Abstract
The purpose of this study was to evaluate the predictive value of monocyte-to-lymphocyte ratio (MLR) on the short-term (28 days) and long-term (365 days) mortality risk in patients with acute pancreatitis (AP) using multiple statistical and machine learning (ML) models. Studies selected 1,044 eligible AP patients from the MIMIC-IV database and divided them into four groups based on their MLR values (MLR<0.32; 0.32 ≤ MLR<0.57; 0.57 ≤ MLR<1; MLR ≥ 1). Findings revealed that MLR demonstrated a U-shaped relationship with patient mortality risk, with the minimal mortality risk occurring at an MLR of approximately 0.57. Cox regression model analysis showed that after adjusting for multiple parameters, MLR was still significantly associated with the risk of death. Moreover, ML model analysis identified that MLR has potential value in predicting AP patient outcomes. This study suggests that MLR can be used as a potential indicator to assess prognostic risk in critically ill patients with AP to support clinical decision-making.
Similar content being viewed by others
Introduction
Acute pancreatitis (AP), which is a common inflammatory disease of the exocrine pancreas, causes severe abdominal pain and multiple organ dysfunction, which can result in pancreatic necrosis, chronic organ failure, and mortality1. Although the mortality rate for AP is only 1–5%, the mortality rate for severe acute pancreatitis (SAP) is close to 20% -30%2. The global incidence of AP is increasing; specifically, it has a global incidence of 30–40 cases per 100,000 people per year and has been demonstrated to be more than twice this rate in some regions, thus contributing to high costs for health care systems3. Currently, there is no specific causal treatment for AP4, although some underlying causes such as hypertriglyceridemia and gallstones can be managed.
Although the pathophysiological mechanisms of AP have become better understood in recent years, its prognostic assessment remains challenging5. The inflammatory response and immune dysregulation are important causes of the onset and progression of AP6. Consequently, the search for biomarkers that reflect the inflammatory status and immune function of patients is important for the early identification of high-risk patients and improvements in patient prognosis.
ML techniques are becoming increasingly important in health care and have demonstrated considerable potential for accurately predicting survival rates and disease progression in cancer patients7. To clarify, while ML techniques are increasingly utilized to predict patient outcomes in AP, there are significant deficiencies in the methodology of recently published ML based prognostic models in AP patients8, and few systems are specifically designed for routine screening of AP-related risks in the ICU. Thus, this study, based on ML algorithms, aimed to construct multiple prediction models and feature analysis methods for data analysis, identify the primary clinical markers influencing AP patients’ prognosis, and provide stronger support for clinical decision-making.
The monocyte-to-lymphocyte ratio (MLR) integrates two key immune pathways: monocytes represent systemic inflammation, while lymphocytes reflect immune competence. MLR has rarely been researched in AP. Given the central role of inflammation and immune dysregulation in AP pathogenesis, we hypothesized that MLR could serve as a novel prognostic biomarker in critically ill AP patients.
We focused specifically on AP patients requiring ICU admission because this subgroup experiences the highest morbidity and mortality, representing a critical population where early and accurate prognostic assessment is most urgently needed to guide intensive care resource allocation and therapeutic strategies.
Methods
Data source
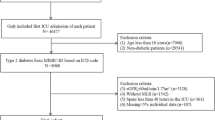
This study utilized data from MIMIC-IV v3.0 (Medical Information Mart for Intensive Care), which is a publicly available critical care database containing deidentified records of 94,458 intensive care unit admissions between 2008 and 2022 (Fig. 1). MIMIC-IV is a publicly available critical care database from the United States, containing data from Beth Israel Deaconess Medical Center in Boston. The Institutional Review Board of the BIDMC approved a waiver of the author’s informed consent. The author (WW) was permitted to access the database (certificate number 56452808).
Inclusion and exclusion criteria
Inclusion criteria: (1) patients aged over 18 years; and (2) patients diagnosed with AP based on the International Classification of Diseases9. Exclusion criteria: (1) patients who had an ICU stay of fewer than twenty-four hours; (2) absent serum monocytes and lymphocytes in the initial laboratory test; and (3) data only being included from a patient’s initial hospitalization in cases where the patient experienced multiple ICU stays. Finally, 1044 individuals who met the criteria were selected for inclusion (Fig. 1).
Outcome
This study primarily focused on 28-day all-cause mortality as the main outcome, and 365-day all-cause mortality was regarded as the secondary outcome.
Data extraction
We retrieved data from the MIMIC-IV, including demographic information, vital signs, comorbidities, therapies, laboratory data, scoring systems, and prognostic data during follow-up. All of the hematological parameters were assessed for the first time following patient admission to the ICU. The following formula was used for determining the MLR: serum monocytes (K/µL)/serum lymphocytes (K/µL).
Statistical analysis
Patients were stratified into four groups based on quartiles of MLR values to explore potential nonlinear associations and ensure balanced group sizes for comparative analysis. The normality of distribution was assessed via Shapiro-Wilk tests supplemented by visual examination of the Q-Q plots. Normally distributed continuous variables are presented as the means ± standard deviations, whereas nonnormally distributed variables are reported as medians (interquartile ranges). Categorical variables are expressed as counts (percentages). Intergroup comparisons were performed via Mann-Whitney U tests for nonparametric continuous data, chi-square tests for categorical variables, or Fisher’s exact tests (when appropriate). A two-tailed p value less than 0.05 was considered to be statistically significant. R software was used for all of the statistical analyses (version 4.4.1).
Kaplan-Meier survival analysis with log-rank tests was used to compare 28-day and 365-day survival rates among the four groups.
To control for potential confounding factors in our survival analysis, we constructed multivariate Cox proportional hazards regression models with progressive covariate adjustments. Model I included only the MLR. Model II was adjusted for age, sex, language, marital status, and race. Subsequently, Model III was adjusted for age, hypertension, myocardial infarction, congestive heart failure, cerebrovascular disease, chronic pulmonary disease, diabetes, renal disease, malignant cancer, liver disease, sepsis, sex, language, marital status, and race.
To investigate potential variations in the associations between the MLR and outcomes across different patient subgroups, as well as to examine possible interaction effects, we conducted stratified analyses by using the following variables: (1) demographic characteristics including sex and age (< 60 vs. ≥60 years) and (2) comorbidities including hypertension and diabetes mellitus (DM). The findings were graphically presented using forest plots. In addition, restricted cubic spline (RCS) plots with four knots were used to identify potential inflection points to assess the linear or nonlinear relationships between the clinical outcomes and the levels of the MLR as continuous variables. The analysis was also adjusted for various confounding factors, including age, hypertension, myocardial infarction, congestive heart failure, cerebrovascular disease, chronic pulmonary disease, diabetes, renal disease, malignant cancer, liver disease, sepsis, sex, language, marital status, and race. For all of the models, the median values of the MLR were set as a reference.
Model development
We conducted a LASSO regression cross-validation analysis to select variables. In the development of the machine learning (ML) algorithm, acceptable variables were carefully selected and integrated into the algorithm. The dataset was subsequently partitioned into training and validation subsets at a 7:3 ratio. All ML analyses were conducted using R software (version 4.4.1) within the mlr3 ecosystem (version 0.20.2). The modeling workflow was constructed using the mlr3pipelines package. The Cox proportional hazards survival learner (Coxph), conditional inference tree (ctree), gradient boosting machine (GBM), neural network (NN), survival random forest SRC learner (rsf), and extreme gradient boosting survival learner (xgboost) algorithms. For algorithms requiring hyperparameter tuning (ctree, rsf, xgboost, GBM, NN), we performed a 5-fold cross-validated grid search using the mlr3 tuning package. A predefined parameter space was established for each learner, and the optimal hyperparameter set was selected based on maximizing Harrell’s C-index. The final model for each algorithm was then refitted on the entire training set using its respective optimal configuration. The key hyperparameters for the final models were as follows: Ctree: alpha = 0.1, minbucket = 25, Rsf: ntree = 200, mtry = 3, nodesize = 21, Xgboost: eta = 0.08917, max_depth = 1, nrounds = 269, GBM: n.trees = 100, interaction.depth = 1, n.minobsinnode = 5, shrinkage = 0.1, NN: num_nodes = c (5, 5, 5), dropout = 0.2631, weight_decay = 0.1331, learning_rate = 0.07773, batch_size = 32. We optimized hyperparameters using grid search and assessed model robustness via five-fold cross-validation. The ROC curve and its corresponding area under the curve (AUC) were used to determine model performance. Clinical effectiveness was evaluated using decision curve analysis (DCA), and the model’s accuracy in forecasting absolute risk was assessed using calibration curves. The Shapley additive explanations (SHAP) method was used to explain the optimal model to understand the impact of individual features on the model predictions (Fig. 1).
Results
Baseline characteristics
Data were available from 1044 patients. Table 1 displays the baseline characteristics of the study participants. There were 433 (41.48%) females and 611 (58.52%) males, as well as 665 (63.70%) patients with hypertension, 118 (11.30%) patients with myocardial infarctions, 229 (21.93%) patients with congestive heart failure, 71 (6.8%) patients with peripheral vascular disease, 69 (6.61%) patients with cerebrovascular disease, 227 (21.74%) patients with chronic pulmonary disease, 395 (37.84%) patients with diabetes, 243 (23.28%) patients with renal disease, 79 (7.57%) patients with malignant cancer, 368 (35.25%) patients with liver disease, 12 (1.15%) patients with AIDS, and 606 (58.05%) patients with sepsis.
Patients were divided into the following four groups: Group 1 (MLR < 0.32), which consisted of 255 individuals; Group 2 (0.32 ≤ MLR < 0.57), which consisted of 261 individuals; Group 3 (0.57 ≤ MLR < 1), which consisted of 243 individuals; and Group 4 (MLR ≥ 1), which consisted of 285 individuals.
Significant variations were observed across MLR groups in multiple clinical parameters (Table 1). The overall patterns suggested that Group 1 tended to have higher values for mean arterial pressure, oxygen saturation, hematocrit, hemoglobin, albumin, calcium, sodium and initial absolute lymphocyte count, while generally showing lower values for prothrombin time, partial thromboplastin time, international normalized ratio, blood cell count, and initial absolute monocyte count. Additionally, the patterns indicated that Group 2 tended to have a greater bicarbonate level and a lower creatinine level. Group 3 was generally associated with a greater platelet count (max) and potassium level (max), alongside a lower anion gap (max), alkaline phosphatase level, and aspartate aminotransferase level (max). Moreover, Group 4 presented the most pronounced pattern, tending to have a greater respiratory rate; greater white blood cell count, anion gap, BUN, creatinine, glucose, prothrombin time, partial thromboplastin time, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, total bilirubin, initial absolute monocyte count, Oxford acute severity of illness score, simplified acute physiology score II and acute physiology score III values; and lower mean arterial pressure, temperature, oxygen saturation, hematocrit, albumin, bicarbonate, calcium, chloride, sodium, and initial absolute lymphocyte count values.
Clinical outcomes
Statistically significant differences in survival distribution among the different MLR groups were observed over time (Table 2). The overall patterns indicated that the highest ICU mortality (10.88%) and hospital mortality (20.70%) were numerically observed in Group 4 (the highest MLR group), while the lowest rates (3.45% and 4.21%, respectively) were noted in Group 2. Regarding length of stay, Group 4 was associated with the longest median ICU stay (2.88 days) and hospital stay (16.04 days), whereas the shortest median durations were observed in Group 1 (1.95 days and 8.19 days, respectively). Mortality rates across various time points (28-day to 365-day) also showed a consistent pattern, with Group 4 demonstrating numerically higher rates and Group 2 showing numerically lower rates compared to other groups.
Kaplan-Meier (K-M) curves demonstrated a significant overall difference in survival probability among the groups at 28 days (log-rank p < 0.001, Fig. 2). Visual inspection of the curves suggested that patients in the fourth quartile had the poorest estimated survival. In contrast, no significant overall difference was observed in the 365-day survival rates across the groups (Supplementary Fig. 1).
In the RCS analysis, both 28-day all-cause mortality (Fig. 3A) and 365-day all-cause mortality (Fig. 3B) exhibited a U-shaped relationship between the MLR and mortality risk. This pattern persisted irrespective of whether the covariates were adjusted (Supplementary Fig. 2).
Cox regression model
The results of the Cox regression analyses for 28-day all-cause mortality are presented in Table 3. Using Q2 as the reference, the mortality risks in Q1 and Q3 were not significantly different across all models (all p > 0.05). In contrast, Q4 was associated with a significantly elevated risk compared to Q2 in the crude model (Model 1, HR: 3.63, 95% CI: 2.01–6.54), and this association remained robust after sequential adjustments for demographic factors (Model 2, HR: 3.03, 95% CI: 1.66–5.52) and comprehensive clinical comorbidities (Model 3, HR: 2.91, 95% CI: 1.59–5.32). Furthermore, a significant positive trend was observed across the quartiles (P for trend < 0.001 in all models), indicating a graded relationship between higher MLR levels and increased 28-day mortality risk.
The associations between MLR quartiles and 365-day all-cause mortality are shown in Table 4. In the fully adjusted model (Model 3), both Q1 (HR: 1.63, 95% CI: 1.04–2.56) and Q3 (HR: 1.63, 95% CI: 1.06–2.52) showed a significantly increased mortality risk compared to Q2. The risk was most pronounced in Q4 (HR: 2.81, 95% CI: 1.89–4.17). This pattern was consistent across all models, with significant positive trends (all p for trend < 0.001), reinforcing a strong, independent association between elevated MLR and long-term mortality. Notably, the emergence of a significant risk in Q1 at 365 days, which was not observed at 28 days, suggests that the predictive power of MLR may extend to long-term outcomes across a broader range of values.
Subgroup analysis
To evaluate the consistency of the association between MLR and mortality across different patient populations, we conducted subgroup analyses for both 28-day (Fig. 4A) and 365-day (Fig. 4B) all-cause mortality. The analyses stratified patients by sex (male/female), age (≥ 60/<60 years), and key comorbidities (hypertension, diabetes, myocardial infarction). The increased mortality risk associated with MLR was consistently observed in all subgroups at both time points (all P < 0.05). Importantly, no significant interaction effects were detected (P for interaction > 0.05 for all variables), demonstrating that the association remained robust regardless of these baseline characteristics.
Feature selection
Feature selection was performed using LASSO (Least Absolute Shrinkage and Selection Operator) regression with 28-day all-cause mortality as the outcome variable. LASSO regression was used to screen the relevant features of the training set, and the characteristics of the variable coefficients are shown in Fig. 5A. The iterative analysis was performed using a ten fold cross-validation method. The 10 variables that were determined to be closely associated with AP included age, temperature_mean, Spo2_min, hemoglobin_max, partial thromboplastin time (PTT_min), bilirubin_total_min, APSIII, heart_failure, cancer, and MLR (Fig. 5B).
Model performance comparisons
A K-M survival curve was constructed after splitting, which compared the survival probabilities of the validation set and the derivation set (Supplementary Fig. 3). The derivation set and the validation set did not significantly differ in terms of survival probability, thus indicating that the split was reasonable.
To determine the risk of AP patients in the ICU, we built six machine learning models. In the derivation group (Fig. 6A), the AUC of the xgboost model was 0.9151, and that of the rsf model was 0.9381, both of which indicated high prediction accuracy. In the validation group (Fig. 6B), the Coxph model demonstrated the highest AUC of 0.8908, whereas the xgboost model had an AUC of 0.8677, and the rsf model had an AUC of 0.8771, both of which were slightly lower than those of the derived populations; however, they still exhibited good predictive performance. Depending on the AUC values in the derivation groups, the Coxph and rsf models may be optimal, especially in applications that require high prediction accuracy.
Model calibration curves were also constructed, which corresponded to the model’s performance in the derivation and validation groups (Fig. 7). In both groups, the calibration curves for most of the models were close to the reference line, thus suggesting that the models performed well in predicting risk and that the predicted values were in good agreement with the actual values. In the derived group (Fig. 7A), the calibration curves for all of the models were relatively close to the reference line, especially in regions with low prediction risk. This result demonstrated that the model was reliable regarding the training data. In the validation group (Fig. 7B), the calibration curve of the model was also close to the reference line, thereby suggesting that the model also demonstrated good generalizability for unseen data. Compared with those of the other models, the calibration curves of the Coxph model in the derived groups and the ctree and Coxph models in the validated groups were closer to the reference line in high-risk regions, thus indicating that the predictions in these regions were more accurate.
The DCA curves of the six ML algorithms on two sets of data (the derivation group and the validation group) are presented in Fig. 8. Every model (with the exception of GBM) demonstrated a strong net benefit in terms of clinical application over a broad range of threshold probabilities; additionally, the models exhibiting the largest net benefits were the rsf and xgboost models (Fig. 8A), as well as the Coxph and xgboost models (Fig. 8B). Therefore, based on the DCA curve, the model with the highest net return within a specific threshold range can be selected.
Figure 9 presents the SHAP summary plots for all models, from which two key observations emerge. First, MLR’s importance is not an artifact of a single model but is a robust finding across multiple, independent algorithms. Furthermore, the high importance assigned to MLR by non-linear models like RSF, XGBoos and GBM aligns perfectly with our previous finding of a significant non-linear relationship via RCS plots. It confirms that these models effectively capture the complex, U-shaped association between MLR and mortality that a linear Cox model may partially mask in its feature importance breakdown. Therefore, the combination of the Cox model’s superior overall AUC and the cross-model validated importance of MLR via SHAP, provides a more comprehensive and compelling argument for MLR’s role as a key prognostic factor. Finally, based on the results, the optimal model was determined to be Coxph. To facilitate clinical application, a nomogram was developed and is presented in supplementary Fig. 4.
Discussion
Our research identified significant associations between the MLR and increased 28-day, 365-day, and in-hospital mortality in SAP patients. These associations remained significant across different age groups, sexes, and subgroups (type 2 diabetes, hypertension, myocardial infarction) after adjusting for covariates. This is the first study to investigate the link between prognosis and the MLR in SAP patients.
Among the clinical outcomes, the K-M survival curve revealed that the MLR ≥ 1 group exhibited the highest mortality. Moreover, the RCS analysis revealed a U-shaped association between the MLR and 28-day mortality in AP patients; additionally, the Cox regression model revealed that a substantially greater risk of death was associated with an MLR ≥ 1. The correlation between the MLR and mortality risk persisted even after controlling for several variables. In addition, the MLR demonstrated a similar trend in the prediction of 365-day mortality, thus suggesting that it has some potential predictive value for long-term prognosis. ML algorithms revealed that the Coxph and rsf models exhibited the highest AUC values in the validation set. Compared with those of the other models, the ctree and Coxph models in the validated groups were closer to the reference line in high-risk regions, thereby indicating that the predictions in these regions were more accurate. Moreover, the DCA curve revealed that the Coxph and xgboost models demonstrated the greatest net benefits in the validation set. The SHAP method was used to visually interpret the Coxph model, and the APS III score and age were determined to be the most important predictive features; additionally, the MLR also had an impact on the model prediction.
Monocytes and lymphocytes are commonly used indicators of disease inflammation and immune status. Monocyte was significantly higher in survivors in sepsis10, And low lymphocyte count serves as an independent marker of progressive inflammation11. However, the prediction performance of monocyte or lymphocyte alone is poor.
Monocyte-lymphocyte ratio (MLR) has shown excellent predictive performance in many diseases. A previous study demonstrated that MLR can provide critical insights into the overall physiological conditions of patients with traumatic brain injury (TBI)12. One study revealed that both the identification of those patients with incident symptomatic TB disease and the prediction of subsequent TB infections may benefit from the utilization of the MLR13. The MLR has also been assessed as a predictor of survival in patients with various malignant diseases14,15. Due to the fact that the MLR has rarely been researched in AP, we investigated the relationship between the MLR and mortality in ICU patients with AP to determine the usefulness of the MLR in predicting ICU AP prognosis.
The MLR is a novel inflammatory biomarker that integrates the effects of both inflammatory markers (monocytes and lymphocytes)16. The MLR has exhibited high predictive and prognostic values in a variety of cancers, including endometrial cancer17, renal cell carcinoma16, and breast cancer18. High expression of MLR was detected in inflammatory diseases. High MLR expression was associated with mortality in acute respiratory distress syndrome19. In addition, the MLR is often used to evaluate the prognosis of pulmonary TB20. The MLR is a convenient and noninvasive inflammatory biomarker12 that provides a more complete representation of the balance between inflammation and immune function. However, few studies have investigated the role of the MLR in predicting the prognosis of AP patients. Therefore, more detailed experiments are needed to investigate the association between the MLR and AP.
We used LASSO regression for feature selection and constructed six models, including Coxph, ctree, GBM, NN, rsf, and xgboost. The SHAP approach was subsequently utilized to evaluate the best model (Coxph) and examine how each feature affected the model predictions. SHAP is a method that is used for interpreting ML model predictions; moreover, it quantifies the contribution of each feature to the model predictions21, and we utilized this method to determine the ten most crucial feature variables pertaining to AP mortality. Recently, research based on ML and SHAP has shown that the six most important characteristic variables are important for the short-term assessment of AP22. By using a greater number of biochemical indicators, our study further investigated the important predictors and prediction models for 28-day and 365-day all-cause mortality in AP patients. The results demonstrated that the 10 characteristic variables (apsIII, age, hemoglobin_max, heart_failure, cancer, temperature_mean, PTT_min, SpO2_min, MLR, and bilirubin_total_min) involved in the model that was proposed in this study are equally important for predicting the poor prognosis of AP patients.
Research has indicated that in AP patients, a lower SpO2 is associated with a greater mortality rate23. Moreover, AP may be the first manifestation of pancreatic cancer, and patients with malignant tumors such as pancreatic cancer are more likely to have a poor prognosis24. Additionally, several studies have also suggested that elderly patients with AP are likely to have atypical clinical presentations and poor prognoses, which are mainly due to existing comorbidities25,26. In rats with AP, hypothermia can decrease pancreatic inflammation and increase survival27. In addition, the death rate of SAP patients has been shown to be closely associated with PTT28. These results indicate that the previously mentioned variables are trustworthy predictors of AP mortality rates.
In our research, the U-shaped association that was observed between the MLR and mortality may be closely related to the body’s inflammatory response and immunomodulatory mechanisms. Monocytes are a vital part of the innate immune system. And lymphocytes possess strong effector mechanisms25. Inflammation is associated with a low lymphocyte count and function26. A higher MLR indicates a high monocyte count, which aggravates local and systemic inflammatory responses, thereby leading to tissue damage24, And the elevated MLR may caused by the decrease in the lymphocyte count, making it difficult to effectively eliminate inflammatory factors, thus leading to worsening of the disease27.
Unlike previous studies, this study not only focused on 28-day mortality but also explored the predictive value of the MLR for long-term prognosis by using 365-day follow-up data, which further expands the potential application of the MLR in the field of AP. In addition, this study used various ML models to analyze the data. These models outperformed traditional statistical methods in accurately identifying key mortality factors and provided robust support for clinical decision-making. Clinically, the MLR (which is a readily accessible biomarker obtained from routine blood tests) demonstrates high utility for early risk stratification, thereby enabling the rapid identification of high-risk AP patients upon admission. Methodologically, the integration of ML algorithms (such as xbgoost, rsf, and NN) can facilitate the development of robust prognostic models and feature analysis frameworks, thus advancing predictive analytics in AP research. These models underscore the role of the MLR in AP.
Despite the use of rigorous methodologies, several limitations existed in this study. First, the data (which were solely derived from the MIMIC-IV database) may have introduced selection bias, thereby limiting generalizability. Second, we were unable to account for certain confounding factors that are potentially result-altering, such as concurrent infections (beyond the diagnosis of sepsis), disease-specific severity scores (e.g., BISAP, Revised Atlanta Criteria), and detailed medication records (e.g., steroids or immunomodulators), which could influence both patient outcomes and MLR values. Future studies should aim to incorporate these critical variables to enhance the robustness and clinical applicability of the prognostic models. Third, when considering model-specific constraints, the Coxph model assumes that the risk ratio is constant over time, which may not be true in some cases, whereas xgboost has high predictive performance but poor model interpretation. Future studies may consider expanding the data sources, optimizing model construction methods, performing in-depth explorations on the interactions between various characteristics, and validating the findings in more health care institutions and patient groups, in order to improve the reliability and usefulness of the results of this study.
Conclusion
This study demonstrated that the likelihoods of short-term and long-term death in AP patients are substantially connected with the MLR, and this association was observed to be U shaped. A comparison of multiple ML models revealed that the Coxph and rsf models performed well in predicting patient prognosis. Via multidimensional data analysis, the MLR was observed to be strongly correlated with AP severity, especially in the prediction of long-term survival. Therefore, the MLR can be used as a potential indicator to assess the prognostic risk of patients with AP in the ICU.
Screening process for AP patients in ICU and the research process.
A 28-day KM survival curve. KM curves showing the survival rates at 28 days for each quartile.MLR: Quartile 1 (<0.32), Quartile 2 (0.32 ≤ MLR<0.57), Quartile 3 (0.57 ≤ MLR<1), and Quartile 4 (MLR ≥ 1).
RCS analysis of 28-day (A) and 365-day (B) all-cause mortality after adjusting the covariates. Adjusted covariates: age, hypertension, myocardial infarct, congestive heart failure, cerebrovascular disease, chronic pulmonary disease, diabetes, renal disease, malignant cancer, liver disease, sepsis, gender, language, marital status, race.
(A) Subgroup forest plot for 28-day all-cause mortality. (B) Subgroup forest plot for 365-day all-cause mortality.
Demographic and clinical feature selection using the least absolute shrinkage and selection operator (LASSO) binary logistic regression model. (A)LASSO coefficient profiles of the 68 texture features; (B) Tuning parameter (λ) selection using LASSO penalized logistic regression with 10-fold cross validation.
ROC curves of the machine learning algorithms. (A) derivation groups; (B)validation groups.
The calibration curve of each model. (A) derivation groups; (B) validation groups.
DCA curves of the machine learning algorithms. (A) derivation groups; (B)validation groups.
The SHAP summary plots for all models. (A)coxph shap (B)gbm shap (C)xgboost shap (D)nn shap (E)rsf shap (F)ctree shap.
Data availability
The datasets are available in the physionet (https://physionet.org/content/mimiciv/0.4/).
References
Horibe, M. et al. Normal saline versus ringer’s solution and critical-illness mortality in acute pancreatitis: a nationwide inpatient database study. J. Intensive Care. 12 (1), 27 (2024).
Iannuzzi, J. P. et al. Global incidence of acute pancreatitis is increasing over time: a systematic review and Meta-Analysis. Gastroenterology 162 (1), 122–134 (2022).
Szatmary, P. et al. Acute pancreatitis: diagnosis and treatment. Drugs 82 (12), 1251–1276 (2022).
Glaubitz, J. et al. Immune response mechanisms in acute and chronic pancreatitis: strategies for therapeutic intervention. Front. Immunol. 14, 1279539 (2023).
Modenbach, J. M. et al. Biochemical analyses of cystatin-C dimers and cathepsin-B reveals a trypsin-driven feedback mechanism in acute pancreatitis. Nat. Commun. 16 (1), 1702 (2025).
Chen, F. et al. Mitochondrial dysfunction in pancreatic acinar cells: mechanisms and therapeutic strategies in acute pancreatitis. Front. Immunol. 15, 1503087 (2024).
Swanson, K., Wu, E., Zhang, A., Alizadeh, A. A. & Zou, J. From patterns to patients: advances in clinical machine learning for cancer diagnosis, prognosis, and treatment. Cell 186 (8), 1772–1791 (2023).
Critelli, B. et al. A systematic review of machine learning-based prognostic models for acute pancreatitis: towards improving methods and reporting quality. PLoS Med. 22 (2), e1004432. https://doi.org/10.1371/journal.pmed.1004432 (2025).
Wang, J. et al. Association between serum creatinine to albumin ratio and short- and long-term all-cause mortality in patients with acute pancreatitis admitted to the intensive care unit: a retrospective analysis based on the MIMIC-IV database. Front. Immunol. 15, 1373371 (2024).
Djordjevic, D. et al. Neutrophil-to-Lymphocyte ratio, Monocyte-to-Lymphocyte ratio, platelet-to-Lymphocyte ratio, and mean platelet Volume-to-Platelet count ratio as biomarkers in critically ill and injured patients: which ratio to choose to predict outcome and nature of bacteremia? Mediators inflamm. 2018, 3758068 (2018).
Chen, X., Lin, Z., Chen, Y. & Lin, C. C-reactive protein/lymphocyte ratio as a prognostic biomarker in acute pancreatitis: a cross-sectional study assessing disease severity. Int. J. Surg. 110 (6), 3223–3229 (2024).
Cheng, Y. W. et al. Predictive value of hematologic indices on weaning from mechanical ventilation and 30-day mortality in patients with traumatic brain injury in an intensive care unit: a retrospective analysis of MIMIC-IV data. Neurotherapeutics 2025, e00559 (2025).
Rees, C. A. et al. The potential of CBC-derived ratios (monocyte-to-lymphocyte, neutrophil-to-lymphocyte, and platelet-to-lymphocyte) to predict or diagnose incident TB infection in Tanzanian adolescents. BMC Infect. Dis. 20 (1), 609 (2020).
Xiang, J. et al. Preoperative Monocyte-to-Lymphocyte ratio in peripheral blood predicts stages, metastasis, and histological grades in patients with ovarian cancer. Transl Oncol. 10 (1), 33–39 (2017).
Asey, B. et al. Peripheral blood-derived immune cell counts as prognostic indicators and their relationship with DNA methylation subclasses in glioblastoma patients. Brain Pathol. 2025, e13334 (2025).
Yildirim, A. et al. Association of baseline inflammatory biomarkers and clinical outcomes in patients with advanced renal cell carcinoma treated with immune checkpoint inhibitors. Ther. Adv. Med. Oncol. 17, 17588359251316243 (2025).
Song, H. et al. Preoperative neutrophil-to-lymphocyte, platelet-to-lymphocyte and monocyte-to-lymphocyte ratio as a prognostic factor in non-endometrioid endometrial cancer. Int. J. Med. Sci. 18 (16), 3712–3717 (2021).
Chen, X. et al. Association of inflammatory blood markers and pathological complete response in HER2-positive breast cancer: a retrospective single-center cohort study. Front. Immunol. 15, 1465862 (2024).
Yang, L. et al. Monocyte-to-lymphocyte ratio is associated with 28-day mortality in patients with acute respiratory distress syndrome: a retrospective study. J. Intensive Care. 9 (1), 49 (2021).
Naranbhai, V. et al. The association between the ratio of monocytes:lymphocytes at age 3 months and risk of tuberculosis (TB) in the first two years of life. BMC Med. 12, 120 (2014).
Lundberg, S. M. et al. From local explanations to global Understanding with explainable AI for trees. Nat. Mach. Intell. 2 (1), 56–67 (2020).
Li, X., Tian, Y., Li, S., Wu, H. & Wang, T. Interpretable prediction of 30-day mortality in patients with acute pancreatitis based on machine learning and SHAP. BMC Med. Inf. Decis. Mak. 24 (1), 328 (2024).
Miller, J. et al. Derivation and validation of the ED-SAS score for very early prediction of mortality and morbidity with acute pancreatitis: a retrospective observational study. BMC Emerg. Med. 21 (1), 16 (2021).
Umans, D. S. et al. Pancreatitis and pancreatic cancer: a case of the chicken or the egg. World J. Gastroenterol. 27 (23), 3148–3157 (2021).
Gardner, T. B. et al. The effect of age on hospital outcomes in severe acute pancreatitis. Pancreatology 8 (3), 265–270 (2008).
Szakacs, Z. et al. Aging and comorbidities in acute pancreatitis II.: a Cohort-Analysis of 1203 prospectively collected cases. Front. Physiol. 9, 1776 (2018).
de Oliveira, C. et al. Multimodal transgastric local pancreatic hypothermia reduces severity of acute pancreatitis in rats and increases survival. Gastroenterology 156 (3), 735–747e10 (2019).
Yang, Y., Du, S., Yuan, W., Kou, Y. & Nie, B. Prolonged activated partial thromboplastin time predicts poor short-term prognosis in patients with acute pancreatitis: a retrospective cohort study. Clin. Transl Sci. 15 (10), 2505–2513 (2022).
Acknowledgements
We acknowledged the contributions of the MIMIC-IV 2.2 program registry for creating and updating the MIMIC-IV database. Thanks to AJE for polishing the article.
Funding
Wei Wu grants from National Natural Science Foundation of China (82302418).
JunYuan Yang grants from Natural Science Foundation of Hubei Province (2024AFB530).
Author information
Authors and Affiliations
Contributions
JYY, CTD and MMG contributed equally to this work. WW designed the study, JYY and MMG drafted the manuscript. CTD, SY extracted the data from the MIMIC-IV database. SY, JDC analyzed the data, HDZ, HG, JX and YL guided the literature review. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The MIMIC-IV database was approved by the Massachusetts Institute of Technology (Cambridge, MA) and consent was obtained for the original data collection.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, J., Dong, C., Guo, M. et al. Association between monocyte-to-lymphocyte ratio and mortality in patients with acute pancreatitis requiring intensive care unit admission: a retrospective cohort study and predictive model establishment based on machine learning. Sci Rep 16, 9157 (2026). https://doi.org/10.1038/s41598-026-37791-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37791-6