Abstract
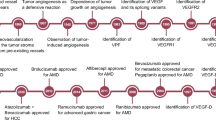
Vascular Endothelial Growth Factor (VEGF) plays a central role in angiogenesis, regulating both physiological processes such as wound healing, tissue repair, and bone formation, and pathological events including tumor progression, metastasis, and diabetic retinopathy. Due to its crucial role in vascular biology, VEGF serves as an important therapeutic target in anti-angiogenic drug development and precision medicine. However, conventional experimental methods for VEGF identification are costly and time-consuming, emphasizing the need for efficient computational approaches. To address this challenge, we introduce DeepStack-VEGF, an advanced deep learning framework designed for accurate and robust VEGF prediction. The model integrates diverse sequence-derived features, including physicochemical descriptors, sequential patterns, evolutionary information, and secondary structure motifs, further enhanced by pretrained embeddings from UniProt and ProtBert. Feature optimization was achieved using Support Vector Machine–Recursive Feature Elimination. DeepStack-VEGF employs a stacking ensemble of three architectures including Feedback Generative Adversarial Network Gated Recurrent Unit and Capsule Convolutional Neural Network each contributing distinct representational capabilities. Comprehensive evaluations demonstrate that the fused feature set and stacking ensemble substantially outperform individual models, achieving superior accuracy, robustness, and generalization. By combining deep learning with biological insight, DeepStack-VEGF provides a reliable and scalable computational framework for VEGF identification, supporting rational drug discovery, anti-angiogenic therapy design, and precision medicine applications.
Similar content being viewed by others
Data availability
We have made all datasets, feature extraction sets, and classifier codes freely available on GitHub at the following link: https://github.com/Farman335/DeepStack-VEGF
References
A. M. Byrne, D. J. Bouchier‐Hayes, J. H. J. J. o. c. Harmey, and m. medicine, "Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF)," vol. 9, no. 4, pp. 777–794, 2005.
Hu, K. & Olsen, B. R. J. The roles of vascular endothelial growth factor in bone repair and regeneration. Bone 91, 30–38 (2016).
Johnson, K. E. & Wilgus, T. A. J. Vascular endothelial growth factor and angiogenesis in the regulation of cutaneous wound repair. Adv. Wound Care 3(10), 647–661 (2014).
Shinkaruk, S., Bayle, M., Lain, G. & Deleris, G. J. C. Vascular endothelial cell growth factor (VEGF), an emerging target for cancer chemotherapy. Curr. Med. Chem. Anti-Cancer Agents 3(2), 95–117 (2003).
M. L. Formica, H. G. Awde Alfonso, S. D. J. P. R. Palma, and Perspectives, "Biological drug therapy for ocular angiogenesis: Anti‐VEGF agents and novel strategies based on nanotechnology," vol. 9, no. 2, p. e00723, 2021.
K. R. Khrapko et al., "Methods of DNA sequencing by hybridization based on optimizing concentration of matrix-bound oligonucleotide and device for carrying out same," ed: Google Patents, 1996.
Pagano, K. et al. Identification of a novel extracellular inhibitor of FGF2/FGFR signaling axis by combined virtual screening and NMR spectroscopy approach. Bioorg. Chem. 136, 106529 (2023).
Zaha, D. C. J. Significance of immunohistochemistry in breast cancer. World J. Clin. Oncol. 5(3), 382 (2014).
Alsini, R. et al. Deep-VEGF: Deep stacked ensemble model for prediction of vascular endothelial growth factor by concatenating gated recurrent unit with two-dimensional convolutional neural network. J. Biomol. Struct. Dyn. https://doi.org/10.1080/07391102.2024.2323144 (2024).
Ali, F. et al. VEGF-ERCNN: A deep learning-based model for prediction of vascular endothelial growth factor using ensemble residual CNN. J. Comput. Sci. 83, 102448 (2024).
M. Khalid, F. Ali, W. Alghamdi, A. Alzahrani, R. Alsini, and A. Alzahrani, "An ensemble computational model for prediction of clathrin protein by coupling machine learning with discrete cosine transform," J. Biomol. Struct. Dyn., pp. 1–9, 2024.
Ali, F. et al. Identification of defensins using transformer-derived protein embeddings and discrete cosine transformation-enhanced evolutionary features with generative adversarial capsule bidirectional temporal convolutional neural network. Int. J. Biol. Macromol. https://doi.org/10.1016/j.ijbiomac.2025.148336 (2025).
Ali, F. et al. DBPPred-PDSD: Machine learning approach for prediction of DNA-binding proteins using Discrete Wavelet Transform and optimized integrated features space. Chemometr. Intell. Lab. Syst. 182, 21–30 (2018).
Hasegawa, K., Moriwaki, Y., Terada, T., Wei, C. & Shimizu, K. Feedback-AVPGAN: Feedback-guided generative adversarial network for generating antiviral peptides. J. Bioinform. Comput. Biol. 20(06), 2250026 (2022).
Madhu, G., Mohamed, A. W., Kautish, S., Shah, M. A. & Ali, I. Intelligent diagnostic model for malaria parasite detection and classification using imperative inception-based capsule neural networks. Sci. Rep. 13(1), 13377 (2023).
S.-Y. Lin, "A Comparative Evaluation of Long Short-Term Memory (LSTM), Gated Recurrent Units (GRU), and Transformer-Based Informer Model for Predicting Rice Leaf Blast," Purdue University, 2024.
U. Consortium. UniProt: a hub for protein information. Nucleic Acids Res. 43(D1), D204–D212 (2015).
S. K. Burley, H. M. Berman, G. J. Kleywegt, J. L. Markley, H. Nakamura, and S. Velankar, Protein Data Bank (PDB): the single global macromolecular structure archive," Protein crystallography: methods and protocols, pp. 627-641, 2017.
Mistry, J. et al. Pfam: The protein families database in 2021. Nucleic Acids Res. 49(D1), D412–D419 (2021).
Alghamdi, W. et al. A deep learning model for epidermal growth factor receptor prediction using ensemble residual convolutional neural network. Sci. Rep. 15(1), 33676 (2025).
He, X. et al. PhaseNet: A computational framework for identifying phase-separating proteins based on protein language model. Int. J. Biol. Macromol. https://doi.org/10.1016/j.ijbiomac.2025.149044 (2025).
Almusallam, N. et al. Multi-headed ensemble residual CNN: A powerful tool for fibroblast growth factor prediction. Results Eng. 24, 103348 (2024).
Ali, F. et al. AFP-CMBPred: Computational identification of antifreeze proteins by extending consensus sequences into multi-blocks evolutionary information. Comput. Biol. Med. 139, 105006 (2021).
Ali, F. et al. DEEP-EP: Identification of epigenetic protein by ensemble residual convolutional neural network for drug discovery. Methods 226, 49–53 (2024).
A. Ahmad, S. Akbar, M. Hayat, F. Ali, and M. Sohail, "Identification of antioxidant proteins using a discriminative intelligent model of k-space amino acid pairs based descriptors incorporating with ensemble feature selection," Biocybernet. Biomed. Eng., 2020.
Ahmad, A. et al. Deep-AntiFP: Prediction of antifungal peptides using distanct multi-informative features incorporating with deep neural networks. Chemometr. Intell. Lab. Syst. 208, 104214 (2021).
Khan, A. et al. Prediction of antifreeze proteins using machine learning. Sci. Rep. 12(1), 1–10 (2022).
Sikander, R., Ghulam, A. & Ali, F. XGB-DrugPred: Computational prediction of druggable proteins using eXtreme gradient boosting and optimized features set. Sci. Rep. 12(1), 1–9 (2022).
R. Alsini et al., "Deep-VEGF: deep stacked ensemble model for prediction of vascular endothelial growth factor by concatenating gated recurrent unit with two-dimensional convolutional neural network," pp. 1–11, 2024.
S. Hasnat, F. A. Shifa, S. Murshed, S. T. Ahmed Rumee, and M. M. Mahbub, "A Framework for Accurate Prediction of Plastic-Degrading Enzymes using Convolutional Neural Networks," bioRxiv, p. 2024.10. 20.619257, 2024.
Z. Chen et al., 2018 iFeature: a python package and web server for features extraction and selection from protein and peptide sequences Bioinformatics vol. 34, no. 14, pp. 2499–2502,
Wang, H. & Hu, X. J. B. Accurate prediction of nuclear receptors with conjoint triad feature. BMC Bioinformatics 16(1), 1–13 (2015).
Li, F. et al. "Computational analysis and prediction of PE_PGRS proteins using machine learning. Comput. Struct. Biotechnol. J. 20, 662–674 (2022).
Zhao, L., Li, J., Zhang, B. & Jiang, X. Combining knowledge distillation and neural networks to predict protein secondary structure. Sci. Rep. 15(1), 32031 (2025).
Ayimbila, F. et al. Exploring the binding interaction of 1, 4-naphthoquinone derivative–human serum albumin complex by biophysics and molecular simulation. Sci. Rep. 15(1), 19249 (2025).
Chowdhury, A. S., Reehl, S. M., Kehn-Hall, K., Bishop, B. & Webb-Robertson, B.-J.M. Better understanding and prediction of antiviral peptides through primary and secondary structure feature importance. Sci. Rep. 10(1), 1–8 (2020).
Chowriappa, P., Dua, S., Kanno, J. & Thompson, H. W. Protein structure classification based on conserved hydrophobic residues. IEEE/ACM Trans. Comput. Biol. Bioinform. 6(4), 639–651 (2008).
Rayhan, F. et al. iDTI-ESBoost: Identification of drug target interaction using evolutionary and structural features with boosting. Sci. Rep. 7(1), 17731 (2017).
A. Ghulam, Z. N. K. Swati, F. Ali, S. Tunio, N. Jabeen, and N. Iqbal, "DeepImmuno-PSSM: Identification of Immunoglobulin based on Deep learning and PSSM-Profiles," 2023.
M. Khalid et al., "An ensemble computational model for prediction of clathrin protein by coupling machine learning with discrete cosine transform," pp. 1–9, 2024.
Mall, R., Kaushik, R., Martinez, Z. A., Thomson, M. W. & Castiglione, F. Benchmarking protein language models for protein crystallization. Sci. Rep. 15(1), 2381 (2025).
S. Rahu et al., "UBI-XGB: Identification of ubiquitin proteins using machine learning model," vol. 8, pp. 14–26, 2022.
Ali, F. et al. Leveraging deep learning for epigenetic protein prediction: A novel approach for early lung cancer diagnosis and drug discovery. Health Inf. Sci. Syst. 13(1), 28 (2025).
A. Khan, J. Uddin, F. Ali, A. Banjar, and A. Daud, "Comparative analysis of the existing methods for prediction of antifreeze proteins," Chemometrics Intell. Lab. Syst., p. 104729, 2022.
Lin, X. et al. AFPDeepPred: A deep learning framework for accurate identification of antifreeze proteins. J. Chem. Inf. Model. 65(22), 12256–12267 (2025).
Ali, F. et al. Deep-CABPred: Deep learning model for predicting functional chlorophyll ab binding proteins in trait-based plant ecology using hybrid embedding with semi-normalized temporal convolutional networks. Ecol. Inform. https://doi.org/10.1016/j.ecoinf.2025.103400 (2025).
Khan, Z. U., Ali, F., Ahmad, I., Hayat, M. & Pi, D. iPredCNC: Computational prediction model for cancerlectins and non-cancerlectins using novel cascade features subset selection. Chemometr. Intell. Lab. Syst. 195, 103876 (2019).
Sikander, R., Ghulam, A. & Ali, F. XGB-DrugPred: Computational prediction of druggable proteins using eXtreme gradient boosting and optimized features set. Sci. Rep. 12(1), 5505 (2022).
S. Zouari et al., "Deep‐GB: A novel deep learning model for globular protein prediction using CNN‐BiLSTM architecture and enhanced PSSM with trisection strategy," 2024.
M. Ullah, A. Iltaf, Q. Hou, F. Ali, and C. Liu, "A foreground extraction approach using convolutional neural network with graph cut," in 2018 IEEE 3rd International Conference on Image, Vision and Computing (ICIVC), 2018, pp. 40–44: IEEE.
Kabir, M. et al. Prediction of membrane protein types by exploring local discriminative information from evolutionary profiles. Anal. Biochem. 564, 123–132 (2019).
Ali, F., Ahmed, S., Swati, Z. N. K. & Akbar, S. DP-BINDER: Machine learning model for prediction of DNA-binding proteins by fusing evolutionary and physicochemical information. J. Comput.-Aided Mol. Des. 33(7), 645–658 (2019).
Ali, F. et al. AFP-CMBPred: Computational identification of antifreeze proteins by extending consensus sequences into multi-blocks evolutionary information. Comput. Biol. Med. https://doi.org/10.1016/j.compbiomed.2021.105006 (2021).
Zouari, S. et al. Deep-GB: A novel deep learning model for globular protein prediction using CNN-BiLSTM architecture and enhanced PSSM with trisection strategy. IET Syst. Biol. 18(6), 208–217 (2024).
Hasegawa, K. et al. Feedback-AVPGAN: Feedback-guided generative adversarial network for generating antiviral peptides. J. o. B. 20(06), 2250026 (2022).
M. Huh, S.-H. Sun, and N. Zhang, "Feedback adversarial learning: Spatial feedback for improving generative adversarial networks," in Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, 2019, pp. 1476–1485.
Almusallam, N. et al. An omics-driven computational model for angiogenic protein prediction: Advancing therapeutic strategies with Ens-deep-AGP. Int. J. Biol. Macromol. 282, 136475 (2024).
F. Ali, A. Masmoudi, T. Alkhalifah, F. Alturise, W. Alghamdi, and M. Khalid, "IR-MBiTCN: Computational prediction of insulin receptor using deep learning: A multi-information fusion approach with multiscale bidirectional temporal convolutional network," Int. J. Biol. Macromol., p. 143844, 2025.
Mahdi, E. T., Al-Barzinji, S. M. & Awad, W. K. Object detection using capsule neural network: An overview. Babylon. J. Mach. Learn. 2024, 157–164 (2024).
Vijayakumar, T. Comparative study of capsule neural network in various applications. J. Artif. Intell. 1(01), 19–27 (2019).
K. Sun et al., "CACNN: Capsule attention convolutional neural networks for 3D object recognition," IEEE Transac. Neural Netw. Learn. Syst., 2023.
R. Dey and F. M. Salem, "Gate-variants of gated recurrent unit (GRU) neural networks," in 2017 IEEE 60th international midwest symposium on circuits and systems (MWSCAS), 2017, pp. 1597–1600: IEEE.
Merkelbach, K., Schaper, S., Diedrich, C., Fritsch, S. J. & Schuppert, A. Novel architecture for gated recurrent unit autoencoder trained on time series from electronic health records enables detection of ICU patient subgroups. Sci. Rep. 13(1), 4053 (2023).
R. J. a. p. a. Rana, "Gated recurrent unit (GRU) for emotion classification from noisy speech," 2016.
A. Banjar, F. Ali, O. Alghushairy, and A. Daud, "iDBP-PBMD: A machine learning model for detection of DNA-binding proteins by extending compression techniques into evolutionary profile," Chemometrics Intell. Lab. Syst., p. 104697, 2022.
Xie, P. et al. Toward high-efficiency, low-resource, and explainable neuropeptide prediction with MSKDNP. Brief. Bioinf. 26(5), bbaf466 (2025).
Acknowledgements
1. National Natural Science Foundation of China, Grant No. 62501110 2. China Postdoctoral Science Foundation, Grant No. 2020TQ0138
Author information
Authors and Affiliations
Contributions
**Farman Ali:** Drafted and composed the manuscript, contributed to the interpretation of results. **Majdi Khalid:** Performed the experiments, assisted in data acquisition. **Abdulmohsen Algarni:** Conducted validation analyses, contributed to data verification. **Naif Waheb Rajkhan:** Substantively revised the manuscript, contributed to critical editing. **Othman Asiry:** Reviewed the manuscript, contributed to critical feedback, and interpretation. **Meng-Ze Du:** Supervision.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
No human or animal subjects are involved in this work.
Consent for publication
All authors agree on the publication of the paper.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ali, F., Khalid, M., Algarni, A. et al. DeepStackVEGF a stacking ensemble deep learning framework for vascular endothelial growth factor prediction. Sci Rep (2026). https://doi.org/10.1038/s41598-026-40134-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-40134-0