Abstract
Prior structural magnetic resonance imaging (MRI) studies that investigated structural alterations in focal dystonia have reported inconsistent findings, potentially due to methodological limitations or sample heterogeneity. This work investigates gray and white matter changes in patients with upper limb dystonia using T1-weighted images and diffusion tensor imaging (DTI). Imaging data collected with MRI at 3T from 28 right-handed individuals with right upper limb dystonia and 29 healthy controls were analyzed. T1-weighted images were analyzed using FreeSurfer to examine cortical thickness, volume and area. DTI data were assessed using tract-based spatial statistics, regions of interest (ROI) analysis and probabilistic tractography. ROI included the basal ganglia, thalamus and cerebellum. Between-group differences were tested using permutation-based statistics. Whole-brain vertex-wise analyses revealed no significant differences in cortical morphology between groups. Tract and ROI-based analyses showed no changes in DTI metrics. These findings indicate preserved gray and white matter microstructural integrity in individuals with focal upper limb dystonia compared to controls, although finer abnormalities might still become evident using higher-resolution imaging methods. The absence of significant findings suggests potential for neuromodulation therapies and network-based approaches in this condition. Larger, multi-modal studies are warranted to further characterize anatomical substrates in focal dystonia.
Similar content being viewed by others
Introduction
Dystonia is a complex movement disorder characterized by involuntary muscle contractions, leading to repetitive movements, twisting, and abnormal postures1, which significantly impact the quality of life through pain, functional impairment, and motor disability. Despite extensive research, the pathophysiology of dystonia remains elusive2, though it is increasingly recognized as a network disorder. This perspective is supported by evidence showing that deep brain stimulation (DBS), in dystonia and Parkinson’s disease, can restore altered connectivity within cortico-striato-thalamo-cerebellar-cortical circuitry, thus improving clinical outcomes as demonstrated by both symptomatic relief and functional magnetic resonance (MRI) findings3,4.
Nevertheless, structural MRI studies exploring dystonia present a fragmented picture, reporting variable changes in gray matter volume (GMV), cortical thickness, and white matter integrity, particularly in regions related to motor control and sensorimotor integration5. While some studies reported changes like gray matter volume increases in sensorimotor cortices6,7 or the basal ganglia8, others found GMV decreases in regions like the thalamus and cerebellum9. Similarly, diffusion MRI studies reveal disparate findings regarding white matter changes in dystonia, with findings of decreased fractional anisotropy (FA) in the corticospinal tract10,11 or no significant differences12.
This inconsistency likely stems from dystonia’s inherent heterogeneity, which manifests across various subtypes and etiologies, complicating the task of characterizing and categorizing patients effectively. This diversity among subtypes, each potentially exhibiting distinct functional or structural peculiarities13, underscores the challenge of deriving generalized conclusions from the existing body of research. By concentrating on focal dystonia—particularly focal upper limb dystonia—a more consistent pattern of structural changes might be discerned. This focused approach can help minimize the variability introduced by the broad spectrum of dystonia subtypes, providing clearer insights into the pathophysiology underlying these disorders.
Moreover, most structural MRI studies in dystonia have utilized voxel-based morphometry (VBM) method6,7,8,9,13, criticized for its susceptibility to false positives14. In response to these limitations, recent dystonia research has begun to incorporate alternative morphometry techniques, such as surface-based morphometry (SBM) and tractography using diffusion tensor imaging (DTI), to explore white matter tracts10,11,12,13,15, however, the results remain inconsistent11. Table 1 summarizes key MRI studies focusing on upper limb dystonia, showcasing the diversity of findings and methodologies applied.
Given the complexities and existing gaps in dystonia research, there’s a pressing need for studies with greater methodological rigor, utilizing standardized techniques and well-defined, uniform samples to better understand dystonia’s varied presentations. Building upon our previous work16, our study seeks to illuminate the structural changes in a homogeneous group of patients with upper limb dystonia using DTI and T1-weighted surface-based morphometry MRI. By conducting a comprehensive analysis that includes whole-brain exploratory assessments, region-of-interest (ROI), and tract-of-interest analyses, we aim to delve into the structural alterations in white and gray matter, thereby advancing our understanding of focal upper limb dystonia’s pathophysiology.
Methods
Subjects
Thirty patients with focal right-upper limb dystonia were enrolled by neurologists at specialized movement disorders centers or through advertisements between 2017 and 2021. Additionally, 30 healthy controls were recruited via advertisements and convenience sampling. Inclusion criteria included an age range of 18 to 60 years and a minimum of 8 years of formal education. All subjects were right-handed, as determined by self-report and confirmed using the Edinburgh Handedness Inventory. The diagnosis of idiopathic focal dystonia was established according to current recommendations1, including a prerequisite of normal brain image exam (MRI or computer tomography scan) prior to recruitment. Exclusion criteria encompassed neurological conditions (except dystonia for the patient group), history of neurosurgery, moderate to severe head or upper limb trauma, cancer, uncontrolled metabolic disorders, current use of specific medications (anticholinergics, neuroleptics, antidepressants, benzodiazepines, vasodilators, or vasoconstrictors), and substance abuse history. Patients undergoing botulinum toxin treatment were required to wait for at least three months post-injection to participate. Furthermore, the severity and duration of dystonia in the patients were assessed using the Burke-Fahn-Marsden Dystonia Rating Scale. This study was approved by the Institutional Review Boards of all participating centers: Hospital Israelita Albert Einstein, Hospital Servidor Público Estadual de São Paulo, and Department of Neurology and Neurosurgery of Universidade Federal de São Paulo (UNIFESP), obtaining approval number 2.004.139. All methods were conducted in accordance with the relevant ethical guidelines and regulations, including the applicable national standards for research involving human participants. The datasets generated and analysed during the current study are available in the Open Neuro repository, [https://openneuro.org/datasets/ds006395/versions/1.0.1].
Imaging acquisition
MRI images were acquired on a Siemens 3.0T Tim Trio system [102 × 32 RF channels] using a 32-channel head coil. Structural images T1 sequence: Rapid gradient-echo (MPRAGE), repetition time (TR): 2500 ms, echo time (TE) 3.47 ms, Inversion Time (TI): 1.100 ms, 192 slices of 1 mm of thickness without gap, and matrix of 256 × 256 resulting in an isotropic image of 1.0 × 0.5 × 0.5 × mm, acquisition time (TA) 2:34 min.
DTI images were obtained from 60 non-collinear directions with a b value of 1000 s/mm2, along with nine non-diffusion weighted (b0) images. We used the maximal gradient strength of 45 mT/m in a monopolar (phase encoding direction: anterior-to-posterior) single shot, spin-echo diffusion-weighted echoplanar pulse sequence with 60 axial slices, covering the whole brain at 2-mm slice thickness (TR/TE: 3151/84.2 ms; acquisition matrix 128 × 128; FOV: 260 mm2; slice spacing: 2 mm; average: 1; TA: 3:25 min). Description of data preprocessing is available in the Supplementary Material.
Whole brain analysis
Comparisons between groups for DTI and T1-weighted images covering the entire brain were conducted using a non-parametric permutation-based method via the Permutation Analysis of Linear Models (PALM)19, with 10,000 permutations. Data were controlled for the effects of age and sex. In the dystonia group, we investigated the influence of disease duration and severity as measured by the Burke-Fahn-Marsden (BFM) scale. The test level was set at α = 0.05, with false discovery rate (FDR) correction17. Given that the joint combination of cortical thickness and area has been shown to be a more sensitive method to detect cortical morphological changes18, we used the nonparametric combination (NPC) of area and cortical thickness to investigate group differences on cortical morphology. For DTI, voxelwise statistical analysis of the AD, FA, MD and RD data was carried out using Tract-based spatial statistics (TBSS).
Volumetric region of interest (ROI) analysis
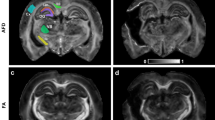
For non-cortical ROI, we examined the volumetric differences in the bilateral putamen, caudate, globus pallidus, thalamus, and cerebellar gray matter (Fig. 1). Factors such as estimated intracranial volume, sex, and age were incorporated as covariates in our analytical model. We employed permutation testing for statistical inference, with a total of 10,000 permutations conducted. The significance level was established at α = 0.05, adjusted for false discovery rate (FDR) correction.
FreeSurfer Subcortical Segmentation Regions of Interest (ROI) of one subject. ROI include bilateral Cerebellum cortex (orange), Thalamus (green), Caudate (light blue), Putamen (pink), and Pallidum (purple). The top-left image presents a sagittal view, the top-right displays a coronal view, the bottom-left shows an axial view.
Probabilistic tractography
We employed XTRACT, a command-line tool based on FSL, for automated probabilistic tractography (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/XTRACT). This allowed us to compare the mean Fractional Anisotropy (FA) values extracted from subjects’ diffusion data. XTRACT software streamlined the automated extraction of ROIs from DTI data19. The resulting tracts were transformed to MNI space for group-level analysis. These ROIs were specifically chosen to encompass critical white matter tracts relevant to the pathophysiology of dystonia, based on both anatomical and functional knowledge of the disease, as documented in previous research9,11. The targeted tracts included bilateral anterior thalamic radiation, superior thalamic radiation, superior longitudinal fasciculus, corticospinal tract, middle cerebellar peduncle, and corpus callosum (including the forceps major and minor), as depicted in Fig. 2. Age and sex were included in the model as covariates. Statistical analysis used permutation tests, with 10,000 permutations, to test group differences. Significance threshold was set at α = 0.05, with FDR correction.
Analyzed Tracts with FSL’s XTRACT. Tracts under investigation include Superior Thalamic Radiation (light blue), Anterior Thalamic Radiation (orange), Longitudinal Fascicle (dark blue), Middle Cerebellar Peduncle (yellow), Forceps Minor (pink), Forceps Major (dark red), and Corticospinal Tract (green). Tracts are overlaid on a standard MNI template across axial, coronal, and sagittal planes.
Results
Subjects’ characteristics
From our dataset of 30 patients and 30 healthy controls, two patients with dystonia were excluded due to incidental MRI findings (microangiopathy and a brain tumor). Additionally, one control was excluded due to technical acquisition problems. Consequently, our MRI sample comprised 28 patients (19 females) and 29 controls (16 females). The median age for dystonia patients was 45.5, ranging from 19 to 60 years, while the control group had a median age of 37, ranging from 19 to 58 years. Importantly, no significant age differences were observed between patients and controls (p = 0.11). Among the patients, eleven had task-specific dystonia (writer’s cramp). The median disease duration was 10 years, ranging from 1 to 45 years, and the median score for the Burke–Fahn–Burk–Marsden dystonia scale was 2, ranging from 1 to 12.
Whole brain analysis
For T1, the vertexwise analysis did not yield significant results for area, volume or thickness at p-value < 0.05. Notably, neither disease duration nor dystonia severity were significantly associated with cortical differences in these metrics. For DTI, TBSS yielded no statistically significant differences between groups were found based on FA, AD, MD and RD at p < 0.05.
Volumetric ROI analysis
No volume differences between patients and controls were observed in the analyzed ROI. Detailed descriptive and statistical test results for each ROI can be found in Supplementary Table S1. No correlations between ROI volume and Burke-Fahn-Marsden scores were observed.
Probabilistic tractography
Two subjects were excluded due to diffusion tractography failure attributed to motion-related distortion during acquisition. No significant differences were detected between patients and controls in terms of mean FA in the analyzed tracts-of-interest. Detailed descriptive and statistical test results for each tract can be found in Supplementary Tables S1 and S2.
Discussion
Our study aimed to compare structural changes in individuals with upper limb dystonia and healthy controls in terms of both white matter tracts and gray matter. We conducted whole-brain surface analysis of gray matter and employed tract-based spatial statistics for the white matter tracts. Additionally, we used ROI analysis to investigate differences in the basal ganglia volume and diffusion metrics in the corpus callosum, the thalamic radiations (anterior and posterior), as well as in the superior longitudinal fasciculus using probabilistic tractography.
Our findings align with other studies indicating that there are no significant structural changes in focal upper limb dystonia12. Notably, to account for potential variations within each focal dystonia subtype, we employed a homogeneous sample of individuals with focal upper limb dystonia affecting the right limb. We also utilized optimal preprocessing and rigorous permutation-based methods for statistical inference. Moreover, we meticulously controlled for sex and age variables across all analyses. Specifically, in our analyses of subcortical ROIs, we carefully adjusted for total intracranial volume to ensure the accuracy of our findings.
Our study builds upon the pioneering research by Tomić et al.11 and stands as one of the few endeavors to apply a surface-based analysis approach in the exploration of focal upper limb dystonia, albeit featuring a notably larger sample size. Despite conducting a comprehensive whole-cortex analysis, we did not identify any significant differences between patients and controls in metrics such as cortical area, volume, or thickness. Our choice of FreeSurfer as the analysis tool was motivated by its proven efficiency in reducing false positives, a known limitation of voxel-based morphometry (VBM)20. Prior research on focal upper limb dystonia has predominantly relied on VBM, which may have contributed to inconsistent findings regarding volumetric changes in key areas such as the thalamus, basal nuclei, cerebellum, and cortex—regions recognized as nodes within the dystonic network5,13. Furthermore, when both FreeSurfer and VBM techniques were applied to the same sample, they produced divergent findings11, underscoring the substantial impact of the chosen analysis tool on study outcomes.
Regarding DTI, our whole brain analysis using TBSS and tractography-based analysis did not reveal significant differences. White matter tract alterations in focal upper limb dystonia have been the focus of previous studies, yielding likewise varying and occasionally conflicting findings6,7,8,9. For example, Tomić et al.11 reported decreased mean FA in several white matter tracts in patients with focal upper limb dystonia, while Merchant et al.12 found no differences between patients and controls.
Although our current findings did not uncover any structural brain alterations in focal dystonia, there is a potential avenue for future research to delve into the microstructural changes within the basal ganglia. A recent neuroimaging study suggests the feasibility of in vivo parcellation of the matrix and striosome striatal compartments using DTI tractography21. Imbalances between striosome and matrix functions have been implicated in basal ganglia disorders22. Notably, a previous investigation utilizing DTI tractography to assess striosome and matrix integrity in X-linked dystonia-parkinsonism reported increased striosomal afferents to the putamen23. However, it remains unclear whether non-degenerative dystonias are similarly affected. Higher resolution MRI techniques, such as Diffusion Kurtosis Imaging, aimed at detailed examination of the basal ganglia’s microstructure, could provide fresh perspectives on the underlying mechanisms of dystonia.
While some evidence, such as anomalies in the volume of specific brain regions, suggests potential structural alterations8,11,13 a growing number of studies consistently emphasize disruptions in functional connectivity across various brain regions as the most pivotal factor in the pathophysiology of dystonias16,24,25,26. These functional alterations contribute to the hypothesis of an altered network that influences dystonia symptoms, underscoring the necessity of therapeutic tools such as DBS to reestablish balance. In this context, the absence of significant structural differences in focal upper limb dystonia can be viewed as a positive perspective, particularly in the context of neuromodulation therapies4. DTI markers may serve as predictors for DBS intervention, as preserved structural connectivity to subcortical nuclei, sensorimotor regions, and frontal regions has been associated with improved motor outcomes4, whereas subcortical atrophy has often been linked to poorer motor outcomes27.
Limitations
In the field of neuroimaging, achieving statistically robust findings often demands a sufficiently sized sample28. While our study had a larger sample compared to prior investigations focusing on structural disparities in upper limb dystonia, it is important to emphasize that our sample is still relatively small. Studies with larger samples and meta-analyses could further elucidate the possibility of focal imaging changes in dystonia. Moreover, while the BFM scale is a standard instrument for the clinical evaluation of dystonia, it lacks sufficient detail to capture the complexity of upper limb dystonia, and its severity scores may not accurately reflect the degree of disability. Dystonia heterogeneity is another issue to be considered: task-specific forms such as musician’s dystonia may show increased gray matter volume due to extensive training leading to sensorimotor reorganization rather than dystonia-related primary pathology29. As only 11 of 30 patients had task-specific dystonia, this variability should be considered when interpreting the findings.
Conclusions
Our study demonstrates that individuals with upper limb dystonia do not exhibit significant structural brain alterations compared to controls. Leveraging state-of-the-art neuroimaging processing and rigorous statistical analyses, we were unable to replicate previously reported findings in the literature. This observation can be viewed as a positive outcome, as the preservation of microstructure may represent a favorable characteristic for treatment based on neuromodulation.
Data availability
The datasets generated and analysed during the current study are available in the Open Neuro repository, [[https://openneuro.org/datasets/ds006395/versions/1.0.1](https://openneuro.org/datasets/ds006395/versions/1.0.1)].
References
Albanese, A. et al. Phenomenology and classification of dystonia: A consensus update. Mov. Disord. 28, 863–873 (2013).
Breakefield, X. O. et al. The pathophysiological basis of dystonias. Nat. Rev. Neurosci. 9, 222–234 (2008).
Shen, L. et al. Subthalamic nucleus deep brain stimulation modulates 2 distinct neurocircuits. Ann. Neurol. 88, 1178–1193 (2020).
Andrews, L., Keller, S. S., Osman-Farah, J. & Macerollo, A. A structural magnetic resonance imaging review of clinical motor outcomes from deep brain stimulation in movement disorders. Brain Commun. 5, fcad171 (2023).
Zoons, E., Booij, J., Nederveen, A. J., Dijk, J. M. & Tijssen, M. A. J. Structural, functional and molecular imaging of the brain in primary focal dystonia—a review. Neuroimage 56, 1011–1020 (2011).
Garraux, G. et al. Changes in brain anatomy in focal hand dystonia. Ann. Neurol. 55, 736–739. https://doi.org/10.1002/ana.20113 (2004).
Egger, K. et al. Voxel based morphometry reveals specific Gray matter changes in primary dystonia. Mov. Disord. 22, 1538–1542 (2007).
Zeuner, K. E. et al. Increased volume and impaired function: The role of the basal ganglia in writer’s cramp. Brain Behav. 5, e00301 (2015).
Delmaire, C. et al. Structural abnormalities in the cerebellum and sensorimotor circuit in writer’s cramp. Neurology 69, 376–380 (2007).
Berndt, M., Li, Y., Gora-Stahlberg, G., Jochim, A. & Haslinger, B. Impaired white matter integrity between premotor cortex and basal ganglia in writer’s cramp. Brain Behav. 8, e01111 (2018).
Tomić, A. et al. Brain structural changes in focal dystonia—what about task specificity? A multimodal MRI study. Mov. Disord. 36, 196–205. https://doi.org/10.1002/mds.28304 (2021).
Merchant, S. H. I. et al. The role of the inferior parietal lobule in writer’s cramp. Brain 143, 1766–1779 (2020).
Bianchi, S., Fuertinger, S., Huddleston, H., Frucht, S. J. & Simonyan, K. Functional and structural neural bases of task specificity in isolated focal dystonia. Mov. Disord. 34, 555–563 (2019).
Bookstein, F. L. Voxel-based morphometry should not be used with imperfectly registered images. Neuroimage 14, 1454–1462 (2001).
Delmaire, C. et al. Diffusion abnormalities in the primary sensorimotor pathways in writer’s cramp. Arch. Neurol. 66, 502–508 (2009).
de Faria, D. D. et al. Task-related brain activity and functional connectivity in upper limb dystonia: A functional magnetic resonance imaging (fMRI) and functional near-infrared spectroscopy (fNIRS) study. Neurophotonics 7, 45004 (2020).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. Roy. Stat. Soc. Ser. B (Methodol.). 57, 289–300 (1995).
Winkler, A. M. et al. Joint analysis of cortical area and thickness as a replacement for the analysis of the volume of the cerebral cortex. Cereb. Cortex. 28, 738–749. https://doi.org/10.1093/cercor/bhx308 (2018).
Warrington, S. et al. XTRACT—Standardised protocols for automated tractography in the human and macaque brain. Neuroimage 217, 116923. https://doi.org/10.1016/j.neuroimage.2020.116923 (2020).
Greve, D. N. & Fischl, B. False positive rates in surface-based anatomical analysis. Neuroimage 171, 6–14 (2018).
Waugh, J. L. et al. An MRI method for parcellating the human striatum into matrix and striosome compartments in vivo. Neuroimage 246, 118714 (2022).
Crittenden, J. R. & Graybiel, A. M. Basal ganglia disorders associated with imbalances in the striatal striosome and matrix compartments. Front. Neuroanat. 5, 59 (2011).
Blood, A. J. et al. Increased insula-putamen connectivity in X-linked dystonia-parkinsonism. Neuroimage Clin. 17, 835–846 (2018).
Delnooz, C. C. S., Helmich, R. C., Toni, I. & van de Warrenburg, B. P. C. Reduced parietal connectivity with a premotor writing area in writer’s cramp. Mov. Disord. 27, 1425–1431 (2012).
Dresel, C. et al. Multiple changes of functional connectivity between sensorimotor areas in focal hand dystonia. J. Neurol. Neurosurg. Psychiatry. 85, 1245–1252 (2014).
Gallea, C. et al. Loss of Inhibition in sensorimotor networks in focal hand dystonia. Neuroimage Clin. 17, 90–97 (2018).
Hamed, M. A., Ponce, F. A., Lambert, M. & Moguel-Cobos, G. Subcortical atrophy and motor outcomes in pallidal deep brain stimulation for Parkinson disease. World Neurosurg. 142, e89–e94 (2020).
Szucs, D. & Ioannidis, J. P. A. Sample size evolution in neuroimaging research: An evaluation of highly-cited studies (1990–2012) and of latest practices (2017–2018) in high-impact journals. Neuroimage 221, 117164 (2020).
Mantel, T. et al. Abnormalities in grey matter structure in embouchure dystonia. Parkinsonism Relat. Disord. 65, 111–116 (2019).
Acknowledgements
This study was supported by grant #2021/14108-4, São Paulo Research Foundation (FAPESP) and Coordination for the Improvement of Higher Education Personnel (CAPES).
Funding
This study was supported by grant São Paulo Research Foundation (FAPESP).
Author information
Authors and Affiliations
Contributions
Author contributions included conception and study design (DDF and PCA), data collection or acquisition (DDF, AJMP, VB, SMA, HBF and PCA), statistical analysis (DDF, AJMP, JPQP and AMW), interpretation of results (All authors), drafting the manuscript work or revising it critically for important intellectual content (All authors) and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (All authors).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical compliance statement
This study was approved by the Institutional Review Boards of all participating centers: Hospital Israelita Albert Einstein, Hospital Servidor Público Estadual de São Paulo, and Department of Neurology and Neurosurgery of Universidade Federal de São Paulo (UNIFESP), obtaining approval number 2.004.139. All subjects provided informed written consent. We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
de Faria, D.D., Paulo, A.J.M., de Paiva, J.P.Q. et al. Structural brain analysis in focal upper limb dystonia. Sci Rep 16, 9112 (2026). https://doi.org/10.1038/s41598-026-39542-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-39542-z