Abstract
We analyzed how conditional survival (CS) after radiofrequency ablation (RFA) for early-stage hepatocellular carcinoma (HCC) predicts survival estimates over time. We enrolled 791 patients with newly diagnosed early‐stage HCC (i.e., within Milan criteria) and Child–Pugh class A liver disease undergoing RFA. Three-year CS, calculated as CS = S(x + 3)/S(x), was the probability of surviving an additional three years, given that the patient had already survived x years. One-, three-, and five-year overall survival rates were 96.3, 72.3, and 59.7%, respectively, and were lower for patients with a MELD score of > 9, BCLC stage A disease, and AFP level of ≥ 20 ng/ml. However, three-year CS showed that patients ablated for BCLC stage A or AFP ≥ 20 ng/ml had the same survival probabilities as patients with BCLC stage 0 or AFP < 20 ng/ml from the third year onward after RFA. In contrast, the three-year CS of patients with a MELD score of ≤ 9 was always better than that of patients with a MELD score of > 9. We concluded that CS showed that the impact of different variables on survival after RFA is not linear over time. Information derived from CS can be used to dynamically predict the prognosis of patients with HCC.
Similar content being viewed by others
Introduction
Percutaneous radiofrequency ablation (RFA) has been introduced in clinical guidelines as a potentially curative treatment for early-stage hepatocellular carcinoma (HCC) since the early 2000s1,2,3. Survival estimates are typically reported from the time of diagnosis of HCC and are estimated via several clinical and pathological risk factors such as tumor number, size, vascular invasion, and extrahepatic metastasis and α-fetoprotein (AFP) level, age, and liver function reserve1,2,3. However, similar to the trends in mortality of patients with other malignancies, hazard ratios for death of patients with HCC are higher in the first few years and decrease with time thereafter4,5. This indicates that the risk of HCC recurrence and, therefore, overall survival (OS), changes over time, and thus survival estimates made at the time of diagnosis become inaccurate. Real-time survival probability can be estimated using conditional survival (CS) analysis, which predicts the survival probability of a patient who has already survived for a period of time6.
Multiple studies have reported changes over time in the CS of patients undergoing resection for HCC7,8,9,10. However, only one study reported the CS of patients undergoing RFA for HCC7. It enrolled patients with Child–Pugh class B liver disease. However, life expectancy is limited in patients with decompensated cirrhosis11. Therefore, the present study aimed to define the CS probabilities of patients with Child–Pugh class A liver disease undergoing RFA for HCC.
Materials and methods
Definition of conditional survival
Conditional survival can be calculated using the Kaplan–Meier method. The mathematical definition of CS is CS (y/x), which is the probability of surviving for an additional y years, given that the person has already survived x years. Let S(t) be the traditional actuarial life table survival at time t. Then, conditional survival can be expressed as CS (y/x) = S(x + y)/S(x)6.
HCC registry data at our institution
For this study, data were extracted from the Kaohsiung Chang Gung Memorial Hospital’s HCC registry. The data for this registry are prospectively collected. The reference is the Surveillance, Epidemiology, and End Results (SEER) Program (https://seer.cancer.gov/), which is an authoritative source for cancer statistics in the US. The HCC registry has adopted the original Barcelona Clinic Liver Cancer (BCLC) staging12. BCLC stage 0 is defined as a single tumor of ≤ 2.0 cm without macrovascular invasion or extrahepatic metastasis, with an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0 and Child–Pugh class A liver disease. BCLC stage A is defined as a single tumor of 2–5 cm or < 3 tumor nodules of ≤ 3 cm without macrovascular invasion or extrahepatic metastasis, with an ECOG-PS of 0 and Child–Pugh class A or B liver disease.
Study population and inclusion criteria
Patients with newly diagnosed early‐stage HCC (i.e., BCLC stage 0 or A) and Child–Pugh class A liver disease undergoing RFA between 2011 and 2021 at our institution were enrolled. We excluded patients with incomplete ablation. Complete ablation was obtained if no nodular, focal or irregular enhancement after RFA at the ablation zone was visible during the arterial phase by contrast-enhance computed tomography (CT) or magnetic resonance imaging (MRI)13. This follow-up scan was typically done 1 month after RFA.
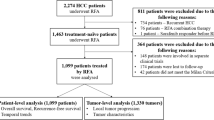
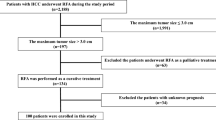
The flowchart of patient enrollment is shown in Fig. 1.
Flowchart of patient enrollment.
Overall survival was defined as the time from the date of RFA to the date of last follow-up or death; patients were censored at last follow-up. Recurrence-free survival (RFS) was defined as the time from the date of RFA to the date of last follow-up or recurrence; patients were censored at last follow-up. Follow-up of patients subjected to salvage transplantation was censored the day before the procedure. Recurrence was defined according to image studies (i.e., liver imaging reporting and data system [LI-RADS] category 514) or pathology diagnosis1,2,3.
Definition of well-preserved liver function in Child–Pugh class A liver disease
We assumed that compensated liver function could be stratified with additional granularity using a MELD score of > 9 for patients with HCC15.
RFA procedure
All procedures were carried out under general anesthesia or sedation. RFA was performed percutaneously with ultrasound guidance using intra-tumoral approach.
The details of the RFA procedure are described in our previous publication16.
Statistical analyses
Data are presented as number (percentage) or median (interquartile range [IQR]). Survival rates were calculated using the Kaplan–Meier method, compared using the log-rank test, and applied to the calculation of three-year CS. Only variables that were significantly related to patient survival in the Kaplan–Meier analysis were used for calculation of three-year CS. The differences in CS between subgroups were compared using standardized differences (d). A d value < 0.2 indicated no meaningful difference between means; 0.2–0.5 indicated a small difference; 0.5–0.8 indicated a moderate difference; and > 0.8 indicated a large difference or clear separation17. All p-values were two-tailed, and a p < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS Statistics software (version 25; IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics of the study cohort
In the cohort of patients in this study, 389 (49.2%) were aged > 65 years; 495 (62.6%) were male; 162 (20.5%) were with multiple tumors; 459 (58.0%) were with tumors > 20 mm in size; 289 (36.5%) were BCLC stage 0; 502 (63.5%) were BCLC stage A; 289 (36.5%) had an AFP level of ≥ 20 ng/ml; 330 (41.7%) were hepatitis B surface antigen (HBsAg) positive; 376 (47.5%) were anti-hepatitis C virus (HCV) positive; and 378 (47.8%) were with HCCs diagnosed by pathology. The median (IQR) body mass index was 25.2 (22.8–27.9) and the median (IQR) MELD score was 8.2 (6.8–10.1).
Five-year OS and RFS of this cohort
The median (IQR) follow-up period was 2.3 (0.9–4.9) years. Twenty patients (2.5%) underwent salvage transplantation. The five-year OS was 59.7% (Fig. 2) and the five-year RFS was 34% (Fig. 3).
Kaplan–Meier curves for overall survival.
Kaplan–Meier curves for recurrence-free survival.
Assessment of OS based on different clinical factors
The five-year OS of patients with BCLC stage A cancer was lower than that of patients with stage 0 disease (p = 0.024). The OS of patients with AFP ≥ 20 ng/ml was inferior to that of patients with AFP < 20 ng/ml (p = 0.01). In addition, the OS of patients with a MELD score of > 9 was also inferior to that of patients with a MELD score of ≤ 9 (p < 0.001). Thus, the expected actuarial patient survival was determined by both factors associated with liver function reserve and tumor-related variables (Table 1).
Comparison of CS estimates between different subgroups over time
CS analysis demonstrated that unconditional OS estimates may underestimate prognosis for patients who have already survived beyond the initial treatment period. For example, the unconditional 5-year OS rate for all patients was 59.7%, whereas patients who had already survived 2 years had a 3-year conditional OS of 88.1% (Fig. 4). Similar trends were observed across clinical subgroups: for patients with BCLC stage 0, the 5-year OS was 67.1%, increasing to 91.0% after 2 years (Fig. 5); for BCLC stage A, the 5-year OS was 55.0%, rising to 86.0% (Fig. 6); for AFP < 20 ng/mL, the 5-year OS was 65.1%, increasing to 88.6% (Fig. 7); and for AFP ≥ 20 ng/mL, the 5-year OS was 51.1%, with a conditional OS of 87.6% (Fig. 8). Among patients with a MELD score ≤ 9, the 5-year OS was 67.0%, improving to 92.1% after 2 years (Fig. 9), while those with MELD > 9 had a 5-year OS of 48.0%, which increased to 81.6% conditionally (Fig. 10). These findings highlight that CS provides a more accurate and encouraging prognosis for patients who have already surpassed early survival milestones.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for all patients.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with BCLC stage 0.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with BCLC stage A.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with AFP < 20 ng/mL.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with AFP ≥ 20 ng/mL.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with MELD score ≤ 9.
Three-year conditional survival (CS3) compared with actuarial overall survival (OS) for patients with MELD score > 9.
The CS of the entire study population showed an increase in the first three years and a decrease from the fourth year onward. The three-year CS of patients with a MELD score of ≤ 9 was always higher by at least 6.3% than that of patients with a MELD score of > 9 except in the third year after RFA. Unlike the MELD score, which is related to liver function reserve, tumor factors were associated with shortening survival only in the first two years. From the third year after RFA, the probability of survival of patients with a more advanced tumor stage (i.e., BCLC stage A) or aggressive tumor biology (i.e., AFP ≥ 20 ng/ml) was very similar to that of patients with less advanced tumor stages (i.e., BCLC stage 0) or with indolent tumor biology (i.e., AFP < 20 ng/ml). Differences in three-year CS of patients with BCLC stage A cancer started to diminish from the third year onward: the probability of surviving an additional three years, given that such patients have already survived three years, was only 0.5% less than that of BCLC stage 0 patients. Similarly, two years after RFA, the probability of patients with AFP ≥ 20 ng/ml surviving an additional three years was only 0.8% less than that of patients with AFP < 20 ng/ml and remained stable in the following years (Table 2).
Discussion
Radiofrequency ablation is a curative treatment option for early-stage HCC, but the rate of recurrence is high after RFA1,2,3. Recurrence (except local recurrence) in the first two years after RFA is presumed to represent metastatic dissemination of the initially ablated tumor18,19,20 and carry a worse prognosis. In contrast, recurrence beyond two years is presumed to be de novo tumors, which suggests better prognosis21. Therefore, the survival estimates made at the time of HCC diagnosis do not accurately predict the survival of a given patient as time passes.
To the best of our knowledge, this is the largest study to date to examine the CS pattern of early-stage HCC in patients undergoing RFA. We showed that tumor features, such as BCLC stage and AFP level, were indeed predictors of survival as expected, but only in the first two years, which is compatible with the concept of early recurrence having a strong impact on OS. In contrast, surrogate markers of liver function reserve, such as the MELD score, predicted survival at all time points. Cucchetti et al. obtained similar results. They enrolled 300 consecutive cirrhotic patients who underwent HCC resection. The five-year CS of patients with a MELD score of < 9 was always higher by at least 5.5% than that of patients with a MELD score of ≥ 9. In contrast, tumor factors shortened survival only in the first two years. On the other hand, two years after liver resection, the probability of survival of patients with adverse pathological tumor characteristics (i.e., tumor–node–metastasis [TNM]22 T3, tumor grade23 3 or 4, or the presence of microscopic vascular invasion [MVI]) was very similar to that of patients without adverse pathological tumor characteristics (i.e., TNM T1–2, tumor grade 1 or 2, or absence of MVI)8.
Facciorusso et al. enrolled 125 very early/early HCC patients treated with RFA.
Age, Child–Pugh class, AFP, and Cancer of the Liver Italian Program (CLIP) score24 were significant predictors of OS. Age and Child–Pugh class were significant predictors of CS at all time points, while the CS of subgroups stratified by AFP and CLIP did not differ significantly from the third year onward after RFA7. The results of Facciorusso et al.7 were similar to our findings. While surrogate markers of liver function reserve (i.e., MELD score in our study and Child–Pugh class in Facciorusso et al.7) were significant predictors of CS at all time points, tumor factors (i.e., BCLC stage and AFP level in our study and AFP level and CLIP stage in Facciorusso et al.7) did not differ significantly from the third year onward after RFA.
In Facciorusso et al.’s study, the distribution of patients with CLIP scores of 0, 1, and 2 was 70 (56%), 48 (38%), and 7 (6%), respectively7. Their enrollment of Child–Pugh class B patients may explain why the CLIP score was associated with OS7. We did not enroll Child–Pugh class B patients due to limited life expectancy in patients with decompensated cirrhosis if they did not undergo liver transplantation. Due to the extreme shortage of deceased organ donors in Taiwan, only 20 (2.5%) patients underwent salvage transplantation in our study.
Several studies have investigated post-ablation outcomes beyond the use of CS. Prior research has proposed predictive models for early recurrence following RFA for hepatocellular carcinoma (HCC)25,26,27,28. For example, Cha et al.27 developed a model incorporating variables such as age, albumin–bilirubin (ALBI) grade, AFP, protein induced by vitamin K absence or antagonist-II (PIVKA-II), and imaging features suggestive of microvascular invasion (MVI). Ni et al.28 included tumor number, neutrophil-to-lymphocyte ratio, contrast-enhanced ultrasound findings, and AFP level in their predictive model. Xin et al.26 used the systemic inflammation response index (defined as neutrophil count × monocyte count / lymphocyte count), AFP, tumor number, and tumor size. Yang et al.25 identified multiple tumors, along with AFP, gamma-glutamyltransferase, and serum albumin levels, as key predictors. These studies consistently emphasize that early recurrence—defined as recurrence within the first two years after RFA—is a major determinant of long-term survival18,19,20.
The strength of the present study is its large sample size and our enrollment of only Child–Pugh class A patients. However, it is also limited by being a retrospective single-center study.
Conclusion
The three-year CS calculation showed that patients ablated for BCLC stage A or with AFP ≥ 20 ng/ml experienced the same survival probabilities as patients with BCLC stage 0 or AFP < 20 ng/ml from the third year onward after RFA. The three-year CS of patients with a MELD score of ≤ 9 was always better than that of patients with a MELD score of > 9. This information could be used for dynamically predicting the prognosis of patients undergoing RFA for early-stage HCC.
Data availability
This cohort’s raw data are available via the following digital object identifier: [https://www.dropbox.com/scl/fi/wffcuxk7zc79pt9vn22ub/conditional-survival-RFA.xlsx?rlkey=ylf00pc2heuxubbseb5zks239%26dl=0].
References
Marrero, J. A. et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the study of liver diseases. Hepatology 68, 723–750. https://doi.org/10.1002/hep.29913 (2018).
European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 56, 908–943. https://doi.org/10.1016/j.jhep.2011.12.001 (2012).
Reig, M. et al. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J. Hepatol. 76, 681–693. https://doi.org/10.1016/j.jhep.2021.11.018 (2022).
Dikken, J. L. et al. Conditional probability of survival nomogram for 1-, 2-, and 3-year survivors after an R0 resection for gastric cancer. Ann. Surg. Oncol. 20, 1623–1630. https://doi.org/10.1245/s10434-012-2723-6 (2013).
Kim, Y. et al. Conditional survival after surgical resection of gastric cancer: a multi-institutional analysis of the US gastric cancer collaborative. Ann. Surg. Oncol. 22, 557–564. https://doi.org/10.1245/s10434-014-4116-5 (2015).
Henson, D. E. & Ries, L. A. On the estimation of survival. Semin. Surg. Oncol. 10, 2–6. https://doi.org/10.1002/ssu.2980100103 (1994).
Facciorusso, A. et al. Conditional survival analysis of hepatocellular carcinoma patients treated with radiofrequency ablation. Hepatol. Res. 45, E62-72. https://doi.org/10.1111/hepr.12458 (2015).
Cucchetti, A. et al. Conditional survival after hepatic resection for hepatocellular carcinoma in cirrhotic patients. Clin. Cancer Res. 18, 4397–4405. https://doi.org/10.1158/1078-0432.CCR-11-2663 (2012).
Shah, M. M. et al. Conditional survival analysis of hepatocellular carcinoma. J. Surg. Oncol. 122, 684–690. https://doi.org/10.1002/jso.26049 (2020).
Elfadaly, A. N. et al. Impact of tumor burden score on conditional survival after curative-intent resection for hepatocellular carcinoma: a multi-institutional analysis. World J. Surg. 45, 3438–3448. https://doi.org/10.1007/s00268-021-06265-3 (2021).
European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J. Hepatol. 69, 406–460. https://doi.org/10.1016/j.jhep.2018.03.024 (2018).
Llovet, J. M., Bru, C. & Bruix, J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin. Liver Dis. 19, 329–338. https://doi.org/10.1055/s-2007-1007122 (1999).
Nguyen, N. et al. Percutaneous radiofrequency ablation for hepatocellular carcinoma developed on non-alcoholic fatty liver disease. Liver Int. 42, 905–917. https://doi.org/10.1111/liv.15129 (2022).
Tang, A. et al. Evidence supporting LI-RADS major features for CT- and MR imaging–based diagnosis of hepatocellular carcinoma: a systematic review. Radiology 286, 29–48. https://doi.org/10.1148/radiol.2017170554 (2018).
Citterio, D. et al. Hierarchic interaction of factors associated with liver decompensation after resection for hepatocellular carcinoma. JAMA Surg. 15, 846–853. https://doi.org/10.1001/jamasurg.2016.1121 (2016).
Yen, Y. H. et al. Overall survival was inferior in octogenarians with early-stage hepatocellular carcinoma undergoing percutaneous radiofrequency ablation. Am. J. Med. Sci. S0002–9629(25), 01063–01068. https://doi.org/10.1016/j.amjms.2025.05.006 (2025).
Cohen, J. Statistical power analysis for the behavioral sciences. Routledge https://doi.org/10.4324/9780203771587 (2013).
Chen, Y. J. et al. Chromosomal changes and clonality relationship between primary and recurrent hepatocellular carcinoma. Gastroenterology 119, 431–440. https://doi.org/10.1053/gast.2000.9373 (2000).
Finkelstein, S. D. et al. Microdissection-based allelotyping discriminates de novo tumor from intrahepatic spread in hepatocellular carcinoma. Hepatology 37, 871–879. https://doi.org/10.1053/jhep.2003.50134 (2003).
Ng, I. O., Guan, X. Y., Poon, R. T., Fan, S. T. & Lee, J. M. Determination of the molecular relationship between multiple tumour nodules in hepatocellular carcinoma differentiates multicentric origin from intrahepatic metastasis. J. Pathol. 199, 345–353. https://doi.org/10.1002/path.1287 (2003).
Cucchetti, A. et al. Comparison of recurrence of hepatocellular carcinoma after resection in patients with cirrhosis to its occurrence in a surveilled cirrhotic population. Ann. Surg. Oncol. 16, 413–422. https://doi.org/10.1245/s10434-008-0232-4 (2009).
United Network for Organ Sharing Policy 3.6.4.4. http://optn.transplant.hrsa.gov/. (2010).
Edmondson, H. A. & Steiner, P. E. Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 7, 462–503. https://doi.org/10.1002/1097-0142(195405)7:3%3c462::aid-cncr2820070308%3e3.0.co;2-e (1954).
Capuano, G. et al. A new prognostic system for hepatocellular carcinoma: a retrospective study of 435 patients: the Cancer of the Liver Italian Program (CLIP) investigators. Hepatology 28, 751–755. https://doi.org/10.1002/hep.510280322 (1998).
Yang, Y. et al. Early recurrence after radiofrequency ablation for hepatocellular carcinoma: a multicenter retrospective study on definition, patterns and risk factors. Int. J. Hyperthermia. 38, 437–446. https://doi.org/10.1080/02656736.2020.1849828 (2021).
Xin, Y. et al. A Systemic inflammation response index (SIRI)-based nomogram for predicting the recurrence of early stage hepatocellular carcinoma after radiofrequency ablation. Cardiovasc. Intervent. Radiol. 45, 43–53. https://doi.org/10.1007/s00270-021-02965-4 (2022).
Cha, D. I. et al. Risk group stratification for recurrence-free survival and early tumor recurrence after radiofrequency ablation for hepatocellular carcinoma. Cancers (Basel) 15, 687. https://doi.org/10.3390/cancers15030687 (2023).
Ni, Z. et al. Prediction model and nomogram of early recurrence of hepatocellular carcinoma after radiofrequency ablation based on logistic regression analysis. Ultrasound Med. Biol. 48, 1733–1744. https://doi.org/10.1016/j.ultrasmedbio.2022.04.217 (2022).
Acknowledgements
The authors thank the Cancer Center at Kaohsiung Chang Gung Memorial Hospital for providing the HCC registry data. The authors also thank Chih-Yun Lin, Nien-Tzu Hsu, and the Biostatistics Center at Kaohsiung Chang Gung Memorial Hospital for their assistance with the statistical analysis.
Funding
This study was supported by Grant CMRPG8N1131 from the Chang Gung Memorial Hospital-Kaohsiung Medical Center, Taiwan.
Author information
Authors and Affiliations
Contributions
Study conception and design: YHY Acquisition of data: all authors Analysis and interpretation of data: SHM, CYL Drafting of manuscript: YHY.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The Institutional Review Board of Kaohsiung Chang Gung Memorial Hospital approved this study (Reference number: 202201189B0) and waived the need for informed consent. The research was conducted in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yen, YH., Moi, S., Kee, KM. et al. Conditional survival after radiofrequency ablation for early-stage hepatocellular carcinoma. Sci Rep 16, 7461 (2026). https://doi.org/10.1038/s41598-026-38771-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38771-6