Abstract
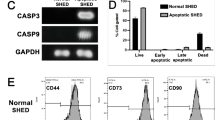
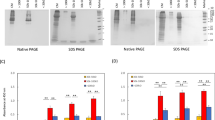
The developmental dysfunction of cartilage progenitor cells (CPCs) causes dwarfism. Radical therapies for dwarfism remain underdeveloped. Recently, the therapeutic benefits of extracellular vesicles (EVs) released from human exfoliated deciduous tooth-derived stem cells (SHED) have been investigated for their potential to restore disease-target cells. However, the effects of EVs on human CPCs for the treatment of dwarfism remain unclear. We investigated the impact of EVs on cell proliferation, telomerase activity, telomerase reverse transcriptase (TERT) expression, cell cycle, and extracellular signal-regulated protein kinase 1/2 (ERK1/2) levels in human CPCs and in our established osteogenesis imperfecta (OI)-specific SHED (OI-SHED). EVs enhanced the proliferation and G1/S phase transition of CPCs, which was associated with increased TERT expression and telomerase activity. However, RNase-preconditioned EVs did not attenuate the efficacy of EVs in CPCs. ERK1/2 inhibitor tests demonstrated that CPCs exhibited suppressed proliferation, telomerase activity, and G1/S phase progression. EV-transferring CD29 induced ERK1/2 phosphorylation in CPCs, subsequently activating telomerase activity to induce proliferation. Interestingly, EV stimulation restored the reduction in cell proliferation, cell cycle progression, phosphorylated ERK1/2 levels, and TERT expression in OI-SHED. In conclusion, the present findings provide new insights into ERK1/2-mediated proliferation of human CPCs using EVs.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Information files.
References
Kronenberg, H. M. Developmental regulation of the growth plate. Nature 423, 332–336 (2003).
Mizuhashi, K. et al. Resting zone of the growth plate houses a unique class of skeletal stem cells. Nature 563, 254–258 (2018).
Jiang, Y. et al. Human cartilage-derived progenitor cells from committed chondrocytes for efficient cartilage repair and regeneration. Stem Cells Transl Med. 5, 733–744 (2016).
Winograd-Katz, S. E., Fässler, R., Geiger, B. & Legate, K. R. The integrin adhesome: From genes and proteins to human disease. Nat. Rev. Mol. Cell Biol. 15, 273–288 (2014).
Ren, K. et al. Periodic mechanical stress activates integrinβ1-dependent src-dependent PLCγ1-independent Rac1 mitogenic signal in rat chondrocytes through ERK1/2. Cell. Physiol. Biochem. 30, 827–842 (2012).
Geister, K. A. & Camper, S. A. Advances in skeletal dysplasia genetics. Annu. Rev. Genomics Hum. Genet. 16, 199–227 (2015).
Götherström, C. et al. Pre- and postnatal transplantation of fetal mesenchymal stem cells in osteogenesis imperfecta: A two-center experience. Stem Cells Transl. Med. 3, 255–264 (2014).
Memeo, A. et al. Mesenchymal stem cells as adjuvant therapy for limb lengthening in achondroplasia. J. Pediatr. Orthop. B 28, 221–227 (2019).
Sonoda, S. et al. Extracellular vesicles from deciduous pulp stem cells recover bone loss by regulating telomerase activity in an osteoporosis mouse model. Stem Cell Res Ther. 11, 296. https://doi.org/10.1186/s13287-020-01818-0 (2020).
Sonoda, S. et al. Targeting of deciduous tooth pulp stem cell-derived extracellular vesicles on telomerase-mediated stem cell-niche and immune regulation in systemic lupus erythematosus. J. Immunol. 206, 3053–3063 (2021).
Jiang, Y. & Tuan, R. S. Origin and function of cartilage stem/progenitor cells in osteoarthritis. Nat. Rev. Rheumatol. 11, 206–212 (2015).
Bruno, S., Kholia, S., Deregibus, M. C. & Camussi, G. The role of extracellular vesicles as paracrine effectors in stem cell-based therapies. Adv. Exp. Med. Biol. 1201, 175–193 (2019).
Gao, S. et al. MicroRNA-197 regulates chondrocyte proliferation, migration, and inflammation in pathogenesis of osteoarthritis by targeting EIF4G2. Biosci. Rep. 40, BSR20192095. https://doi.org/10.1042/BSR20192095 (2020).
Wan, S. et al. Extracellular vesicles from hypoxic pretreated urine-derived stem cells enhance the proliferation and migration of chondrocytes by delivering miR-26a-5p. Cartilage. 13, 19476035221077400. https://doi.org/10.1177/19476035221077401 (2022).
Sun, C., Teng, F. & Xia, Y. Extracellular vesicles in osteoarthritis: Mechanisms, therapeutic potential, and diagnostic applications. Front. Immunol. 16, 1595095. https://doi.org/10.3389/fimmu.2025.1595095 (2025).
Ghorbaninejad, M., Kamrani, S., Ghorbaninejad, Z. & Hosseini, S. Epigenetic crosstalk in bone and cartilage disorders: Emerging role of extracellular vesicles. Biomed. Pharmacother. 193, 118706. https://doi.org/10.1016/j.biopha.2025.118706 (2025).
Ayariga, J. A., Huang, H. & Dean, D. Decellularized avian cartilage, a promising alternative for human cartilage tissue regeneration. Materials 15, 1974. https://doi.org/10.3390/ma15051974 (2022).
Zhang, R. et al. Mxra8 is a receptor for multiple arthritogenic alphaviruses. Nature 557, 570–574 (2018).
Su, T., Zhu, Y., Wang, X., Zhu, Q. & Duan, X. Hereditary dentin defects with systemic diseases. Oral Dis. 29, 2376–2393 (2023).
Chen, C. et al. Telomerase governs immunomodulatory properties of mesenchymal stem cells by regulating FAS ligand expression. EMBO Mol. Med. 6, 322–334 (2014).
Izquierdo, E. et al. Extracellular vesicles and PD-L1 suppress macrophages, inducing therapy resistance in TP53-deficient B-cell malignancies. Blood 139, 3617–3629 (2022).
Zhang, S. et al. MSC exosomes mediate cartilage repair by enhancing proliferation, attenuating apoptosis and modulating immune reactivity. Biomaterials 156, 16–27 (2018).
Yonekura, A. et al. Transforming growth factor-β stimulates articular chondrocyte cell growth through p44/42 MAP kinase (ERK) activation. Endocr J. 46, 545–553 (1999).
Lai, L. P., Dasilva, K. A. & Mitchell, J. Regulation of indian hedgehog mRNA levels in chondrocytic cells by ERK1/2 and p38 mitogen-activated protein kinases. J. Cell. Physiol. 203, 177–185 (2005).
Beier, F. et al. TGFβ and PTHrP control chondrocyte proliferation by activating cyclin D1 expression. Mol. Biol. Cell. 12, 3852–3863 (2001).
Jirmanova, L., Afanassieff, M., Gobert-Gosse, S., Markossian, S. & Savatier, P. Differential contributions of ERK and PI3-kinase to the regulation of cyclin D1 expression and to the control of the G1/S transition in mouse embryonic stem cells. Oncogene 21, 5515–5528 (2002).
Long, F., Zhang, X. M., Karp, S., Yang, Y. & McMahon, A. P. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development 128, 5099–5108 (2001).
Aszodi, A., Hunziker, E. B., Brakebusch, C. & Fässler, R. β1 integrins regulate chondrocyte rotation, G1 progression, and cytokinesis. Genes Dev. 17, 2465–2479 (2003).
Shi, S. et al. Bone formation by human postnatal bone marrow stromal stem cells is enhanced by telomerase expression. Nat. Biotechnol. 20, 587–591 (2002).
Chueaphromsri, P., Kunhorm, P., Phonchai, R., Chaicharoenaudomrung, N. & Noisa, P. Cordycepin enhances SIRT1 expression and maintains stemness of human mesenchymal stem cells. In Vivo 37, 596–610 (2023).
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009).
Li, Y., Cheng, H. S., Chng, W. J., Tergaonkar, V. & Cleaver, J. E. Activation of mutant TERT promoter by RAS-ERK signaling is a key step in malignant progression of BRAF-mutant human melanomas. Proc. Natl. Acad. Sci. U S A. 113, 14402–14407 (2016).
Nicholson, I. P., Gault, E. A., Foote, C. G., Nasir, L. & Bennett, D. Human telomerase reverse transcriptase (hTERT) extends the lifespan of canine chondrocytes in vitro without inducing neoplastic transformation. Vet. J. 174, 570–576 (2007).
Yang, J. et al. Establishment and characterization of an immortalized human chondrocyte cell line. Biotechnol. Lett. 42, 707–716 (2020).
Morita, M., Nakanishi, K., Kawai, T. & Fujikawa, K. Telomere length, telomerase activity, and expression of human telomerase reverse transcriptase mRNA in growth plate of epiphyseal articular cartilage in femoral head during normal human development and in thanatophoric dysplasia. Hum. Pathol. 35, 403–411 (2004).
Zhou, B., Chen, D., Xu, H. & Zhang, X. Proliferation of rabbit chondrocyte and inhibition of IL-1β-induced apoptosis through MEK/ERK signaling by statins. In Vitro Cell Dev. Biol. Anim. 53, 124–131 (2017).
Becker, K. A. et al. Self-renewal of human embryonic stem cells is supported by a shortened G1 cell cycle phase. J. Cell. Physiol. 209, 883–893 (2006).
Lanske, B. et al. PTH/PTHrP receptor in early development and Indian hedgehog-regulated bone growth. Science 273, 663–666 (1996).
Vortkamp, A. et al. Regulation of rate of cartilage differentiation by Indian Hedgehog and PTH-related protein. Science 273, 613–622 (1996).
Tsukasaki, M. et al. Periosteal stem cells control growth plate stem cells during postnatal skeletal growth. Nat. Commun. 13, 4166. https://doi.org/10.1038/s41467-022-31592-x (2022).
Lv, Z. et al. Impaired proliferation of growth plate chondrocytes in a model of osteogenesis imperfecta. Biochem. Biophys. Res. Commun. 613, 146–152 (2022).
Otsuru, S. et al. Extracellular vesicles released from mesenchymal stromal cells stimulate bone growth in osteogenesis imperfecta. Cytotherapy 20, 62–73 (2018).
Lee, A. E. et al. DPSC-derived extracellular vesicles promote rat jawbone regeneration. J. Dent. Res. 102, 313–321 (2023).
Lu, Y. et al. Rab27a-mediated extracellular vesicle secretion contributes to osteogenesis in periodontal ligament-bone niche communication. Sci. Rep. 13, 8479. https://doi.org/10.1038/s41598-023-35172-x (2023).
Wang, J. et al. Exosomes derived from human gingival mesenchymal stem cells induce metabolic reprogramming of inflammatory macrophages. J. Clin. Periodontol. 52, 1196–1210 (2025).
Iwanaka, T. et al. A model study for the manufacture and validation of clinical-grade deciduous dental pulp stem cells for chronic liver fibrosis treatment. Stem Cell Res Ther. 11, 134. https://doi.org/10.1186/s13287-020-01630-w (2020).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Acknowledgements
We thank the Research Support Center, Research Center for Human Disease Modeling, Kyushu University Graduate School of Medical Sciences, for the technical assistance with cell sorting using FACSAria Fusion cell sorter and FACDiva software (BD Biosciences). This was partially supported by the Mitsuaki Shiraishi Fund for Basic Medical Research. Finally, we thank Editage (https://www.editage.jp) for the English editing of our manuscript.
Funding
This work was supported by JSPS KAKENHI grants JP22K19565 (T.Y.), JP23H03071 (T.Y.), JP23K27762 (T.Y.), JP21K16932 (S.S.), JP24K12873 (S.S.), and JP21J10881 (S.M.).
Author information
Authors and Affiliations
Contributions
SM and SS: study design, collection, analysis and interpretation of data, and statistical analysis. Y.K-N., M.M.S., L.Y., R.A., E.Y–T., M.F.Z., H.K., N.U., and H.Y.: assembly of data and statistical analysis. T.Y.: study conception and design, manuscript drafting, and critical revision of the manuscript for important intellectual content. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Murata, S., Sonoda, S., Kyumoto-Nakamura, Y. et al. Deciduous pulp stem cell-derived extracellular vesicles stimulate the proliferation of cartilage progenitor cells via extracellular signal-regulated protein kinase 1/2 activation. Sci Rep (2026). https://doi.org/10.1038/s41598-026-37380-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-37380-7