Abstract
Keratinocytes, the most important cell type constituting the epidermis, migrate to restore the epithelial barrier during wound healing and are a crucial step in wound healing. This study utilized bioinformatics analysis of comprehensive expression datasets of aberrantly expressed genes in wound healing to identify the abnormal expression of the critical transcription factor Fos-like antigen-1 (FOSL1), which is involved in various diseases. Currently, there is limited research on the role of FOSL1 in wound healing, and its molecular mechanisms remain unclear. This study explores the role and regulatory mechanisms of FOSL1 in the wound-healing process. A comprehensive expression dataset of abnormal genes in wound repair was constructed by bioinformatics analysis. Mouse trauma models and mouse wound splint models were constructed to verify the role of FOSL1 in vivo. Real-time quantitative polymerase chain reaction (qRT-PCR), immunoblot, immunofluorescence staining, and HE staining were used to confirm the analysis, and FOSL1 was used as the target in the wound healing process. At the cellular level, using 5ʹ-ethynyl-2ʹ-deoxyuridine (EdU) assay, Transwell assay, Migration assay, western blotting and immunofluorescence, FOSL1 promoted the molecular mechanism of wound repair by regulating the proliferation and migration of keratinocytes through IL-17 signaling pathway. Bioinformatics analysis revealed differential expression of FOSL1 during wound healing. In the mouse back wound model, qRT-PCR, western blotting (WB), and immunofluorescence staining showed significant upregulation of FOSL1 and IL-17 expression during wound tissue healing, indicating a close association between FOSL1 and mouse wound healing. In the mouse wound splinting model, subcutaneous injection of recombinant FOSL1 protein contributed to wound surface healing. Overexpression of FOSL1 in HaCaT cells promoted their proliferation and migration abilities. When IL-17 inhibitor was added to HaCaT cells, both FOSL1 overexpression and knockdown inhibited the proliferation and migration abilities of HaCaT cells. Thus, this study confirms that FOSL1 promotes keratinocyte proliferation and migration through the IL-17 signaling pathway, facilitating wound healing in epidermal wound repair. The results of this study indicate that FOSL1 plays a key role in epidermal wound healing, and elucidate a new molecular mechanism by which FOSL1 promotes keratinocyte proliferation and migration through IL-17 signaling pathway in epidermal wound repair, thereby promoting wound healing.
Similar content being viewed by others
Introduction
The skin is the largest organ of the human body. As the outermost barrier of our body, injuries to the skin increase the risk of infection, disease and scarring. Effective wound repair is critical to individual health and survival. Mammalian skin wound healing is an evolutionarily conserved process that restores the skin’s barrier function and maintains homeostasis1. The wound repair process is mainly achieved through the regulation of different cellular and molecular pathways by the continuous activation of local cells and systemic cells, which work synergistically to restore skin integrity. As the primary cell type constituting the epidermis, Keratinocytes play various important roles in skin wound repair. After skin damage, keratinocytes (Keratinization, KC) re-form the epithelial layer through migration, proliferation, and differentiation processes. This process is called re-epithelialization. Re-epithelialization is critical for wound repair, preventing infection and restoring the normal skin barrier2. Keratinocyte migration is a critical step in the re-epithelialization process. The transformation of different cell states of keratinocytes is regulated by various wound microenvironmental factors, including growth factors, cytokines, chemokines, and matrix metalloproteinases3. Enhanced migration of keratinocytes during wound healing indicates a positive effect on the healing process, whereas delayed migration indicates a negative effect. Therefore, finding the mechanism that regulates keratinocyte proliferation, migration and differentiation plays an important role in wound healing.
Fos-like antigen-1 (FOSL1) is a basic leucine zipper (bZIP) protein and a member of the AP1 complex. It heterodimerizes with proteins of JUN family members to form an active transcription factor complex. AP-14. Fos-like antigen-1 (FOSL1) is an important transcription factor related to organ development5. In embryonic development, Fosl1 is critical for transforming embryonic stem cells into trophoblast cells and the development of the placenta. It regulates key cellular functions related to the pathogenesis of AKI, such as proliferation, differentiation, survival, epithelial-mesenchymal transition, and inflammation , cell fate and stem cell reprogramming6,7. Conventional gene deletion of Fosl1 will lead to increased embryonic lethality7,8. Fosl1 controls the osteogenic activity of osteoblasts. Fosl1-deficient mice have longer bones and reduced bone mass, and the bone phenotype of Fosl1 knockout mice is similar to low-turnover osteoporosis in humans9. FOSL1 belongs to the FOS gene family and regulates cell differentiation, proliferation and transformation10. The mRNA and protein expression of FOSL1 are upregulated in various tumor types. This molecule plays a role in proliferation, differentiation, invasion, epithelial-mesenchymal transition (EMT) and drug resistance by regulating various cellular processes. Related studies have shown that high expression of FOSL1 is characteristic of the most aggressive forms of cancer. In addition to higher AP1 transcriptional activity in malignant gliomas, the expression level of FOSL1 is also upregulated. In TNBC and malignant glioma, specimens from patients with liver11 and lymph node12 metastasis showed more intense FOSL1 staining than tumor samples from patients without metastasis, suggesting that higher FOSL1 levels may indicate the presence of metastasis. FOSL1 is differentially expressed in psoriasis and is closely related to the occurrence of psoriasis13,14. Previous studies have reported that FOSL1 regulates the proliferation of psoriatic keratinocytes and the nlrp3-mediated inflammatory response by transcriptionally activating the NF-kB signaling pathway of TRAF315. Recent studies have found that FOSL1 has a protective effect on adaptive kidneys. During acute kidney injury AKI, Fosl1 is rapidly upregulated in proximal renal tubular cells, inhibiting pro-inflammatory responses and reducing pro-inflammatory factors by promoting the expression of the renal protective protein Klotho, thereby exerting an adaptive renal protection effect16.
In previous studies, we found through bioinformatics that FOSL1 is abnormally expressed in keratinocytes during wound repair. However, current research on the role of FOSL1 in wound repair needs to be clarified. This study will explore the interaction between FOSL1 and IL-17 downstream signaling pathways. Furthermore, further explore the role and mechanism of FOSL1 in the mouse wound repair model.
Methods
Bioinformatics analysis of differentially expressed genes in keratinocytes during wound healing
Download the data set GSE30355 from the gene expression omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/geo/). Use the limma software package of R 4.1.3 software to analyze GES30355 and obtain differentially expressed genes with a set threshold of |logFC|> 2 and adjPValue < 0.05. Use the WGCNA package of R 4.1.3 software to perform WGCNA (weighted gene co-expression network) analysis to analyze 807 genes expressed explicitly in sorted keratinocytes. Set the optimal threshold to 25 to find the MEpink module closely related to the sorted keratinocyte and extract genes with network connectivity greater than 0.3 in the MEpink module. For the genes with network connectivity greater than 0.3 in the MEpink module, the “clusterprofiler” R package was used to perform GO (Gene Ontology) enrichment analysis and KEGG (Kyoto Encyclopedia of Genes) enrichment analysis17,18,19. The GO enrichment analysis included biological processes, molecular function and cellular composition.
Cell culture and treatment
Permanent keratinocytes HaCaT were purchased from Guangzhou all-perfect Biological Technology Co., Ltd. Culture in DMEM medium (SK5070, GZYXbio) containing 10% fetal bovine serum (FS0997, FUSHENBIO) at 37℃ and 5% carbon dioxide. When the number of cells reaches one million levels, digest the cells, collect them into a centrifuge tube, and centrifuge at 1000 g for 5 min at room temperature. Discard the supernatant and precipitate it into cells. Adjust the density of the cell suspension to 1 × 105 cells/mL, add 500uL of the cell suspension to each well for plating, resuspend the cells in 2 mL of an entire culture, and plate them into a 6-well plate. We used Invitrogen BLOCK-iT™ RNAi Designer to design the shRNA sequence of FOSL1 and handed it over to Shandong Weizhen Biotech for construction and synthesis. The FOSL1 overexpression vector was also constructed and synthesized by Shandong Weizhen Biotechnology and packaged into lentiviral particles. After confirming that the cells are in good condition and the density reaches 30–40%, add the virus according to the following formula: Amount of virus added to each well (uL) = MOI × number of cells/titer at infection (TU/mL) × 103. Twelve hours after virus transfection, replace with fresh complete cell culture medium. 72 h after transfection, the cells were observed and photographed under a fluorescence microscope. To block IL17, HaCaT cells were incubated with the IL17 inhibitor Secukinumab (10 μM; AbMole, China) for 24 h, and cells in the standard group were not treated.
The full-thickness skin incision trauma model was used to investigate the expression changes of FOSL1 in wound healing
Animal experiments used 4–6 weeks-old C57BL/6 J mice (Beijing HFK Bio, Beijing, China). The animals used were kept in an SPF environment. All our animal procedures followed the Guide for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Jinan University (IACUC-20220719-07). The animals were anesthetized using 1% isoflurane gas, and a circular full-thickness skin incision with a diameter of 10 mm was made in the center of the mouse’s back under sterile conditions to establish a trauma model. On days 0, 1, 3, 7 and 14, take photos to observe the wound healing and extract epidermal tissue at different healing time points.
The full-thickness skin incision trauma model was used to investigate the role of FOSL1 in wound healing
The C57BL/6 J mice (Beijing HFK Bio, Beijing, China) aged 4–6 weeks were used in the experiment. The animals used were raised in the SPF-level animal room of Guangzhou Miles Biotechnology Co., Ltd. All our animal procedures followed the Guide for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Jinan University. The animals were anesthetized using 1% isoflurane gas, and a circular full-thickness skin incision with a diameter of 10 mm was made in the center of the mouse’s back under sterile conditions. A circular splint was fixed on the surface of the incision to prevent skin retraction. The experimental group was subcutaneously injected with 0.2 mL of FOSL1 recombinant protein daily (5ug/mL FOSL1 was dissolved in 0.1 mL of pure water and then 0.1 mL of glycerol was added). The control group was injected subcutaneously with 0.1 ml of pure water and 0.1 mL of glycerin daily. On days 0, 1, 3, 7 and 14, take photos to observe the wound healing and extract epidermal tissue at different healing time points.After the experiment, we used an overdose of isoflurane gas to anesthetize the animals and euthanize them, and then took samples.
Hematoxylin–eosin staining for wound clearance detection
Mouse epidermal tissue was fixed using 4% paraformaldehyde (GS539A, GSbioth, Guangzhou), and then embedded in paraffin. Sections with a thickness of 5 mm were prepared and analyzed with hematoxylin and eosin kit (GS700A, GSbioth, Guangzhou) for dyeing. Observe the morphological changes of HE-stained sections of wound tissue under a microscope.
Western blotting to detect the expression of FOSL1-related pathway proteins
Proteins were extracted from HaCaT cells and tissues using pre-chilled RIPA buffer (GS504A, GSbioth, Guangzhou) and protein phosphatase inhibitor cocktail (GS1439A, GSbioth, Guangzhou). Protein concentration was measured using the BCA protein quantification kit (BCA02, Dingguo Biotech, Shanghai). Then, proteins (50 μg/lane) were separated using a 12% or 10% SDS-PAGE Electrophoresis kit (BeyotimeBiotech, Shanghai, China) and transferred to a 0.22um PVDF membrane. The membrane was blocked with 3% blocking buffer (Beyotime) for 1 h at room temperature and incubated with GAPDH (affinity, T0004, 1:5000), IL-17A (affinity, DF6127, 1:1500), FOSL1 (Abcam, ab252421; 1:1000), IL-17RA (affinity, DF3602, 1:1000), and incubate overnight at 4 °C. The membrane was incubated with secondary antibody horseradish peroxidase-labeled IgG (1:5,000, S0001 or S0002, affinity) for 2 h. Next, the membrane was visualized using an enhanced chemiluminescence reagent (Millipore). Western blots were analyzed using ImageJ 1.54 software (National Institutes of Health), with GAPDHn as the internal reference.
Quantitative real-time PCR (qPCR) to detect the expression of FOSL1-related pathway mRNA
Tissues and HaCaT cells were lysed in total RNA extraction reagent (BL258A, biosharp, China), and RNA was extracted according to the instructions of the RNA extraction kit (RN70, Aidlab, China). Primers were designed using Primer Primer5.0 software. PrimeScript™ RT Master Mix (RR036A, TAKARA, Japan) was then used to reverse-transcribe the RNA, and 2 × UniTaq SYBR qPCR Master Mix (ROX-) (P1504, TIANYAbiotech, Guangzhou) was used to detect the expression of the target gene. Guangzhou All-Perfect Biological Technology Co., Ltd designed and synthesized the target gene primers. Please see Table 1 for the primer sequence.
Cell migration assay
Inoculate 5 × 105 cells/well to evaluate cell migration into a 12-well plate. After fully grown cells, use a 200ul pipette tip to make a scratches perpendicular to the marking line. Remove the culture medium, gently wash it once with PBS, and put it under a microscope. Take photos, record the initial scratch status, mark the photo location, add 5% complete culture medium, and take photos at 0 and 24 h. Then, use ImageJ 1.54 software for evaluation. We set the initial scratch width to 100%, and the results are expressed as a percentage of wound closure.After washing the cells three times with PBS, trypsin was added to digest the cells. After centrifugation at 1000 r/min for 5 min, the supernatant was discarded and the suspended cells were counted. The cell concentration was adjusted to 5 × 105/mL. Add 200 μL of cell suspension to the upper chamber of the Transwell chamber. Add 600 μL of complete culture medium to the lower chamber. Place the Transwell chamber at 37 °C and continue to culture for 24 h. Remove the chamber and fix it with paraformaldehyde in PBS (4%) for 15 min. Stain with 0.1 crystal violet for 20 min. Use a cotton swab to gently wipe off the cells on the upper layer of the microporous membrane. Wash 3 times with PBS. Take pictures under a microscope and manually count the cells that migrate under the filter. Cell numbers were quantified from randomly selected areas of the same area.
EDU assay to detect cell proliferation ability
HACAT cells were plated in a 96-well plate, and experimental treatments were performed. Each group was set with three duplicate wells. After the cells adhered to the wall, the EdU cell proliferation test kit (Apollo) was used to detect the proliferation ability of HaCaT cells according to the kit’s protocol. Cell nuclei were stained using a Hoechst 33,342 staining solution. The staining results were observed under a fluorescence microscope (Keyence, Osaka, Japan). The proliferation ability was evaluated based on the proportion of edu-positive cells.
Statistical analysis
Each experiment was repeated three times independently, and the experimental results were statistically analyzed using the independent sample T test using IBM SPSS Statistics. P value < 0.05 was considered to indicate statistical significance.
Results
Differential expression of FOSL1 in wound healing
In this paper, we analysed the differentially expressed genes of normal skin epidermal cells versus sorted keratinocytes and normal human epidermal keratinocytes versus sorted keratinocytes in wound healing according to the GSE30355 dataset, and plotted a clustered heat map (Fig. 1A,B). analysed and clustered heat maps were drawn (Fig. 1A,B), then the intersection of the two analyses was taken to obtain the significantly expressed genes specific to sorted keratinocytes (Fig. 1C,D), and by WCDNA analysis, we found that the MEpink module of sorted keratinocytes was the hub gene module (Fig. 1E, and the list of hub genes is provided in Additional Material 1). We then subjected the genes in the MEpink module to protein interaction network analysis and found that FOSL1 is an important central node in wound healing, closely related to other DEGs-derived proteins (Fig. 1F,G). We then subjected the hub genes to GO and KEGG enrichment analysis (Fig. 1H,I). The results indicated that the hub genes are closely related to the IL-17 signalling pathway.There are six members of the IL-17 family, including IL-17A, IL-17B, IL-17C, IL-17D, IL-17E and IL-17F. IL-17A is the prototype of the IL-17 family and its binding to the receptor IL-17RA is required to initiate signalling. The members of the IL-17 family are known as homodimers or heterodimers to carry out their functions. Signalling begins with the binding of the cytokine to its receptor IL-17RA, the absence of which results in ineffective signalling. Therefore, we chose FOSL1 as an object of study in the promotion of wound healing by keratinocytes and hypothesised that it may regulate the IL-17 signalling pathway and play a role in promoting keratin wound healing.
(A, B) Screening of differential genes during wound healing. (C, D) Cluster heat map analysis of the differentially expressed genes of normal skin epidermal cells and sorted kereatinocytes and normal human epidermal keratinocyte and sorted kereatinocyte from the GSE30355 dataset. (E) Intersection of up-regulated and down-regulated genes during wound healing. (F) WCDNA analysis using MEpink module of kereatinocytes and genes in the hub gene module. MEpink module for protein interaction network analysis (G) FOSL1 and protein interaction network analysis derived from other DEGs (H), The GO enrichment analysis of the key genes in Figure (G, I), KEGG enrichment analysis of the key genes in panel (G).
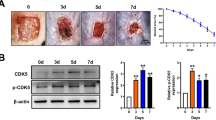
FOSL1 expression is up-regulated during trauma repair
In order to study the effect of FOSL1 on wound healing in vivo, we established a mouse back full-thickness skin excision wound model and observed the expression of wound repair on days 1, 3, 7, and 14 after mouse skin injury (Fig. 2A). Subsequently, we performed Western blot and qPCR experiments to confirm the expression changes of FOSL1 during the wound healing process. The results showed that the expression level of FOSL1 increased significantly during the wound repair process, then slowly decreased. FOSL1 reached its peak expression on day three after skin injury (Fig. 2B–D). These results prove that FOSL1 is differentially expressed in wound healing and may play a specific role in the healing process. Preliminary bioinformatics analysis showed that the wound-healing process in mice is not only related to FOSL1 but may also be closely related to the IL-17 signaling pathway; therefore, in order to explore whether the wound repair process is through activation of the IL-17 signaling pathway. We examined the expression level of IL-17 and IL-17RA through a mouse full-thickness skin excision model. We extracted the epidermal tissue from the back of mice at different healing time points on Days 0, 1, 3, 7, and 14. Furthermore, the expression levels of IL-17A and IL-17RA were detected by Western blot and qpc. The results showed that the expression of IL-17A and IL-17RA was significantly up-regulated during the wound repair process, with the highest expression levels on the 7th day, indicating that the IL-17 signaling pathway is involved in the wound repair process (Fig. 2E–G).
FOSL1 was upregulated in wound tissues. (A) Macroscopy view of wound healing on days 0, 1, 2, 3, 7, 14 mice. Scale bar: 1 mm. (B) A mouse back full-thickness skin resection trauma model in days 0, 1, 2, 3,7, The FOSL1 mRNA expression was determined by qPCR in 14. (C) A mouse back full-thickness skin resection trauma model in days 0, 1, 2, 3, 7, 14 FOSL1 protein expression was determined by Western Blot. (D) Quantification of C. All bars represent mean ± SD (n = 6). (E) qPCR detection of IL17A and IL17RA mRNA expression in traumatic epidermal tissue. (F, G) Western Blot test for the expression of IL17A and IL17RA proteins in traumatic epidermal tissue; Quantification of F. All bars represent mean ± SD (n = 3). *, P < 0.05; **, P < 0.01; ***, P < 0.001.
FOSL1 promotes wound healing
In order to explore the role of FOSL1 in wound repair, we established a wound excision splint model on the back of mice and injected FOSL1 subcutaneously to overexpress it. Photographs were taken on days 0, 1, 3, 7, and 14 to observe the wound healing. Based on the macromorphological analysis of the photos during the wound healing process, we evaluated the wound area. Compared with the control group, the healing area in the FOSL1 injection group showed a significant difference within the same time (Fig. 3A,B). At the same time, we took the epidermal tissue from the wound site and performed hematoxylin–eosin staining (HE). The wound gap was detected under a microscope, and the results showed that compared with the control group, the wound gap in the FOSL1 injection group decreased significantly at each testing time point (Fig. 3C,D). These research results indicate that FOSL1 is mainly involved in the wound repair process in vivo, and increasing FOSL1 accelerates wound healing.
FOSL1 promotes wound healing. (A) Macroscopy view of wound healing on days 0, 1, 3, 7 and 14 in FOSL1 and Control mice. (B, C) Quantification of (A–D). All bars represent mean ± SD (n = 3). (D) Wound healing as monitored by histological staining of skin sections at the wound edges at days 0, 1, 3, 7 and 14 after injury in WT, FOSL1 mice.*, P < 0.05; *, P < 0.01; ***, P < 0.001.
FOSL1 expression promotes the proliferation of HACAT cells and reduces HACAT apoptosis
In mice, we found that FOSL1 contributes to wound repair. Further, we want to understand the molecular mechanism through which FOSL1 performs wound repair at the cellular level. We constructed HaCaT cell lines that stably overexpressed FOSL1 (over-FOSL1) and stably knocked down FOSL1 (sh-FOSL1) through lentiviral technology (Fig. 4A,B). The EDU experiment was used to verify the effect of FOSL1 on keratinocyte proliferation. The experimental results showed that compared with the control group after FOSL1 overexpression, the number of EDU fluorescent cells in HaCaT was significantly reduced, and the fluorescence intensity became weaker. On the contrary, in the FOSL1 inhibition group, the number of EDU fluorescent cells increased significantly, and the fluorescence intensity did not weaken (Fig. 4C,D). It shows that overexpression of FOSL1 contributes to HaCaT cell proliferation. The scratch healing assay verified the effect of FOSL1 on the migration ability of keratinocytes. It was found that the migration ability of keratinocytes was significantly enhanced after FOSL1 overexpression, while the cell migration ability was significantly weakened after FOSL1 was silenced (Fig. 4E,F). It shows that overexpression of FOSL1 benefits HACAT cell proliferation and migration. In addition, to study the effect of FOSL1 on keratinocyte apoptosis, we performed tunnel staining on keratinocytes. The results show that after plating the same number of cells, normal HaCaT is at an average level of proliferation and apoptosis, with both proliferating and apoptotic cells (brown indicates DAB-positive cells). When FOSL1 is overexpressed, the number of brown cells in HaCaT decreases, and the level of apoptosis significantly decreases; when FOSL1 is silenced, cell proliferation slows down, and the level of apoptosis significantly increases (Fig. 4G,H). These findings indicate that FOSL1 affects the cell proliferation rate and migration ability of HACAT.
Expression of FOSL 1 promoted HACAT cell proliferation and decreased the apoptosis of HACAT. (A, B) Expression levels of FOSL 1 overexpression and knockdown mRNA were determined by qPCR. (C, D) Cell apoptosis was assessed via EdU staining assay. Green represents EdU staining and blue represents nuclear staining. Scale bar: 50 μm. (E, F) migration of FOSL1 HaCat cells for overexpression or knockdown by Transwell assays. (G, H) immunohistochemica. *, P < 0.05; *, P < 0.01; ***, P < 0.001.
FOSL1 activates IL-17 signalling pathway
Previously, we confirmed that the IL-17 signaling pathway is activated in animal wound models. In order to further explore the relationship between the IL-17 signaling pathway and FOSL1, we verified the expression of IL-17A and IL-17RA under different FOSL1 expression levels through qRT-PCR and western blot. The results showed that when the expression level of FOSL1 increased, the expression levels of IL-17A and IL-17RA increased significantly. When the expression level of FOSL1 was significantly inhibited, the expression levels of IL-17A and IL-17RA decreased significantly (Fig. 5A–D). The above shows that FOSL1 can regulate the IL-17 signaling pathway. So, does FOSL1 affect the migration of keratinocytes through the IL-17 signaling pathway? We added IL-17 inhibitor to cells with different FOSL1 expression levels and observed cell migration through the Transwell migration assay and scratch healing assay. The results showed that after using an IL-17 inhibitor, regardless of FOSL1 overexpression or knockdown, HaCaT’s migration and repair abilities were reduced (Fig. 5E–H). These results confirm that FOSL1 stimulates keratinocyte migration and promotes wound repair by activating IL-17.
FOSL 1 promoted the expression of IL-17A / IL-17A. (A, B) The FOSL1/IL-17A/IL-17RA/GAPDH level was examined by Western blot. (C, D) The IL-17A and IL-17RA mRNA level was examined by qPCR. Data are shown as mean ± S.E. (E, F) Wound healing assays were shown in HaCat cells after transfection with FOSL1-Over/shRNA and IL-17 inhibitor or NC. Cell migration was observed using a microscope (original magnification × 100). (G, H) Transwell assays were used to detect cell invasion capacities of HaCaT cells after transfection with FOSL1-Over/shRNA and IL-17 inhibitor or NC (dyeing with crystal violet; original magnification, × 200).*, P < 0.05; *, P < 0.01; ***, P < 0.001.
Discussion
Effective wound healing is a critical biological process essential for maintaining the skin’s barrier function, preventing infection, and promoting tissue regeneration. The skin is the body’s first line of defense against environmental insults, pathogens, and physical trauma. When the skin is injured, it triggers a series of complex events to facilitate wound healing20, including the activation of the inflammatory response, recruitment of immune cells, formation of new blood vessels (angiogenesis), and the proliferation and migration of various cell types21,22,23, such as keratinocytes, fibroblasts, and endothelial cells. Disruption of any of these processes can impair wound healing, leading to increased infection risk, delayed tissue repair, and the development of chronic wounds24,25.
Cell proliferation plays a crucial role in wound healing because it enables the regeneration of damaged or lost cells, ultimately leading to the restoration of the skin’s structure and function26. Understanding the molecular mechanisms that regulate skin wound healing and cell proliferation is essential for developing effective therapies to promote tissue regeneration and improve patient outcomes. FOSL1 is a transcription factor involved in regulating various cellular processes, including cell proliferation and wound repair27,28,29. Studies have shown that after nerve injury, FOSL1 is highly expressed around the injury site, enhancing Schwann cell proliferation and migration, thus facilitating nerve repair30. Furthermore, FOSL1 also helps endothelial cells differentiate into the correct tubular shape, promoting angiogenesis31,32. Through dataset analysis, we observed that the expression of FOSL1 also changes during skin wound healing. Further research revealed that overexpression of FOSL1 in HACAT cells enhanced their proliferation and migration abilities. In animal experiments, researchers induced skin wounds in animal models and investigated the impact of FOSL1 upregulation on the wound healing process. They found that FOSL1 promotes wound healing, consistent with its role in nerve injury repair and angiogenesis.
The IL-17A signaling pathway plays a crucial role in wound healing and tissue regeneration. Under normal physiological conditions, IL-17A promotes wound repair by regulating the metabolic adaptation of epithelial cells and protecting tissues by modulating the inflammatory response of neutrophils33,34,35. During the inflammatory phase of wound healing, IL-17A secreted by Th17 cells and other immune cells helps recruit and activate neutrophils, macrophages, and other inflammatory cells to the wound site36,37. The influx of these immune cells, along with the subsequent release of growth factors and cytokines, facilitates the transition to the proliferative phase, during which cell proliferation, angiogenesis, and extracellular matrix deposition occur. Additionally, IL-17A has been shown to stimulate the migration and proliferation of keratinocytes, fibroblasts, and endothelial cells, which is essential for reepithelialization, granulation tissue formation, and neovascularization during wound repair38,39,40. Therefore, the IL-17A signaling pathway plays a key role in coordinating the various stages of wound healing and promoting tissue regeneration. In this study, we also explored the relationship between FOSL1 and the IL-17A signaling pathway. We found that when FOSL1 was overexpressed, the expression of both IL-17A and IL-17RA increased. Inhibition of the IL-17A signaling pathway using an IL-17A pathway inhibitor abolished the regulatory effect of FOSL1 on the proliferation and migration abilities of HACAT cells. This suggests that FOSL1 regulates the proliferation and migration abilities of keratinocytes through the IL-17A signaling pathway.
Our findings indicate that targeting the FOSL1/IL-17A axis has therapeutic potential to enhance skin regeneration and promote wound healing. Modulating the FOSL1-mediated IL-17A pathway may offer a promising approach for developing new therapies to accelerate wound healing and improve skin regeneration. This could be particularly beneficial for the treatment of chronic or hard-to-heal wounds, which present significant challenges in clinical practice. Further research is needed to fully elucidate the potential mechanisms by which FOSL1 regulates the IL-17A pathway and explore potential therapeutic applications targeting this axis.
Data availability
The data provided in this study are stored in ScienceDB. The data can be obtained by sending an email to the corresponding author.The data link: https://www.scidb.cn/s/aae6Rf.
References
Eming, S. A., Martin, P. & Tomic-Canic, M. Wound repair and regeneration: mechanisms, signaling, and translation. Sci. Transl. Med. 6(265), 265sr266 (2014).
Werner, S. & Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 83(3), 835–870 (2003).
Pastar, I. et al. Epithelialization in wound healing: A comprehensive review. Adv. Wound Care (New Rochelle) 3(7), 445–464 (2014).
Zhang, L., Xiang, J., Zhang, F., Liu, L. & Hu, C. Mscs can be a double-edged sword in tumorigenesis. Front. Oncol. 12, 1047907 (2022).
Sobolev, V. V. et al. Role of the transcription factor Fosl1 in organ development and tumorigenesis. Int. J. Mol. Sci. 23(3), 1521 (2022).
Pecce, V. et al. The role of Fosl1 in stem-like cell reprogramming processes. Sci. Rep. 11(1), 14677 (2021).
Lee, B. K. et al. Fosl1 overexpression directly activates trophoblast-specific gene expression programs in embryonic stem cells. Stem Cell Res. 26, 95–102 (2018).
Schreiber, M. et al. Placental vascularisation requires the Ap-1 component Fra1. Development 127(22), 4937–4948 (2000).
Eferl, R. et al. The fos-related antigen Fra-1 is an activator of bone matrix formation. Embo J. 23(14), 2789–2799 (2004).
Zhang, L. Z. et al. Overexpression of Fra-1, C-Jun and C-Fos in odontogenic keratocysts: Potential correlation with proliferative and anti-apoptotic activity. Histopathology 73(6), 933–942 (2018).
Diesch, J. et al. Widespread Fra1-dependent control of mesenchymal transdifferentiation programs in colorectal cancer cells. PLoS ONE 9(3), e88950 (2014).
Dong, J. et al. Transcriptional super-enhancers control cancer stemness and metastasis genes in squamous cell carcinoma. Nat. Commun. 12(1), 3974 (2021).
Adil, S. et al. A computational systems analyses to identify biomarkers and mechanistic link in psoriasis and cutaneous squamous cell carcinoma. Front. Immunol. 12, 662528 (2021).
Benhadou, F. et al. Epidermal autonomous Vegfa/Flt1/Nrp1 functions mediate psoriasis-like disease. Sci. Adv. 6(2), eaax5849 (2020).
Liang, Y., Han, D., Zhang, S. & Sun, L. Fosl1 regulates hyperproliferation and Nlrp3-mediated inflammation of psoriatic keratinocytes through the Nf-Kb signaling via transcriptionally activating Traf3. Biochim. Biophys. Acta Mol. Cell Res. 1871(4), 119689 (2024).
Cuarental, L. et al. The transcription factor Fosl1 preserves klotho expression and protects from acute kidney injury. Kidney Int. 103(4), 686–701 (2023).
Kanehisa, M. & Goto, S. Kegg: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28(1), 27–30 (2000).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28(11), 1947–1951 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Kawashima, M. & Ishiguro-Watanabe, M. Kegg for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res. 51(D1), D587-d592 (2023).
Sorg, H., Tilkorn, D. J., Hager, S., Hauser, J. & Mirastschijski, U. Skin wound healing: An update on the current knowledge and concepts. Eur. Surg. Res. 58(1–2), 81–94 (2017).
Ren, H., Zhao, F., Zhang, Q., Huang, X. & Wang, Z. Autophagy and skin wound healing. Burns Trauma 10, tkac003 (2022).
Larouche, J., Sheoran, S., Maruyama, K. & Martino, M. M. Immune regulation of skin wound healing: Mechanisms and novel therapeutic targets. Adv. Wound Care (New Rochelle) 7(7), 209–231 (2018).
Peña, O. A. & Martin, P. Cellular and molecular mechanisms of skin wound healing. Nat. Rev. Mol. Cell Biol. 25(8), 599–616 (2024).
Raziyeva, K. et al. Immunology of acute and chronic wound healing. Biomolecules 11(5), 700 (2021).
Wilkinson, H. N. & Hardman, M. J. Wound healing: Cellular mechanisms and pathological outcomes. Open Biol. 10(9), 200223 (2020).
Qiu, H. et al. Prospective application of exosomes derived from adipose-derived stem cells in skin wound healing: A review. J. Cosmet. Dermatol. 19(3), 574–581 (2020).
Maurus, K. et al. The Ap-1 transcription factor Fosl1 causes melanocyte reprogramming and transformation. Oncogene 36(36), 5110–5121 (2017).
Banerjee, J., Lodhi, N. & Nguyen, B. N. The role of Poly(Adp-Ribose) polymerase-1 in cutaneous wound healing. Adv. Wound Care (New Rochelle) 8(12), 634–643 (2019).
Balli, M. et al. Activator protein-1 transcriptional activity drives soluble micrograft-mediated cell migration and promotes the matrix remodeling machinery. Stem Cells Int. 2019, 6461580 (2019).
Chen, Q., Zhang, L., Zhang, F. & Yi, S. Fosl1 modulates schwann cell responses in the wound microenvironment and regulates peripheral nerve regeneration. J. Biol. Chem. 299(12), 105444 (2023).
Wang, M. et al. Transcription factor Fosl1 promotes angiogenesis of colon carcinoma by regulating the vegf pathway through activating Timp1. Biochem. Genet. 62(5), 3389–3402 (2024).
Galvagni, F., Orlandini, M. & Oliviero, S. Role of the Ap-1 transcription factor Fosl1 in endothelial cells adhesion and migration. Cell Adh. Migr. 7(5), 408–411 (2013).
Wang, J. & Ding, X. Il-17 signaling in skin repair: Safeguarding metabolic adaptation of wound epithelial cells. Signal Transduct. Target. Ther. 7(1), 359 (2022).
Dhamija, B. et al. Il-17a orchestrates reactive oxygen species/Hif1α-mediated metabolic reprogramming in psoriasis. J. Immunol. 212(2), 302–316 (2024).
Bechara, R., McGeachy, M. J. & Gaffen, S. L. The metabolism-modulating activity of Il-17 signaling in health and disease. J. Exp. Med. 218(5), e20202191 (2021).
Shibabaw, T. Inflammatory cytokine: Il-17a signaling pathway in patients present with Covid-19 and current treatment strategy. J. Inflamm. Res. 13, 673–680 (2020).
Takagi, N. et al. Il-17a promotes neutrophilic inflammation and disturbs acute wound healing in skin. Exp. Dermatol. 26(2), 137–144 (2017).
Zepp, J. A. et al. Il-17a-induced Plet1 expression contributes to tissue repair and colon tumorigenesis. J. Immunol. 199(11), 3849–3857 (2017).
Piipponen, M., Li, D. & Landén, N. X. The immune functions of keratinocytes in skin wound healing. Int. J. Mol. Sci. 21(22), 8790 (2020).
Furue, M., Furue, K., Tsuji, G. & Nakahara, T. Interleukin-17a and keratinocytes in psoriasis. Int. J. Mol. Sci. 21(4), 1275 (2020).
Acknowledgements
We appreciate each participant providing assistance in this study.
Funding
This work was supported by Science and Technology Projects in Guangzhou (2024A03J1041).
Author information
Authors and Affiliations
Contributions
All authors participated in the conception and design of the study. Mao Haoran and Jiang Xiao were responsible for the experimental operations, data compilation, and formal analysis. Xie Guanghui, Liu Hongwei, and Xiao Liling were responsible for funding and project management. Liang Jiaji, Zhang Lei, Yang Zixian, and Chen Zhijing were responsible for methodology, and Qiao Jinlong, An Xifeng, and Li Xuangu were responsible for paper writing. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Yes, the first animal experiment was approved by the Experimental Animal Ethics Committee of Jinan University with the approval number IACUC-20220719-07. The second animal experiment was approved by the Guangzhou Myers Biological Animal Center Experimental Committee with the approval number IACUC-MIS20230074. In conducting the project, we followed the policies of the Nature Portfolio journal and the ARRIVE Guidelines 2.0.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mao, H., Jiang, X., Liang, J. et al. FOSL1 promotes keratinocyte migration and wound repair by modulating the IL17 signaling pathway. Sci Rep 15, 16457 (2025). https://doi.org/10.1038/s41598-025-99128-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-99128-z
Keywords
This article is cited by
-
Aucklandiae Radix Ameliorates Sepsis-Associated Acute Gastrointestinal Injury by Enhancing Intestinal Barrier Function: An Integrated Multi-omics Study
Innovations in Acupuncture and Medicine (2025)
-
Enhancing peripheral nerve regeneration in aging: the role of Schwann cells, c-Jun, and emerging therapeutic strategies
GeroScience (2025)