for the sex \(\times\) physical activity interaction for one of the structural brain outcomes, including age, sex, ICV and physical activity effects as covariates in the model.
In a post-hoc analysis, we used AIC model selection to distinguish among three possible models describing the relationship between age, sex, ICV, physical activity and volumetric brain outcomes (Appendix F). For all outcomes, there was no evidence in favour of including the interaction term (physical activity*sex). For three cohorts, the best-fit model included physical activity as a parameter in relationship to the left (AICweight (LISA) = 0.45, AICweight (NESDA) = 0.43) or right (AICweight (LCBC) = 0.50, AICweight (NESDA) = 0.54) hippocampus. For all other outcomes, the best fitting model did not include physical activity, or its interaction, as a parameter.
Exploratory analyses, including additional regions of interest, showed that sex moderated the association between physical activity and volume of the medial temporal lobe, with women showing a stronger association between physical activity and volume in the medial temporal lobe. However, this was not significant after correcting for multiple comparisons (b = 0.06; 95% CI 0.01–0.11; p = 0.023). No other pooled estimates for the sex*physical activity interaction were observed (Appendix B).
Sensitivity analyses
Older adults
Analyses were repeated with samples restricted to participants aged 60 years or older (n = 1,647). Given the low number of older participants in the LCBC (n = 5) and NESDA (n = 0) cohorts, these samples were not included in this sensitivity analysis. Again, across cohorts, there was no evidence for an association between grey matter volumes and physical activity, nor for the interaction between sex and physical activity (Appendix G). An additional exploratory analysis of the three-way interaction between sex, age and physical activity is also presented in Appendix G.
What’s in a MET?
To test the validity of total MET mins/week as a measure of physical activity, we conducted post-hoc correlational analyses in the LISA cohort (n = 401), which included multiple indices of physical function, physical activity, and physical fitness (Fig. 4; Appendix H). Total MET mins/week was associated with accelerometer-measured step count (r = 0.31, p < 0.001), but not with gait speed (r = 0.04, p = 0.42) or estimated cardiorespiratory fitness (r = 0.03, p = 0.55).
Heatmap illustrating correlation coefficients from the associations between measures of physical function, physical activity and estimated physical fitness in the LISA cohort (n = 401). eCRF estimated cardiorespiratory fitness.
Discussion
Using data pooled from five European cohorts, the present analyses aimed to test whether sex moderated associations between physical activity (PA) levels and brain structure. Contrary to our hypotheses, there was no evidence of such an interaction. Our interpretations are, however, limited by the finding that there was also no significant relationship between physical activity levels and grey matter volumes. Accordingly, this analysis did not reveal any of the expected associations.
Previous observational studies have repeatedly associated physical activity with volumetric indices of brain structure43,44,45. For example, in the UK Biobank an accelerometer-derived estimate of physical activity, was associated with larger total grey matter volume as well as right hippocampal volume in 10,083 adults aged 50 years or older. In the same analysis, sex was a significant moderator - whereby the physical activity relationship was only significant in men46. In contrast, and in a much smaller sample (n = 92), Varma and colleagues found that daily step count was associated with larger hippocampal volumes in older women, but not in older men (≥ 60 years;17). Unlike these two studies, our samples covered a much wider age range (18–87 years old). Others have noted that the link between physical activity and grey matter volume was only significant in older adults19 and that physical activity may have different associations with brain structure at different stages of the lifespan47, raising a plausible explanation for the discrepancy in our findings. However, our results remained unchanged (i.e., null) when restricting the sample to those aged 60 years or older.
In contrast to the above-described studies, our PA outcome measure was derived from self-reported PA questionnaires. Others have, like us, also not found a significant association when using self-reported outcomes48,49, with some exceptions50,51. Here, physical activity levels were defined by METs, or the “metabolic equivalent of task”, as estimated from PA questionnaires. Physical activity questionnaires are cheap and easy to administer, making them a common feature of large datasets. However, estimated METs have several confounds. First, MET intensity values are typically obtained from a compendium of physical activities, which was developed based on data from healthy adults aged 18 to 65 years old. Importantly, these values may not reflect energy costs of older adults or children performing the same task52. While this can be circumvented by using age-adjusted energy cost values, as done in questionnaires specifically designed for older adults (e.g., community healthy activities model program for seniors (CHAMPS)), this limits the comparability of values across age groups. Regarding comparability across studies, it is also important to note that the included MET estimates were calculated from different physical activity questionnaires (PASE, CHAMPS, IPAQ and EPAQ). All values were z-scored in the analyses to increase the comparability between studies. As a result, we have lost the ability to interpret how much physical activity (in METs) corresponds to a change in brain volume. Finally, as recently argued by Gill and colleagues53, questionnaires overestimate physical activity during vigorous efforts but underestimate physical activity during intermittent activities of daily living (e.g. going up a flight of stairs).
Further, as observed in our sensitivity analysis in the LISA study, the correlations between objectively-measured physical activity (e.g., accelerometer-based step count, estimated cardiorespiratory fitness) and MET values estimated from self-report were modest at best, a limitation which has been well-described elsewhere54. In older adults, a tendency to under-report physical activity has previously been noted55, and the accuracy of self-reported physical activity levels may even vary with sex and BMI56. Accordingly, and in light of previous null results48,49, these limitations raise the question of whether the use of objective measures may be preferable to PA questionnaires for assessing the brain-PA relationship. In the likely event that a full adoption of objective measures is not feasible for larger studies (but see57), an in-sample validation of the applied physical activity questionnaires relative to more objective measures would be a step forward for the field. The literature on this topic is unlikely to become any clearer before ensuring the validity of our physical activity measures.
Once the quality of the measurements has been strengthened, the next step will be to identify the key ingredients of physical activity and fitness that most effectively modify brain structure. For example, when looking at the functional integrity of ageing-related brain networks, cardiorespiratory fitness (CRF), but not physical activity, has been shown to play a role58. Sex differences in the relationship between CRF and brain structure have also been reported—as increased CRF was associated with greater bilateral subiculum volume in older women, but not men59. As new cohort studies require a decision about which variables to include in their protocols, we encourage researchers to broaden their scope and include measures beyond conventional physical activity questionnaires, such as fitness (or resting heart rate as a proxy for fitness e.g., Jackson, Sui60) and physical function.
Pooling data across multiple countries also invites questions around the cross-cultural validity of the applied questionnaires. For example, using data from five countries, a study examining the level of agreement between accelerometer- and questionnaire-based physical activity found language-specific differences in the level of agreement when comparing tests administered in English, Czech or Danish. There may, therefore, also be systematic linguistic (or cultural) differences in the interpretation of questionnaire items61.
In line with these differences, variations between study samples may have contributed to a potential loss in sensitivity when combining across cohorts. In a previous report, we observed that despite an overall similarity in the association of lifestyle factors and brain structure, the underlying pattern of associations between lifestyle factors and brain structure outcomes differed substantively between two cohorts of older adults also included in the present analyses (Whitehall and LISA)62. If associations between gray matter volumes and physical activity are small, pooling across varied populations with self-reported measurements may wash out any (predominantly modest) lifestyle-brain associations. Combining data from five European cohorts also had its advantages. First, the access to individual-level data enabled us to run harmonised analyses in each cohort before estimating the overall effect of physical activity, and physical activity by sex, on structural brain outcomes. Second, it resulted in a larger sample size. No a priori power analysis was conducted, raising the question of whether we were powered to detect potential interactions. Unlike analyses of main effects, interactions require much larger samples63. Even so, previous significant findings in a sample of 92 individuals17, suggest this was not the case in the present paper.
Conclusion
Understanding factors that influence the relationship between self-reported physical activity and brain structure may enable the development of tailored, and ultimately more effective, physical activity interventions for brain health. Here, using data from five European datasets, we found no evidence to suggest that the relationship between physical activity and indices of brain structure was moderated by biological sex. We also did not observe an association between grey matter volumes and physical activity. We propose that the reliance on self-report measures may not be sufficiently sensitive to probe associations, much less interactions, between physical activity levels and brain outcomes across the lifespan.
Data availability
The data analysed in this study is subject to the following restrictions: data may be made available upon reasonable request, given appropriate ethical and data protection approvals. Requests to access these datasets should be directed to the PI from each cohort: Klaus P Ebmeier (Whitehall II), Carl-Johan Boraxbekk (LISA), Kristine B Walhovd (LCBC), Rik Henson (Cam-CAN), Brenda Penninx (NESDA). For more information, see: www.lifebrain.uio.no/about/access-to-data/.
References
Xue, Y., Yang, Y. & Huang, T. Effects of chronic exercise interventions on executive function among children and adolescents: a systematic review with meta-analysis. Br. J. Sports Med. 53 (22), 1397–1404 (2019).
Chaddock, L. et al. A neuroimaging investigation of the association between aerobic fitness, hippocampal volume, and memory performance in preadolescent children. Brain Res. 1358, 172–183 (2010).
Dunas, T. et al. Multimodal image analysis of apparent brain age identifies physical fitness as predictor of brain maintenance. Cereb. Cortex 31 (7), 3393–3407 (2021).
Boraxbekk, C. J. et al. Physical activity over a decade modifies age-related decline in perfusion, gray matter volume, and functional connectivity of the posterior default-mode network-A multimodal approach. Neuroimage 131, 133–141 (2016).
Raji, C. A. et al. Exercise-related physical activity relates to brain volumes in 10,125 individuals. J. Alzheimers Dis. 97 (2), 829–839 (2024).
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the lancet commission. Lancet 396 (10248), 413–446 (2020).
Firth, J. et al. Effect of aerobic exercise on hippocampal volume in humans: A systematic review and meta-analysis. Neuroimage 166, 230–238 (2018).
Gogniat, M. A., Robinson, T. L. & Miller, L. S. Exercise interventions do not impact brain volume change in older adults: a systematic review and meta-analysis. Neurobiol. Aging 101, 230–246 (2021).
Sexton, C. E. et al. A systematic review of MRI studies examining the relationship between physical fitness and activity and the white matter of the ageing brain. Neuroimage 131, 81–90 (2016).
Demnitz, N. et al. Hippocampal maintenance after a 12-month physical activity intervention in older adults: The REACT MRI study. Neuroimage Clin. 35, 102762 (2022).
Barha, C. K. et al. Biological sex: A potential moderator of physical activity efficacy on brain health. Front. Aging Neurosci. 11, 329 (2019).
Strain, T. et al. National, regional, and global trends in insufficient physical activity among adults from 2000 to 2022: a pooled analysis of 507 population-based surveys with 5.7 million participants. Lancet Glob. Health 12, e1232–e1243 (2024).
Barha, C. K. et al. Sex differences in aerobic exercise efficacy to improve cognition: A systematic review and meta-analysis of studies in older rodents. Front. Neuroendocrinol. 46, 86–105 (2017).
Colcombe, S. & Kramer, A. F. Fitness effects on the cognitive function of older adults: a meta-analytic study. Psychol. Sci. 14 (2), 125–130 (2003).
Kramer, A. F. & Colcombe, S. Fitness effects on the cognitive function of older adults: A Meta-analytic study-revisited. Perspect. Psychol. Sci. 13 (2), 213–217 (2018).
Zotcheva, E. et al. Effects of 5 years aerobic exercise on cognition in older adults: The generation 100 study: A randomized controlled trial. Sports Med. 52 (7), 1689–1699 (2022).
Varma, V. R. et al. Low-intensity daily walking activity is associated with hippocampal volume in older adults. Hippocampus 25 (5), 605–615 (2015).
Casaletto, K. B. et al. Sexual dimorphism of physical activity on cognitive aging: Role of immune functioning. Brain Behav. Immun. 88, 699–710 (2020).
Hamer, M., Sharma, N. & Batty, G. D. Association of objectively measured physical activity with brain structure: UK biobank study. J. Intern. Med. 284, 439–443 (2018).
Walhovd, K. B. et al. Healthy minds 0–100 years: Optimising the use of European brain imaging cohorts (“Lifebrain”). Eur. Psychiatr. 50, 47–56 (2018).
Binnewies, J. et al. Lifestyle-related risk factors and their cumulative associations with hippocampal and total grey matter volume across the adult lifespan: a pooled analysis in the European lifebrain consortium. Brain Res. Bull. 200, 110692 (2023).
Anaturk, M. et al. Prediction of brain age and cognitive age: Quantifying brain and cognitive maintenance in aging. Hum. Brain Mapp. 42 (6), 1626–1640 (2021).
Duzel, E., van Praag, H. & Sendtner, M. Can physical exercise in old age improve memory and hippocampal function?. Brain 139 (Pt 3), 662–673 (2016).
ten Brinke, L. F. et al. Aerobic exercise increases hippocampal volume in older women with probable mild cognitive impairment: a 6-month randomised controlled trial. Br. J. Sports Med. 49 (4), 248–254 (2015).
Erickson, K. I. et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 108 (7), 3017–3022 (2011).
Ritchie, S. J. et al. Sex differences in the adult human brain: Evidence from 5216 UK biobank participants. Cereb Cortex 28 (8), 2959–2975 (2018).
Fjell, A. M. et al. One-year brain atrophy evident in healthy aging. J. Neurosci. 29 (48), 15223–15231 (2009).
Colcombe, S. J. et al. Aerobic exercise training increases brain volume in aging humans. J. Gerontol. A Biol. Sci. Med. Sci. 61 (11), 1166–1170 (2006).
Northey, J. M. et al. Objectively measured physical activity is associated with dorsolateral prefrontal cortex volume in older adults. Neuroimage 221, 117150 (2020).
Weinstein, A. M. et al. The association between aerobic fitness and executive function is mediated by prefrontal cortex volume. Brain Behav. Immun. 26 (5), 811–819 (2012).
Jonasson, L. S. et al. Aerobic exercise intervention, cognitive performance, and brain structure: Results from the physical influences on brain in aging (PHIBRA) study. Front. Aging Neurosci. 8, 336 (2016).
Shafto, M. A. et al. The Cambridge centre for ageing and neuroscience (Cam-CAN) study protocol: a cross-sectional, lifespan, multidisciplinary examination of healthy cognitive ageing. BMC Neurol. 14, 204 (2014).
Walhovd, K. B. et al. Neurodevelopmental origins of lifespan changes in brain and cognition. Proc. Natl. Acad. Sci. USA 113 (33), 9357–9362 (2016).
Eriksen, C. S. et al. Physical activity as intervention for age-related loss of muscle mass and function: protocol for a randomised controlled trial (the LISA study). BMJ Open 6 (12), e012951 (2016).
Filippini, N. et al. Study protocol: the Whitehall II imaging sub-study. Bmc Psychiatr. https://doi.org/10.1186/1471-244X-14-159 (2014).
Penninx, B. W. et al. The Netherlands study of depression and anxiety (NESDA): rationale, objectives and methods. Int. J. Methods Psychiatr. Res. 17 (3), 121–140 (2008).
Fischl, B. et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33 (3), 341–355 (2002).
RStudio Team, RStudio: Integrated Development Environment for R. (2020)
R Core Team, R: A language and environment for statistical computing. R.F.F.S. Computing (2020).
Balduzzi, S., Rucker, G. & Schwarzer, G. How to perform a meta-analysis with R: a practical tutorial. Evid. Based Ment. Health 22 (4), 153–160 (2019).
Wagenmakers, E. J. & Farrell, S. AIC model selection using Akaike weights. Psychon. Bull. Rev. 11 (1), 192–196 (2004).
Nyberg, L. et al. Individual differences in brain aging: heterogeneity in cortico-hippocampal but not caudate atrophy rates. Cereb. Cortex 33 (9), 5075–5081 (2023).
Killgore, W. D., Olson, E. A. & Weber, M. Physical exercise habits correlate with gray matter volume of the hippocampus in healthy adult humans. Sci. Rep. 3, 3457 (2013).
Arenaza-Urquijo, E. M. et al. Distinct effects of late adulthood cognitive and physical activities on gray matter volume. Brain Imaging Behav. 11 (2), 346–356 (2017).
Fox, F. A. U. et al. Association between accelerometer-derived physical activity measurements and brain structure: A population-based cohort study. Neurology 99 (11), e1202–e1215 (2022).
Brown, B. M. et al. Non-modifiable factors as moderators of the relationship between physical activity and brain volume: A cross-sectional UK biobank study. J. Alzheimers Dis. 88 (3), 1091–1101 (2022).
Erickson, K. I. et al. Physical activity, cognition, and brain outcomes: A review of the 2018 physical activity guidelines. Med. Sci. Sports Exerc. 51 (6), 1242–1251 (2019).
Smith, J. C. et al. Interactive effects of physical activity and APOE-epsilon4 on BOLD semantic memory activation in healthy elders. Neuroimage 54 (1), 635–644 (2011).
Rosano, C. et al. Psychomotor speed and functional brain MRI 2 years after completing a physical activity treatment. J. Gerontol. A Biol. Sci. Med. Sci. 65 (6), 639–647 (2010).
Floel, A. et al. Physical activity and memory functions: are neurotrophins and cerebral gray matter volume the missing link?. Neuroimage 49 (3), 2756–2763 (2010).
Walhovd, K. B. et al. Blood markers of fatty acids and vitamin D, cardiovascular measures, body mass index, and physical activity relate to longitudinal cortical thinning in normal aging. Neurobiol. Aging 35 (5), 1055–1064 (2014).
Ainsworth, B. E. et al. 2011 compendium of physical activities: a second update of codes and MET values. Med. Sci. Sports Exerc. 43 (8), 1575–1581 (2011).
Gill, J. M. et al. Potential impact of wearables on physical activity guidelines and interventions: opportunities and challenges. Br. J. Sports Med. 57 (19), 1223–1225 (2023).
Prince, S. A. et al. A comparison of direct versus self-report measures for assessing physical activity in adults: a systematic review. Int. J. Behav. Nutr. Phys. Act. 5, 56 (2008).
Northey, J. M. et al. Can the intensity of physical activity be accurately measured in older adults using questionnaires?. J. Sci. Med. Sport 22 (7), 803–807 (2019).
Quinlan, C. et al. The accuracy of self-reported physical activity questionnaires varies with sex and body mass index. PLoS ONE 16 (8), e0256008 (2021).
Doherty, A. et al. Large scale population assessment of physical activity using wrist worn accelerometers: The UK biobank study. PLoS ONE 12 (2), e0169649 (2017).
Voss, M. W. et al. Fitness, but not physical activity, is related to functional integrity of brain networks associated with aging. Neuroimage 131, 113–125 (2016).
Kern, K. L., Storer, T. W. & Schon, K. Cardiorespiratory fitness, hippocampal subfield volumes, and mnemonic discrimination task performance in aging. Hum. Brain Mapp. 42 (4), 871–892 (2021).
Jackson, A. S. et al. Longitudinal cardiorespiratory fitness algorithms for clinical settings. Am. J. Prev. Med. 43 (5), 512–519 (2012).
Cerin, E. et al. Correlates of agreement between accelerometry and self-reported physical activity. Med. Sci. Sports Exerc. 48 (6), 1075–1084 (2016).
Demnitz, N. et al. Characterising the covariance pattern between lifestyle factors and structural brain measures: a multivariable replication study of two independent ageing cohorts. Neurobiol. Aging 131, 115–123 (2023).
Giner-Sorolla, R. et al. Power to detect what? Considerations for planning and evaluating sample size. Pers. Soc. Psychol. Rev. 28 (3), 276–301 (2024).
Acknowledgements
The Lifebrain study is funded by the EU Horizon 2020 (Grant Nr. 732592). The LISA study was supported by the Nordea Foundation (Grant from Center for Healthy Aging, University of Copenhagen, Denmark) and the Whitehall MRI Sub-study was funded by the UK Medical Research Council (G1001354) and the HDH Wills 1965 Charitable Trust (Nr. 1117747). ND is supported by funding from the Lundbeck Foundation (Grant Nr. R380-2021-1269). Hartwig R. Siebner is supported by a collaborative project grant “ADAPT-PD” from the Lundbeck Foundation (Grant Nr. R186-2015-2138).
Author information
Authors and Affiliations
Contributions
ND analysed data, visualised results and prepared a draft of the manuscript, and CJB supervised the analyses and edited the manuscript. ND, CJB and RK were involved in the study conception. AMF, KBW, KPE, WFCB, HRS and LN were involved in funding acquisition and data curation. AMB, ØS and RK advised on the methodology. WFCB, JB, AMB, ANF, ATG, RK, MK, KSM, LN, SP, HRS, SS, KBW, ØS, KPE and CJB reviewed and edited the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Disclosures
HRS has received honoraria as speaker and consultant from Lundbeck AS, Denmark, and as editor (Neuroimage Clinical) from Elsevier Publishers, Amsterdam, The Netherlands. He has received royalties as book editor from Springer Publishers, Stuttgart, Germany, Oxford University Press, Oxford, UK, and from Gyldendal Publishers, Copenhagen, Denmark.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Demnitz, N., Baaré, W.F.C., Binnewies, J. et al. No significant association between self-reported physical activity and brain volumes in women and men from five European cohorts. Sci Rep 15, 19067 (2025). https://doi.org/10.1038/s41598-025-98601-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-98601-z
Keywords
This article is cited by
-
Randomized controlled trial of resistance exercise and brain aging clocks
GeroScience (2026)