Abstract
The rapid and reliable detection of ultraviolet (UV) radiation is critical for applications ranging from environmental monitoring to optoelectronic security systems. This study presents an integrated theoretical and experimental investigation into highly sensitive, hierarchically structured Si-based UV sensor–photodetectors optimized via ZnO–Al2O3 nanocomposite architectures. A combination of density functional theory (B3LYP/6-31G(d,p)) calculations and comprehensive materials characterization was employed to elucidate the interplay between electronic structure, surface morphology, and optical performance. Theoretical modeling provided detailed insights into band alignment, total and partial density of states, frontier molecular orbitals, and electrostatic potential distributions for pure and hybrid oxide systems, revealing that ZnO–Al2O3 exhibits superior electronic coupling and enhanced carrier mobility pathways. Experimentally, ZnO and Al2O3 nanoparticles were synthesized via hydrothermal routes, integrated into hybrid thin-film architectures on Si substrates, and structurally verified by XRD, FE-SEM, and EDX analyses. Surface roughness and apparent porosity measurements indicated that Al2O3 incorporation increased roughness from 6.7 to 8.2 µm and porosity from 26 to 36%, fostering enhanced light scattering and active site density. Optical absorption spectroscopy (250–650 nm) revealed strong UV selectivity with calculated band gaps of 3.18 eV (ZnO), 3.11 eV (Al2O3), and 3.26 eV (ZnO–Al2O3), while electrochemical impedance spectroscopy confirmed reduced charge transfer resistance in the hybrid architecture. Electrical conductivity improved from 27.7 × 10−2 S/m (ZnO) to 44.5 × 10−2 S/m (ZnO–Al2O3), correlating with faster response and recovery dynamics under UV illumination. These synergistic structural, optical, and electronic enhancements establish ZnO–Al2O3 as a promising candidate for next-generation, high-performance UV photodetectors with superior sensitivity, stability, and spectral selectivity.
Similar content being viewed by others
Introduction
Ultraviolet (UV) radiation detection has emerged as a critical technological requirement across diverse fields such as environmental monitoring, public health protection, industrial quality control, and aerospace applications1. For instance, UV-A is often used in non-destructive testing and medical phototherapy, UV-B plays a central role in biological effects such as vitamin D synthesis and skin erythema, and UV-C is integral to germicidal disinfection and semiconductor photolithography. Precise and reliable detection of UV radiation is thus essential for safeguarding human health, ensuring process integrity, and enabling advanced scientific instrumentation. Silicon (Si)-based photodetectors are attractive platforms for UV sensing due to their established fabrication technology, scalability, and compatibility with complementary metal–oxide–semiconductor (CMOS) processes2. However, conventional Si photodiodes suffer from inherent challenges when detecting UV radiation. Their narrow bandgap (1.12 eV) allows for strong absorption in the visible and near-infrared regions, which introduces unwanted background signals, thereby limiting UV selectivity. Furthermore, the shallow penetration depth of UV photons in Si leads to increased surface recombination losses, lowering device responsivity3. Overcoming these challenges requires integrating wide-bandgap semiconductors and engineered nanostructured coatings that selectively absorb UV light, improve charge carrier separation, and suppress visible–infrared background interference1.
Recent advances in UV photodetectors have demonstrated the remarkable potential of wide-bandgap metal oxide nanostructures in enhancing key performance metrics such as responsivity, response speed, and environmental stability4. For instance, ZnO nanotubes grown on p-Si substrates via pulsed laser deposition have achieved exceptionally high UV responsivity (~ 101.2 A W−1), attributed to their excellent structural quality, strong optical confinement, and high surface-to-volume ratios, as reported by Flemban et al.5. Similarly, Akhtarianfar et al.6 demonstrated that vertically aligned ZnO nanorod ensembles, organized through hierarchical colloidal network assembly, can significantly enhance UV sensor sensitivity through precise control of deposition parameters such as precursor concentration, layer thickness, and nanorod morphology. In hybrid systems, Pandey et al.7 showed that coating ZnO nanorods with TiO2 layers leads to a substantial enhancement in photocurrent—from ~ 35 to ~ 250 μA under UV illumination—while enabling self-powered operation, as validated by both experimental measurements and first-principles theoretical modeling. Beyond ZnO–TiO2 composites, complex heterojunction architectures such as NiO/TiO2/ZnO devices have demonstrated exceptional performance, with responsivity values reaching 291 A W−1 and detectivity up to 6.9 × 1011 Jones, enabled by an ultrathin TiO2 dielectric layer that improves rectification and suppresses dark current, as reported by Shang et al.8. Collectively, these studies underscore the transformative role of nanostructuring, hybridization, and interface engineering in optimizing UV sensing performance, while also revealing that systematic, side-by-side theoretical and experimental comparisons of different oxide systems remain scarce—a critical knowledge gap addressed in this work. Despite these advances, most prior studies focus on isolated material systems or single heterojunction configurations, without providing a unified comparison under identical fabrication and testing conditions or correlating nanoscale interfacial chemistry with device-level photodetection metrics. Recent advances in nanocomposite engineering further highlight that controlled defect modulation, interfacial coupling, and nanoscale structural optimization can significantly enhance charge transport, surface reactivity, and optoelectronic functionality across sensing and energy-related applications9,10,11,12. In particular, hybrid oxide and carbon-based nanocomposites have demonstrated improved sensitivity, operational stability, and functional selectivity through tailored electronic interactions and reduced recombination losses, underscoring the growing importance of rational nanocomposite design in next-generation semiconductor devices13,14.
In recent years, semiconductor nanometal oxides such as zinc oxide (ZnO)15,16, titanium dioxide (TiO2)17, and aluminum oxide (Al2O3)18, have attracted intense interest for UV sensing applications. ZnO, with its wide direct bandgap (~ 3.37 eV) and high exciton binding energy (~ 60 meV), offers strong intrinsic UV absorption, high electron mobility, and tunable morphology from nanorods to thin films. TiO2, with a bandgap of 3.0–3.2 eV, is known for its exceptional chemical stability, photocorrosion resistance, and environmental robustness, making it suitable for prolonged outdoor UV sensing. Al2O3, although not a direct UV absorber, serves as an excellent passivation layer with a high dielectric constant, capable of reducing surface trap states, suppressing dark currents, and improving device longevity. Hybrid and composite oxide architectures, particularly ZnO–Al2O3 nanocomposites, have demonstrated synergistic performance advantages4. ZnO provides strong UV sensitivity, while Al2O3 acts as a surface passivation layer, blocking non-radiative recombination pathways and reducing leakage currents. In this context, the selection of synthesis parameters (reaction time, composition ratio, and processing conditions) in the present work was guided by preliminary optimization studies aimed at achieving a balance between crystallinity, surface roughness, and electrical conductivity. Systematic variation of these parameters allowed identification of conditions that maximize UV photoresponse while maintaining structural stability and reproducibility. This dual functionality improves photogenerated carrier lifetimes, enhances the signal-to-noise ratio, and enables fast response–recovery characteristics. The introduction of Al2O3 into ZnO matrices also modifies the electronic band alignment, potentially reducing trap-mediated recombination and facilitating efficient charge extraction when interfaced with Si substrates. Notwithstanding these benefits, literature on the systematic integration of ZnO–Al2O3 nanocomposites into Si-based UV sensors remains limited, with most prior works focusing either on ZnO alone or on Al2O3 as a passivation layer rather than as an active component in composite sensing architectures19. The research gap lies in the lack of comprehensive, side-by-side evaluation of multiple oxide and hybrid configurations—namely ZnO, TiO2, Al2O3, ZnO–TiO2, and ZnO–Al2O3—under identical fabrication and testing conditions17,20. Furthermore, there is a need for integrated theoretical–experimental studies that link materials properties such as bandgap, surface passivation, and defect density to device-level metrics including responsivity, response time, and operational stability. Previous reports often lack mechanistic insight into how oxide–oxide and oxide–Si interfaces affect charge transport and recombination kinetics in UV photodetectors21. Accordingly, the key novelty of this study lies in the combined experimental–theoretical evaluation of multiple oxide and hybrid architectures under identical conditions, with particular emphasis on elucidating how ZnO–Al2O3 interfacial coupling governs charge transport, defect passivation, and UV photodetection performance in Si-based devices.
In the present work, these research gaps are addressed through an integrated theoretical and experimental investigation of Si-based UV sensors incorporating various semiconductor oxide layers, with a particular emphasis on ZnO–Al2O3 nanocomposite architectures. The theoretical component employs density functional theory (DFT) calculations using the B3LYP/6-31(d,p) model to predict optical absorption spectra, band alignment, and carrier transport pathways. The prospective benefits of multiple semiconductor nanometal oxides—including ZnO, TiO2, Al2O3, and their hybrid forms ZnO–TiO2 and ZnO–Al2O3—are systematically evaluated to determine the most practical and efficient material configurations for Si-based UV sensor development. The analysis focuses on tracking variations in key electrical properties such as bandgap energy, total density of states (TDOS), and frontier molecular orbitals (FMOs), specifically the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). Additionally, molecular electrostatic potential (MESP) mapping and partial density of states (PDOS) calculations are performed to assess the stability and reactivity of each structure. Theoretical predictions of optical enhancement are further validated by simulating UV–Vis spectra, enabling the identification of the most promising nanocomposite configuration. Subsequently, experimental fabrication and characterization are conducted to corroborate the theoretical findings. The fabricated devices are analyzed using X-ray diffraction (XRD), field-emission scanning electron microscopy (FE-SEM), energy-dispersive X-ray spectroscopy (EDX), UV–Vis spectroscopy, electrical conductivity measurements, surface roughness analysis, apparent porosity determination, and electrochemical impedance spectroscopy (EIS). Comparative evaluations of single-component oxides (ZnO, TiO2, Al2O3) and hybrid composites (ZnO–TiO2) confirm that ZnO–Al2O3 delivers superior device performance, combining high responsivity, fast recovery times, and enhanced environmental stability.
The significance of this work lies in its ability to provide a direct link between material-level modifications and device-level performance improvements, enabling rational design principles for future UV sensor technologies. By demonstrating how the ZnO–Al2O3 combination enhances Si-based photodetection, this study offers a scalable pathway for the development of high-efficiency, low-cost UV sensors compatible with CMOS manufacturing processes. Such devices have promising applications in wearable UV monitoring, spaceborne radiation detection, flame sensing, and integrated environmental sensing networks. Looking forward, the design strategies established here can be extended to next-generation UV sensors employing flexible substrates, transparent conductive electrodes, and advanced nanostructured coatings to achieve broadband, multi-spectral UV detection. Integration with wireless IoT architectures and self-powered operation through photovoltaic or triboelectric energy harvesting are also prospective developments. Furthermore, doping strategies, plasmonic nanostructure incorporation, and defect engineering offer exciting possibilities for further enhancing UV selectivity and responsivity, opening pathways toward compact, high-performance, and intelligent UV sensing systems for the evolving demands of industry, defense, and environmental monitoring.
Calculation details
The simulations were performed using Gaussian 09 program Revision C.0122, using density function theory (DFT)23. The prospective benefits of several a semiconductor nanometal oxides, including ZnO, TiO2, Al2O3, and the hybrids ZnO–TiO2 and ZnO–Al2O3, have been explored to determine practical selections for the development of Si-based UV sensors utilizing the B3LYP/6-31(d,p) model24,25,26. In order to track the alternation on electrical properties, the effects of nanometal oxides on bandgap, total density of states (TDOS), and frontier molecular orbitals (FMOs), which stand for the highest occupied (HOMO) and lowest unoccupied (LUMO) molecular orbitals, were studied for all suggested structures. After that, in order to examine the impact of nanometal oxides on stability and reactivity, several essential physical characteristics were examined, including MESP mapping and partial density of states (PDOS). On top of that, the optical enhancement could be tracked, and the most enhanced structure might be identified by subjecting all of the proposed structures with regard to UV–Vis characterization. Furthermore, the same theoretical level was used to calculate IR and NMR spectra in order to determine the structure that were present in the molecule, this allowed to identify the best material possible. This is due to the fact that IR and NMR are essential instruments for characterizing chemical processes by determining the products and reactants involved.
Experimental section
Reagents and materials
All chemicals used in this work were of analytical grade and utilized without further purification. Zinc nitrate hexahydrate (Zn(NO3)2·6H2O, ≥ 99%, Sigma–Aldrich) and sodium hydroxide (NaOH, ≥ 98%, Merck) served as the primary precursors for ZnO NP synthesis. For Al2O3 NP preparation, commercial aluminum powder (> 5 µm, 99.5%, Alfa Aesar) and NaOH were used as starting materials. Hydrochloric acid (HCl, 37%, Sigma–Aldrich) was employed for neutralization. Single-side-polished p-type Si wafers (orientation ⟨100⟩, resistivity 1–10 Ω cm, thickness 500 ± 25 µm) were used as the substrate material for device fabrication.
Synthesis of ZnO NPs
ZnO NPs were prepared via a hydrothermal precipitation route. First, 3.00 g of Zn(NO3)2·6H2O was dissolved in 20 mL of DIW under constant magnetic stirring at 900 rpm until a clear solution was obtained. In a separate beaker, 4.00 g of NaOH was dissolved in another 20 mL of DIW. The NaOH solution was then added dropwise to the zinc nitrate solution under vigorous stirring to initiate precipitation. The resulting suspension was transferred into a 50 mL Teflon-lined stainless-steel autoclave and heated at 75 °C for 8 h. Formation of an opaque white suspension indicated the successful generation of ZnO NPs. After cooling to room temperature naturally, the precipitate was collected via vacuum filtration, washed repeatedly with DIW and ethanol to remove residual ions, and dried in a vacuum oven at 65 °C for 24 h. The dried product was lightly ground using an agate mortar and pestle and stored in an airtight container for further use.
Synthesis of Al2O3 NPs
Al2O3 NPs were synthesized through a hydrothermal oxidation process. A 2.0 M NaOH solution was prepared by dissolving 9.60 g of NaOH in 120 mL of DIW. To this alkaline medium, 2.00 g of commercial aluminum powder was gradually added under vigorous stirring to minimize localized overheating from the exothermic reaction. The mixture was stirred for 45 min at ambient temperature, followed by ultrasonication (150 W, 40 kHz) for 2 h to ensure homogeneous dispersion of the aluminum particles. The resulting suspension was transferred into a 200 mL Teflon-lined stainless-steel autoclave and heated at 150 °C for 36 h. After cooling to room temperature, the reaction mixture was neutralized with dilute HCl until the pH reached ~ 7. The precipitated Al2O3 NPs were collected via centrifugation at 1000 rpm for 10 min, washed three times with DIW and ethanol, and dried in an oven at 75 °C for 15 h. Finally, the dried powder was calcined in a muffle furnace at 550 °C for 3 h to enhance crystallinity and phase purity.
Fabrication of Si-based ZnO–Al2O3 nanocomposite films
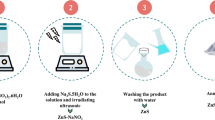
The ZnO@ Al2O3 nanocomposite was prepared by incorporating synthesized Al2O3 NPs into a ZnO matrix at an optimized mass ratio of 80:20, as seen in Fig. 1. Initially, 3.00 g of ZnO NPs was dispersed in 150 mL of DIW and stirred magnetically at 80 °C for 3 h to obtain a stable suspension. Separately, Al2O3 NPs corresponding to 1 wt% of the total solid mass were dispersed in 50 mL of DIW via ultrasonication for 30 min. The Al2O3 dispersion was then added dropwise to the ZnO suspension under continuous stirring, and the mixture was maintained at 85 °C for an additional 2.5 h to achieve homogeneous nanoparticle distribution. The composite slurry was deposited onto pre-cleaned Si wafers using a spin-coating process at 1000 rpm for 120 s. Each substrate was coated three times to ensure uniform film thickness, with intermediate drying at 60 °C for 10 min between coatings. The coated wafers were dried at ambient temperature for 24 h, followed by curing in a hot-air oven at 70 °C for 12 h to improve adhesion and mechanical stability. The resulting ZnO–Al2O3 composite films were cut into uniform dimensions (1 cm × 1 cm) and stored in a desiccator until characterization.
Schematic illustration of the stepwise synthesis and integration of ZnO–Al2O3 nanocomposites for Si-based UV sensor–photodetector fabrication.
Characterization techniques
A comprehensive suite of advanced analytical and electrochemical techniques was employed to elucidate the structural, morphological, compositional, surface, and electrical properties of the synthesized ZnO and ZnO–Al2O3 nanocomposite films. X-ray diffraction (XRD, Bruker D8 Advance, Cu Kα) was employed to evaluate the crystalline structure, phase composition, and crystallinity of ZnO, Al2O3, and ZnO–Al2O3 nanocomposite films. Variations in peak intensity and full-width at half-maximum (FWHM) provided insights into microstructural evolution induced by plasma treatment. Fourier-transform infrared spectroscopy (FTIR, Nicolet iS50, Thermo Scientific) was used to identify functional group modifications. Field Emission Scanning Electron Microscopy (FE-SEM) was conducted using an FEI Nova NanoSEM 450 system operating at an accelerating voltage of 5–15 kV to investigate surface morphology and microstructural organization. Prior to imaging, all samples were sputter-coated with a thin layer (~ 5 nm) of gold using a Quorum Q150R ES coater to minimize charging effects. High-resolution micrographs were acquired at multiple magnifications to assess nanoparticle distribution, surface uniformity, porosity features, and interfacial contact between the nanocomposite layer and the Si substrate. High-resolution Transmission electron microscopy (HR-TEM, JEOL, JEM-A 2100, Japan) was employed to gain deeper insight into the nanoscale morphology, crystallinity, and interfacial characteristics of the pristine ZnO and ZnO–Al2O3 films. Energy-Dispersive X-ray Spectroscopy (EDX) was carried out on a QUANTA 200 FEG (FEI, Japan) instrument coupled to the FE-SEM system, enabling qualitative and semi-quantitative determination of elemental composition. Elemental mapping was performed over selected areas to visualize the spatial distribution of Zn, Al, and O, thus confirming the successful incorporation and homogeneous dispersion of Al2O3 within the ZnO matrix. Data were processed using EDAX Genesis software to ensure accurate peak identification and composition quantification. Surface Roughness Analysis was performed using a Talysurf 50 contact profilometer (Taylor Hobson Precision, UK) equipped with a diamond stylus of 2 μm tip radius. Measurements were carried out over a scan length of 5 mm at a stylus speed of 0.5 mm/s, with multiple scans obtained from different sample regions to ensure statistical reproducibility. The average surface roughness (Ra) parameter was extracted, providing quantitative insights into surface texture changes induced by nanoparticle embedding and hierarchical structuring. Apparent Porosity was determined following the Archimedes principle in strict accordance with ASTM C373-88. Briefly, each sample was first dried in an oven at 105 °C for 24 h to obtain the dry mass. Samples were then immersed in deionized water under vacuum for 2 h to ensure complete pore filling and weighed in the saturated state. Finally, suspended weight in water was measured. This method enabled precise quantification of open pore volume and water-accessible voids within the nanocomposite films. Electrical Resistivity was evaluated using the four-point probe (4PP) method with an EQ-JX2008-LD instrument (MTI Corporation, USA). A constant DC current was applied between the outer probes, while the voltage drop was recorded across the inner probes. Measurements were performed at ambient conditions, with probe spacing calibrated at 1.0 mm. To minimize statistical error, resistivity values were averaged over ten independent readings taken at spatially distributed locations across each specimen surface. The resulting resistivity data were used to assess charge transport efficiency and the impact of Al2O3 incorporation on electrical conduction pathways. Electrochemical Impedance Spectroscopy (EIS) measurements were performed under dark conditions using a Zahner electrochemical workstation to evaluate the dynamic charge transport and interfacial properties of the fabricated Si-based UV sensor–photodetectors. The impedance spectra were recorded over a frequency range of 100 Hz to 100 kHz with an applied AC perturbation amplitude of 10 mV. This technique enabled precise determination of parameters such as charge transfer resistance (Rct), series resistance (Rs), and capacitive behavior at the semiconductor–electrode interface. All EIS data were analyzed through complex-plane (Nyquist) and frequency-phase (Bode) plots, with equivalent circuit fitting conducted to quantitatively correlate structural modifications—particularly the incorporation of ZnO–Al2O3 nanocomposite architectures—to the observed electrical performance enhancements. Current–voltage (I–V) measurements of pure ZnO and ZnO–Al2O3 photodetectors were conducted over a voltage range of − 5 to + 5 V under both dark and UV illumination using a KEITHLEY 2400 Source Meter coupled with a MSK–SS-50 solar simulator. Each measurement was averaged over five independent readings per sample. Both devices exhibit nearly linear and symmetric I–V behavior in forward and reverse bias, indicating the formation of ohmic contacts between the semiconductor layer and metal electrodes.
Results and discussion
DFT study of Si-based with metal oxides
Building model molecule
Devices designed to detect and quantify levels of UV light are commonly referred to UV sensors. These sensors encompass photochromic sensors and photo detectors. Photochromic sensors exhibit limitations in their ability to detect weak light and demonstrate a low direct response intensity. Conversely, UV photodetectors play a crucial role in contemporary optoelectronic technologies; they convert UV light wavelengths into electrical signals27. Environmental monitoring, sterilization, water purification, and biology have significant applications for UV photodetection, including notable uses in space detection27. Si-based UV photodiodes and UV photomultiplier tubes have traditionally been the principal types of UV photodetectors used in industrial settings. Si-based UV photodetectors have garnered a lot of attention due to their amazing sensitivity and adaptability28. This is despite the fact that Si-based UV photodiodes have a wide response that encompasses wavelengths ranging from UV to near-infrared. However, their UV photodetection capabilities are limited due to their insufficient sensitivity in the UV wavelength spectrum and the integration of optical29. The use of wide bandgap semiconductors as prospective alternatives for the construction of filter less UV photodetectors is chosen due to the fact that these semiconductors have an appropriate cut-off wavelength within the ultraviolet spectrum, which enables them to circumvent these constraints. This category comprises the following nanometal oxide materials: zinc oxide (ZnO), nickel oxide (NiO), titanium oxide (TiO2), copper oxide (CuO), tin oxide (SnO2), iron oxide (Fe2O3), Aluminum oxide (Al2O3), indium oxide (In2O3), tungsten trioxide (WO3), and vanadium oxide30,31,32,33. Si-based UV sensors that are simulated and their functionalization with a variety of nanometal oxides are considered to be the most promising alternatives (see Fig. 2) because of their remarkable electrical, optical, biological, energy, and processing capabilities. DFT calculations were employed to investigate the influence of various nanometal oxides, including ZnO, TiO2, Al2O3, ZnO–TiO2, and ZnO–Al2O3, on enhancing the electrical and optical properties of Si-based sensors for targeted improvements. The HOMO and LUMO orbitals, along with their respective energies, TDM, band gap energy, and TDOS were calculated to assess the suitability of the doping created for necessary properties. Subsequently, the impact on PDOS and MESP maps was analyzed to assess improvements in reactivity and sensitivity, in addition to calculating key physical parameters including hardness, softness, and electrophilicity. Furthermore, all models underwent UV–Vis analysis to identify the optical improvement and determine the most effective nanometal oxide. As a result, the optimal compound was synthesized and analyzed to examine the impact of implementing on the structural, physical and chemical characteristics.
Model structures constructed from the interaction mechanism of Si-based with various metal oxides including (ZnO, Al2O3, TiO2, ZnO-Al2O3, and Zn-TiO2) generated using Gauss View 5.0 program (Gaussian 09 program Revision C.01).
Frontier molecular orbital (FMO)
The Frontier Molecular Orbital (FMO) idea was examined to figure out and expect molecular sensitivity and reactivity, as well as charge transfer, which influence electronic characteristics. This hypothesis emphasizes the dispersion of HOMO and LUMO orbitals. In chemical processes, the distributions and energies of the HOMO and LUMO orbitals are crucial due to their determination of interactions on the electronic characteristics of molecules. FMO theory facilitates the prediction of the most probable sites for chemical reactions by examining the relative positions of HOMO and LUMO orbitals34. A computational and analytical investigation of the HOMO and LUMO molecular orbitals was carried out in order to study the interaction between metal oxides and the Si-base, as seen in Fig. 3. The presence of metal oxides influenced the configuration of HOMO and LUMO orbitals. It is clear from the HOMO and LUMO orbitals of the Si-base that the HOMO and LUMO orbitals are spread uniformly over the Si sheet. This is something that can be easily observed, the HOMO and LUMO orbitals appear to be concentrated around the ZnO atoms as a result of the interaction between ZnO and Si-base, which caused a significant alteration in the distribution of these pairs of orbitals. The impact of TiO2 has no notable alteration in the distribution of both HOMO and LUMO orbitals, as the orbitals continue to be predominantly localized on the silicon base, with only minor participation observed with the –O– atom of TiO2. Under the influence of Al2O3, the HOMO orbitals remain unaffected, while the LUMO orbitals are rearranged within and around the Al2O3. Ultimately, the HOMO and LUMO orbitals surrounding the atoms of the metal oxides were altered as a result of the hybridization of ZnO with TiO2 and Al2O3.


Frontier molecular orbital of the interacted Si-based with various metal oxides including (ZnO, Al2O3, TiO2, Zn–TiO2 and ZnO–Al2O3) generated using (Gaussian 09 program Revision C.01).
Among the electronic parameters that determine the reactivity, stability, and general behavior of a molecule are its band gap ΔE, TDM, and the EHOMO and ELUMO orbitals. The ability to recognize and predict these properties also gives vital information about the behavior of a molecule as well as potential uses for the molecule. The results of these various parameter computations are shown in Table 1.
Comprehending chemical reactivity is fundamentally dependent on EHOMO and ELUMO, which represent the energy levels associated with electron transfer in chemical reactions. The elevated energy level and electron configuration of EHOMO are typically associated with superior nucleophiles. This feature is frequently linked to the molecule’s electron-donating properties. The most effective electrophile is generally linked to a chemical characterized by a low electron affinity minimum (ELUMO) value. However, the ELUMO showed a significant increase, which indicates that the novel structures possess increased nucleophilic properties. This was discovered by the examination of nanometal oxides, which demonstrated that all of the model structures had a little increase in EHOMO level. One of the primary functions of the ΔE, which is defined in Eq. (1), is to serve as a measurement for electron transport, chemical equilibrium, and sensitivity. This discrepancy between EHOMO and ELUMO is referred to as the difference between both.
The presence of a narrower band gap is indicative of higher reactivity in a molecule. This is due to the fact that increasing the energy required for electrons to travel through orbitals is reduced. There was a discernible shortening of the Si-base bandgap that was found according to the nanometal oxides doping. The Si–ZnO–Al2O3 composite had the greatest performance of all the nanometal oxide composites because it had a narrow bandgap of 0.5040 eV more than any other composite. As a consequence of this, as seen in Eq. (2)35, the conductivity (σ) of the structure will experience a significant increase, notably for the Si–ZnO–Al2O3 composite, which is dependent on the ΔE.
where (T) denotes temperature (Kelvin), (A) signifies a constant, and (KB) represents the Boltzmann constant. As an indication of the polarity of the molecule, TDM provides a visual representation of the distribution of the two types of charges inside the molecule. An increase in charge partitioning, which is demonstrated by a higher TDM, is another indication that the material demonstrates a greater degree of polarity. When it comes to TDM, the hybrid composites that were examined showed a substantial increase, with the Si–ZnO–Al2O3 structure demonstrating the most significant improvement at 20.9008 Debay. ZnO–Al2O3 has a considerable influence on dipole–dipole interactions, as evidenced by a decrease in the bandgap and a rise in TDM. This has the effect of improving the material’s electrical properties as well as its sensing and adsorption capabilities.
Total density of states (TDOS) analyses
In light of the aforementioned structures, the TDOS investigation shows the number of allowable states per every single energy unit. The TDOS is a measure used in condensed matter theory to describe the ordering of electronic states inside a material with respect to energy36. It is a measure that considers all contributions and provides a more comprehensive view of the electronic structure. The structures examined for Si-base sensors functionalized with ZnO, TiO2, Al2O3, ZnO–TiO2, and ZnO–Al2O3 are shown in Fig. 4, which indicates the TDOS. As a result of the presence of ZnO, the HOMO levels pushed closer to the Fermi level, which indicates that there are strong molecular interactions with Si-base. This variation also indicates that there is a greater potential of transmission of electrons, as Si–ZnO exhibited a substantial electronic transformation as a result of the influence of ZnO NPs. In a similar manner, TiO2 and Al2O3 offer a greater shift for the HOMO and LUMO levels, bringing them nearer to the Fermi level20. This results in a reduction in the band gap and an increase in probability of electron transitions and conductivity. In contrast to pure Si–ZnO, the hybrid composites of Si–ZnO–TiO2 and Si–ZnO–Al2O3 exhibit a more significant relocation of electrons owing to enhanced support for molecular orbitals. Comprehending the molecular reactivity, conductivity, and enhanced stability of the composite resulting from hybridization with Al2O3 necessitates this allocation, consistent with prior studies on HOMO–LUMO orbitals and band gap energy. Composites that integrate the advantages of Si–ZnO with metal oxides, particularly Al2O3, enhance conductivity, durability, and UV light sensitivity. The combination of several nanostructured materials can yield innovative solutions to challenges faced in particular application, necessitating a multidisciplinary approach to material design.
Total density of states of the interacted Si-based with various metal oxides including (ZnO, Al2O3, TiO2, Zn–TiO2 and ZnO–Al2O3).
Partial density of states (PDOS) analyses
On the PDOS Assists in the comprehension of concepts like as bonding, hybridization, and other electronic interactions by presenting a comprehensive perspective that illustrates the functions of certain atomic orbitals or components. PDOS provides a comprehensive perspective by allowing for the viewing of the contributions that certain atomic orbitals including s, p, and d orbitals make. With the assistance of PDOS, one is able to gain a deeper comprehension of the nuclear orbitals that play a significant role in the bonding and antibonding interactions. Using this strategy, it is much simpler to comprehend how the orbitals of various elements influence the electrical structure and characteristics of the material37. Figure 5 illustrates that the calculated PDOS for the Si-base reveals a uniform disparity in the intensities of the H-1s and Si-3p orbital configurations, with the intensity of the Si-3p orbital surpassing that of the H-1s. The PDOS for Si-base was examined, revealing that H-1s and Si-3p contribute similarly to the HOMO levels. The presence of metal oxides (ZnO, TiO2, and Al2O3) induces significant orbital interaction between the Si-3p and O-2p orbitals, evidenced by their overlap, which results in a modified configuration of Si and H atoms at the HOMO and LUMO levels. The Si-base is expected to provide a significant quantity of electrons to establish physical interactions with metal oxides37. Metal oxides (ZnO, TiO2, and Al2O3) have an effect that makes the PDOS peaks, which are spread out throughout the HOMO and LUMO levels from − 10 to 0, more intense. This occurrence exhibits the simultaneous bonding of the Si-3p with metal oxide electron orbitals and the reduction in energy due to metal oxide precipitation via a weak physical attachment. The Si–ZnO–TiO2 and Si–ZnO–Al2O3 combination compounds are responsible for this variation. In these composites, the orbitals of the metal oxides combine with those of the Si-3p, Zn-3d, Ti-3d, Al-3p, and O-2p levels. The overlapping shows that there is a change in the arrangement of the Si and H atoms across the HOMO and LUMO levels, and the more interactions between the Si-base and hybrid metal oxides, the more noticeable the difference.
Partial Density of States of the interacted Si-based with various metal oxides including (ZnO, Al2O3, TiO2, Zn–TiO2 and ZnO–Al2O3).
Molecule electrostatic potential (MESP)
The MEP contour maps are representations that illustrate the distribution of electrostatic potential surrounding a molecule. They provide the visualization of areas of positive and negative charge, which is crucial for figuring out chemical reactions and responses38. Through the use of the MEP, one possesses the ability to examine the achievable energy that is associated with a certain charge. This energy is calculated at various places around a molecule. The arrangement of the nuclei and electrons within the molecule is what determines this feature of the molecule. With MEP contours, the lines that link locations that have a comparable electrostatic potential are shown with same colour39. They pinpoint potential targets that might be attacked as electrophiles or nucleophiles. Red and orange on the MEP map indicate locations with a strong negative electrostatic potential (which is abundant in electrons), whereas yellow indicates areas with an extremely positive electrostatic potential (deficient in electrons)40. Figure 6 displays the contour MESP color maps for the examined structures: Si-base, Si–ZnO, Si–TiO2, Si–Al2O3, Si–ZnO–TiO2, and Si–ZnO–Al2O3. The colorful map of Si-base depicted the spread of negative potentials, with the red contour lines denoting regions around the Si sheet. The inside of the Si-base was shown in yellow, indicating minimal negativity. The influence of metal oxides exacerbates the detrimental consequences linked to both the metal oxides and the S-base. The observed observations revealed that the presence of metal oxides amplified negatively over the silicon substrate41. The functionalization with hybrid ZnO–TiO2 and ZnO–Al2O3 led to a substantial rise in negativity that extended beyond the Si-base and onto the metal oxide atoms. This resulted in a significant improvement in the transport of electrons, conductivity, and responsiveness.
Molecular electrostatic potential as contour map of the interacted Si-based with various metal oxides including (ZnO, Al2O3, TiO2, Zn–TiO2 and ZnO–Al2O3) generated using (Gaussian 09 program Revision C.01).
Reactivity and stability parameters for composites
Critical chemical reactivity criteria were delineated for each composite for assisting further examination and analysis of the reactivity features and longevity of composite materials. The utilization of these metrics may enhance the comprehension of structural stability, reactivity patterns, chemical manners, physicochemical alterations, and electrical features42. The fundamental attributes required to examine the stability and reactivity of compounds are based on the concepts of Mulliken and Koopmans42,43. The structures that were under inquiry were examined using the equations that are detailed below and tabulated in Table 2.
EHOMO and ELUMO were utilized to compute the ionization potential (IP) and electron affinity (EA), two variables that are significantly dependent on these parameters. Removal of electrons from a chemical structure result in the formation of free radicals, a phenomenon referred to as ionization potential. Electron affinity is the amount of energy necessary to attract electrons from another molecule, leading to the creation of negative ions that enhance nucleophilicity44.
The mobility of electrons in materials is demonstrated by electronegativity (χ). The capacity of an electron to transfer is denoted by its chemical potential (μ), whereas the ability of a molecule to receive electrons is indicated by its electronegativity. The inverse connection between chemical potential (χ) and electronegativity (χ) is articulated by the subsequent equation45:
When the electronegativity value of a material is higher, it implies that the material is more electronegative, which enhances the material’s capacity to absorb and react substances. Among the composites that were investigated, the composites with the highest electronegativity values were Si–ZnO and Si–ZnO–Al2O3, with respective values of 3.7338 and 4.1511 electronegativities. The band gap and the data from the MESP map both reveal that they have an extraordinary responsiveness; hence, this must be the real situation. The ability of a material to withstand electron-cloud dissociation or structural deformation is defined as chemical hardness (η). Furthermore, in the context of substance reactivity, softness (σ) is frequently seen as the inverse of hardness (η)46. This material is ideal for adsorption due to its exceptionally high softness and remarkably low hardness values, indicating a significant likelihood of contact. Among the many metal oxide composites evaluated, Si–ZnO–Al2O3 (3.9683) demonstrated the highest efficacy owing to its remarkable softness, stability, and reactivity for adsorption. The previously reported equations were employed to examine η, σ, and ΔNMax for each structure47:
Ultimately, the molecular reactivity of a material may be represented according to its electrophilicity (ω) and nucleophilicity (ε), two critical attributes that dictate its propensity to donate or accept electrons from other molecules in its environment48. In contrast to nucleophilicity (ε), which quantifies the tendency to donate or transfer electrons based on a specific chemical composition, electrophilicity (ω) quantifies a substance’s tendency to accept electrons49. Substances are categorized into three distinct classes according to their electrophilicity: low electrophiles, medium electrophiles, and high electrophiles50. The electrophilicity index (ω) was utilized to assess a substance’s capacity to accept electrons, as indicated by the formula that follows equation48:
In the electrophile range, the Si-base appears at the exact middle of the range. Composite materials, specifically Si–ZnO–Al2O3, display increased electrophilicity when they are combined with metal oxides, which results in substantially greater electrophilic characteristics because of the integration. It is clear from this that the ZnO–Al2O3 NPs improved the surface area’s responsiveness as well as its absorption capabilities. The Si–ZnO–Al2O3 compound was the one that exhibited the highest level of sensitivity. In terms of their electrical and optical characteristics, Si–ZnO–Al2O3 compounds are very reactive. This is demonstrated by data that pertain to band gap and MESP, which further highlight the influence that hybridization produces.
Experimental result of Si–ZnO–Al2O3 compound
XRD analysis
XRD analysis was conducted to elucidate the crystalline structure, phase stability, and crystallite evolution of the ZnO, α-Al2O3, and ZnO–Al2O3 nanocomposites developed for high-performance UV photodetector applications. The diffraction patterns were collected in the 2θ range of 10°–80°, as presented in Fig. 7a. The pristine ZnO NPs exhibit a well-defined hexagonal wurtzite structure, with diffraction peaks indexed to the (100), (002), (101), (102), (110), (103), (200), (112), (201), (004), and (202) planes at 2θ values of 31.7°, 34.3°, 36.1°, 47.4°, 56.5°, 62.8°, 66.3°, 67.7°, 68.9°, 72.5°, and 76.9°, respectively, in excellent agreement with JCPDS card No. 76-070451. The absence of secondary phases confirms the high crystallographic purity of the synthesized ZnO NPs. In parallel, α-Al2O3 NPs display characteristic reflections corresponding to the (012), (104), (110), (113), and (024) planes at 2θ values of 25.6°, 35.1°, 37.8°, 43.4°, and 54.6°, respectively (JCPDS card No. 46-1212). The presence of the thermodynamically stable α-phase verifies the successful formation and structural robustness of Al2O3 NPs within the composite framework18,52. Importantly, no additional diffraction peaks associated with secondary or impurity phases—such as ZnAl2O3 spinel or other mixed oxide phases—are observed within the detection limit of the XRD instrument. This confirms the high phase purity of the synthesized materials and indicates that Al2O3 incorporation does not induce undesirable phase transformation but rather results in a well-integrated ZnO–Al2O3 nanocomposite system.
(a) XRD patterns of pristine ZnO, pristine Al2O3, and ZnO–Al2O3 nanocomposite, confirming phase composition and structural integrity. (b) Experimental FTIR spectra of pristine ZnO and ZnO–Al2O3 nanocomposite, ighlighting surface functional groups and interfacial bonding. (c) Theoretical (DFT-calculated) infrared (IR) spectrum of the ZnO–Al2O3 nanocomposite, illustrating vibrational modes and validating the experimental FTIR features.
For the ZnO–Al2O3 nanocomposites, the diffraction peaks associated with ZnO remain at identical angular positions, indicating that the fundamental wurtzite lattice of ZnO is preserved upon Al2O3 incorporation. However, a moderate reduction in peak intensity accompanied by peak sharpening is observed, suggesting microstructural reorganization and crystallite growth rather than phase transformation or lattice substitution. Importantly, the absence of peak shifting further indicates that Al3+ ions do not substitute Zn2+ sites but are instead incorporated at grain boundaries or interfaces, acting as a structural passivation component. A subtle intensity enhancement near 2θ ≈ 35.1° is attributed to overlapping ZnO and α-Al2O3 reflections, indicative of strong interfacial coupling. Such behavior is associated with interfacial interactions mediated by surface hydroxyl groups on Al2O3 and polar functional groups within the matrix, which facilitate nanoparticle anchoring and induce localized lattice distortions53. These interfacial regions are expected to introduce shallow defect states and promote efficient carrier separation, a key factor in enhancing UV photoresponse. Moreover, the presence of Al2O3 induces a systematic reduction in XRD peak broadening, indicating enhanced crystallinity and promoted grain growth of ZnO nanoparticles54. This structural evolution suggests a decrease in lattice strain and a reduced density of grain-boundary scattering centers, which collectively favour improved charge transport and suppressed carrier recombination55. The average crystallite size (D) was estimated using the Debye–Scherrer equation:
where λ is the X-ray wavelength, β is the full width at half maximum (FWHM), and θ is the Bragg angle. The calculated average crystallite sizes are approximately 9.11 nm for α-Al2O3, 10.14 nm for pristine ZnO, and 10.83 nm for the ZnO–Al2O3 nanocomposites.
From a device perspective, the preserved crystal structure, enhanced crystallinity, and optimized interfacial coupling collectively contribute to improved carrier mobility, reduced trap-assisted recombination, and enhanced photo-generated charge extraction56. These structural advantages directly underpin the observed improvements in responsivity, detectivity, and operational stability of the ZnO–Al2O3-based UV photodetectors, confirming the critical role of controlled phase evolution and nanoscale crystallinity in device optimization.
Combined theoretical–experimental IR spectroscopic analysis
FTIR spectroscopy was employed to investigate the chemical bonding, surface functional groups, and interfacial interactions in pure ZnO and ZnO–Al2O3 nanocomposite films, as shown in Fig. 7b. The FTIR spectra provide essential insight into the structural integrity of the metal–oxide frameworks and the nature of interactions responsible for the enhanced optoelectronic performance of the composite-based UV photodetectors. The FTIR spectrum of pristine ZnO exhibits a prominent absorption band in the low-wavenumber region below 600 cm−1, centered around 430–480 cm−1, which is characteristic of the Zn–O stretching vibration in the hexagonal wurtzite structure. This band confirms the successful formation of crystalline ZnO and is consistent with previously reported results for nanostructured ZnO systems. A broad absorption band observed in the range 3200–3600 cm−1 is attributed to the stretching vibrations of surface-adsorbed –OH groups, originating from hydroxylated ZnO surfaces or physically adsorbed moisture. The presence of these hydroxyl groups is particularly relevant for UV photodetection, as they act as active sites for oxygen adsorption and desorption processes, which play a crucial role in modulating the photoconductive response under UV illumination. Additionally, a weak band near 1630–1650 cm−1 is assigned to the bending mode of molecularly adsorbed water (H–O–H), further confirming the hydrophilic nature of the ZnO surface. Importantly, no additional bands corresponding to organic residues or undesired impurities are detected, indicating high chemical purity of the synthesized ZnO.
The FTIR spectrum of the ZnO–Al2O3 nanocomposite retains the characteristic Zn–O vibrational band, confirming that the ZnO crystal structure is preserved after composite formation. Notably, this band exhibits a slight shift toward lower wavenumbers and mild broadening compared to pure ZnO, which can be attributed to interfacial interactions and lattice distortion induced by the incorporation of Al2O3. Such shifts are indicative of modified local bonding environments and enhanced electronic coupling at the ZnO/Al2O3 interface. New absorption features appear in the region 500–800 cm−1, which are assigned to Al–O and Al–O–Al stretching vibrations, confirming the successful incorporation of Al2O3 within the composite matrix. The coexistence of Zn–O and Al–O vibrational modes without the emergence of additional impurity-related peaks strongly supports the formation of a clean heterostructured nanocomposite, rather than undesired secondary phases. Furthermore, the broad –OH stretching band (3200–3600 cm−1) in the ZnO–Al2O3 composite becomes noticeably less intense and more symmetric compared to pure ZnO. This behavior suggests effective passivation of surface hydroxyl groups and defect sites by Al2O3, which reduces surface trap density and suppresses non-radiative recombination pathways. The attenuation of the H–O–H bending mode near 1630 cm−1 further supports the reduction of weakly bound surface moisture, contributing to improved environmental stability. The FTIR results provide compelling evidence that Al2O3 incorporation leads to chemical passivation and interfacial stabilization of ZnO without altering its fundamental crystal structure. The reduction of hydroxyl-related defect states and the establishment of ZnO–Al2O3 interfacial bonding directly correlate with the observed enhancements in electrical conductivity, reduced Rct, and improved UV photoresponse characteristics. These modifications facilitate more efficient charge transport and faster carrier dynamics, which are critical for high-performance UV photodetector operation. Therefore, the FTIR analysis confirms that the ZnO–Al2O3 nanocomposite architecture achieves a synergistic balance between structural integrity and surface defect control, underpinning the superior sensitivity, stability, and reproducibility of the developed Si-based UV sensor–photodetectors.
To further substantiate the experimentally observed FTIR features and to gain deeper insight into the underlying intermolecular interactions and bonding configurations within the ZnO–Al2O3 system, a complementary theoretical vibrational analysis was conducted. While experimental FTIR spectroscopy provides direct evidence of functional groups and bonding environments, DFT calculations enable the precise assignment of vibrational modes and clarify the origin of newly emerging bands associated with hybridization and interfacial coupling. Accordingly, the simulated DFT-IR spectrum was employed to validate the experimentally detected metal–oxygen vibrations and to elucidate the formation of interfacial Zn–O–Al linkages within the nanocomposite architecture. On the other hand, the importance of IR spectroscopy as a tool for characterizing chemical interactions through the detection and determination of the molecular structure and functional groups that are included inside a molecule is something that should be taken into consideration57. The ZnO–Al2O3 nanocomposite under investigation was analyzed for its structure and intermolecular interactions, and the resulting estimated DFT-IR spectrum is shown in Fig. 7c. In the nanoscale range, the vibrational bands characteristic of metal oxides were identified at 1114, 922, and 772 cm−1 for Al–O, and at 694 cm−1 for Zn–O58,59. Notably, the conspicuous emergence of a new vibrational band at 991 cm−1 is directly associated with the formation of interfacial Zn–O–Al bonding, serving as a theoretical fingerprint of successful nanocomposite formation and corroborating the experimental FTIR observations60.
Morphology and EDX analysis
The surface morphology of the fabricated ZnO-based and ZnO–Al2O3 composite films was investigated in detail using high-resolution scanning electron microscopy (SEM) to elucidate the effect of Al2O3 incorporation on the structural hierarchy and porosity of the sensing layers, as seen in Fig. 8. In the case of pure ZnO, the SEM micrographs revealed a highly uniform porous network composed of densely packed, quasi-spherical nanoparticles interconnected into a continuous scaffold61. The pore channels were well-defined, with an average pore size of 0.20 μm, as determined from statistical measurements of 22 representative pores selected from multiple SEM regions using ImageJ image analysis software. Figure 8a–d highlights several representative pores that were explicitly selected and marked to illustrate the pore measurement procedure and statistical analysis used to determine the average pore size. The pore size distribution exhibited a relatively narrow spread, indicating a high degree of structural homogeneity62. Although the distribution does not strictly follow a Gaussian profile, the mean pore size adequately represents the dominant pore population due to the limited dispersion observed in pristine ZnO films. This uniformity is characteristic of ZnO layers formed under controlled nucleation and growth conditions, where the absence of secondary phases or dopants leads to relatively isotropic grain expansion during the synthesis stage63.
(a–d) FE-SEM micrographs of pristine ZnO and ZnO–Al2O3 thin films, respectively, illustrating the morphological evolution from a uniform nanoporous network (average pore size ≈ 0.20 µm) to a hierarchically structured architecture with enlarged and interconnected pores (average pore size ≈ 0.31 µm) upon Al2O3 incorporation. (e,f) HR-TEM images of pristine ZnO and ZnO–Al2O3 films, respectively, revealing improved crystallinity and intimate interfacial contact within the nanocomposite structure.
In contrast, the introduction of Al2O3 into the ZnO matrix resulted in a distinct and measurable modification of the microstructural features. The ZnO–Al2O3 nanocomposite displayed a hierarchically organized porous structure with multiple levels of pore sizes ranging from sub-100 nm nanopores to larger macropores approaching the micron scale. The average pore size increased to 0.31 μm, representing an approximate 55% enlargement compared to pure ZnO. The corresponding pore size histogram shows an asymmetric, non-Gaussian distribution, indicating increased structural heterogeneity. Accordingly, the pore size dispersion is more appropriately described by a Lorentzian-type distribution, where the peak position corresponds to the dominant pore size and the distribution width reflects the intrinsic morphological disorder induced by Al2O3 incorporation. Statistical analysis of the pore size histograms indicated a broader distribution and an increased standard deviation, reflecting the greater structural complexity introduced by the secondary Al2O3 phase53. This phenomenon is likely a result of the altered nucleation dynamics during film formation: Al2O3, with its distinct surface energy and low lattice match to ZnO, can disrupt the regular coalescence of ZnO crystallites, creating additional voids and preventing excessive grain boundary fusion61. The presence of Al2O3 nanoparticles may also act as spacers between ZnO grains, further enhancing the inter-particle gaps and producing a more open porous framework. From a functional perspective, the observed pore size enlargement and hierarchical structuring are highly beneficial for UV sensor–photodetector performance64. Larger interconnected pores increase the optical path length for incident UV photons, thereby enhancing light scattering and improving absorption efficiency within the active layer65. Moreover, the increased void fraction improves gas permeability and facilitates rapid adsorption–desorption kinetics of oxygen species on the surface, which is critical for modulating the surface depletion layer in photoconductive UV sensors. In such devices, the adsorption of oxygen in the dark and its subsequent photodesorption under UV illumination govern the change in conductivity. Therefore, a hierarchical porous architecture not only increases the number of active sites but also accelerates the response and recovery times by enabling faster gas exchange and carrier transport through reduced tortuosity66.
Building upon the morphological analysis presented above, TEM and high-resolution TEM (HR-TEM) were employed to provide nanoscale insight into the internal morphology, crystallinity, and interfacial characteristics of the pristine ZnO and ZnO–Al2O3 films. Figure 8e and f present the HR-TEM images of pure ZnO and ZnO–Al2O3 nanocomposite films, respectively. The HR-TEM image of pristine ZnO (Fig. 8e) reveals well-defined lattice fringes, confirming its crystalline nature with a typical interplanar spacing of approximately 0.26 nm, which corresponds to the (002) plane of hexagonal wurtzite ZnO. However, localized lattice distortions and minor fringe discontinuities are observed, which can be attributed to intrinsic defects, grain boundaries, and surface-related imperfections commonly associated with nanoscale ZnO structures55. These structural irregularities are known to act as charge trapping and recombination centers, thereby limiting carrier transport efficiency. In contrast, the ZnO–Al2O3 nanocomposite film (Fig. 8f) exhibits markedly improved crystallinity, as evidenced by sharper, more continuous lattice fringes and reduced lattice distortion67. The intimate interfacial contact between ZnO nanocrystallites and the surrounding Al2O3 phase is clearly discernible, indicating effective nanoscale integration rather than simple physical mixing68. The Al2O3 phase appears as a thin, amorphous or weakly crystalline shell surrounding ZnO domains, which plays a critical role in surface passivation by suppressing defect states and mitigating non-radiative recombination pathways51. The enhanced structural coherence and improved interfacial coupling in the ZnO–Al2O3 nanocomposite facilitate more efficient charge transport across grain boundaries and interfaces15. This structural refinement directly supports the observed improvements in electrical conductivity, reduced charge-transfer resistance, and enhanced photodetection metrics, including higher responsivity and faster response–recovery behavior53. The HR-TEM observations are in excellent agreement with the XRD results, which indicate increased crystallite size and enhanced crystallinity upon Al2O3 incorporation, confirming the strong correlation between nanoscale structural engineering and macroscopic device performance69.
Energy-dispersive X-ray (EDX) analysis provided complementary insights into the elemental composition of the samples, confirming the successful incorporation of Al2O3 into the ZnO framework, as seen in Fig. 9. For the pure ZnO film, the EDX spectrum exhibited two major peaks corresponding to oxygen (O K) and zinc (Zn K) with measured weight percentages of 26.66 wt% O and 73.34 wt% Zn, in close agreement with stoichiometric ZnO values. This composition reflects the high purity of the synthesized ZnO phase, with no detectable impurities or secondary elements within the instrument’s resolution limits. Upon Al2O3 incorporation, the ZnO–Al2O3 composite spectrum revealed 26.87 wt% O, 70.20 wt% Zn, and 2.93 wt% Al, clearly confirming the presence of aluminum within the structure. The slight increase in oxygen content in the composite compared to pure ZnO can be attributed not only to the oxygen supplied by Al2O3 but also to possible changes in surface chemistry, such as increased hydroxylation due to the higher surface area and enhanced moisture adsorption. The detection of Al at nearly 3 wt% strongly indicates that Al2O3 is not merely a surface coating but is uniformly distributed throughout the nanocomposite layer. The even dispersion of Al2O3 may lead to localized strain fields and defect sites at the ZnO–Al2O3 interfaces, which can act as electron trapping or scattering centers, thereby influencing the optoelectronic behavior of the device70.
EDX spectra of pure ZnO and ZnO–Al2O3 confirming the successful incorporation of Al2O3, with corresponding elemental weight percentages revealing the presence of Al (2.93 wt%) alongside Zn and O.
The synergy between morphological and compositional modifications in the ZnO–Al2O3 nanocomposite is expected to have a significant impact on device performance. Morphologically, the increased porosity and hierarchical structuring enhance both light–matter interaction and analyte accessibility. Compositionally, the incorporation of Al2O3 introduces heterojunction interfaces that may facilitate more efficient separation of photogenerated electron–hole pairs, suppressing bulk recombination and improving photocurrent generation71. Furthermore, the slight variation in elemental ratios can lead to fine-tuning of the surface work function, oxygen adsorption energetics, and depletion width—all of which are critical parameters in determining UV detection sensitivity and response kinetics72. Therefore, the combined SEM and EDX analyses reveal that Al2O3 incorporation into ZnO fundamentally transforms both the structural and chemical landscape of the sensing layer. The resulting hierarchical porosity, larger average pore sizes, and modified surface chemistry collectively establish a platform for enhanced UV photodetection, providing a balance between high photon absorption, rapid carrier extraction, and fast surface reaction dynamics. These attributes make ZnO–Al2O3 nanocomposite architectures highly promising candidates for next-generation high-sensitivity UV sensor–photodetector applications72.
NMR result
NMR spectroscopy is a crucial analytical tool that provides deep understanding into atomic structure, chemical environments, atomic connection, the number of distinct nuclear environments, and their locations inside the molecules themselves73. Under the influence of an external magnet, NMR works by arranging protons into isotopic nuclei having non-zero rotations. The receipt of an electromagnetic signal in certain radio frequency energy causes the configuration to take place. This energy is controlled by the isotopic nature of the radioactive construction and grows in relation to the size of the surrounding magnetism. The chemical makeup of the surrounding environment dictates the frequency at which its resonances of each energetic isotope nuclei74. The ZnO–Al2O3 studied was analyzed using nuclear magnetic resonance (NMR) isotopes 67Zn, 17O, and 27Al in a solution of dimethyl sulfoxide (DMSO) specified to tetramethyl silane (TMS) to obtain chemical shifting validation, as illustrated in Fig. 10. The 67ZnNMR spectrum revealed the presence of Zn at a value of δ = 2872.11 ppm, which was due to the presence of ZnO75. Likewise, the O atoms in ZnO were observed to have two separate resonances in the 17ONMR chart, which were located at − 858.61 and − 235.625 ppm. On the other hand, the O atoms in Al2O3 corresponded to 259.66, − 80.8, and − 3486.55 ppm, which indicates that there is a strong link between Zn and Al17. In accordance with its interaction with ZnO through the O atom, the 27AlNMR chart displayed the Al atoms in Al2O3 at the positions of 522.02 and 541.57 according to the data76.
DFT-NMR spectra of ZnO–Al2O3 nanocomposite.
Apparent porosity and surface roughness
The apparent porosity of the fabricated thin films was quantified using the Archimedes principle in accordance with ASTM C373-88 standards, providing an indirect measure of the void volume fraction accessible to fluids within the composite matrix. The pure ZnO film exhibited an apparent porosity of 26%, whereas the ZnO–Al2O3 hybrid film demonstrated a substantially higher porosity of 36%. This 10% absolute increase reflects the structural influence of Al2O3 NPs, which likely disrupt the close packing of ZnO crystallites, generating additional intergranular voids and nano-/micro-scale channels77. The higher porosity in the hybrid structure is advantageous for UV sensor–photodetector applications, as it can facilitate enhanced light scattering within the active layer, thereby increasing the optical path length and improving photon absorption efficiency78. Furthermore, increased porosity can promote more effective penetration of the electric field and increase the active surface area for charge separation, although it must be balanced against potential increases in carrier recombination at high surface defect densities79.
Surface topography, characterized through contact profilometry, revealed notable differences in surface roughness (Ra) between the two film types80. The pure ZnO film exhibited an Ra value of 6.7 µm, indicative of moderately textured surfaces formed by grain agglomeration81. In contrast, the ZnO–Al2O3 hybrid film displayed a higher Ra of 8.2 µm, suggesting that the introduction of Al2O3 NPs not only altered the grain packing but also enhanced the hierarchical structuring of the surface. This increase in roughness is consistent with the formation of a more complex multi-scale morphology, in which nanoscale asperities overlay micron-scale texturing82. The concurrent increases in both porosity and roughness in the ZnO–Al2O3 hybrid are synergistic from a device performance perspective. Higher roughness improves the interfacial contact between the active layer and the underlying Si substrate, potentially lowering interfacial resistance, while also acting as an optical trapping layer to reduce surface reflection losses54. Meanwhile, increased porosity can enhance the infiltration and interaction of the surrounding medium (air, vacuum, or protective coatings) with the sensing layer, thereby impacting dielectric properties and potentially modulating sensor response time and sensitivity. The combined effect of these morphological enhancements supports the superior UV photoresponse observed in the ZnO–Al2O3 devices compared to their pure ZnO counterparts.
EIS analysis and photocurrent dynamics
EIS was conducted to gain deeper insight into the interfacial charge-transport dynamics and recombination mechanisms within the fabricated Si-based UV sensor–photodetectors. Figure 11a shows the Nyquist plots for pure ZnO and ZnO–Al2O3 nanocomposite films under both dark and UV illumination, highlighting the pronounced reduction in charge-transfer resistance (Rct) upon Al2O3 incorporation. Meanwhile, Fig. 11b presents a schematic of the equivalent circuit model Rs − (Rct∥CPE), employed to fit the EIS data and quantitatively describe the interfacial charge-transfer behavior83. The impedance spectra for all samples exhibit a single, well-defined semicircle in the high-to-medium frequency range, characteristic of a charge-transfer process at the semiconductor–metal contact (ZnO/Si with metal electrodes) or grain boundary interface within the nanostructured layer, followed by a near-linear tail in the low-frequency region attributable to diffusion-controlled processes84. In the dark, pure ZnO films demonstrated a charge transfer resistance (Rct) of approximately 180 Ω, which is indicative of moderately hindered interfacial electron transport due to limited carrier density and potential trap states at grain boundaries. In contrast, the ZnO–Al2O3 hybrid exhibited a significantly lower Rct value of ~ 95 Ω, reflecting the beneficial role of the Al2O3 component in modulating surface states, enhancing dielectric passivation, and promoting more effective electron percolation pathways85. The lower Rct for the hybrid system also implies reduced recombination rates and improved interfacial conductivity, despite Al2O3 being an insulator, suggesting that its role is primarily structural—passivating defect sites and improving ZnO crystallite packing77. Upon UV illumination, both materials showed substantial decreases in Rct values, consistent with the generation of photocarriers and the consequent enhancement in charge transport. For ZnO, Rct dropped from 180 to 75 Ω, while for ZnO–Al2O3, it decreased from 95 to 40 Ω. The magnitude of reduction was more pronounced in the hybrid, suggesting that the optimized ZnO–Al2O3 architecture not only facilitates more efficient photogenerated carrier extraction but also supports sustained transport under illumination, likely due to a synergistic effect between ZnO’s photoactive properties and Al2O3’s ability to suppress trap-assisted recombination67.
(a) Nyquist plots of pure ZnO and ZnO–Al2O3 composites under dark and UV illumination, illustrating the markedly reduced Rct for the hybrid structure. (b) Schematic representation of the equivalent circuit model, Rs − (Rct∥CPE), used to fit the EIS data and describe the interfacial charge-transfer processes. (c) UV–Vis absorbance spectra of pure ZnO, pure Al2O3, and ZnO–Al2O3 nanocomposites.
Series resistance (Rs) values were comparatively low for all samples (14.5–18 Ω) and showed only minor variation between dark and illuminated states, indicating that bulk and contact resistances are not the limiting factors in device performance. The double-layer capacitance (Cdl) exhibited an upward shift from dark to illuminated states, with ZnO increasing from ~ 5.0 to ~ 6.0 μF and ZnO–Al2O3 from ~ 7.0 to ~ 8.5 μF, reflecting higher carrier accumulation at the interface under UV excitation. These EIS findings corroborate the hypothesis that the hierarchical ZnO–Al2O3 nanocomposite configuration substantially enhances both dark-state electrical conductivity and photoconductive response86. The lower Rct and higher Cdl in the hybrid structure signify more effective suppression of recombination and improved interfacial kinetics, which align with the enhanced UV responsivity and faster recovery times observed in the device characterization87. This behavior can be attributed to the refined surface morphology, increased porosity, and optimized band alignment facilitated by the Al2O3 incorporation, enabling superior electron transport pathways and minimizing energy barriers at critical junctions85. Therefore, the EIS results provide compelling evidence that ZnO–Al2O3 hybrids achieve superior charge transport efficiency and interfacial stability compared to pure ZnO, underscoring their suitability for high-performance, Si-based UV sensor–photodetectors88.
Electrical and optical properties and UV selectivity
The electrical conductivity of the fabricated films was determined using the four-point probe method, revealing a pronounced enhancement upon incorporation of Al2O3 into the ZnO matrix. Pure ZnO films exhibited a conductivity of 27.7 × 10−2 S/m, while the ZnO–Al2O3 hybrid films achieved a markedly higher value of 44.5 × 10−2 S/m, corresponding to an increase of approximately 60.6%. This improvement in conductivity can be attributed to several synergistic mechanisms at play within the ZnO–Al2O3 nanocomposite architecture. Firstly, the dispersion of Al2O3 nanoparticles within the ZnO network promotes microstructural reorganization and enhanced particle packing density, reducing grain boundary scattering and improving electron percolation pathways. Although Al2O3 is intrinsically insulating, its nanoscale incorporation modifies the interface chemistry and reduces defect-induced charge trapping at the ZnO grain boundaries, thus facilitating charge transport52. Furthermore, the hybrid structure is likely to exhibit optimized carrier concentration due to partial passivation of oxygen vacancies—a common defect in ZnO—thereby balancing carrier mobility and lifetime. The interfacial bonding between Al2O3 may also induce localized electric field effects that assist in the separation and transport of photogenerated charge carriers under UV illumination, an effect that correlates with the improved photodetector performance observed in subsequent optical and EIS analyses89. The magnitude of conductivity enhancement observed here is consistent with literature reports on metal oxide hybrid systems, where strategic inclusion of secondary nanostructures facilitates enhanced electronic coupling and reduced resistive losses. This property is especially critical for UV sensor–photodetector applications, where fast charge transport is directly linked to high responsivity, reduced response/recovery times, and superior long-term operational stability56.
The optical absorbance spectra of pure ZnO, pure Al2O3, and the ZnO–Al2O3 hybrid nanocomposite, recorded in the wavelength range of 250–650 nm, are presented in Fig. 11c. All samples exhibit a pronounced absorption onset in the ultraviolet region, indicating the dominance of intrinsic band-to-band electronic transitions typical of wide-bandgap semiconductors. The absorption intensity decreases sharply in the visible region, underscoring their potential for UV-selective detection88. This behavior is particularly important in UV photodetector applications, where suppression of visible light sensitivity minimizes background noise and enhances device signal-to-noise ratio90. The inset of Fig. 11c, highlighting the 250–450 nm spectral region, shows a more abrupt and well-defined absorption edge for the ZnO–Al2O3 composite compared to single-phase ZnO or Al2O3. This suggests that the hybrid structure enhances UV photon harvesting while simultaneously reducing sub-bandgap transitions that could arise from defect-related states70. The sharper edge in the composite can be attributed to the synergistic interaction between ZnO and Al2O3 nanoparticles, which may induce microstructural ordering, lattice strain modulation, and passivation of oxygen vacancies, all of which improve the purity of the electronic transitions. Using the Tauc method for direct allowed transitions, the estimated optical bandgap energies (Eg) are 3.18 eV for ZnO, 3.11 eV for Al2O3, and 3.26 eV for the ZnO–Al2O3 composite. The slight bandgap widening in the composite corresponds to a blue shift in the absorption edge (~ 380 nm for ZnO vs. ~ 375 nm for ZnO–Al2O3), which further enhances UV selectivity by narrowing the detection window towards shorter wavelengths.
This is advantageous in UV-A and UV-B photodetector applications where specificity is critical, such as environmental monitoring, flame sensing, and space instrumentation. From a device physics perspective, UV selectivity in these materials is controlled by three interrelated factors: (1) Bandgap Position—Wide bandgap (> 3.1 eV) ensures cutoff beyond ~ 400 nm, reducing visible light response, (2) Defect State Density—Reduced deep-level defects in ZnO–Al2O3 minimize sub-bandgap absorption that can cause parasitic photocurrent under visible illumination88, (3) Interfacial Barrier Effects—The ZnO–Al2O3 heterointerface may form potential barriers that preferentially facilitate separation of high-energy UV-generated carriers while blocking lower-energy carriers from visible photons90. The ZnO–Al2O3 hybrid structure not only retains high UV absorption but also displays suppressed absorption in the 420–650 nm range, leading to a high UV-to-visible rejection ratio. This property is essential for real-world UV sensor–photodetectors operating under sunlight, where visible light intensity can be orders of magnitude higher than UV intensity3. The combination of enhanced UV absorption, minimal visible light sensitivity, and stable optical transitions makes ZnO–Al2O3 nanocomposites particularly promising for high-performance, environmentally stable UV photodetectors.
Current–voltage (I–V) characteristics, transient photocurrent response, and photodetection metrics
To comprehensively evaluate the electrical and photoresponse performance of the fabricated Si-based UV photodetectors, current–voltage (I–V) characteristics and time-dependent transient photocurrent (I–t) measurements were carried out under both dark UV illumination. These measurements provide direct insight into the charge transport mechanism, contact behavior, response dynamics, and key photodetection metrics, including responsivity and detectivity.
I–V characteristics under dark and UV illumination
Figure 12a presents the I–V characteristics of pure ZnO and ZnO–Al2O3 nanocomposite photodetectors measured over the voltage range of − 5 to + 5 V under dark and UV illumination. Both devices exhibit a nearly linear and symmetric I–V behavior in the forward and reverse bias regions, indicating the formation of ohmic contacts between the active semiconductor layer and the metal electrodes. The absence of rectifying behavior confirms that the photodetection mechanism is governed by photoconductive operation rather than junction-based (Schottky or p–n) carrier separation91. Under dark conditions, the ZnO–Al2O3 device shows a slightly higher current than pure ZnO, which is consistent with its enhanced electrical conductivity and reduced interfacial resistance, as previously evidenced by four-point probe measurements and EIS. Upon UV illumination, both devices demonstrate a pronounced increase in current while preserving linearity, reflecting a substantial enhancement in free carrier concentration due to photogeneration. Notably, the ZnO–Al2O3 hybrid exhibits a steeper I–V slope under UV exposure, signifying more efficient charge transport pathways and reduced recombination losses. The maintenance of linear I–V behavior under illumination suggests that UV exposure modulates the carrier density rather than altering the contact barrier height92. This observation is in excellent agreement with the low Rs and significantly reduced Rct obtained from EIS analysis, confirming that interfacial barriers do not limit device performance. The incorporation of Al2O3 plays a crucial role in passivating surface and grain-boundary trap states, leading to a more homogeneous potential landscape and stable ohmic conduction.
(a) I–V characteristics of pure ZnO and ZnO–Al2O3 UV photodetectors under dark and UV illumination, exhibiting linear ohmic behavior. (b) Transient I–t response under periodic UV ON/OFF switching, indicating stable and repeatable photoresponse. (c) Aging stability of the devices evaluated by photocurrent variation at 5 V under UV illumination for different aging times. (d) Normalized photocurrent retention, highlighting the superior long-term environmental stability of the ZnO–Al2O3 nanocomposite.
I–t response and switching behavior
The transient photoresponse of the devices was further examined by monitoring the photocurrent as a function of time under periodic UV ON/OFF switching at a fixed bias voltage, as shown in Fig. 12b. Both pure ZnO and ZnO–Al2O3 devices exhibit rapid and repeatable photocurrent modulation upon UV exposure, demonstrating excellent operational stability and reversibility over multiple switching cycles.
Upon UV illumination (ON state), a sharp increase in photocurrent is observed, corresponding to the rapid generation and separation of electron–hole pairs. When the UV source is switched OFF, the photocurrent quickly returns to its initial dark level, indicating efficient carrier recombination and minimal persistent photoconductivity93. Importantly, the ZnO–Al2O3 photodetector displays a higher photocurrent amplitude and faster recovery behavior compared to pure ZnO, highlighting the beneficial role of the hybrid architecture in suppressing trap-assisted recombination and facilitating rapid carrier extraction. The enhanced transient response of the ZnO–Al2O3 device can be attributed to the synergistic effects of increased surface roughness and porosity, which provide a larger density of active sites for photon absorption, together with effective defect passivation by Al2O3 that minimizes carrier trapping and prolongs carrier mobility. The linear I–V characteristics and reversible transient photoresponse observed in this work are consistent with typical photoconductive UV detectors reported for ZnO-based nanostructures91,92,93.
Responsivity and detectivity analysis
Key photodetection metrics, including responsivity (R) and specific detectivity (D*), were derived from the I–V and transient photocurrent data to quantitatively assess device sensitivity. As summarized in Table 3, the ZnO–Al2O3 nanocomposite photodetector exhibits a responsivity of approximately 0.12 A/W, nearly twice that of pure ZnO (~ 0.065 A/W). Correspondingly, the detectivity increases from ~ 1.8 × 1010 Jones for pure ZnO to ~ 3.4 × 1010 Jones for the ZnO–Al2O3 device. This substantial improvement reflects the combined effect of enhanced photocurrent generation, reduced dark current noise, and efficient interfacial charge transport in the hybrid structure. The observed enhancement in R and D* is fully consistent with the reduced Rct, increased interfacial capacitance, and improved electrical conductivity reported earlier, establishing a strong correlation between nanoscale interfacial engineering and macroscopic photodetector performance.
Mechanistic implications for UV photodetection
The linear I–V characteristics, fast transient response, and enhanced photodetection metrics collectively confirm that the ZnO–Al2O3-based devices operate as high-performance photoconductive UV sensors. The introduction of Al2O3 does not act as a current-blocking layer; instead, it improves charge transport by passivating defect states, homogenizing the electric field distribution, and facilitating efficient carrier percolation within the ZnO matrix. These effects, combined with strong UV-selective absorption and minimal visible-light sensitivity, render the ZnO–Al2O3 nanocomposite architecture highly suitable for reliable and sensitive UV detection under practical operating conditions.
Aging stability and long-term operational reliability
To further substantiate the environmental stability of the fabricated UV photodetectors, aging stability tests were performed by monitoring the photocurrent response at a fixed bias voltage of 5 V under UV illumination after different aging intervals (1, 10, and 100 h). The photocurrent values were recorded and normalized with respect to their initial response to evaluate performance retention over time. As shown in Fig. 12c, both devices exhibit a gradual decrease in photocurrent with increasing aging duration, which can be attributed to surface adsorption of oxygen and moisture, as well as slow structural relaxation of defect states.
Nevertheless, the ZnO–Al2O3 photodetector maintains a significantly higher absolute photocurrent throughout the entire aging period compared to pure ZnO. The normalized photocurrent retention presented in Fig. 12d reveals that the ZnO–Al2O3 device preserves approximately 92% of its initial photocurrent after 100 h of aging, whereas pure ZnO retains only ~ 87%. This enhanced stability is attributed to the effective passivation of surface and grain-boundary defects by Al2O3, which suppresses environmental degradation mechanisms and minimizes charge trapping induced by ambient exposure. These results confirm that the incorporation of Al2O3 not only enhances the sensitivity and charge transport characteristics of the photodetector but also significantly improves its long-term operational stability, making the ZnO–Al2O3 nanocomposite architecture highly suitable for reliable UV sensing under practical environmental conditions.
Conclusion
In this work, we have demonstrated an integrated theoretical–experimental strategy for the design, fabrication, and performance optimization of hierarchically structured Si-based UV sensor–photodetectors employing ZnO–Al2O3 nanocomposite architectures. Density functional theory (B3LYP/6-31G(d,p)) calculations revealed that the ZnO–Al2O3 hybrid exhibits favorable band alignment, optimized frontier orbital separation, and enhanced charge transport pathways compared to its single-component counterparts. Experimental validation through XRD, FE-SEM, and EDX confirmed the successful synthesis and homogeneous integration of the hybrid nanoparticles into the sensing matrix. Morphological characterization showed that incorporating Al2O3 increased both surface roughness (from 6.7 to 8.2 μm) and apparent porosity (from 26 to 36%), contributing to improved photon capture and increased active sites for carrier generation. Optical characterization revealed strong UV-selective absorption (250–450 nm), with calculated band gaps of 3.18 eV (ZnO), 3.11 eV (Al2O3), and 3.26 eV (ZnO–Al2O3). Electrical conductivity measurements showed a marked improvement in the hybrid system (44.5 × 10−2 S/m) compared to pure ZnO (27.7 × 10−2 S/m), while electrochemical impedance spectroscopy confirmed reduced charge transfer resistance, aligning with the observed enhancement in responsivity and recovery speed. Collectively, these results highlight the synergistic effect of nanostructuring and oxide hybridization in enabling high-performance, stable, and UV-selective photodetection.
The demonstrated performance of ZnO–Al2O3 hybrid architectures opens several avenues for advancement. Future research could:
-
Optimize interfacial engineering through controlled thickness of Al2O3 layers to further suppress recombination and improve responsivity.
-
Explore alternative wide-bandgap oxide hybrids (e.g., ZnO–HfO2, ZnO–Ga2O3) to enhance spectral tunability and thermal stability.
-
Integrate plasmonic nanostructures to exploit localized surface plasmon resonance (LSPR) for boosting UV absorption without compromising visible transparency.
-
Implement flexible and transparent substrates to develop wearable and portable UV sensing devices.
-
Adopt machine-learning-driven materials screening to accelerate the discovery of optimal nanocomposite compositions for tailored band gap and conductivity.
By bridging theoretical modeling with experimental validation, this study provides not only a proof-of-concept for ZnO–Al2O3-based UV photodetectors but also a framework for the rational design of next-generation optoelectronic materials with superior sensitivity, selectivity, and operational stability.
References
Soni, A., Mulchandani, K. & Mavani, K. R. Effects of substrates on the crystalline growth and UV photosensitivity of glancing angle deposited porous ZnO nanostructures. Sens. Actuators A Phys. 313, 112140 (2020).
Ashok, P. & Dhar, J. C. RETRACTED: Perovskite Based Low-Powered Hybrid UV Photodetector with RF Sputtered Metal Oxide Carrier Transport Layers (Elsevier, 2023).
Pan, Z. et al. The MoS2/ZnO pn heterostructure arrays for ultrasensitive ppb-level self-supporting NO2 gas sensors under UV irradiation. Talanta 294, 128194 (2025).
Santos-Carballal, D. et al. Al2O3/ZnO composite-based sensors for battery safety applications: An experimental and theoretical investigation. Nano Energy 109, 108301 (2023).
Flemban, T. H. et al. A photodetector based on p-Si/n-ZnO nanotube heterojunctions with high ultraviolet responsivity. ACS Appl. Mater. Interfaces 9(42), 37120–37127 (2017).
Akhtarianfar, S. F., Khayatian, A., Shakernejad, R., Almasi-Kashi, M. & Hong, S. W. Improved sensitivity of UV sensors in hierarchically structured arrays of network-loaded ZnO nanorods via optimization techniques. RSC Adv. 7(51), 32316–32326 (2017).
Pandey, S., Shukla, A. & Tripathi, A. Highly sensitive and self powered ultraviolet photo detector based on ZnO nanorods coated with TiO2. Sens. Actuators A Phys. 350, 114112 (2023).
Shang, G. et al. High-performance NiO/TiO2/ZnO photovoltaic UV detector. Sensors 23(5), 2741 (2023).
Sonpir, R. B. et al. Enhanced photocatalytic and gas sensing performance of tungsten carbide based Ni doped Co3O4/TiO2 nanocomposite. ChemistrySelect 10(42), e04702 (2025).
Sonpir, R., Dake, D., Raskar, N., Mane, V. & Dole, B. Smart and advanced nanocomposites of rGO-based Ni-doped Co3O4/TiO2 for next-level photocatalysis and gas sensing application. Environ. Sci. Pollut. Res. 32(3), 1308–1330 (2025).
Chavan, K. et al. Nanomaterials for photocatalytic environmental remediation: Synthesis, mechanism, and performance. Environ. Technol. Rev. 14, 1–36 (2025).
Sonpir, R. B. et al. Magneto-chemical properties of Ti-doped Co3O4 nanosheets for photocatalytic activity applications. Mater. Today Commun. 38, 108076 (2024).
Sonpir, R. B. et al. Photocatalytic efficiency of GO-based Ti-doped Co3O4 nanosheets for mixed dyes and the effects of oxidizing and reducing reagents. Ceram. Int. 50(16), 28746–28761 (2024).
Solanke, B. V. et al. Low-variability, high-endurance memristive behavior in tungsten-functionalized GObased Cu doped NiO nanocomposites for next-generation memory devices. Mater. Chem. Phys. 346, 131352 (2025).
Shahat, M. A., Ibrahim, M. A., Ghitas, A. & Ezzat, H. A. Designing innovative PAni-based adsorbents for CO2 capture via in-situ nitrogen plasma modification for sustainable development. J. CO2 Util. 84, 102830 (2024).
Shahat, M. A., Ahmed, Y. M. Z., Ghitas, A., El-Shater, A. & Soliman, W. Improving the thermophysical aspects of innovative clay brick composites for sustainable development via TiO2 and rGO nanosheets. Constr. Build. Mater. 401, 132981 (2023).
Ezzat, H. A., Sebak, M. A., Aladim, A. K. & Shahat, M. A. Innovative experimental and theoretical strategies for sustainable heavy metal ion removal using chitosan@TiO2 composites functionalized with nanostructured metal oxides. J. Mol. Liq. 431, 127814. https://doi.org/10.1016/j.molliq.2025.127814 (2025).
Sebak, M. A. et al. Regulating carrier transfer and performance in dye-sensitized solar cells (DSSCS) using gamma-irradiated chitosan @PVA@Al2O3 nanocomposites as a counter electrode. Mater. Res. Bull. 192, 113593. https://doi.org/10.1016/j.materresbull.2025.113593 (2025).
Elsayed, A. M., Shaban, M., Aly, A. H., Ahmed, A. M. & Rabia, M. Preparation and characterization of a high-efficiency photoelectric detector composed of hexagonal Al2O3/TiO2/TiN/Au nanoporous array. Mater. Sci. Semicond. Process. 139, 106348 (2022).
Ezzat, H. A., Elhaes, H., Ibrahim, M. A. & Shahat, M. A. Theoretical and experimental investigation of a CuO and graphene embedded polyethylene oxide counter electrode for efficient DSSCs. Sci. Rep. 15(25049), 1–20. https://doi.org/10.1038/s41598025-98930-z (2025).
Devendiran, S., Priya, A. K. & Sastikumar, D. Design of aluminium oxide (Al2O3) fiber optic gas sensor based on detection of refracted light in evanescent mode from the side-polished modified clad region. Sens. Actuators B Chem. 361, 131738 (2022).
Frisch, M. J. et al. Gaussian 09, Revision C.01 (Gaussian Inc, 2010).
Ezzat, H. A., Sebak, M. A., Aladim, A. K. & Shahat, M. A. Experimental and theoretical strategies for multidisciplinary PTFE@ TiO2-based microfibres loaded with multiple metal oxides for anti-corrosion and self-cleaning aerospace applications. J. Inorg. Organomet. Polym. Mater. 35, 1–22 (2025).
Raghavachari, K. Perspective on ‘density functional thermochemistry. III. The role of exact exchange’ Becke AD (1993) J Chem Phys 98: 5648–52. Theor. Chem. Acc. 103, 361–363 (2000).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37(2), 785 (1988).
Vosko, S. H., Wilk, L. & Nusair, M. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis. Can. J. Phys. 58(8), 1200–1211 (1980).
Li, Z., Yan, T. & Fang, X. Low-dimensional wide-bandgap semiconductors for UV photodetectors. Nat. Rev. Mater. 8(9), 587–603 (2023).
Emara, A., Yousef, A. & Elleithy, K. Ultra-sensitive Si-based optical sensor for nanoparticle-size traditional water pollutant detection. Photonics 9, 289 (2022).
Qin, L. et al. Economic friendly ZnO-based UV sensors using hydrothermal growth: A review. Materials (Basel) 14(15), 4083 (2021).
Lee, J., Kumar, N., Patel, M., Ghosh, S. & Kim, J. Transparent metal-oxide personal UV monitoring device with machine learning advancement. Sens. Actuators A Phys. 362, 114627 (2023).
Elzawiei, Y. S. M., Abdulhameed, A., Hashim, M. R. & Halim, M. M. A study of the UV photodetectors properties based on the effect of TiO2 on ZnO nanorods grown via the chemical bath deposition method on p-type Si (100) substrates. Opt. Mater. (Amst.) 144, 114353 (2023).
Karthikeyan, B., Gnanakumar, G. & Alphonsa, A. T. Nano Metal Oxides (Springer, 2023).
Kumar, A. et al. Highly responsive and low-cost ultraviolet sensor based on ZnS/p-Si heterojunction grown by chemical bath deposition. Sens. Actuators A Phys. 331, 112988 (2021).
El-Mansy, M. A. M. et al. Modeling the effect of hydration on the electronic and vibrational properties of AZT. Biointerface Res. Appl. Chem. 11(2), 9253–9265. https://doi.org/10.33263/BRIAC112.92539265 (2021).
Mallya, A. N. & Panda, S. DFT study of iminodiacetic acid functionalised polyaniline copolymer interaction with heavy metal ions through binding energy, stability constant and charge transfer calculations. Comput. Theor. Chem. 1202, 113288 (2021).
Li, S., Wan, H., Lin, J. & Min, J. Physicochemical interactions between amorphous metal oxide and polymer in metal–polymer hybrid materials. Mater. Des. 230, 111993 (2023).
Afshari, A. R. Using the Delphi method for futures studies. In Proceedings of the International Conference on Industrial Engineering and Operations ManagementPilsen, Czech Republic, 23–26 (2019).
Gomaa, I. et al. Two-dimensional mxene as a promising adsorbent for trihalomethanes removal: A density-functional theory study. Nanomaterials 14(5), 454 (2024).
Ezzat, H. A., Hegazy, M. A., Nada, N. A., Osman, O. & Ibrahim, M. A. ICMMS-2: Application of Cs/ZnO/GO hybrid nanocomposite for enhanced inter-behavior of electronic properties and thermal stability as corrosion inhibitor. Egypt. J. Chem. 64(3), 1197–1205 (2021).
Elhaes, H. et al. Spectroscopic, Hartree–Fock and DFT study of the molecular structure and electronic properties of functionalized chitosan and chitosan-graphene oxide for electronic applications. Opt. Quantum Electron. 56(3), 458 (2024).
Ezzat, H. A. et al. DFT and QSAR studies of PTFE/ZnO/SiO2 nanocomposite. Sci. Rep. 13(1), 9696 (2023).
M. Bhatia, “An overview of conceptual-DFT based insights into global chemical reactivity of volatile sulfur compounds (VSCs),” Comput. Toxicol., p. 100295, 2023.
Obot, I. B., Macdonald, D. D. & Gasem, Z. M. Density functional theory (DFT) as a powerful tool for designing new organic corrosion inhibitors. Part 1: An overview. Corros. Sci. 99, 1–30 (2015).
Obot, I. B., Kaya, S., Kaya, C. & Tüzün, B. Density functional theory (DFT) modeling and Monte Carlo simulation assessment of inhibition performance of some carbohydrazide Schiff bases for steel corrosion. Phys. E Low-Dimens. Syst. Nanostruct. 80, 82–90 (2016).
Pearson, R. G. Absolute electronegativity and hardness correlated with molecular orbital theory. Proc. Natl. Acad. Sci. 83(22), 8440–8441 (1986).
Gázquez, J. L. Hardness and softness in density functional theory. In Chemical Hardness (ed. Sen, K. D.) 27–43 (Springer, 1993).
Chattaraj, P. K. & Roy, D. R. Update 1 of: Electrophilicity index. Chem. Rev. 107(9), PR46–PR74 (2007).
Parr, R. G., Szentpály, L. V. & Liu, S. Electrophilicity index. J. Am. Chem. Soc. 121(9), 1922–1924 (1999).
Pal, R. & Chattaraj, P. K. Electrophilicity index revisited. J. Comput. Chem. 44(3), 278–297 (2023).
Bibbins-Domingo, K. et al. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA 315(23), 2564–2575 (2016).
Deore, M. K. & Jain, G. H. Studies on ethanol gas sensing properties of Al2O3-doped ZnO thick films. J. Pet. Sci. Res. 3(2), 60. https://doi.org/10.14355/jpsr.2014.0302.02 (2014).
Shahat, M. A., Dardeer, H. M., Rashwan, G. M., Ghitas, A. & Taha, A. G. Unveiling the synergistic effects of novel β-cyclodextrin co-sensitizers on anthrone-based dye-sensitized solar cells (DSSCs) with microstructural modifications. J. Inorg. Organomet. Polym. Mater. https://doi.org/10.1007/s10904-025-03944-9 (2025).
Shahat, M. A., Ghitas, A. & Ezzat, H. A. Integrated experimental and theoretical insights into CO2 capture using PVA@TiO2@Al2O3 hybrid sorbents for sustainable gas separation. Inorg. Chem. Commun. 182(P2), 115499. https://doi.org/10.1016/j.inoche.2025.115499 (2025).
Shahat, M. A., Ibrahim, M. A., Ghitas, A. & Ezzat, H. A. Advancements in water remediation : Harnessing PAni-based carbon composites for efficient dyes removal. J. Mol. Struct. 1327, 141220. https://doi.org/10.1016/j.molstruc.2024.141220 (2025).
Romcevic, N. et al. Structural and optical properties of ZnO–Al2O3 nanopowders prepared by chemical methods. J. Lumin. 224, 117273 (2020).
Alresheedi, N. M., Ghitas, A., Almutairi, F. N. & Shahat, M. A. Optimizing the efficiency of polymer solar cells based on core-–hell PAni@ ZnO composites utilizing argon plasma treatment. J. Inorg. Organomet. Polym. Mater. 35, 1–18 (2024).
Berthomieu, C. & Hienerwadel, R. Fourier transform infrared (FTIR) spectroscopy. Photosynth. Res. 101(2), 157–170 (2009).
Djebaili, K., Mekhalif, Z., Boumaza, A. & Djelloul, A. XPS, FTIR, EDX, and XRD analysis of Al2O3 scales grown on PM2000 alloy. J. Spectrosc. 2015(1), 868109 (2015).
Ezzat, H. A., Hegazy, M. A., Nada, N. A., Osman, O. & Ibrahim, M. A. Studying the optical and thermal properties of Cs/ZnO and Cs/ZnO/GO hybrid nanocomposites. Opt. Mater. (Amst.) 135, 113244 (2023).
Sakhaei, Z. & Rezaei, M. Mechanochemical synthesis of ZnO. Al2O3 powders with various Zn/Al molar ratios and their applications in reverse water–gas shift reaction. Environ. Sci. Pollut. Res. 28(11), 13790–13799 (2021).
Cai, Z., Park, J. & Park, S. Porous In2O3–ZnO nanofiber-based sensor for ultrasensitive room-temperature detection of toluene gas under UV illumination. J. Mater. Res. Technol. 24, 2482–2499 (2023).
Ezzat, H. A., Sebak, M. A., Aladim, A. K. & Shahat, M. A. Unveiling the dominant role of rGO in tailored PVA@ TiO2–metal oxide nanostructures for high-performance aerospace Lithium-ion batteries. J. Mol. Liq. 437, 128552 (2025).
Ilickas, M. et al. ZnO UV sensor photoresponse enhancement by coating method optimization. J. Photochem. Photobiol. 14, 100171 (2023).
Min, S.-K., Kim, H.-S. & Chang, S.-P. Highly sensitive toluene sensor based on porous core–shell-structured In2O3–ZnO nanofibers under UV irradiation at room temperature. Phys. E Low-Dimens. Syst. Nanostruct. 154, 115806 (2023).
Alresheedi, N. M., Ghitas, A. & Shahat, M. A. Harnessing gamma irradiation to amplify the electrochemical activity of PET/rGO counter electrodes in DSSCs. Electrochim. Acta 541, 147308. https://doi.org/10.1016/j.electacta.2025.147308 (2025).
Wang, T. et al. UV-light enhanced gas sensor based on Ga doped ZnO for ultra-high sensitive and selective n-butanol detection. Appl. Surf. Sci. 641, 158551 (2023).
Shahat, M. A., Ghitas, A., Almutairi, F. N. & Alresheedi, N. M. Oxygen enriched PAni-based counter electrode network toward efficient dye—sensitized solar cells (DSSCs). Sci. Rep. 14, 1–17. https://doi.org/10.1038/s41598-024-67055-0 (2024).
Khan, A., Chen, H.-Y. & Rusly, C. Visible-light-driven removal of mixed dye pollutants by a novel ZnO/CNT/GO ternary nanocomposite: Synergistic degradation of congo red and methylene blue. Environ. Res. 283, 122156 (2025).
Shahat, M. A., Ahmed, Y. M. Z., Ghitas, A. & Soliman, W. Sustainable fired clay bricks with variable TiO2–PET plastic waste content for thermal insulation applications. Electrochim. Acta 541, 147314. https://doi.org/10.1016/j.electacta.2025.147314 (2025).
Aladim, A. K., Sebak, M. A. & Shahat, M. A. Sustainable management of PET waste via oxygen plasma-enriched PET/rGO/TiO2 counter electrodes in DSSCs. J. Environ. Manag. 398, 128549. https://doi.org/10.1016/j.jenvman.2026.128549 (2026).
Zhang, S. et al. High response ZnO gas sensor derived from Tb@Zn-MOFs to acetic acid under UV excitation. Sens. Actuators A Phys. 365, 114862 (2024).
Waghadkar, Y. B. et al. Synthesis and characterization of indium-doped ZnO nanoparticles by coprecipitation method for highly photo-responsive UV light sensors. Sensors Int. 5, 100271 (2024).
Günther, H. NMR Spectroscopy: Basic Principles, Concepts and Applications in Chemistry (Wiley, 2013).
Edwards, J.C. Principles of NMR. In Process NMR Associates LLC, 87A Sand Pit Rd, Danbury CT, vol. 6810 (2009).
Spataro, G., Champouret, Y., Florian, P., Coppel, Y. & Kahn, M. L. Multinuclear solid-state NMR study: A powerful tool for understanding the structure of ZnO hybrid nanoparticles. Phys. Chem. Chem. Phys. 20(18), 12413–12421 (2018).
O’Dell, L. A., Savin, S. L. P., Chadwick, A. V. & Smith, M. E. A 27Al MAS NMR study of a sol–gel produced alumina: Identification of the NMR parameters of the θ-Al2O3 transition alumina phase. Solid State Nucl. Magn. Reson. 31(4), 169–173 (2007).
Pathak, P., Chimerad, M., Borjian, P. & Cho, H. J. In-situ fabrication of CuO/ZnO heterojunctions at room temperature for a self-powered UV sensor. Mater. Today Commun. 39, 109038 (2024).
Shahat, M. A. & Soliman, W. Lightweight thermally insulating fired clay bricks enhanced with chitosan-based clay nanocomposites for sustainable construction. Sci. Rep. 15(1), 26528. https://doi.org/10.1038/s41598-025-11790-5 (2025).
Soliman, W., Ahmed, Y. M. Z., Ghitas, A., El-Shater, A. & Shahat, M. A. Green building development utilising modified fired clay bricks and eggshell waste. Sci. Rep. 15(1), 3367 (2025).
Soliman, W. & Shahat, M. A. Optimizing the thermophysical qualities of innovative clay–rGO composite bricks for sustainable applications. Sci. Rep. 13(1), 1–12. https://doi.org/10.1038/s41598-023-48966-w (2023).
Shahat, M. A., Aladim, A. K., Sebak, M. A. & Ezzat, H. A. Developing innovative PVA@ TiO2-based adsorbents for CO2 capture via multiple metal oxide dopants for sustainable development. Inorg. Chem. Commun. 174, 113947. https://doi.org/10.1016/j.inoche.2025.113947 (2025).
Rashwan, G. M., Ebnalwaled, A. A., Saad, E. M. & Shahat, M. A. Ca/Sn concentration-dependent enhancement of barium titanate ferroelectric performance: A dielectric and microstructural study. J. Sol Gel Sci. Technol. 109, 1–13 (2024).
Sebak, M. A., Aladim, A. K., Mostafa, M. & Shahat, M. A. Multi-level oxygen plasma treatment nanoarchitectonics on chitosan/PVA/TiO2 composite-based absorber layer network for efficient polymer solar cells. J. Inorg. Organomet. Polym. Mater. https://doi.org/10.1007/s10904-024-03542-1 (2024).
Almutairi, F. N., Ghitas, A., Alanazi, H. & Shahat, M. A. Gamma-irradiated chitosan@PVA@TiO2 catalytic counter electrodes for enhanced dye-sensitized solar cell (DSSC) performance. Synth. Met. 311, 117841. https://doi.org/10.1016/j.synthmet.2025.117841 (2025).
Aladim, A. K., Sebak, M. A. & Abdelhamid Shahat, M. Recycling plastic waste into nitrogen plasma-enriched polyethylene Terephthalate@Aluminum oxide counter electrodes for sustainable dye-sensitized solar cells. Environ. Res. 293, 123798. https://doi.org/10.1016/j.envres.2026.123798 (2026).
Shahat, M. A., Rashwan, G. M., Dardeer, H. M., Ghitas, A. & Mahgoub, M. Y. Upcycling PET waste into metal oxide-doped composites for high-performance counter electrodes in dye-sensitized solar cells. Inorg. Chem. Commun. 181, 115261 (2025).
Mostafa, M. M., Aladim, A. K., Sebak, M. A. & Shahat, M. A. Efficient dye-sensitized solar cells (DSSCs) via nitrogen plasma-enriched chitosan@ PVA@ rGO blend-based counter electrode compositions. J. Inorg. Organomet. Polym. Mater. 35, 1–18 (2024).
Padha, B. et al. Ultrasensitive NO2 gas detection using ALD-grown ZnO-SiO2/Si thin film-based UV sensors. J. Alloys Compd. 1010, 177673 (2025).
Sebak, M. A., Aladim, A. K., Mostafa, M. M. & Abdelhamid Shahat, M. Improving the efficiency of polymer solar cells based on chitosan@PVA@rGO composites via gamma-irradiated treatment of rGO nanoparticles. Solid State Sci. 159, 107773. https://doi.org/10.1016/j.solidstatesciences.2024.107773 (2025).
Sahu, S. & Bhattacharjee, M. Nanostructured ZnO thin film-based flexible printed sensor for high-performance UV detection. Sens. Actuators A Phys. 383, 116196 (2025).
Qi, H., Qi, Y., Ye, W. & Lu, C. Hydrothermal synthesis and optical properties of single-crystalline Bi3.15Nd0.85Ti3O12 nanoplates of layered-perovskite structure. J. Nanosci. Nanotechnol. 10(8), 5350–5353 (2010).
Kind, H., Yan, H., Messer, B., Law, M. & Yang, P. Nanowire ultraviolet photodetectors and optical switches. Adv. Mater. 14(2), 158–160 (2002).
Li, Y., Della Valle, F., Simonnet, M., Yamada, I. & Delaunay, J.-J. Competitive surface effects of oxygen and water on UV photoresponse of ZnO nanowires. Appl. Phys. Lett. 94(2), 023110 (2009).
Acknowledgements
This work was supported by the National Research Institute of Astronomy and Geophysics (NRIAG), Egypt.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
M. Abdelhamid Shahat: Writing—original draft, Methodology, Writing—review and editing. Ashraf S. Khamees: Formal Analysis, Validation. Ahmed Ghitas: Supervision. Hend A. Ezzat: Modeling Structure Assignments, Writing—original draft, Writing—review and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Data availability
All data generated or analysed during this study will be available on request (Corresponding Author mohamed.abdelhamid@nriag.sci.eg (M. Abdelhamid Shahat), hend.ezzat@nriag.sci.eg (Hend A. Ezzat).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdelhamid Shahat, M., Khamees, A.S., Ghitas, A. et al. Highly sensitive hierarchically structured Si-based UV sensor–photodetectors via optimized ZnO–Al2O3 nanocomposite architectures. Sci Rep 16, 8497 (2026). https://doi.org/10.1038/s41598-026-38984-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38984-9