Abstract
An aminothiazole-based chromogenic chemosensor, (4-methyl-2-(phenylamino)thiazol-5-yl)(phenyl)methanone (MPTP), was synthesized via an iodine-catalyzed one-pot reaction and tailored for the selective and sensitive detection of Fe3+ ions. The sensor exhibited a distinct chromogenic transition from pale yellow to brown upon Fe3+ binding in ethanol. MPTP demonstrated pronounced positive solvatochromism, Job’s plot analysis confirms a 1:1 binding stoichiometry between MPTP and Fe3+, while Benesi-Hildebrand method reveals a high binding constant, indicative of strong complex formation, a low detection limit (LOD = 0.268 µM), and broad pH stability (2–12), with optimal performance in the physiologically relevant range of pH 6–10. The Fe3+-induced response was reversible through EDTA-mediated chelation, enabling facile sensor regeneration. A portable paper-based test strip incorporating MPTP reproduced the solution-phase chromogenic response, allowing rapid and visual detection of Fe3+ without instrumentation. Environmental and pharmaceutical Fe3+ analysis using simulated water and ferric citrate tablets confirmed the sensor’s applicability, achieving a 98% and 102% recovery rate. DFT calculations support the experimental data, attributing the 361 nm band to a π-π* transition with pronounced intramolecular charge transfer, and HOMO-LUMO/MEP analysis highlights nitrogen-rich sites as preferred Fe3+ binding centres, further underscoring MPTP as a robust probe for Fe3+ detection.
Similar content being viewed by others
Introduction
The reliable detection of metal ions, particularly trivalent iron (Fe3+), is of critical importance in environmental surveillance, industrial process control, and biomedical diagnostics1,2,3. Elevated Fe3+ levels are implicated in oxidative stress, neurodegenerative diseases, and metabolic disorders, while their accumulation in natural water systems contributes to contamination, infrastructure degradation, and reduced ecological quality4,5,6. Therefore, sensitive, selective, and practical detection of Fe3+ is essential across environmental and healthcare domains to mitigate both biological and ecological risks.
Conventional analytical techniques such as atomic absorption spectroscopy (AAS), inductively coupled plasma mass spectrometry (ICP-MS), inductively coupled plasma optical emission spectroscopy (ICP-OES), X-ray fluorescence (XRF), and various electrochemical methods offer excellent precision, sensitivity, and quantitative capabilities7,8,9,10. However, their widespread application is often hindered by substantial capital and operational costs, bulky instrumentation, labor-intensive sample preparation protocols, and the requirement for highly skilled personnel. Additionally, their reliance on centralized laboratory settings makes them unsuitable for real-time, point-of-need analysis, particularly in remote or resource-constrained environments.
In contrast, chemosensors, especially colorimetric sensors, have emerged as versatile and efficient alternatives11,12. These sensors operate on a simple principle, converting molecular recognition events into visible color changes, thus allowing for naked-eye detection without the need for sophisticated analytical instruments13. Colorimetric chemosensors are highly attractive for point-of-care diagnostics, environmental field testing, and on-site industrial monitoring due to their operational simplicity, fast response time, low energy demands, and minimal infrastructure requirements14,15. They can be easily integrated into flexible formats such as test kits, dipsticks, microfluidic platforms, or paper-based analytical devices, further enhancing their portability and user-friendliness16,17. This feature is particularly advantageous for decentralized testing, where immediate decision-making is necessary. Through careful molecular design, such as incorporating heteroatoms (N, S, O), modulating electronic properties, or introducing conjugated π-systems, these sensors can achieve high degrees of selectivity and sensitivity toward specific metal ions, including Fe3+18.
Among various chemosensor scaffolds, thiazole derivatives offer a particularly promising platform. The thiazole ring contains nitrogen and sulfur atoms capable of bidentate metal ion coordination, enhancing binding specificity19. Additionally, its aromatic, conjugated structure promotes intramolecular charge transfer (ICT), which facilitates a pronounced chromogenic response upon metal interaction20,21. Thiazole-based compounds also exhibit favorable physicochemical properties such as high thermal and photochemical stability, tunable electronic characteristics, and synthetic accessibility22,23. Compared to other heterocyclic frameworks like imidazoles or pyridines, thiazoles allow greater flexibility in ligand design while retaining strong affinity for transition metals, making them excellent candidates for the development of robust and selective colorimetric Fe3+ sensors24.
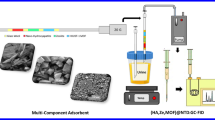
In this study, we report the design and synthesis of a novel aminothiazole-based colorimetric chemosensor, (4-methyl-2-(phenylamino)thiazol-5-yl)(phenyl)methanone (MPTP), developed via an iodine-catalyzed one-pot strategy. The sensor exhibits a distinct chromogenic shift from pale yellow to brown upon interaction with Fe3+, attributed to selective Fe-N coordination and enhanced ICT. MPTP demonstrates excellent selectivity, broad pH tolerance, and reversibility through EDTA-mediated regeneration. Furthermore, its successful integration into a paper-based platform enables portable, equipment-free detection. These findings establish MPTP as a promising next-generation chromogenic sensor, combining solution and solid-phase adaptability for reliable Fe3+ detection in pharmaceutical, clinical, and environmental settings. Figure 1 shows the schematic representation illustrating the overall workflow and key outcomes of this study.
Overview of MPTP colorimetric sensing of Fe3+ ions.
Experimental section
Materials and methods
All chemicals, solvents, and reagents were acquired from Sigma Aldrich, Merck Pvt. Ltd (India) SD fine and used as is, with no additional purification. All reactions and product purity were evaluated with thin-layer chromatography (TLC) on aluminium-backed plates. The pharmaceutical supplement FERYXIA™ (ferric citrate tablets) was purchased from a local medical store in Mysuru, Karnataka, India. The resulting product is characterized using HRMS for structural analysis. The UV-Visible spectrum was acquired using a Shimadzu UV-1900 spectrometer and a quartz cell with a path length of 1.0 cm. The FT-IR spectra were recorded using a Shimadzu IRSpirit FT-IR spectrophotometer. The1H and13C NMR spectra were obtained using a Varian 400 MHz spectrometer, and chemical shifts were measured in ppm. All (Density functional theory) DFT calculations were performed using the B3LYP functional with the 6–311 + + G(d, p) basis set in the gas phase as implemented in Gaussian 09.
Synthesis of MPTP
A 50 mL round-bottom flask was charged with a combination of 1-phenyl butane-1,3-dione (1.0 equiv.), aniline (1.0 equiv.), NaSCN (2.5 equiv.), I2 (1.0 equiv.), and DMSO (10 mL), as illustrated in Fig. 2. The reaction mixture was left to stir at 90 °C until the reaction was complete, which was monitored by TLC. After cooling the reaction mixture to room temperature, sodium thiosulfate was added to stop the reaction, followed by 10 mL of water to dilute the solution. The aqueous layer was extracted with ethyl acetate (3 × 15 mL) and dried on Na2SO425. To attain the highest purity, the product is dried and recrystallized before being characterized using several spectroscopic techniques. FT-IR (ν, cm− 1, KBr): 1775 (C = N), 1628 (C = O), 1373 (CH3), 1282 (C-N), 845 (C-S), 601 (C-H); 1H NMR (400 MHz, DMSO-d6, δ 10.83 (s, 1H, NH), 7.71–7.07 (m, 10 H, Ar-H), 2.36 (s, 3 H, CH3); 13C NMR (400 MHz, DMSO-d6, δ, ppm): 187.6, 159.2, 140.8, 132.4, 128.2, 128.6, 128.6, 129.8, 128.9, 132.1, 140, 129.9, 129.6, 123.4, 118.7, 118.6, 19.2; HRMS: m/z calculated for C17H15N2OS [M + H]⁺ = 294.37, found = 294.1366; base peak at m/z = 293.1330 ([M-H]⁺).
Synthetic route of MPTP.
Solvatochromic effect
The solution of MPTP (300 µM) was prepared in different solvents, such as DMSO, DMF, ethanol, methanol, dioxane, chloroform, and acetonitrile26. The UV-Vis absorption spectra were analysed through a UV-vis spectrometer.
Selectivity analysis of MPTP toward metal ions
To assess the selectivity of MPTP as a colorimetric sensor, MPTP was evaluated in the presence of various metal ions under identical conditions. A series of UV-Visible absorption spectra at 361 nm were recorded for MPTP (100 µM in ethanol) upon addition of 100 µM of different cations, including Ferric chloride (Fe3+), Mercury acetate (Hg2+), Ferrous ammonium sulphate (Fe2+), Manganous sulphate (Mn2+), Nickel chloride (Ni2+), Zinc chloride (Zn2+), Chromium (IV) oxide (Cr4+), Thorium nitrate (Th4+), Zirconium nitrate (Zr4+), Magnesium chloride (Mg2+), Aluminium sulphate (Al3+), Cadmium carbonate (Cd2+), Cobaltous chloride (Co2+), Copper sulphate (Cu2+)27,28.
Interference tolerance test
MPTP (100 µM) was dissolved in ethanol, and an amount of 100 µM of Hg2+, Fe2+, Mn2+, Ni2+, Zn2+, Cr4+, Th4+, Zr4+, Mg2+, Al3+, Cd2+, Co2+, Cu2+ was dissolved in ethanol. An amount of 1 mL of each metal (100 µM) was obtained and combined with 1 mL of each MPTP sensor solution (100 µM) prepared previously. Then, 1 mL of the Fe3+ solution was added to the combined solution containing metal ions and MPTP. UV-vis spectra were obtained at room temperature after a brief mixing period29.
Job’s method
To determine the binding stoichiometry between MPTP and Fe3+, Job’s method of continuous variation was employed. MPTP (100 µM) and FeCl3 (100 µM) were prepared in ethanol. A series of mixtures was prepared by varying the mole fractions of MPTP and Fe3+. Specifically, 1.5 mL of MPTP (3.0, 2.7, 2.4, 2.1, 1.8, 1.5, 1.2, 0.9, 0.6, 0.3, 0 mL) were mixed with 1.5 mL of Fe3+ (0, 0.3, 0.6, 0.9, 1.2, 1.5, 1.8, 2.1, 2.4, 2.7, 3.0 mL), respectively. The solutions were thoroughly mixed, equilibrated at room temperature, and the UV-Visible absorption spectra were recorded at 361 nm. The absorbance values were plotted against the mole fraction of Fe3+ to generate the Job plot. The maximum of the plot corresponds to the stoichiometric ratio of complexation30,31.
Results and discussion
The structure of the product was confirmed using FT-IR, 1H NMR, 13C NMR, and HRMS, shown in Supplementary Information as Section S1. FT-IR spectral analysis (Fig. S1a) confirms the presence of a carbonyl (C = N) stretch at 1775 cm− 1, C = O stretch at 1628 cm− 1, and methyl (CH3) vibrations at 1373 cm−132,33,34. The aromatic C-N stretch at 1282 cm− 1, aromatic ring deformation at 907 cm− 1, C-S stretch at 845 cm− 1, and C-H out-of-plane bending at 601 cm− 1 further support the presence of thiazole and aromatic functionalities in the compound35,36. The 1H NMR spectrum of the compound supports its proposed structure (Fig. S1b), the singlet at 10.83 ppm (1H) corresponds to the phenylamino N-H proton37,38. Aromatic signals appear between 7.71 and 7.07 ppm, consistent with protons on substituted phenyl rings showing typical splitting patterns due to neighbouring proton coupling39,40. A singlet at 2.34 ppm (3 H) is assigned to the methyl group on the thiazole ring, confirming its isolated environment41,42. The observed chemical shifts and splitting patterns are in agreement with the expected electronic environments. The C NMR spectrum (Fig. S1c) shows a distinct downfield resonance at 187.6 ppm, corresponding to the carbonyl (C = O) of the benzoyl group. The C = N carbon of the thiazole ring is observed at 159.2 ppm, while the carbon adjacent to the sulfur atom in the thiazole ring appears at 132.4 ppm. The aromatic carbons of the phenyl ring attached to the carbonyl resonate at 128.2, 128.6, 129.8, 128.9, 132.1, and 140.3 ppm. The phenyl ring of the phenylamino group shows signals at 140.8, 129.9, 129.6, 129.8, 123.4, 118.7, and 118.6 consistent with a para-substituted aromatic system. The methyl carbon attached to the thiazole ring is clearly identified at 19.235 ppm32,43,44,45. HRMS analysis revealed a molecular ion peak at m/z = 294.1366 [M + H]+, consistent with the calculated molecular mass of C17H14N2OS (294.37 g/mol), while a base peak at m/z = 293.1330 [M-H]+ further supported the molecular identity (Fig. S1d).
Plausible mechanism
The proposed mechanism for the synthesis of MPTP proceeds via a multistep iodine-mediated pathway, as illustrated in Fig. 3. The process initiates with the activation of 1-phenylbutane-1,3-dione (I), which undergoes keto-enol tautomerism due to the acidity of the methylene proton flanked by two carbonyl groups. This tautomerization generates an enolate species stabilized by resonance. I2 then reacts with the enolate, yielding an α-iodinated intermediate (II) through electrophilic iodination at the activated methylene carbon. Subsequent nucleophilic substitution of the iodine atom by thiocyanate anion (SCN−), generated from sodium thiocyanate (NaSCN), furnishes the thiocyanate intermediate (III). This electrophilic thiocyanate is then attacked by the nucleophilic amino group of aniline via a Michael-type addition, forming the phenylcarbamimidothioate intermediate (IV). Intermediate IV undergoes intramolecular cyclization via a 5-exo-trig mechanism, forming the thiazolidine ring system (V), specifically (4-hydroxy-4-methyl-2-(phenylimino)thiazolidin-5-yl)(phenyl)methanone. Dehydration of the hydroxyl group leads to the elimination of water and formation of the dihydrothiazole intermediate (V). Finally, aromatization of intermediate (V) occurs through spontaneous electron delocalization, yielding the thermodynamically stable aromatic thiazole-fused product (IV), characterized as (4-methyl-2-(phenylamino)thiazol-5-yl)(phenyl)methanone. The aromatization enhances conjugation and provides a rigid, planar structure favorable for metal coordination in sensing applications.
Proposed iodine-mediated multistep reaction mechanism for the synthesis of MPTP25.
Solvatochromic effect
The solvatochromic behavior of MPTP was systematically investigated in solvents of varying polarity to evaluate its sensitivity to environmental dielectric properties. The absorption spectra were recorded in seven solvents: DMSO, DMF, ethanol, methanol, dioxane, chloroform, and acetonitrile and are shown in Fig. 4a, while the full spectral range is included in Fig. S2. A distinct bathochromic shift (red shift) in the absorption maxima (λmax) was observed with increasing solvent polarity, characteristic of positive solvatochromism46. This behavior indicates that the excited state of MPTP is more stabilized in polar solvents than the ground state, resulting in a reduced HOMO-LUMO energy gap and, consequently, a red-shifted absorption47,48,49.
To quantify solvent polarity, Reichardt’s empirical polarity scale (ETN) was employed, and the corresponding transition energies (ET) were calculated using the Eq. (1)50:
The calculated ET values and corresponding molar absorptivity (ε) values are summarized in Table 1, where ε varies significantly across solvents, indicating altered transition probabilities linked to electronic reorganization. This systematic solvent-dependent variation in absorption characteristics reaffirms the strong ICT behavior of MPTP and highlights its utility as a potential polarity-sensitive probe in various sensing environments. The variations in λmax and ET across different solvents reflect pronounced solvent-solute interactions, confirming that MPTP undergoes significant electronic reorganization in the excited state51. These results suggest an ICT mechanism, which is common in donor-acceptor systems52,53,54.
In MPTP, the thiazole ring functions as an electron donor, while the adjacent phenyl ketone moiety acts as an electron acceptor. Upon excitation, electron density shifts from the donor to the acceptor, leading to a more polar excited state. This polar excited state is better stabilized in polar solvents, giving rise to the red-shifted absorption maximum27,47,55.
The correlation between ETN and λmax is further illustrated in Fig. 4b. The graph shows a consistent increase in λmax with increasing values ETN, reinforcing the conclusion that MPTP exhibits positive solvatochromism. This observation supports the ICT mechanism and highlights the sensitivity of MPTP to microenvironmental polarity, making it a promising candidate as a polarity-sensitive optical probe.
(a) Absorption spectra of MPTP (300 µM) in different solvents for solvatochromism at room temperature, and (b) Correlation between maximum absorption wavelength (λₘₐₓ) and solvent polarity (ETN values).
Selectivity analysis of MPTP
UV-Visible spectral response
To substantiate the visually observed selectivity, the UV-Visible absorption spectra of MPTP were recorded in the presence of various metal ions (Fig. S3). The spectrum of free MPTP displayed a well-defined absorbance profile, which remained virtually unchanged upon the addition of all tested metal ions, with the notable exception of Fe3+. Upon addition of Fe3+ ions to the MPTP solution, the λmax undergoes a hypsochromic shift from 363.81 nm (MPTP) to 359.10 nm (MPTP-Fe3+ complex shown in Fig. 5a29. This blue shift of 4.71 nm indicates a change in the electronic environment of the ligand, consistent with metal-ligand complexation and a potential decrease in conjugation or electron delocalization upon Fe3+ binding. Additionally, the absorbance intensity increases significantly, from 0.65 a.u. (free MPTP) to approximately 1.90 a.u. in the presence of Fe3+, highlighting the strong binding affinity and formation of a stable MPTP-Fe3+ complex. This combination of spectral shift and intensity enhancement strongly supports the effective coordination of Fe3+ to the sensor molecule.
Naked-eye detection
The colorimetric response was further validated by visual inspection of MPTP solutions treated with different metal ions. As seen in Fig. 5b, only the vial containing Fe3+ produced an intense brown coloration, readily visible to the naked eye56,57. All other metal ions failed to induce any significant color change, with the solutions retaining their pale-yellow appearance, essentially identical to the control sample containing MPTP alone.
This distinctive color development in the presence of Fe3+, coupled with the spectral evidence, confirms the formation of a specific MPTP-Fe3+ complex. The strong color contrast makes the probe a highly effective candidate for the simple, equipment-free detection of Fe3+ ions.
(a) Absorption variation of MPTP (100 µM) with cations (100 µM) at 361 nm, and (b) Colour change of MPTP (200 μm in ethanol) in the presence of various cations (200 μm in ethanol).
Confirmation of MPTP-Fe3+ complex formation
Fourier transform infrared (FT-IR) spectral analysis
FT-IR spectroscopy was utilized to elucidate the interaction between MPTP and Fe3+ ions. The comparative FT-IR spectra of free MPTP and the MPTP-Fe3+ complex are presented in Fig. 6, providing compelling evidence for complexation via the nitrogen coordination site. As discussed above, the spectrum of free MPTP displays characteristic bands of the ligand framework, including N-H stretching at 3214 cm− 1, a C = N band at 1775 cm− 1, C = O stretching at 1628 cm− 1, aromatic C-N at 1200 cm− 1, aliphatic C-N at 1071 cm− 1, and C-S at 845 cm− 1. Upon complexation with Fe3+, the FT-IR spectrum of the MPTP-Fe3+ complex exhibits significant changes indicative of coordination. The disappearance of the N-H stretching band at 3214 cm− 1 suggests the involvement of the N-H stretch and possible deprotonation during complex formation. The absence of the C = N stretching band at 1775 cm− 1 implies direct coordination of the imine nitrogen with, while the retention of the C = O stretching band at 1628 cm− 1 indicates that the carbonyl group is not involved in binding. The aliphatic C-N stretch at 1071 cm− 1, initially weak in the free ligand, becomes markedly more intense in the complex, likely due to conformational changes upon coordination. Other characteristic bands, including the aromatic C-N stretch at 1200 cm− 1, and the C-S stretch at 845 cm− 1, remain largely unchanged, reflecting the preservation of the ligand’s aromatic structure. Notably, a new band at 455 cm− 1, assigned to Fe-N stretching, confirms the formation of a coordination bond with a nitrogen donor, supporting the successful complexation of MPTP with Fe3+58.
FT-IR absorption band of MPTP and MPTP-Fe3+ complex.
HRMS analysis for MPTP-Fe3+ complex
To further confirm the formation and stoichiometry of the MPTP-Fe3+ complex, HRMS analysis was performed, as detailed in Section S4. An ion at m/z = 454.6322 (Fig. S4a) corresponds to the species [MPTP + Fe3+ + 3Cl−]+, confirming a 1:1 metal-ligand complex with three counter ions, supports the proposed coordination model shown in Fig. 7, and provides direct experimental evidence for the presence of this ion and its proposed complex structure. As shown in (Fig. S4b), the Job plot displays a clear maximum at a mole fraction of 0.5, indicating a 1:1 binding stoichiometry (Fig. S5) between MPTP and Fe3+18.
A proposed complex formed between MPTP-Fe3+.
Competitive interference assay
To examine the selectivity of MPTP in the presence of potential competing metal ions, mixed-ion solutions were prepared by adding Fe3+ (100 µM) to MPTP (100 µM) in the presence of other interfering cations (100 µM each). This unique spectral and visual response remained unchanged even in the presence of metal ions such as Hg2+, Fe2+, Mn2+, Ni2+, Zn2+, Cr4+, Th4+, Zr4+, Mg2+, Al3+, Cd2+, Co2+, Cu2+ (Fig. 8a)27. This robustness against interference further highlights the exceptional selectivity of MPTP for Fe3+. The persistence of the dark brown coloration is visually evident in Fig. 8b, with other metal ions. Additionally, to validate the sensor under more stringent conditions, interference studies were also performed using excess concentrations of competing metal ions (200 µM each), as shown in Fig. S6.
(a) Bar graph comparing absorbance at the λmax at 361 nm of MPTP (100 µM) with metal ions (100 µM each), and (b) Color changes of MPTP (200 µM) to Fe3+ (200 µM) with diverse cations (200 µM).
Determination of binding constant using the Benesi-Hildebrand method
The binding affinity of MPTP toward Fe3+ was quantitatively evaluated using the Benesi-Hildebrand method59. UV-Vis absorption titrations were performed by incremental addition of 5, 10, 15, 20, 25 µM of Fe3+ to a fixed concentration of MPTP 30 µM, and the resulting absorbance changes were monitored at the characteristic complexation wavelength. The experimental data were fitted to the linearized Benesi-Hildebrand equation, a linear plot of 1/A-A0 versus 1/[Fe3+] (Fig. 9a) with an excellent correlation coefficient (R2 = 0.9969), confirming the validity of the 1:1 binding model. From the slope and intercept, the Binding constant (Ka) was calculated to be 0.81 × 106 M− 1, indicating a strong and stable interaction between MPTP and Fe3+. This high binding affinity can be attributed to the strong electrostatic attraction between the electron-deficient Fe3+ ion and the electron-rich donor sites in MPTP.
Detection limit determination
To quantify the detection capability of MPTP, UV-Visible titrations were performed with increasing concentrations of Fe3+ ranging from 0 to 10 µM. A linear relationship was observed between the absorbance at 361 nm and Fe3+ concentration, as illustrated in Fig. 9b29. The detection limit (LOD) was calculated using the 3.3σ/k method, where σ is the standard deviation of the blank and k is the slope of the calibration curve60. The LOD was determined to be 0.2687 µM, which is significantly lower than the U.S. EPA’s permissible limit for Fe3+ in drinking water (5.37 µM), thereby validating the probe’s environmental relevance61. The MPTP detection limit for Fe3+ is noteworthy since it is similar to the previously published values (Table S1).
Effect of pH
We examined how pH affected the MPTP and MPTP-Fe3+ complex absorption response in a range of pH between 2 and 12 (Fig. 9c). The pH experiments showed that the ligand MPTP was stable across the investigated pH range, since it did not show any discernible absorbance increase62. The MPTP-Fe3+ combination, on the other hand, showed a noticeable pH dependency in its absorption. The absorbance was very moderate at low pH values, but it rose dramatically between pH 6 and pH 10, suggesting that complex formation was improved in this range. The absorbance dropped above pH 10, indicating that the complex’s stability in very alkaline circumstances may be compromised63.
(a) Linear plot of Benesi-Hildebrand method absorbance at 361 nm, (b) MPTP detection limit for Fe3+ at 50µM absorbance at 361 nm, (c) pH impact on MPTP and MPTP-Fe3+ absorbance at 361 nm, and (d) Visual response of MPTP-loaded test strips upon exposure to increasing concentrations of Fe3+ (50µM, 100µM, 200µM).
Reversibility and reusability via EDTA-mediated regeneration
For practical sensor applications, reversibility and reusability are essential attributes64. To evaluate the recyclability of MPTP as a Fe3+ chemosensor, we investigated its ability to undergo reversible coordination and decoordination cycles using ethylenediaminetetraacetic acid (EDTA) as a competitive chelating agent.
Visual and spectral response
As illustrated in Fig. 10a, the brown-colored solution of the MPTP-Fe3+ complex was treated with EDTA (100 µM), resulting in an immediate color change to pale yellow, resembling the original uncomplexed MPTP. This change indicates the successful extraction of Fe3+ from the complex due to EDTA’s higher chelating affinity. Upon reintroduction of Fe3+, the brown coloration was restored, signifying reformation of the MPTP-Fe3+ complex31,62. To support this observation, UV-Vis absorption measurements were recorded at 361 nm during each cycle. As shown in Fig. 10b, a marked decrease in absorbance was observed upon EDTA treatment, followed by a recovery upon reintroduction of Fe3+. This absorbance switching pattern confirms the reversibility of the complexation process. This suggests that the MPTP molecule remains structurally intact over multiple binding cycles and is not degraded during metal binding or chelator treatment. The sensor’s durability and ease of regeneration position it as an attractive candidate for deployment in disposable test kits or low-cost continuous monitoring systems.
(a) Reversible color changes of MPTP with Fe3+ after the addition of EDTA, and (b) Reversible UV-Vis response of MPTP-Fe3+ complex (30 µM) with EDTA (30 µM) at 361 nm.
Kinetic study of MPTP-Fe3+ complex formation
To assess the temporal dynamics of complex formation, time-resolved UV-Vis measurements were performed at 361 nm using equimolar concentrations of MPTP and Fe3+ (80 µM each). The absorbance exhibited a minor but highly linear decrease over 300 s, with an initial value of 1.514 and a rate of −0.0015 a.u./min. The data revealed that complexation occurs nearly instantaneously, with the minimal change suggesting a stable, post-equilibrium signal suitable for rapid and real-time Fe3+ sensing applications. Full kinetic trace and regression analysis are provided in the Fig. S7.
Practical significance
Paper-based strip sensor for on-site Fe3+ detection
To enhance the field-deployment ability and user-friendliness of the MPTP sensor, a paper-based colorimetric test strip was developed. This format enables rapid, instrument-free detection of Fe3+ in aqueous environments59.
Fabrication of test strips
Filter paper strips were prepared by immersing in a 200 µM ethanolic solution of MPTP, then air-drying under ambient conditions. The dried strips retained their original pale yellow coloration in the absence of metal ions, indicating the stability of the probe on a solid substrate65,66.
Colorimetric response to Fe3+
Upon exposure to increasing concentrations of Fe3+ (50, 100, and 200 µM), the MPTP-loaded paper strips underwent a gradual and distinct color transition from off-white to progressively deeper shades of brown, as shown in Fig. 9d. This visible response is concentration-dependent and observable to the naked eye, enabling semi-quantitative detection. The observed chromogenic progression is attributed to Fe3+-induced complexation with MPTP immobilized on the cellulose matrix, reproducing the solution-phase behavior in a solid-state system.
Environmental and pharmaceutical sample analysis
The practical applicability of MPTP was validated in a ferric citrate tablet and in simulated water27. In tablet extracts containing 2 µM Fe3+, MPTP (40 µM, λmax = 361 nm) yielded a found concentration of 2.04 µM (102% recovery) based on the calibration curve. Similarly, 2 µM Fe3+ spiked into simulated water was recovered at 1.96 µM (98% recovery), demonstrating the accurate determination of Fe3+ in a representative aqueous matrix, as shown in Table 2. This result indicates that MPTP can reliably detect Fe3+ in complex environmental and pharmaceutical samples. The complete experimental procedure, dilution details are provided in Section S812,29,67,68.
Computational validation
To further corroborate the experimental findings and investigate the electronic behaviour of MPTP at a molecular level, DFT calculations were performed using the B3LYP functional with the 6–311 + + G(d, p) basis set. These computations provide detailed insights into the compound’s electronic transitions, frontier molecular orbitals, charge distribution, and donor-acceptor interactions69,70.
The optimized structure of MPTP (Fig. 11a) revealed a planar and conjugated framework that supports extended π-delocalization across the thiazole and aromatic rings, favorable for Fe3+ coordination71. UV-Vis spectrum displayed a prominent π-π* transition centered around 361 nm, in excellent agreement with the experimental maximum absorbance (Section S9)(Fig. S9a)72. The calculated IR spectrum (Fig. S9b) closely replicated the experimental bands, including the imine (C = N) stretch near 1775 cm− 1 and the carbonyl (C = O) stretch at 1628 cm−173,74,75. The electronic circular dichroism (ECD) spectrum showed a distinct bisignate cotton effect, confirming the molecule’s chiral electronic environment (Fig. S9c). Hole electron distribution mapping (Fig. S9d) revealed partial charge separation across the molecule76. A moderate overlap index (0.634) suggested significant ICT, which is crucial for solvatochromism and metal-ion interaction. The HOMO and LUMO distributions (Fig. 11b) showed that the HOMO is primarily localized on the thiazole and adjacent phenyl rings, while the LUMO spans the electron-deficient region, including the carbonyl-bearing phenyl ring77.
This orbital configuration facilitates donor-acceptor interactions during Fe3+ binding. The charge density difference map (Fig. 11c) illustrated electron accumulation and depletion zones, highlighting potential binding sites. The MEP surface (Fig. 11d) revealed electron-rich regions around the nitrogen atoms, indicating their likely involvement in Fe3+ coordination78. This spatial localization of electron density is consistent with the electrophilic nature of Fe3+, which selectively binds to nucleophilic, electron-rich centers. Hence, the electron-rich nitrogen sites on MPTP facilitate strong electrostatic and orbital interactions, underpinning its selective affinity for Fe3+78. Furthermore, the frontier molecular orbital (FMO) analysis and global reactivity descriptors derived from the DFT calculations summarized in Table 3 provide quantitative insight into the molecule’s stability and reactivity. The relatively low energy gap (ΔE = 3.79 eV) and high electrophilicity index (ω = 4.33 eV) suggest that MPTP possesses both good stability and a strong tendency to accept electrons, making it a suitable ligand for transition metal binding80.
(a) MPTP structure optimization, (b) The HOMO-LUMO gap of MPTP, (c) Charge density difference, and (d) MEP of MPTP.
Conclusion
In this study, an aminothiazole-based colorimetric sensor, MPTP was synthesized via a simple iodine-catalysed one-pot route and shown to act as an efficient chemosensor for Fe3+. The probe exhibits a rapid and distinct colour/absorbance response at 361 nm, with excellent selectivity over competing metal ions, broad pH tolerance (2–12), and a low detection limit of 0.268 µM. Job’s plot and Benesi-Hildebrand analyses confirm a 1:1 MPTP-Fe3+ complex with a high association constant, while FT-IR (loss of N-H and C = N bands and appearance of an Fe-N vibration) and kinetic studies highlight nitrogen-centred coordination and fast complex formation, respectively. DFT, frontier orbital, hole-electron, and MEP analyses corroborate the experimental findings, revealing a π-π* transition with pronounced ICT, nitrogen-rich coordination sites, and a low HOMO-LUMO gap with high electrophilicity, which together rationalize the strong and selective Fe3+ binding. The sensor is reversible upon EDTA treatment, enabling reuse, and can be translated into a simple paper-based strip for instrument-free detection. Finally, real-sample measurements yielded recoveries close to 100%, demonstrating accurate Fe3+ determination with negligible matrix interference. Overall, MPTP offers a robust, regenerable, and field-deployable platform for routine spectrophotometric and colorimetric monitoring of Fe3+ in pharmaceutical and environmental matrices.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Adampourezare, M., Hasanzadeh, M., Hoseinpourefeizi, M. A. & Seidi, F. Iron/iron oxide-based magneto-electrochemical sensors/biosensors for ensuring food safety: recent progress and challenges in environmental protection. RSC Adv. 13, 12760–12780 (2023).
Ayodhya, D. Recent progress on detection of bivalent, trivalent, and hexavalent toxic heavy metal ions in water using metallic nanoparticles: A review. Results Chem. 5, 100874 (2023).
Nirma Maurya, S. & Bhardwaj, A. K. S. Selective and sensitive colorimetric sensor for CN- in absence and presence of metal ions (Cu2+/Ni2+). Mimicking Log. Gate Behav. https://doi.org/10.1039/C6RA14237K (2016).
Carocci, A., Catalano, A., Sinicropi, M. S. & Genchi, G. Oxidative stress and neurodegeneration: the involvement of iron. BioMetals 31, 715–735 (2018).
Xia, X., Teng, Y. & Zhai, Y. Biogeochemistry of iron enrichment in groundwater: an indicator of environmental pollution and its management. Sustain 14, 7059 (2022).
Maurya, N. & Singh, A. K. Indirect approach for CN- detection via Cu2 + induced turn-off sensor: using novel AIEE fluorophore with logic gate and antimicrobial application. Dye Pigment. 147, 484–490 (2017).
Sardans, J., Montes, F. & Peñuelas, J. Determination of As, Cd, Cu, hg and Pb in biological samples by modern electrothermal atomic absorption spectrometry. Spectrochim Acta - Part. B Spectrosc. 65, 97–112 (2010).
Rakshitha, G. S. et al. Green synthesized nanocomposites for Mercury, Lead, and cadmium detection. In Biocompatible Nanomaterials for Smart Sensors: Environmental and Biomedical Applications (eds Naik, R., Nagaswarupa, H. P., Koyyada, G. et al.) 67–90 (Springer Nature Switzerland, 2025). https://doi.org/10.1007/978-3-032-01745-1_5 (& Basava Kumar, K. G.)).
Hareesha, N. et al. Honeycomb polypore biomass-derived activated porous carbon nanosheets/graphite/nafion composite: green and sensitive electrocatalyst for nanomolar detection of Hg2 + ions and water-splitting reactions. J. Environ. Chem. Eng. 12, 113584 (2024).
Nagarajappa, H. et al. Carbon-Based electrochemical sensors and voltammetry for the detection of biologically active molecules: Fundamentals, theoretical Aspects, and emerging perspectives. Langmuir 42, 10–31 (2026).
Li, Z., Askim, J. R. & Suslick, K. S. The optoelectronic nose: colorimetric and fluorometric sensor arrays. Chem. Rev. 119, 231–292 (2019).
Maurya, N., Bhardwaj, S. & Singh, A. K. Selective colorimetric and fluorescence ‘turn-on’ sensor for Ag + and in-situ sensing of CN– (off – on-off) via displacement approach. Mater. Sci. Eng. C. 74, 55–61 (2017).
Zhou, Y. & Yoon, J. Recent progress in fluorescent and colorimetric chemosensors for detection of amino acids. Chem. Soc. Rev. 41, 52–67 (2012).
Kumar, V. et al. Recent advances in fluorescent and colorimetric chemosensors for the detection of chemical warfare agents: a legacy of the 21st century. Chem. Soc. Rev. 52, 663–704 (2022).
Alberti, G., Zanoni, C., Magnaghi, L. R. & Biesuz, R. Disposable and low-cost colorimetric sensors for environmental analysis. Int. J. Environ. Res. Public. Health. 17, 1–23 (2020).
Kap, Ö., Kılıç, V., Hardy, J. G. & Horzum, N. Smartphone-based colorimetric detection systems for glucose monitoring in the diagnosis and management of diabetes. Analyst 146, 2784–2806 (2021).
Sousa, L. R. et al. A portable tool for colorimetric detection of corrosion inhibitors using paper-based analytical devices. Microchem J. 183, 108024 (2022).
Roy, N., Dutta, A., Mondal, P., Paul, P. C. & Singh, T. S. A new coumarin based dual functional chemosensor for colorimetric detection of Fe3 + and fluorescence turn-on response of Zn2+. Sens. Actuators B Chem. 236, 719–731 (2016).
Pothulapadu, C. A. S., Jayaraj, A., Swathi, N., Priyanka, R. N. & Sivaraman, G. Novel Benzothiazole-Based highly selective ratiometric fluorescent Turn-On sensors for Zn2 + and colorimetric chemosensors for Zn2+, Cu2+, and Ni2 + Ions. ACS Omega. 6, 24473–24483 (2021).
Pylova, E. K., Sukhikh, T. S., Prieto, A., Jaroschik, F. & Konchenko, S. N. Chemistry of 2-(2′-Aminophenyl)benzothiazole derivatives: Syntheses, photophysical properties and applications. Molecules 30(8), 1659 (2025).
Wu, J. et al. Chromogenic/Fluorogenic ensemble chemosensing systems. Chem. Rev. 115, 7893–7943 (2015).
Dessì, A. et al. Thiazolo[5,4-d]thiazole-based organic sensitizers with strong visible light absorption for transparent, efficient and stable dye-sensitized solar cells. RSC Adv. 5, 32657–32668 (2015).
Dondoni, A. & Marra, A. Thiazole-mediated synthetic methodology. Chem. Rev. 104, 2557–2599 (2004).
Mahesh, K. & Karpagam, S. Thiophene-thiazole functionalized oligomers-excellent fluorescent sensing and selective probe for copper and iron ion. Sens. Actuators B Chem. 251, 9–20 (2017).
Philips, A., Arumugam, A. & Eswaramoorthy, Y. Iodine-Mediated Three-Component strategy to synthesize 2-Aminothiazoles from β -Diketones / β -ketoesters, arylamines and ammonium thiocyanate. https://doi.org/10.1002/ejoc.202201233
Hanif, M. et al. Complexes of 2-Amino-3-methylpyridine and 2-Amino-4-methylbenzothiazole with ag (I) and Cu (II): structure and biological applications. Inorganics, 11(4), 152 (2023).
Kim, A., Chae, J. B., Rha, C. J. & Kim, C. A colorimetric chemosensor for selective detection of copper ions. Color. Technol. 136, 459–467 (2020).
Maurya, N., Bhardwaj, S. & Singh, A. K. A modest colorimetric chemosensor for investigation of CN- in semi-aqueous environment with high selectivity and sensitivity. Sens. Actuators B Chem. https://doi.org/10.1016/j.snb.2016.02.014 (2016).
Alorabi, A. Q. A new colorimetric chemosensor based on 1,3,4-oxadiazole derivative for the high selectivity and sensitivity of Fe3 + ion detection. J. Mol. Struct. 1251, 132019 (2022).
Sarkar, D., Chowdhury, M. & Das, P. K. Naphthalimide based fluorescent organic nanoparticles in selective sensing of Fe3 + and as a diagnostic probe for Fe2+/Fe3 + transition. J. Mater. Chem. B. 9, 494–507 (2021).
Kim, Y. S., Lee, J. J., Lee, S. Y., Jo, T. G. & Kim, C. A highly sensitive benzimidazole-based chemosensor for the colorimetric detection of Fe(II) and Fe(III) and the fluorometric detection of Zn(II) in aqueous media. RSC Adv. 6, 61505–61515 (2016).
Donnakatte Neelalochana, V. et al. Anion exchange membranes based on chemical modification of recycled PET bottles. ACS Appl. Polym. Mater. 5, 7548–7561 (2023).
Delrue, C. et al. Serum-Based assessment of alopecia areata response to treatment using ATR-FTIR spectroscopy. Diagnostics, 15(11), 1369 (2025).
Donnakatte Neelalochana, V. et al. Sustainable conversion of PET waste bottle into proton exchange membranes for fuel cells. ACS Appl. Energy Mater. https://doi.org/10.1021/acsaem.4c03313 (2025).
Vieira, V. F. D., Morais, E. M., Beserra Júnior, J. E. A. & da S. Araújo, F. D. Discrimination of plant pathogenic fungi using attenuated total reflectance fourier transform infrared spectroscopy and chemometrics. Eur. J. Plant. Pathol. 168, 787–797 (2024).
Ren, Z. et al. Phosphorus-sulfur co-doped carbon nitride quantum Dots for enhanced photocatalytic reduction and fluorescence detection of cr (VI). Carbon Lett. 35, 1445–1459 (2025).
Yurttaş, L., Evren, A. E. & Özkay, Y. Synthesis and in Silico evaluation of some new 2, 4-disubstituted thiazole derivatives. 8–19 (2022).
Thi, N., Mai, N., Huan, T. T., Van Thanh, L. & Huan, N. V. Synthesis and antimicrobial activities of hydrazones derived from 4-hydroxy-3-nitrobenzaldehyde. 61(3), 373–381 (2023).
Martínez-López, J. C. et al. Dibenzodioxin-Based polymers of intrinsic microporosity with enhanced transport properties for lithium ions in aqueous media. Macromolecules 57, 9442–9456 (2024).
Neelalochana, V. D. et al. Impact of functionalized titanium oxide on anion exchange membranes derived from chemically modified PET bottles. Electrochim. Acta. 507, 145170 (2024).
Chaban, T. I. et al. Synthesis, anti-inflammatory and antioxidant activities of novel 3H-thiazolo[4,5-b]pyridines. Acta Chim. Slov. 66, 103–111 (2019).
Mohana, K. N. & Kumar, C. B. P. Synthesis and Antioxidant Activity of 2-Amino-5-methylthiazol Derivatives Containing 1, 3, 4-Oxadiazole-2-thiol Moiety. (2013). (2013).
Drexler, M., Francesconi, Z., Arnold, V., Zevaco, U., Sauer, J. & T. A. & Production of oxymethylene ethers (OME) as sustainable diesel fuel substitutes: continuous synthesis from dimethyl ether and trioxane and evaluation of catalyst stability. RSC Sustain. 1941–1956. https://doi.org/10.1039/d4su00818a (2025).
Boutiddar, R. et al. In Silico structural Study, design and efficacy evaluation of fluoro Isoxazolidine derivatives as potential antibacterial agents. J. Fluoresc. https://doi.org/10.1007/s10895-025-04302-1 (2025).
Byeon, C. H. et al. Ultrasensitive characterization of native bacterial biofilms via dynamic nuclear Polarization-Enhanced Solid-State NMR. Angew Chemie - Int. Ed. 202418146, 1–9 (2025).
Nigam, S. & Rutan, S. Principles and applications of solvatochromism. Appl. Spectrosc. 55, 362A (2001).
Uahengo, V., Hamukwaya, E. N., Endjala, P. T. & Naimhwaka, J. H. A potential naphthyl-thiazole-based organic dye and a ditopic chromogenic probe for CN-and Fe3 + with molecular logic functions. New. J. Chem. 44, 18588–18600 (2020).
Reichardt, C. A colorimetric chemosensor for selective detection of copper ions. Chem. Rev. 94, 2319–2358 (1994).
Sucre-Rosales, E., Fernández-Terán, R., Urdaneta, N., Hernández, F. E. & Echevarria, L. Solvatochromism and intramolecular charge transfer in dialkylamino-substituted halogenated Thienyl chalcone analogues. Chem. Phys. 537, 110854 (2020).
Baliarsingh, S., Patel, S. & Mishra, B. K. Solvent effect on the photophysical behavior of some bischromophoric dyes. 52, 9–18 (2013).
Heitele, H., Finckh, P., Weeren, S., Pöllinger, F. & Michel-Beyerle, M. E. Solvent Polarity effects on intramolecular electron transfer. 1. Energetic aspects. J. Phys. Chem. 93, 5173–5179 (1989).
Mondal, A., Mondal, P. & Chattopadhyay, P. Regulatory principles, parameters and probe design to ascertain the interconnection between ICT, ESIPT and PET mechanisms. Inorganica Chim. Acta. 571, 122194 (2024).
de Melo, C. E. A., Domínguez, M., Rezende, M. C. & Machado, V. G. Solvatochromism of dyes inspired in effenberger’s probe. Dye Pigment 184, 108757 (2021).
Wildervanck, M. J. & Hecht, R. & Nowak-Król, A. Synthesis and strong solvatochromism of Push-Pull Thienylthiazole Boron complexes. Molecules 27(17), 5510 (2022).
Breitung, E. M., Shu, C. F. & McMahon, R. J. Thiazole and thiophene analogues of donor-acceptor stilbenes: molecular hyperpolarizabilities and structure-property relationships. J. Am. Chem. Soc. 122, 1154–1160 (2000).
Chereddy, N. R., Suman, K., Korrapati, P. S., Thennarasu, S. & Mandal, A. B. Design and synthesis of Rhodamine based chemosensors for the detection of Fe 3 + ions. Dye Pigment. 95, 606–613 (2012).
Nirma Maurya, A. K. S. Effective Ensemble System for Identification of CN¯, Based on Cobalt (II) Complex: Mimicking Logic Gate. (2017). https://doi.org/10.1039/C6NJ03926J
Saremi, M., Kakanejadifard, A., Ghasemian, M. & Namdari, M. A colorimetric and turn-on fluorescent sensor for rapid and selective detection of Fe3 + ion based on Azo compound of 4-((4-(dimethylamino) phenyl)diazenyl)-N-(pyridin-2-yl)benzamide. J. Mol. Struct. 1283, 135289 (2023).
Mohammadi, A., Khalili, B. & Haghayegh, A. S. A novel Chromone based colorimetric sensor for highly selective detection of copper ions: Synthesis, optical properties and DFT calculations. Spectrochim Acta - Part. Mol. Biomol. Spectrosc. 222, 117193 (2019).
Prasad, G. D., Niranjan, R., Arockiaraj, M., Rajeshkumar, V. & Mahadevegowda, S. H. Synthesis of Di(thiophen-2-yl) substituted Pyrene-Pyridine conjugated scaffold and DFT insights: A selective and sensitive Colorimetric, and ratiometric fluorescent sensor for Fe(III) ions. J. Fluoresc. 35, 789–803 (2024).
Parab, D. D., Yadav, V. O. & Kohli, H. K. Recent progress in water soluble fluorescent sensors for detection of Al + 3, Fe + 3 and Cu + 2 ions. 9(1), 167–185 (2025).
Kim, H. et al. A single colorimetric sensor for multiple target ions: the simultaneous detection of Fe2 + and Cu2 + in aqueous media. RSC Adv. 4, 22463–22469 (2014).
Jang, H. J., Chae, J. B., Jung, J. M., So, H. & Kim, C. Colorimetric detection of Co2+, Cu2+, and Zn2 + by a multifunctional chemosensor in aqueous solution. Bull. Korean Chem. Soc. 40, 650–657 (2019).
Ozmen, P., Demir, Z. & Karagoz, B. An easy way to prepare reusable rhodamine-based chemosensor for selective detection of Cu2 + and Hg2 + ions. Eur Polym. J 162, 110922 (2022).
Kianfar, M. & Mohammadi, A. A simple and sensitive thiazole-based colorimetric chemosensor for detection of CN–, AcO – and Cu2 + ions. J. Iran. Chem. Soc. 17, 1429–1438 (2020).
Wang, J. et al. Novel phenothiazine-based fast-responsive colori/fluorimetric sensor for highly sensitive, selective and reversible detection of Cu2 + in real water samples and its application as an efficient solid-state sensor. Microchem J. 157, 104990 (2020).
Varun, D. N. et al. Simple and sensitive electrochemical analysis of riboflavin at functionalized carbon nanofiber modified carbon nanotube sensor. Monatshefte Fur Chemie. 152, 1183–1191 (2021).
Itagi, S. A. et al. Fast and facile voltammetric detection of acetaminophen at Poly (DL-phenylalanine) modified Dysprosium-Copper oxide Nanoparticle/Carbon composite paste electrode. Open. Chem. Eng. J. 15, 31–40 (2021).
Bhardwaj, S., Maurya, N. & Singh, A. K. Chromone based fluorescent organic nanoparticles for High-Precision in-situ sensing of Cu2 + and CN‾ ions in 100% aqueous solutions. Sens. Actuators B Chem. https://doi.org/10.1016/j.snb.2018.01.003 (2018).
Kumar, A. et al. DFT and experimental insights into a colorimetric pyridine-2, 6-dicarboxamide chemosensor for selective Cu 2 + detection. 282, 117787 (2025).
Mariammal, B., Shylaja, A., Kumar, S. V., Rubina, S. R. & Kumar, R. R. Thiazole-tethered biaryls as fluorescent chemosensors for the selective detection of Fe3 + ions. J. Heterocycl. Chem. 57, 3882–3889 (2020).
Normaya, E., Ahmad Hamdan, M. F., Ahmad, M. N., Aziz, Y. F. A. & Bulat, K. H. K. DFT/TD-DFT study on development and optimization of 1- anilino-3-phenyliminourea as a colorimetric chemosensor for Hg2 + recognition in aqueous medium. J. Mol. Struct. 1206, 127699 (2020).
Ţucureanu, V., Matei, A. & Avram, A. M. FTIR spectroscopy for carbon family study. Crit. Rev. Anal. Chem. 46, 502–520 (2016).
Gunasekaran, S., Sailatha, E., Seshadri, S., Kumaresan, S. & FTIR FT Raman spectra and molecular structural confirmation of Isoniazid. Indian J. Pure Appl. Phys. 47, 12–18 (2009).
Schneider, I. L. et al. FTIR analysis and evaluation of carcinogenic and mutagenic risks of nitro-polycyclic aromatic hydrocarbons in PM1.0. Sci. Total Environ. 541, 1151–1160 (2016).
Menke, S. M. et al. Order enables efficient electron-hole separation at an organic heterojunction with a small energy loss. Nat. Commun. 9, 1–7 (2018).
Sable, Y. R., Adole, V. A., Pithawala, E. A. & Amrutkar, R. D. Design, synthesis, and antitubercular evaluation of piperazinyl-pyrazolyl-2- Hydrazinyl thiazole derivatives: Experimental, DFT and molecular Docking insights. J. Sulfur Chem. https://doi.org/10.1080/17415993.2025.2461582 (2025).
Suresh, C. H. Molecular electrostatic potential analysis: A powerful tool to interpret and predict chemical reactivity. (2022). https://doi.org/10.1002/wcms.1601
Bachs-Herrera, A., Vidal-Daza, I., Boz, E. B., Forner-Cuenca, A. & Martin-Martinez, F. J. Understanding the role of nitrogen-doping and surface topology in the binding of Fe(iii)/Fe(ii) to biobased carbon electrodes. Energy Adv. 3, 1271–1282 (2024).
Sumrra, S. H., Hassan, A. U., Zafar, W., Chohan, Z. H. & Alrashidi, K. A. Molecular engineering for UV-Vis to NIR Absorption/Emission bands of Pyrazine-based A-π-D- π-A switches to design TiO2 tuned dyes: DFT insights. J. Fluoresc. https://doi.org/10.1007/s10895-024-03891-7 (2024).
Acknowledgements
The authors, NS and CSK, thankfully acknowledge JSS Science and Technology University, Mysuru, for their funding support. One of the authors, KK, thanks Khalifa University of Science and Technology for their financial support.
Funding
Khalifa University of Science and Technology, Abu Dhabi, United Arab Emirates, has supported this work.
Author information
Authors and Affiliations
Contributions
G.S. Rakshitha: synthesis, Formal analysis, and Writing - original draft. S Nanjundaswamy: Conceptualization, Supervision, Writing - review and editing. C.S. Karthik, K. Karuppasamy, P. Mallu, and Prof. Akram: Supervision, Writing - review and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rakshitha, G.S., Karthik, C.S., Karuppasamy, K. et al. A robust aminothiazole-based colorimetric sensor for visual detection of Fe3+ ions in environmental and pharmaceutical samples. Sci Rep 16, 9399 (2026). https://doi.org/10.1038/s41598-026-38683-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38683-5