Abstract
Adipose tissue-derived stem cells (ADSCs) possess multipotent differentiation potential and significant immunomodulatory properties, making them valuable in regenerative medicine. However, their adipogenic differentiation can lead to triglyceride accumulation, chronic inflammation, and metabolic dysfunction. This study evaluated the effects of Radio Electric Asymmetric Conveyer (REAC) technology tissue optimization regenerative adipogenesis reprogramming (TO RGN-AR) on ADSC differentiation, focusing on its ability to preserve stemness, suppress adipogenesis, and promote beneficial phenotypes. REAC TO RGN-AR treatment significantly increased the expression of stemness-related genes (Oct-4, Sox2, and Nanog) while downregulating the expression of adipogenic markers (PPAR-γ, LPL, and ACOT2). Additionally, REAC TO RGN-AR treated cells presented a phenotypic shift toward beige adipocytes, characterized by increased TMEM26 expression and reduced ASC-1 expression. These findings underscore the novelty of using REAC TO RGN-AR to modulate cellular endogenous bioelectrical activity, presenting a noninvasive and operator-independent approach to enhance ADSC-based therapies. This work highlights the potential of this treatment to address metabolic disorders and chronic inflammation while advancing regenerative medicine.
Similar content being viewed by others
Introduction
The introduction does not include a heading and should expand on the background of the topic, typically including in-text citations1. Adipose tissue-derived stem cells (ADSCs) have emerged as promising tools in regenerative medicine because of their abundance, ease of isolation, and differentiation potential1,2. As a subset of mesenchymal stem cells (MSCs), ADSCs are capable of differentiating into multiple lineages3, including adipogenic4, osteogenic, and chondrogenic lineages5, and exhibit significant immunomodulatory properties. These features position ADSCs as key candidates for therapies targeting tissue regeneration6, immune modulation7,8, and metabolic dysfunction8.
Despite their versatility, ADSC differentiation into adipogenic lineages poses challenges for therapeutic applications9, particularly in conditions associated with metabolic disorders10. Adipogenic differentiation involves lipid accumulation and the activation of transcriptional regulators such as peroxisome proliferator-activated receptor gamma (PPAR-γ)11 and CCAAT-enhancer-binding proteins (C/EBPs)12, leading to the formation of mature adipocytes4. This process is closely associated with metabolic dysfunctions, including insulin resistance and chronic low-grade inflammation13, which are hallmarks of obesity-related diseases such as type 2 diabetes and cardiovascular disorders4.
White, brown, and beige adipocytes differ in morphology, function, and metabolic relevance. White adipocytes mainly store triglycerides and contribute to the release of pro-inflammatory adipokines. Brown adipocytes, characterized by abundant mitochondria and the expression of uncoupling protein 1 (UCP1), dissipate energy as heat. Beige adipocytes are inducible within white fat depots and exhibit thermogenic and anti-inflammatory properties, representing a metabolically favorable phenotype. Understanding and modulating the shift between these cell types has become crucial for therapeutic approaches addressing metabolic and inflammatory pathologies.
The potential to modulate ADSC differentiation while preserving their stemness is of significant clinical interest. Endogenous bioelectrical modulation, an emerging field in regenerative medicine, has demonstrated promise in influencing cellular behavior through the restoration of endogenous bioelectrical homeostasis. Radio electric asymmetric conveyer (REAC) technology represents a novel approach in this domain, delivering asymmetrically conveyed radio electric fields to noninvasively modulate cellular activity14,15,16. This asymmetric conveyance ensures the selective targeting of the endogenous bioelectrical imbalances, spares unaffected areas, and promotes cellular responses that restore physiological homeostasis.
Previous REAC studies have demonstrated that specific exposure protocols can prime differentiation and reprogramming processes. For example, REAC stimulation enhanced cardiac, neuronal, and skeletal muscle gene expression in mouse embryonic stem cells17; directly reprogrammed human fibroblasts toward these same lineages without chemical or genetic vectors18; and optimized multipotency and lineage commitment in human adipose-derived stem cells obtained via Lipogems19. These findings consolidate the methodological foundation and biological rationale of the present study.
Research has shown that Specific therapeutic protocols of REAC technology are capable of promoting tissue regeneration20,21,22, reduces inflammation23,24,25, and enhances metabolic responses26,27.
The latter include improvements in oxidative metabolism, mitochondrial efficiency, and restoration of anabolic–catabolic equilibrium, as previously documented in REAC-treated cellular systems.
This study investigated the impact of REAC tissue optimization regenerative adipogenesis reprogramming (TO RGN-AR) on ADSCs, focusing on its ability to modulate molecular markers and phenotypic outcomes associated with adipogenic differentiation.
By exploring the early stages of adipogenic commitment and the induction of beige phenotypic traits under REAC-induced bioelectrical modulation, this work aims to provide new mechanistic insight into how restoring bioelectrical homeostasis may counteract metabolic dysfunction and promote regenerative processes.
Methods
Cell isolation and culture
ADSCs were isolated after written informed consent was obtained from the subcutaneous adipose tissue of male and female patients during general surgery procedures, who were not diagnosed with obesity, diabetes, or other related metabolic diseases (n = 6, age = 45 ± 15 years, BMI: 22 ± 3 kg/m2). The study was approved by the Review Board of the Human Studies Ethics Committee of Sassari (n° ETIC 240I/CE 26 July 2016, Ethical Committee, ASL Sassari) All experimental procedures were performed in accordance with relevant guidelines and regulations, including the Declaration of Helsinki and institutional protocols. Samples of adipose tissue were washed in PBS (Euroclone, Milan, Italy), minced into small fragments and digested with type I collagenase solution for 1 h at 37 °C (Gibco Life Technologies, Grand Island, NY, USA). ADSCs were immunomagnetically separated from the total population isolated and characterized by flow cytometry, as previously described28. The cells were then grown in basic growth medium consisting of Dulbecco’s modified Eagle’s medium (DMEM) (Life Technologies Grand Island, NY, USA) supplemented with 20% fetal bovine serum (FBS) (Life Technologies, Grand Island, NY, USA), 200 mM L-glutamine (Euroclone, Milan, Italy), and 200 U/mL penicillin and 0.1 mg/mL streptomycin (Euroclone, Milan, Italy).
Adipogenic differentiation
Adipogenic differentiation was induced via specific adipogenic differentiation medium (DM) (StemPro adipocyte differentiation medium; Gibco Life Technologies, Grand Island, NY, USA). Adipogenesis was induced for a total of 14 days and assessed through immunofluorescence analysis of specific adipogenic markers. The experimental groups included untreated cells (negative control), cells exposed to DM (positive control), and cells exposed to both DM and TO RGN-AR.
REAC treatment protocol
The REAC TO-RGN-AR treatment was performed using the REAC BENE mod. 110 medical device (ASMED, Scandicci, Italy), which is specifically designed to deliver asymmetrically conveyed radio electric fields for bioelectrical modulation. Asymmetric Conveyor Probes (ACPs) were positioned inside the culture plates, ensuring appropriate contact with the culture medium.
The device emits extremely weak radio electric fields, which interact with cellular structures to target and modulate areas of altered endogenous bioelectrical activity. The treatment protocol used in this study is a proprietary, manufacturer-defined sequence of impulses designed for tissue optimization and adipogenic reprogramming. The device parameters are factory pre-set and cannot be altered by the operator, ensuring reproducibility and standardization across all experiments.
Control groups were cultured under identical environmental and experimental conditions but were not exposed to the radio electric field. This methodological approach reproduces the same exposure setup described in previous REAC cellular studies17,18,19, which demonstrated consistent and specific bioelectrical modulation effects on stem and somatic cells.
The treatment was applied continuously for 72 h under controlled conditions (37 °C, 5% CO₂), ensuring consistent cellular viability and experimental reproducibility. The cells were then maintained in culture for up to 14 days in the presence of basic growth- or differentiation-conditioned media. The device parameters, fixed by the manufacturer, preclude operator adjustments, standardizing treatment across replicates. The control groups were cultured under identical environmental conditions without exposure to the radio electric field, which served as a baseline for comparison. This approach underscores the unique ability of asymmetrically conveyed radio electric fields to noninvasively modulate cellular processes, warranting further investigation into their molecular effects.
Molecular analysis
Quantitative RT‒qPCR was used to evaluate the expression of stemness-related Oct-4, Sox2, and Nanog29 and adipogenic-related (ACOT230, LPL31, aP231, PPAR-γ11 and UCP132) markers. Gene expression analysis was performed after 14 days of culture. Total RNA was extracted via a ChargeSwitch kit (Thermo Fisher Scientific, Grand Island, NY, USA). Approximately 1 µg of total RNA from each sample was quantified via a NanoDrop™ One/OneC microvolume UV‒Vis spectrophotometer (Thermo Fisher Scientific, Grand Island, NY, USA) and used for qPCR. Real-time quantitative PCR was performed via Luna Universal qPCR Master Mix (Euroclone, Milan, Italy) in a CFX Thermal Cycler (Bio-Rad, Hercules, CA, USA).
Each reaction was performed in triplicate for each experimental condition, and the mean of the technical replicates was used for analysis. Individual data points are therefore not available, as the values represent the averaged results of each experimental condition.
The target Ct values of each sample were normalized to those of hGAPDH, which was considered a reference gene. The relative expression levels of all analyzed genes are expressed as fold change (2^ − ΔΔCt) compared to untreated control ADSCs. This approach allows normalized and reproducible comparison between groups while minimizing intra-assay variability, consistent with standard qPCR practice in REAC cellular studies17,18,19.
The primer sequences used are shown in Table 1.
Immunofluorescence
Immunofluorescence analysis was used to detect ASC-1 (marker of white adipocytes), PAT2 (marker of brown adipocytes), and TMEM26 (marker of beige adipocytes). All antibodies were previously validated for specificity and reactivity in human cell models in published REAC studies17,18,19, under identical culture and fixation conditions, confirming their reliability for the present work.
The immunostaining was performed according to standard protocols. Cells were fixed with 4% paraformaldehyde, permeabilized, and incubated with primary antibodies overnight at 4 °C. After washing, samples were incubated with Alexa Fluor–conjugated secondary antibodies, counterstained with DAPI, and visualized using a fluorescence microscope (Leica Microsystems, Germany).
Statistical analysis
All data were analyzed using GraphPad Prism software (version 10.0). Before choosing the statistical test, data distribution was evaluated using the Shapiro–Wilk test, and homogeneity of variances was assessed using Levene’s test. When normality and homoscedasticity assumptions were met, parametric analyses (two-way ANOVA followed by Tukey’s post hoc test) were applied to evaluate the effects of treatment and differentiation medium. When these assumptions were not satisfied, non-parametric tests (Kruskal–Wallis and Wilcoxon signed-rank tests) were used instead. Each figure legend specifies the statistical test applied. Results are expressed as mean ± standard deviation (SD) for parametric data or as median with interquartile range (IQR) for non-parametric data, depending on the distribution. Statistical significance was set at p < 0.05.
Results
Stemness gene expression
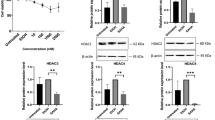
The expression of stemness markers Oct-4, Sox2, and Nanog29 was significantly upregulated in ADSCs treated with REAC TO-RGN-AR compared with that in untreated controls and in cells cultured in the presence of adipogenic differentiation medium alone (DM). These results were consistent across six independent experiments, demonstrating the robustness of the findings.
Each measurement represents the mean of triplicate reactions for each condition, expressed as fold change relative to untreated controls. Importantly, this upregulation persisted even under differentiation-inducing conditions, which typically suppress the expression of stemness markers and promote lineage commitment.
This preservation of stemness indicates that REAC TO-RGN-AR treatment helps maintain cellular plasticity and self-renewal capacity, consistent with previous REAC studies on embryonic stem cells and human fibroblasts17,18, (Fig. 1).
Expression levels of stemness-related genes Oct-4, Sox2, and Nanog in untreated ADSCs, ADSCs cultured with differentiation medium (DM), and ADSCs exposed to REAC TO-RGN-AR. Results are expressed as fold change relative to untreated controls.
Dipogenic marker modulation
Adipogenic differentiation markers, including PPAR-γ11, LPL31, and ACOT230, were significantly downregulated in REAC-treated ADSCs compared with those in cells exposed to adipogenic differentiation medium alone.
This reduction indicates that REAC TO-RGN-AR treatment suppresses the transcriptional program leading to adipogenic commitment and lipid accumulation. Importantly, cell viability remained above 95% in all experimental groups, confirming that the observed modulation was not associated with cytotoxic or inhibitory effects on cell proliferation.
The concurrent upregulation of UCP130, a thermogenic gene characteristic of beige and brown adipocytes, suggests that REAC TO-RGN-AR promotes a shift toward a metabolically active and anti-inflammatory phenotype. This interpretation is consistent with previous REAC studies reporting similar transcriptional trends in adipose-derived stem cells19.
These findings confirm that REAC TO-RGN-AR treatment effectively suppresses adipogenic differentiation, affecting both the molecular and functional hallmarks of this process. Overall, the results indicate that REAC-induced bioelectrical modulation may help redirect ADSC fate away from white adipogenesis toward beige-like differentiation, a process potentially linked to AMPK/mTOR-regulated energy pathways. The selectivity of REAC TO-RGN-AR in modulating adipogenesis without affecting the overall viability or proliferation of ADSCs further underscores its therapeutic potential (Fig. 2).
Relative expression of adipogenic and thermogenic genes (PPAR-γ, LPL, ACOT2, and UCP1) in ADSCs cultured in control medium, DM, or DM plus REAC TO-RGN-AR. Data are presented as mean ± SD from six independent experiments.
Phenotypic analysis
Immunofluorescence analysis revealed striking phenotypic differences between the REAC-treated and control groups. The cells treated with adipogenic differentiation medium alone presented high expression of ASC-133, a white adipocyte marker (Fig. 3). REAC-TO-RGN-AR treatment also effectively suppressed the expression of ASC1 despite the presence of DM, (Fig. 3).
Representative immunofluorescence images showing ASC-1 expression in untreated ADSCs, ADSCs cultured in DM, and ADSCs exposed to REAC TO-RGN-AR. Nuclei were counterstained with DAPI. Scale bars: 40 µm. The fluorescence intensity was calculated using an image software analysis (ImageJ).
Conversely, REAC-treated cells presented an increase in the expression of TMEM2634 (Fig. 4), a beige adipocyte marker, and a reduction in ASC-1 expression. The increased expression of TMEM26, together with PAT234 (Fig. 4), a specific brown adipocyte marker, indicates a shift toward the beige adipocyte phenotype, which is associated with thermogenic and anti-inflammatory properties (Fig. 4 and Fig. 5).
Representative immunofluorescence images showing TMEM26 expression in untreated ADSCs, ADSCs cultured in DM, and ADSCs exposed to REAC TO-RGN-AR. Nuclei were counterstained with DAPI. Scale bar: 40 µm. The fluorescence intensity was calculated using an image software analysis (ImageJ).
Representative immunofluorescence images showing PAT2 expression in untreated ADSCs, ADSCs cultured in DM, and ADSCs exposed to REAC TO-RGN-AR. Nuclei were counterstained with DAPI. Scale bar: 40 µm. The fluorescence intensity was calculated using an image software analysis (ImageJ).
The fluorescence intensity and distribution were consistently observed across independent replicates, and the representative images shown correspond to the most reproducible and well-defined patterns. Although quantitative morphometric analysis and co-staining with Bodipy were not performed, the qualitative consistency of the results supports their reliability. The antibodies used for ASC-1, TMEM26, and PAT2 were previously validated for specificity and localization in earlier REAC studies on human fibroblasts and adipose-derived stem cells18,19, confirming their suitability for the present work.
Importantly, no morphological alterations, abnormal proliferation, or cytotoxic effects were observed in any group, indicating that REAC TO-RGN-AR exposure did not compromise cell viability or phenotype integrity.
Cells treated with REAC TO-RGN-AR alone, without differentiation medium, presented negligible expression of all the markers, demonstrating the specificity of the treatment in modulating adipocyte lineage commitment. Taken together, these findings indicate that REAC TO-RGN-AR not only suppresses white adipogenic differentiation but also promotes the development of a metabolically favorable beige-like phenotype.
Cell morphology
ADSC morphology was evaluated by optical microscopy. ADSCs cultured in the presence of the adipogenic differentiation medium and treated with REAC showed reduced mature adipocytes, as compared to ADSCs cultured in the presence of differentiation medium alone (DM – REAC), that showed an increased number of lipid droplets (Fig. 6). The same Figure showed that ADSCs treated with REAC in the presence of the basic growing medium maintained their typical morphology and proliferation rate, as compared to control untreated cells (Ctrl).
Analysis of REAC-treated ADSC morphology. Images were acquired under an inverted optical microscope. Scale bar 100 µm.
Discussion
This study demonstrated that REAC TO-RGN-AR treatment modulates ADSC differentiation, preserves stemness, and promotes phenotypic shifts toward metabolically favorable beige adipocytes. By leveraging asymmetrically conveyed radio electric fields, this approach offers a novel strategy for influencing endogenous bioelectrical activity and holds promise for both foundational and translational applications in regenerative medicine.
The upregulation of stemness-related genes such as Oct-4, Sox2, and Nanog29 in REAC-treated ADSCs indicates a preserved cellular plasticity, even in the presence of differentiation-inducing stimuli. This property is fundamental to the regenerative potential of ADSCs and is consistent with previous studies demonstrating the influence of bioelectrical modulation on transcriptional networks involved in self-renewal and lineage specification17,18,19.
The concurrent downregulation of key adipogenic markers including PPAR-γ11, LPL31, and ACOT230, alongside an increase in UCP1 expression, suggests a significant shift away from white adipocyte commitment. This is further corroborated by the immunofluorescence analysis, which revealed a decrease in ASC-133 and an increase in TMEM2634 and PAT2 expression, markers indicative of beige/brown adipocyte phenotypes. Beige adipocytes are known for their thermogenic and anti-inflammatory capacities, making them particularly relevant in the context of therapies targeting metabolic dysfunction.
The modulation of adipogenic commitment observed here is particularly relevant in the broader context of metabolic disorders such as obesity, metabolic-associated liver disease (MetALD), and type 2 diabetes. In these conditions, the lipogenic and insulin-resistant environment enhances the adipogenic bias of ADSCs, impairing osteogenic and reparative capacities. Recent findings by Pinto et al. demonstrated that a high lipogenic state negatively affects osteogenic processes and tissue regeneration35, while Sanjabi et al. highlighted that lipid droplet hypertrophy in adipocytes plays a crucial role in metabolic dysregulation and insulin imbalance36. In light of these studies, the REAC TO-RGN-AR–induced suppression of adipogenic differentiation and promotion of beige adipocyte features may contribute to restoring metabolic balance and counteracting endocrine dysfunction.
Although this work was conducted entirely in vitro, the findings are consistent with prior in vivo studies involving REAC technology and provide a strong rationale for advancing to preclinical models. The influence of the native tissue microenvironment on ADSC behavior is well known, and future in vivo investigations will be essential to evaluate long-term outcomes and functional integration in metabolically altered tissues.
Donor metabolic status was not available, and this represents a limitation of the present study.
Protein-level confirmation and lipid staining (e.g., Oil Red O) were not performed and will be included in future analyses to strengthen these observations.
Future directions should also include the evaluation of ADSCs derived from donors with obesity or metabolic syndrome, which more accurately reflect the intended clinical applications. Establishing whether the observed bioelectrical modulation is equally effective in disease-relevant cell populations will be crucial to determining therapeutic applicability.
Furthermore, while the results point toward possible involvement of regulatory pathways such as AMPK and mTOR, both central to cellular energy sensing and mitochondrial dynamics, the present data do not allow for mechanistic confirmation. However, previous REAC studies have suggested that bioelectrical modulation may influence these same pathways, contributing to improved energy homeostasis, oxidative metabolism, and mitochondrial function. Dedicated mechanistic studies will be necessary to elucidate this relationship and confirm whether REAC-induced effects are mediated through AMPK/mTOR axis regulation.
This study also highlights areas for methodological development. For example, expanding the analysis to include dose–response and time-course studies would help define the exposure thresholds and temporal dynamics of response. Incorporating sham control conditions would improve the ability to exclude non-specific effects related to experimental handling. In addition, quantitative assessments of oxidative stress, viability, and potential off-target effects should be included in future investigations to comprehensively address the safety profile of this treatment. Further evaluation including additional beige markers such as CD137 and Ear2 may enhance the phenotypic characterization in future studies.
Overall, the ability of REAC TO-RGN-AR treatment to modulate adipogenic commitment, reduce markers associated with pathological lipid accumulation, and promote phenotypes linked to metabolic resilience reinforces the potential of endogenous bioelectrical modulation as a therapeutic frontier.
By restoring bioelectrical balance, REAC TO-RGN-AR may represent a promising strategy to mitigate the metabolic derangements associated with excessive adipogenesis and to improve the reparative capacity of ADSCs.
Beyond metabolic disorders, this approach may support broader regenerative goals, enhancing the functional integration of ADSCs in various tissue engineering19 contexts.
Conclusions
REAC-TO-RGN-AR treatment is a novel, non-invasive approach to optimize ADSC differentiation. It preserves stemness and promotes beneficial phenotypes with anti-inflammatory and thermogenic properties. Further in vitro and in vivo studies are warranted to explore clinical applications in regenerative medicine and metabolic disorders.
Data availability
All data generated or analyzed during this study are included in this published article. For additional information or to request the dataset, please contact the corresponding author, Dr. Sara Cruciani.
References
Qin, Y. et al. An update on adipose-derived stem cells for regenerative medicine: Where challenge meets opportunity. Adv. Sci. (Weinh) https://doi.org/10.1002/advs.202207334 (2023).
Al-Ghadban, S., Artiles, M. & Bunnell, B. A. Adipose stem cells in regenerative medicine: Looking forward. Front. Bioeng. Biotechnol. 9, 837464. https://doi.org/10.3389/fbioe.2021.837464 (2021).
Naderi, N. et al. The regenerative role of adipose-derived stem cells (ADSC) in plastic and reconstructive surgery. Int. Wound J. 14, 112–124. https://doi.org/10.1111/iwj.12569 (2017).
Cruciani, S. et al. Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs. Int J Mol Sci 21, https://doi.org/10.3390/ijms21176181 (2020).
Gorkun, A. A. et al. The duo of osteogenic and angiogenic differentiation in ADSC-derived spheroids. Front. Cell Dev. Biol. 9, 572727. https://doi.org/10.3389/fcell.2021.572727 (2021).
Dong, L. et al. Adipose stem cells in tissue regeneration and repair: From bench to bedside. Regenerative Therapy 24, 547–560. https://doi.org/10.1016/j.reth.2023.09.014 (2023).
Cumpata, A. J. et al. Towards Regenerative Audiology: Immune Modulation of Adipose-Derived Mesenchymal Cells Preconditioned with Citric Acid-Coated Antioxidant-Functionalized Magnetic Nanoparticles. Medicina (Kaunas) 59, https://doi.org/10.3390/medicina59030587 (2023).
Wang, J. F. et al. Immunomodulation of adipose-derived mesenchymal stem cells on peripheral blood mononuclear cells in colorectal cancer patients with COVID-19. World J. Gastrointestinal oncol. 16, 2113–2122. https://doi.org/10.4251/wjgo.v16.i5.2113 (2024).
Skrypnyk, M. Current progress and limitations of research regarding the therapeutic use of adipose-derived stem cells: literature review. J. Umm Al-Qura Univ. Appl. Sci. https://doi.org/10.1007/s43994-024-00147-9 (2024).
Michurina, S. et al. Decreased UCP-1 expression in beige adipocytes from adipose-derived stem cells of type 2 diabetes patients associates with mitochondrial ROS accumulation during obesity. Diabetes Res. Clin. Pract. 169, 108410. https://doi.org/10.1016/j.diabres.2020.108410 (2020).
Ma, X., Wang, D., Zhao, W. & Xu, L. Deciphering the roles of PPARgamma in adipocytes via dynamic change of transcription complex. Front. Endocrinol (Lausanne) https://doi.org/10.3389/fendo.2018.00473 (2018).
Linhart, H. G. et al. C/EBPalpha is required for differentiation of white, but not brown, adipose tissue. Proc. Natl. Acad. Sci. U S A 98, 12532–12537. https://doi.org/10.1073/pnas.211416898 (2001).
Ertunc, M. E. & Hotamisligil, G. S. Lipid signaling and lipotoxicity in metaflammation: indications for metabolic disease pathogenesis and treatment. J. Lipid. Res. 57, 2099–2114. https://doi.org/10.1194/jlr.R066514 (2016).
Maioli, M. et al. REAC technology and hyaluron synthase 2, an interesting network to slow down stem cell senescence. Sci. Rep. 6, 28682. https://doi.org/10.1038/srep28682 (2016).
Basoli, V. et al. Physical stimulation by REAC and BMP4/WNT-1 inhibitor synergistically enhance cardiogenic commitment in iPSCs. PLoS ONE 14, e0211188. https://doi.org/10.1371/journal.pone.0211188 (2019).
Fontani, V., Cruciani, S., Santaniello, S., Rinaldi, S. & Maioli, M. Impact of REAC regenerative endogenous bioelectrical cell reprogramming on MCF7 breast cancer cells. J. Pers. Med. 13, 1019. https://doi.org/10.3390/jpm13061019 (2023).
Maioli, M. et al. Radiofrequency energy loop primes cardiac, neuronal, and skeletal muscle differentiation in mouse embryonic stem cells: a new tool for improving tissue regeneration. Cell Transplant 21, 1225–1233. https://doi.org/10.3727/096368911X600966 (2012).
Maioli, M. et al. Radio electric conveyed fields directly reprogram human dermal skin fibroblasts toward cardiac, neuronal, and skeletal muscle-like lineages. Cell Transplant 22, 1227–1235. https://doi.org/10.3727/096368912X657297 (2013).
Maioli, M. et al. Radioelectric asymmetric conveyed fields and human adipose-derived stem cells obtained with a nonenzymatic method and device: a novel approach to multipotency. Cell Transplant 23, 1489–1500. https://doi.org/10.3727/096368913X672037 (2014).
Fontani, V., Rinaldi, A. & Rinaldi, S. Restoring function in pediatric neurodegeneration: The impact of radio electric asymmetric conveyor neuroregenerative treatment in a child with canavan syndrome. Cureus 16, e73324. https://doi.org/10.7759/cureus.73324 (2024).
Fontani, V., Rinaldi, A. & Rinaldi, S. Neuroregenerative effects of radio electric asymmetric conveyer (REAC) neuroregenerative (RGN-N) therapy in pediatric adrenoleukodystrophy: A case report. Cureus 16, e74197. https://doi.org/10.7759/cureus.74197 (2024).
Rinaldi, S., Rinaldi, A. & Fontani, V. Functional recovery in a child with adrenoleukodystrophy following neuroregenerative effects of radio electric asymmetric conveyer (REAC) neuro-regenerative (RGN-N) treatment: A detailed case report. Cureus 16, e74283. https://doi.org/10.7759/cureus.74283 (2024).
Lorenzini, L. et al. REAC technology modifies pathological neuroinflammation and motor behaviour in an Alzheimer’s disease mouse model. Sci. Rep. 6, 35719. https://doi.org/10.1038/srep35719 (2016).
Panaro, M. A. et al. Radio electric asymmetric conveyer technology modulates neuroinflammation in a mouse model of neurodegeneration. Neurosci. Bull. 34, 270–282. https://doi.org/10.1007/s12264-017-0188-0 (2018).
Higino Rocha, L., Covallero Renck, A., Fontani, V. & Rinaldi, S. Efficacy of radio electric asymmetric conveyor (REAC) anti-inflammatory, circulatory, and metabolic optimization treatments for managing chronic pain, edema, and lipodystrophy in post-menopausal women: A case series. Cureus 16, e72653. https://doi.org/10.7759/cureus.72653 (2024).
Higino Rocha, L., Fontani, V. & Rinaldi, S. Efficacy of radio electric asymmetric conveyer (REAC) biomodulation treatments in managing chronic pain, edema, and metabolic syndrome: A case report. Cureus 16, e72031. https://doi.org/10.7759/cureus.72031 (2024).
Renck, A. C., Fontani, V. & Rinaldi, S. Radio electric asymmetric conveyer (REAC) biomodulation for chronic pain and functional improvement in rheumatoid arthritis, lipedema, and fibromyalgia: A case study on anti-inflammatory, circulatory, and metabolic optimization. Cureus 16, e74311. https://doi.org/10.7759/cureus.74311 (2024).
Basoli, V. et al. Melatonin and Vitamin D Interfere with the adipogenic fate of adipose-derived stem cells. Int. J. Mol. Sci. https://doi.org/10.3390/ijms18050981 (2017).
Qi, H. & Pei, D. The magic of four: induction of pluripotent stem cells from somatic cells by Oct4, Sox2, Myc and Klf4. Cell Res. 17, 578–580. https://doi.org/10.1038/cr.2007.59 (2007).
Liu, L. et al. The effect of Acot2 overexpression or downregulation on the preadipocyte differentiation in Chinese Red Steppe cattle. Adipocyte 9, 279–289. https://doi.org/10.1080/21623945.2020.1776553 (2020).
Huang, Y. et al. Downregulation of adipose LPL by PAR2 contributes to the development of hypertriglyceridemia. JCI Insight https://doi.org/10.1172/jci.insight.173240 (2024).
Nishimura, T., Katsumura, T., Motoi, M., Oota, H. & Watanuki, S. Experimental evidence reveals the UCP1 genotype changes the oxygen consumption attributed to non-shivering thermogenesis in humans. Sci. Rep. 7, 5570. https://doi.org/10.1038/s41598-017-05766-3 (2017).
Suwandhi, L. et al. Asc-1 regulates white versus beige adipocyte fate in a subcutaneous stromal cell population. Nat. Commun. 12, 1588. https://doi.org/10.1038/s41467-021-21826-9 (2021).
Pilkington, A. C., Paz, H. A. & Wankhade, U. D. Beige Adipose Tissue Identification and Marker Specificity-Overview. Front Endocrinol (Lausanne) 12, 599134, https://doi.org/10.3389/fendo.2021.599134 (2021).
Pinto, T. S. et al. Interaction of high lipogenic states with titanium on osteogenesis. Bone 188, 117242. https://doi.org/10.1016/j.bone.2024.117242 (2024).
Sanjabi, B. et al. Lipid droplets hypertrophy: a crucial determining factor in insulin regulation by adipocytes. Sci. Rep. 5, 8816. https://doi.org/10.1038/srep08816 (2015).
Funding
This work was supported by Fondazione di Sardegna.
Author information
Authors and Affiliations
Contributions
Conceptualization, SR, SC, VF. and MM.; methodology, SR, SC, VF. and MM; validation, SR, SC, VF and MM; formal analysis, SC, MM, SR, and VF; investigation, SR, SC, VF and MM; data curation, SR, SC, VF and MM; writing—original draft preparation, SR, SC, VF. and MM; writing—review and editing, SR, SC, VF. and MM; visualization, SC, MM.; supervision, SR, and MM; project administration, SR, VF and MM.; funding acquisition, SR, VF and MM. All authors have read and agreed to the published version of the manuscript.
Ethics declarations
Competing interests
SR and VF are the authors of the REAC technology patent. The other authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cruciani, S., Rinaldi, S., Fontani, V. et al. REAC RGN-AR treatment modulates adipogenic differentiation in adipose tissue-derived stem cells. Sci Rep 16, 4860 (2026). https://doi.org/10.1038/s41598-026-35204-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-35204-2