Abstract
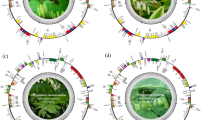
Chloroplast genomes provide conserved yet informative sequences useful for inferring plant evolution and species identification. Genus Aconitum consists of around 300 traditional Indian and Chinese medicinal plant species, many native to mountainous regions, and known to be highly poisonous due to toxic diterpene alkaloids. Their accurate identification and classification are vital for traditional medicine systems, particularly for ensuring their safe use. A consistent quadripartite structure was identified across all chloroplast genomes, comprising the typical large single copy (LSC), small single copy (SSC), and two inverted repeats (IR) regions. Pan-plastome analysis unveiled 72 core and nine accessory genes, indicating an open pan-plastome characteristic. In-depth nucleotide-level homology analysis revealed that homologous genes of all accessory genes are present in all other genomes, implying the need for better chloroplast genome annotation tools that can identify all putative genes from such conserved genomes. Notably, the order of all core and accessory genes remained highly conserved across all analysed genomes, underscoring the overall evolutionary stability despite the diversity of accessory genes. Members of some core pathways are relatively absent on the chloroplast genome, suggesting their potential presence on the nuclear genome, which will be revealed after their nuclear genome sequencing. Our phylogenetic results largely supported the morphological classification, with distinct Lycoctonum and Aconitum subgenera clustering, further validating the gross accuracy except for A. flavum, suggesting a putative morphological classification discrepancy or inaccurate classification. This comparative analysis reveals a highly conserved chloroplast genome architecture across Aconitum while documenting measurable plastome-level variation among 73 species. The chloroplast phylogeny highlights instances of non-monophyletic clustering among conspecific accessions and unexpected placement of certain taxa, indicating complex evolutionary histories within the genus. Such patterns may reflect a combination of processes, including incomplete lineage sorting, chloroplast capture, or sample misidentification, rather than definitive phylogenetic incongruence. While chloroplast genomes alone cannot fully resolve evolutionary relationships in a group shaped by hybridization and polyploidy, the results provide valuable insights into plastome evolution and identify key lineages and taxa that warrant further investigation using nuclear genomic and integrative approaches.
Similar content being viewed by others
Data availability
Data is provided within the manuscript or supplementary information files. Authors have used open-source tools in this analysis. All tool versions have been provided in the methodology.
References
Ameri, A. The effects of Aconitum alkaloids on the central nervous system. Prog. Neurobiol. 56(2), 211–235. https://doi.org/10.1016/S0301-0082(98)00037-9 (1998).
Singh, M. et al. In vitro propagation and phytochemical assessment of Aconitum ferox wall: a threatened medicinal plant of Sikkim Himalaya. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 90(2), 313–321. https://doi.org/10.1007/s40011-019-01104-x (2020).
Rafiq, S. et al. In vitro propagation of Aconitum chasmanthum Stapf Ex Holmes: an endemic and critically endangered plant species of the Western Himalaya. Horticulturae https://doi.org/10.3390/horticulturae7120586 (2021).
Hong, Y. et al. Phylogeny and reclassification of Aconitum subgenus Lycoctonum (Ranunculaceae). PLoS ONE 12(1), e0171038. https://doi.org/10.1371/journal.pone.0171038 (2017).
Kita, Y., Ueda, K. & Kadota, Y. Molecular phylogeny and evolution of the Asian Aconitum subgenus Aconitum (Ranunculaceae). J. Plant Res. 108(4), 429–442. https://doi.org/10.1007/BF02344231 (1995).
Lauener, L. A. A synopsis of Aconitum subgenus paraconitum: I. Notes R. Bot. Gard. Edinburgh 37(1), 113–124. https://doi.org/10.24823/nrbge.1978.3139 (1978).
Luo, Y. & Yang, Q. E. Taxonomic revision of Aconitum (Ranunculaceae) from Sichuan, China. Acta Phytotaxon. Sin. 43(4), 289–386. https://doi.org/10.1360/aps040102 (2005).
Yuan, Q. & Yang, Q. E. Polyploidy in Aconitum subgenus Lycoctonum (Ranunculaceae). Bot. J. Linn. Soc. 150(3), 343–353. https://doi.org/10.1111/j.1095-8339.2006.00468.x (2006).
Wang, W., Liu, Y., Yu, S. X., Gao, T. G. & Chen, Z. D. Gymnaconitum, a new genus of Ranunculaceae endemic to the Qinghai-Tibetan plateau. Taxon 62(4), 713–722. https://doi.org/10.12705/624.10 (2013).
He, J., Ka Lok, W., Shaw, P.-C., Wang, H. & Li, D.-Z. Identification of the medicinal plants in Aconitum L. by DNA barcoding technique. Planta Med. 76, 1622–1628. https://doi.org/10.1055/s-0029-1240967 (2010).
Park, I. et al. The complete chloroplast genome sequence of Aconitum coreanum and Aconitum carmichaelii and comparative analysis with other Aconitum species. PLoS ONE 12(9), e0184257. https://doi.org/10.1371/journal.pone.0184257 (2017).
Sun, J. et al. Species identification and genetic diversity analysis of medicinal plants Aconitum pendulum Busch and Aconitum flavum Hand.-Mazz. Plants https://doi.org/10.3390/plants13060885 (2024).
Kakkar, R. A., Haneen, M. A., Parida, A. C. & Sharma, G. The known, unknown, and the intriguing about members of a critically endangered traditional medicinal plant genus Aconitum. Front. Plant Sci. 14, 1–25. https://doi.org/10.3389/fpls.2023.1139215 (2023).
Wang, G. et al. Molecular marker development and phylogenetic analysis of Aconitum species based on chloroplast genomes. Ind. Crops Prod. 221, 119386. https://doi.org/10.1016/j.indcrop.2024.119386 (2024).
Qu, X.-J., Moore, M. J., Li, D.-Z. & Yi, T.-S. PGA: a software package for rapid, accurate, and flexible batch annotation of plastomes. Plant Methods 15(1), 50. https://doi.org/10.1186/s13007-019-0435-7 (2019).
Lechner, M. et al. Proteinortho: detection of (Co-) orthologs in large-scale analysis. BMC Bioinform. 12(1), 124. https://doi.org/10.1186/1471-2105-12-124 (2011).
Greiner, S., Lehwark, P. & Bock, R. OrganellarGenomeDRAW (OGDRAW) version 1.3.1: expanded toolkit for the graphical visualization of organellar genomes. Nucleic Acids Res. 47(W1), W59–W64. https://doi.org/10.1093/nar/gkz238 (2019).
Wang, X. & Wang, L. GMATA: An integrated software package for genome-scale SSR mining, marker development and viewing. Front. Plant Sci. 7, 1–11. https://doi.org/10.3389/fpls.2016.01350 (2016).
Grant, J. R., Arantes, A. S. & Stothard, P. Comparing thousands of circular genomes using the CGView comparison tool. BMC Genomics 13, 202. https://doi.org/10.1186/1471-2164-13-202 (2012).
Wang, Y. et al. Comparative analysis of codon usage patterns in chloroplast genomes of ten Epimedium species. BMC Genomic Data 24(1), 3. https://doi.org/10.1186/s12863-023-01104-x (2023).
Rice, P., Longden, I. & Bleasby, A. EMBOSS: the European molecular biology open software suite. Trends Genet. 16(6), 276–277. https://doi.org/10.1016/s0168-9525(00)02024-2 (2000).
Puigbò, P., Bravo, I. G. & Garcia-Vallve, S. CAIcal: A combined set of tools to assess codon usage adaptation. Biol. Direct 3(1), 38. https://doi.org/10.1186/1745-6150-3-38 (2008).
Deng, W., Wang, Y., Liu, Z., Cheng, H. & Xue, Y. HemI: a toolkit for illustrating heatmaps. PLoS ONE 9(11), e111988. https://doi.org/10.1371/journal.pone.0111988 (2014).
Xia, X. & Xie, Z. DAMBE: software package for data analysis in molecular biology and evolution. J. Hered. 92(4), 371–373. https://doi.org/10.1093/jhered/92.4.371 (2001).
Shi, S.-L., Liu, Y.-Q., Xia, R.-X. & Qin, L. Comprehensive analysis of codon usage in Quercus chloroplast genome and focus on psbA gene. Genes (Basel) https://doi.org/10.3390/genes13112156 (2022).
Rozas, J. et al. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 34(12), 3299–3302. https://doi.org/10.1093/molbev/msx248 (2017).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32(5), 1792–1797. https://doi.org/10.1093/nar/gkh340 (2004).
Minh, B. Q. et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 37(5), 1530–1534. https://doi.org/10.1093/molbev/msaa015 (2020).
Letunic, I. & Bork, P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 49(W1), W293–W296. https://doi.org/10.1093/nar/gkab301 (2021).
Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 16(2), 111–120. https://doi.org/10.1007/BF01731581 (1980).
Yang, Y. et al. Plastid genome comparative and phylogenetic analyses of the key genera in fagaceae: Highlighting the effect of codon composition bias in phylogenetic inference. Front. Plant Sci. 9, 1–13. https://doi.org/10.3389/fpls.2018.00082 (2018).
Schwarz, E. N. et al. Plastid genome sequences of legumes reveal parallel inversions and multiple losses of rps16 in papilionoids. J. Syst. Evol. 53(5), 458–468. https://doi.org/10.1111/jse.12179 (2015).
Wang, Z.-K., Liu, Y., Zheng, H.-Y., Tang, M.-Q. & Xie, S.-Q. Comparative analysis of codon usage patterns in nuclear and chloroplast genome of Dalbergia (Fabaceae). Genes (Basel) https://doi.org/10.3390/genes14051110 (2023).
Wang, Z. et al. Comparative analysis of codon bias in the chloroplast genomes of Theaceae species. Front. Genet. 13, 1–20. https://doi.org/10.3389/fgene.2022.824610 (2022).
Wright, F. The ‘effective number of codons’ used in a gene. Gene 87(1), 23–29. https://doi.org/10.1016/0378-1119(90)90491-9 (1990).
Li, F. et al. Comparative analysis of chloroplast genome structure and phylogenetic relationships among six taxa within the genus Catalpa (Bignoniaceae). Front. Genet. 13, 1–15. https://doi.org/10.3389/fgene.2022.845619 (2022).
He, B. et al. Analysis of codon usage patterns in Ginkgo biloba reveals codon usage tendency from A/U-ending to G/C-ending. Sci. Rep. 6, 1–11. https://doi.org/10.1038/srep35927 (2016).
Khandia, R. et al. Codon usage bias correlates with gene length in neurodegeneration associated genes. Front. Neurosci. 16, 895607. https://doi.org/10.3389/fnins.2022.895607 (2022).
Kong, W. Q. & Yang, J. H. The complete chloroplast genome sequence of Morus cathayana and Morus multicaulis, and comparative analysis within genus Morus L.. PeerJ 5, e3037. https://doi.org/10.7717/peerj.3037 (2017).
Wang, Z. et al. Comparative analysis of codon usage patterns in chloroplast genomes of six Euphorbiaceae species. PeerJ 8, e8251. https://doi.org/10.7717/peerj.8251 (2020).
Yengkhom, S., Uddin, A. & Chakraborty, S. Deciphering codon usage patterns and evolutionary forces in chloroplast genes of Camellia sinensis var. assamica and Camellia sinensis var. sinensis in comparison to Camellia pubicosta. J. Integr. Agric. 18(12), 2771–2785. https://doi.org/10.1016/S2095-3119(19)62716-4 (2019).
Berg, M. D. & Brandl, C. J. Transfer RNAs: diversity in form and function. RNA Biol. 18(3), 316–339. https://doi.org/10.1080/15476286.2020.1809197 (2021).
Grosjean, H., de Crécy-Lagard, V. & Marck, C. Deciphering synonymous codons in the three domains of life: co-evolution with specific tRNA modification enzymes. FEBS Lett. 584(2), 252–264. https://doi.org/10.1016/j.febslet.2009.11.052 (2010).
Xia, C., Wang, M., Guan, Y. & Li, J. Comparative analysis of the chloroplast genome for Aconitum species: genome structure and phylogenetic relationships. Front. Genet. 13, 878182. https://doi.org/10.3389/fgene.2022.878182 (2022).
Yang, M. et al. Analysis of codon usage patterns in 48 Aconitum species. BMC Genomics 24, 1–13 (2023).
Kong, H., Liu, W., Yao, G. & Gong, W. A comparison of chloroplast genome sequences in Aconitum (Ranunculaceae): a traditional herbal medicinal genus. PeerJ https://doi.org/10.7717/peerj.4018 (2017).
Yanfei, N., Tai, S., Chunhua, W., Jia, D. & Fazhong, Y. Complete chloroplast genome sequences of the medicinal plant Aconitum transsectum (Ranunculaceae): comparative analysis and phylogenetic relationships. BMC Genomics 24(1), 90. https://doi.org/10.1186/s12864-023-09180-0 (2023).
Cauz-Santos, L. A. Beyond conservation: the landscape of chloroplast genome rearrangements in angiosperms. New Phytol. 247(6), 2571–2580. https://doi.org/10.1111/nph.70364 (2025).
A. Gül, M. Karaca, A. N. Onus, and M. Bilgen, Chloroplast matK gene phylogeny of some important species of plants. 18 (2), 157–162. http://www.ncbi.nlm.nih.gov/Genbank/ind. (2005).
Barthet, M. M. & Hilu, K. W. Expression of matK: Functional and evolutionary implications. Am. J. Bot. 94(8), 1402–1412. https://doi.org/10.3732/ajb.94.8.1402 (2007).
Barthet, M. M., Pierpont, C. L. & Tavernier, E. K. Unraveling the role of the enigmatic MatK maturase in chloroplast group IIA intron excision. Plant Direct 4(3), 1–17. https://doi.org/10.1002/pld3.208 (2020).
Lee, K.-H., Kwon, K.-R., Kang, W.-M., Jeon, E.-M. & Jang, J.-H. Identification and analysis of the chloroplast rpoC1 gene differentially expressed in wild ginseng. J. Pharmacopuncture 15(2), 20–23. https://doi.org/10.3831/KPI.2012.15.2.020 (2012).
Shikanai, T. et al. The chloroplast clpP gene, encoding a proteolytic subunit of ATP-dependent protease, is indispensable for chloroplast development in tobacco. Plant Cell Physiol. 42(3), 264–273. https://doi.org/10.1093/pcp/pce031 (2001).
Zhu, Z. et al. Development of molecular markers for marker-assisted breeding and quality evaluation of Aconitum carmichaelii cultivars. BMC Plant Biol. 25(1), 1373. https://doi.org/10.1186/s12870-025-07289-w (2025).
Sutkowska, A., Boroń, P., Warzecha, T., Dębowski, J. & Mitka, J. Hybridization and introgression among three Aconitum (Ranunculaceae) species of different ploidy levels in the Tatra Mountains (Western Carpathians). Plant Species Biol. 32(4), 292–303. https://doi.org/10.1111/1442-1984.12162 (2017).
Gonçalves, D. J. P., Simpson, B. B., Ortiz, E. M., Shimizu, G. H. & Jansen, R. K. Incongruence between gene trees and species trees and phylogenetic signal variation in plastid genes. Mol. Phylogenet. Evol. 138, 219–232. https://doi.org/10.1016/j.ympev.2019.05.022 (2019).
Li, Q. et al. Population genetics and ecological niche modelling shed light on species boundaries and evolutionary history of Aconitum pendulum and A. flavum. Flora 314, 152507. https://doi.org/10.1016/j.flora.2024.152507 (2024).
Lim, C. E. & Park, C.-W. Hybridization in Aconitum subgenus Aconitum at Mt. Sobaek in Korea. Korean J. Plant Taxon. 31(4), 343–358. https://doi.org/10.11110/kjpt.2001.31.4.343 (2001).
Gao, Q., Ren, C. & Yang, Q.-E. Taxonomic status and distributional range of Aconitum angustius (Ranunculaceae) based on cytological evidence. Nord. J. Bot. 30(4), 426–438. https://doi.org/10.1111/j.1756-1051.2012.01506.x (2012).
Uyeki, H. & Sakata, T. Notulae ad Plantas Novae Koreae. Acta Phytotaxon. Geobot. 7(1), 14–19. https://doi.org/10.18942/bunruichiri.KJ00002594457 (1938).
Tamura, M. A synopsis of Aconitum subgenus Lycoctonum: II. Notes R. Bot. Gard. Edinburgh 37(3), 431–466 (1979).
Luo, Y., Zhang, F. M. & Yang, Q. E. Phylogeny of Aconitum subgenus Aconitum (Ranunculaceae) inferred from ITS sequences. Plant Syst. Evol. 252, 11–25. https://doi.org/10.1007/S00606-004-0257-5 (2005).
Xiao, P. G. et al. A pharmacophylogenetic study of Aconitum L. (Ranunculaceae) from China. Acta Phytotaxon. Sin. 44(1), 1–46. https://doi.org/10.1360/aps050046 (2006).
Acknowledgements
GS acknowledges the Department of Science and Technology (DST)-INSPIRE and IIT Hyderabad for supporting his research. RAK is supported by the PhD fellowship from the University Grant Commission, Government of India.
Funding
GS acknowledges the Department of Science and Technology (DST)-INSPIRE and IIT Hyderabad for supporting his research. RAK is supported by the PhD fellowship from the University Grant Commission, Government of India.
Author information
Authors and Affiliations
Contributions
GS generated the idea. RAK performed the analysis and wrote the first draft of the manuscript. RAK and GS edited and finalized the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kakkar, R.A., Sharma, G. Comprehensive analysis of 73 Aconitum chloroplast genomes reveals their structure, codon usage bias, and phylogenetic relationships within family Ranunculaceae. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40105-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-40105-5