Abstract
Cupressales species are widely planted in urban landscapes, where their pollen represents a growing source of biological air pollution and contributes to seasonal allergies. Linking their flowering dynamics with atmospheric allergen loads is essential for understanding ecological flows of airborne particles in cities. We aimed to quantify interspecific differences in allergenic potential among common urban Cupressales taxa and to determine how flowering periods and pollen production shape airborne allergen levels. We specifically assessed variation in Cup a 1 homolog abundance to evaluate its relevance for landscape-scale allergy risk. The study was conducted in Poznań (Poland) during the 2023–2024 pollen seasons. Phenological observations, aerobiological monitoring, and immunoenzymatic assays were combined to relate flowering timing, atmospheric pollen concentrations, and allergen content across ten Cupressales species. Species differed substantially in flowering phenology, pollen output, and allergen levels. Taxus baccata, Thuja occidentalis, and Th. plicata released large amounts of pollen but contained low levels of Cup a 1 homologs. Conversely, Juniperus taxa and Callitropsis nootkatensis showed consistently high allergenicity, contributing disproportionately to peak atmospheric allergen loads despite moderate pollen release. These patterns indicate that allergen exposure risk is driven not only by pollen abundance but also by species-specific allergen potency. Urban planting dominated by high-allergenicity taxa may therefore intensify allergy symptoms at the neighbourhood scale. Our findings demonstrate that allergenicity metrics can inform urban vegetation planning. Selecting species with favourable phenological and allergenic profiles may help reduce airborne allergens and support healthier, more resilient urban landscapes.
Similar content being viewed by others
Introduction
Cupressales is an order of coniferous plants (including two major families, Cupressaceae and Taxaceae) that are commonly found in temperate regions. In Europe, representatives of this group include genera such as Cupressus (cypress), Juniperus (juniper), Thuja (arborvitae), Chamaecyparis (false cypress) and Taxus (yew)1. Taxa belonging to Cupressales are widely used as ornamental plants in horticulture and urban landscaping, owing to their advantageous traits, including year-round greenery, tolerate pruning and shaping, good resistance to air pollution and drought2,3. Numerous new cultivars have been developed, further increasing their popularity. However, their widespread use has an important downside—pollen allergy.
Pollen grains released by species of the order Cupressales are a significant cause of seasonal allergies worldwide4,5,6. The prevalence of Cupressales pollen allergy in the general population usually ranges from 2 to 4% in Southern European countries to as much as 13% in Japan. Among individuals with allergic diseases, however, the proportion of sensitized people is 14–32%, showing a clear upward trend in recent decades7,8. In Mediterranean countries, the emission of Cupressales pollen, primarily from the Arizona cypress (Cupressus arizonica) and Italian cypress (Cupressus sempervirens L.), is a major source of winter allergies, when other plants are not in bloom9,10. The allergenic significance of Cupressales pollen has been confirmed in studies conducted in France11,12,13, Spain14,15, Portugal16, Italy17,18, Greece19, Israel20 and Turkey21. Their pollen grains also pose a serious problem in Japan, where pollen from Japanese cedar (Cryptomeria japonica) is the primary airborne allergen, affecting up to 25% of the population seasonally22. Another significant source of allergenic pollen in Japan is the Hinoki cypress (Chamaecyparis obtusa)23. Meanwhile, in the southern region of North America, three Juniperus species, i.e. cade juniper (Juniperus oxycedrus), mountain cedar (Juniperus ashei), and eastern red cedar (Juniperus virginiana) are significant contributors to seasonal allergies24,25.
The primary allergens (group 1) in pollen grains of species within this order are proteins with a molecular weight of 40–47 kDa, exhibiting pectin lyase activity26,27. This enzyme is believed to be responsible for pectin degradation during pollen tube growth28. In the WHO/IUIS Allergen Nomenclature Database eight species from Cupressales are listed containing pectate lyase type allergen, i.e. Ch. obtusa (Cha o 1), C. japonica (Cry j 1), C. arizonica (Cup a 1), C. sempervirens (Cup s 1), J. ashei (Jun a 1), J. oxycedrus (Jun o 1) and J. virginiana (Jun v 1). The presence of major allergens in pollen grains clearly indicates their potential to cause allergic reactions. However, the allergenicity of pollen is also strongly influenced by the quantitative content of allergens within the grains. Studies analyzing the variability of major pollen allergens in other taxa, such as grasses and birches, have demonstrated significant differences in allergen content even among closely related species29,30. To date, such a comparison has not been conducted within any species of the Cupressales order.
The analysis of airborne pollen concentrations of different Cupressales species is challenging due to their close morphological similarity, which prevents genus-level identification31,32. In general, Cupressales pollen grains are small (20–30 μm), round, and characterized by a thin exine and a very thick intine, often giving the interior of the grain a distinctive star-shaped appearance. Additionally, the exine surface is covered with small, irregularly scattered granules known as orbicules (Ubisch bodies). Since these features are common to most Cupressales pollen grains, standard aerobiological analyses (quantifying airborne pollen concentrations) typically group the pollen of Cupressaceae and Taxaceae together as ‘Cupressales’33. This grouping has further implications, as detailed pollen calendars illustrating the pollination periods of individual Cupressales species are scarce34. It is important to note that the Cupressales pollen season is long, lasting 2–3 months, and consists of several distinct peaks7,34 each associated with the pollination of specific species. However, which species contribute most significantly to these peaks and the allergenic potential of pollen during each peak remain unknown. This lack of detailed knowledge makes it difficult to accurately assess the risk of pollen allergies at specific times within the Cupressales pollen season.
Therefore, our study aimed to address this gap by examining the variability in the quantity of homologs of major pollen allergens in the most common ornamental Cupressales species in Central Europe, particularly in Poznań, Poland. By integrating this data with phenological information (pollination period) and airborne pollen concentration measurements, we identified taxa with the highest allergenic potential, posing the greatest risk to sensitized allergic individuals.
Materials and methods
Plant material and study area
The study focused on 10 species of Cupressales: English yew (Taxus baccata), Nootka cypress (Callitropsis nootkatensis), Lawson’s cypress (Chamaecyparis lawsoniana), northern white cedar (Thuja occidentalis), western red cedar (Thuja plicata), savin juniper (Juniperus sabina), eastern red cedar (Juniperus virginiana), hybrid juniper (Juniperus x pfitzeriana), common juniper (Juniperus communis), and Chinese juniper (Juniperus chinensis).
Aerobiological monitoring, phenological observations, and pollen grain collection were conducted during two Cupressales pollen seasons (2023–2024) in Poznań, Poland (52° 24′ N, 16° 56′ E). The city is located in a temperate climate zone. The highest mean monthly temperatures are recorded in July (18.1 °C), while the lowest occur in January (− 1.6 °C). Annual precipitation ranges from 500 to 550 mm35. Meteorological data were collected from stations belonging to the Polish Institute of Meteorology and Water Management, located at Ławica Airport.
Phenological observations
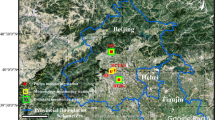
Observations of pollination process were conducted once a week from early February to late May. In 2023, six Cupressales species were monitored: T. baccata, Th. occidentalis, Th. plicata, Ch. lawsoniana, J. communis, and J. sabina. In 2024, in addition to these species, phenological observations were extended to four additional species: C. nootkatensis, J. virginiana, J. × pfitzeriana, and J. chinensis. The observations were carried out in the Botanical Garden of Adam Mickiewicz University, Dendrological Garden of the University of Life Sciences, Campus Morasko, and Cytadela Park in Poznań (Fig. 1). Detailed information on the number of species studied at each site is provided in Supplementary Table S1. Three to five individuals per species were observed. In practice, for each individual during each field visit, approximately 20–30 microsporophylls were assessed every 2–3 days, randomly selected from the available male shoots. Based on the percentage of shedding microsporophylls, three major pollen release stages were distinguished: (1) beginning (Stage 1), when 10% of microsporophylls released pollen, (2) peak (Stage 2), when 50% of microsporophylls released pollen grains, and (3) end of pollination (Stage 3), when the pollen release ended and fruit set became visible36.
Location of the aerobiological station (yellow square) and pollen collection/phenological observation sites (red circles) within the city of Poznań (Poland). The map presents the administrative boundary of the city and general land cover classes for spatial context.
Aerobiological monitoring
Pollen samples used for atmospheric pollen content analysis were collected using a Hirst-type volumetric trap (Burkard Manufacturing Co Ltd) located on the roof of the Faculty of Biology at Adam Mickiewicz University (7.5 km from the city center) at an altitude of 18 m (a.g.l.). The volumetric sampler operates by continuously drawing in 10 L of air per minute. The internal drum rotates at a speed of 2 mm/hour, allowing for hourly pollen grain analysis. A Melinex tape coated with an adhesive is affixed to the drum’s surface, where airborne particles, including pollen grains, are deposited. The device operates on a 7-day cycle. At the end of each week, the tape was cut into seven equal Sects. (14 mm × 48 mm), each representing one day, and stained with fuchsin solution. The preparations were then examined under a light microscope at 400 × magnification.
Daily pollen concentration was calculated using the four-horizontal-transects method according to aerobiological standards37. The obtained value was multiplied by a factor accounting for the total slide surface, the analyzed area on the slide, and the volume of analyzed air38. The result was expressed as pollen grains per cubic meter per day (pollen grains/m3/day). To determine the duration of the Cupressales pollen season, the 95% cumulative percentage method was applied. According to this method, the season starts on the day when the daily pollen concentration reaches 2.5% of the total annual pollen sum and ends when it reaches 97.5%39. The obtained pollen concentration data were then compared with the pollination periods of individual species.
Immunoenzymatic methods
Pollen was collected from flowering individuals that were also included in phenological observations, from the same locations, using a laboratory sieve. Due to the generally low pollen yield and the relatively large amount of pollen required for ELISA analyses, samples from individual species were pooled within the same season. After collection, pollen grains were stored in a refrigerator at –20 °C. To obtain the allergen solution, 0.1 g of pollen was extracted in 1.5 ml of extraction buffer (NH₄HCO₃, H₂O, NH₃). To quantify the Cup a 1 allergen homologs released from pollen grains, a sandwich-type enzyme-linked immunosorbent assay (ELISA) was performed using anti-Cup a 1 monoclonal antibodies and biotinylated monoclonal antibodies supplied by ROXALL GROUP. Streptavidin-peroxidase (Sigma-Aldrich S5512) was used as the enzyme, while 3,3′5,5′-tetramethylbenzidine (Sigma-Aldrich T0440) served as the substrate. The reaction was stopped by adding 2.5 M H₂SO₄ (5 N). The concentration of Cup a 1 in the sample was measured by detecting absorbance at 450 nm. Using a hemocytometer, the number of pollen grains used for sample preparation was counted, and the concentration of Cup a 1 allergen homologs per pollen grain was calculated, allowing for the determination of allergenicity (pg Cup a 1/pollen grain) of selected Cupressales species. Because the samples were pooled within each season, only interannual differences in pollen allergenicity could be assessed (2023 vs. 2024), with no within-season replication. Therefore, no formal statistical analysis of interspecific variability was performed.
Results
Weather background
In 2023, the average temperature in Poznań was 10.6 °C, while in 2024, it increased to 12.3 °C. Annual precipitation and average wind speed were similar in both years—526 mm and 3.7 m/s in 2023, and 534 mm and 3.6 m/s in 2024. Between February and May, corresponding to the pollination period of the observed Cupressales species, total precipitation was 31.5 mm in 2023, whereas in 2024 it increased to 169.2 mm. The average daily temperature during this period was 7.1 °C in 2023 and 10.2 °C in 2024.
Phenological observation of pollination
Among the species studied, T. baccata began pollinating the earliest in both years (Supplementary Table S2). The first open inflorescences (Stage 1) of T. baccata in Poznań were observed on February 16, 2023, and February 10, 2024. The peak pollen release (Stage 2) occurred during the first three weeks of March in 2023 and at the turn of February and March in 2024. The pollination period of T. baccata ended (Stage 3) on March 27, 2023, and March 9, 2024. The pollination periods of the two observed Thuja species overlapped and were mainly limited to March in 2023, and from mid-February to early March in the following year. The Stage 2 phase was recorded in mid-March during the first year of observation and in early March in 2024. On April 1, 2023, pollen release of Juniperus sabina began, with the season ending on April 20, 2023. The Stage 2 phase was recorded between the second and third weeks of April. Similar to the other species studied, the pollination period in 2024 started much earlier than in 2023, with the Stage 2 phase occurring in the second and third weeks of March. J. communis was the latest species to pollinate, with the peak of pollen release in the first week of May 2025 and the beginning of April 2025. In 2024, three other Juniperus species were observed: J. chinensis, J. virginiana, and J. × pfitzeriana. All of them pollinated just before J. sabina, predominantly in the first part of March. In 2024, the pollination period of Callitropsis nootkatensis started on February 25 and ended on March 15, with the peak occurring between the first and second weeks of March. Chamaecyparis lawsoniana is considered a late-flowering species, releasing pollen in the second half of April in 2023 and the second half of March in 2024. Overall, the sequence of pollination among the observed species was similar in both years. It began with Taxus baccata, followed by the two Thuja species, while Juniperus and Chamaecyparis pollinated later in the season. Juniperus communis was the last species to pollinate.
Airborne pollen concentration and its relevance to phenological data
In 2023, the first Cupressales pollen grains were detected in the air in mid-January. The pollen season began on March 4, ended on May 11, and lasted 69 days. The total sum of pollen grains in the air was 2,280 (pollen grains/ m3). The highest daily pollen concentration (308 pollen grains/m3/day) was observed on March 23 (Table 1). The pollen season also had several smaller peaks, specifically on March 14 (298 pollen grains/m3/day), March 19 (210 pollen grains/m3/day), and April 10 (227 pollen grains/m3/day).
In 2024, the first pollen grains in the air were also observed in mid-January. The pollen season began on February 17, ended on April 15, and lasted 61 days. The total sum of pollen grains in the air was 2,345 (pollen grains/ m3). The highest daily pollen concentration (262 pollen grains/m3/day) was recorded on March 2 (Table 1). The 2024 Cupressales pollen season also had several smaller peaks, occurring on February 23 (164 pollen grains/m3/day), March 10 (119 pollen grains/m3/day), and March 15 (198 pollen grains/m3/day).
Based on flowering observations and airborne pollen grain data (Fig. 2), the species contributing most significantly to the seasonal pollen sum were T. baccata, Th. occidentalis, and Th. plicata. In the first half of April 2023, a distinct peak in pollen grain concentration corresponded with J. sabina pollen release. In 2024, this peak also matched the pollen release periods of other junipers, namely J. virginiana and J. x pfitzeriana. Other species, such as J. communis, had a smaller impact on the total pollen sum in the air but significantly prolonged the Cupressales pollen season due to their late flowering period.
Concentration of Cupressales pollen (pink color) compared to the pollen calendar of selected Cupressales species in Poznań, Poland, 2023–2024.
Allergenic protein homolog content
The pollen grains of the studied species exhibited significant variations in the amount of allergenic Cup a 1 homolog (Table 2). The highest allergenicity was observed in the C. nootkatensis, with a mean of 2.085 pg/pollen grain. Relatively high values were also recorded for the Juniperus genus, specifically J. virginiana (1.006 pg/pollen grain), J. communis (0.964 pg/pollen grain), J. x pfitzeriana (0.879 pg/pollen grain), J. sabina (0.836 pg/pollen grain), and J. chinensis (0.471 pg/pollen grain). For the remaining species, namely Th. plicata, Th. occidentalis, T. baccata, and Ch. lawsoniana, Cup a 1 homologues were detected at values near or below the detection limit of the ELISA system used.
Discussion
Allergenic potential of cupressales pollen
Observations that pollen allergenicity can vary substantially even among closely related species highlight the need to integrate molecular and proteomic approaches into classical aerobiological studies. To date, such complementary analyses have been conducted for several major allergenic taxa, including Poaceae30,40, Betulaceae29,41, and Asteraceae42,43. However, despite the clinical relevance of Cupressales pollen, variability in its allergenicity remains poorly characterized. Previous studies typically included only a limited number of species (2–3) and relied on semi-quantitative methods (e.g., SDS-PAGE, Western blotting)44,45 or indirect approaches (e.g., inferring allergen levels from the relationship between airborne pollen counts and measured allergen concentrations)15. Although these studies produced encouraging results—suggesting true species-specific differences in allergenicity within Cupressales—comprehensive and robust investigations have been lacking.
Here, we provide a systematic quantification of homologs of the major cypress pollen allergen, Cup a 1, across multiple Cupressales representatives. Using an enzyme-linked immunosorbent assay (ELISA), we determined allergenic protein levels in pollen grains from several species within the order. In the present study, high levels of proteins cross-reacting with Cup a 1 were detected in all examined juniper species, including Juniperus sabina, J. virginiana, J. communis, J. x media, and J. chinensis. Similarly, very high amount of Cup a 1-homologue was noted in Callitropsis nootkatensis, which is closely related to Juniperus and Cupressus genera46. In contrast to the other species investigated, the group I allergen was detected at much lower levels in the pollen of yew (Taxus baccata), false cypress (Chamaecyparis lawsoniana) and two species of Thuja. Arilla et al.27 showed that the antibodies developed for ELISA were capable of recognizing pectin lyase not only from Cupressus, but also Juniperus and Thuja. In general, in the Cupressaceae family, allergenic proteins exhibit a high degree of sequence identity (≥ 75%) and similarity (≥ 95%)47,48. Pectin lyase activity has been most extensively studied in Japanese cedar (Cryptomeria japonica), where cross-reactivity was demonstrated between the Cry j 1 allergen and the major allergens of Arizona cypress (Cupressus arizonica)—Cup a 1 and Mediterranean cypress (Cupressus sempervirens)—Cup s 149,50. A highly homologous protein has also been characterized in Ashe juniper (Juniperus ashei)—Jun a 126, prickly juniper (Juniperus oxycedrus)—Jun o 1 (EC 4.2.2.2), and eastern red cedar (Juniperus virginiana)—Jun v 151. Cross-reactivity was also demonstrated between the Cry j 1 allergen and the major allergens of Hinoki cypress (Chamaecyparis obtusa)—Cha o 149,50. This suggests that the differences in the level of Cup a 1 homologs in investigated species is related to the immanent characteristic of these pollen grains rather than differences in allergen-antibody recognizing efficiency.
The situation for Taxus baccata is less clear, as no previous data were available on the structure or cross-reactivity of its allergens. T. baccata belongs to the Taxaceae family and is therefore phylogenetically more distant from the other investigated taxa, which belong to the Cupressaceae family. However, Cupressaceae group 1 allergens form a highly conserved protein family with strong sequence and structural similarity, and numerous studies have shown that these allergens are functional homologs with high IgE cross-reactivity, with differences being largely quantitative rather than qualitative47. Consequently, antibodies raised against Cup a 1 are generally expected to recognize shared epitopes across group 1 allergens from different Cupressaceae taxa. To address potential concerns regarding a more phylogenetically distant taxon such as T. baccata, we sequenced its group 1 allergen and compared it with Cup a 1 (Supplementary Fig. S1). This analysis revealed a high amino acid similarity (94%), comparable to or even higher than similarities reported between Cup a 1 and some Cupressaceae genera48. These results indicate that the Taxus group 1 allergen should be efficiently recognized by anti–Cup a 1 antibody.
From this perspective, it is also important to note that phylogenetic relatedness is not always a reliable predictor of allergen similarity or ELISA cross-reactivity. For example, as shown by Radauer et al.52, highly conserved allergenic proteins can be present in species that are relatively distant phylogenetically, while closely related species may show significant divergence in specific allergen sequences. Antibody binding depends primarily on the conservation and surface accessibility of specific linear or conformational epitopes, which may remain preserved even between phylogenetically distant taxa. Moreover, protein folds can remain highly conserved despite substantial sequence divergence53,54. Therefore, although Taxus (Taxaceae) is taxonomically distant from Juniperus and Thuja (Cupressaceae), structurally constrained Cupressales group 1 allergens (Cup a 1–type pectate lyases) may still share conserved epitopes across both families. This interpretation is consistent with our sequence results and provides a plausible explanation for the observed ELISA reactivity, where differences in ELISA signals likely reflect true quantitative differences in allergen content.
Pollen concentration and pollination phenology
Thanks to the combination of direct pollination observations and aerobiological data, we were able to determine which Cupressales species contributed most significantly to atmospheric pollen levels. Based on the collected data, it is evident that the primary sources of pollen are Taxus baccata, Thuja occidentalis, and Th. plicata. These species flower earliest in the season, with their pollination period spanning from mid-February to the end of March. Fortunately for allergy sufferers, T. baccata, Th. occidentalis, and Th. plicata exhibited the lowest levels of Cup a 1 homologs. In contrast, high concentrations of airborne pollen were recorded during the flowering of Juniperus sabina, J. virginiana, and J. × pfitzeriana, which showed relatively high pollen allergenicity among all taxa studied. The widespread planting of these species—particularly J. sabina and J. × media—combined with their high allergenic potential, suggests that they may pose a significant health risk to residents of Poznań, especially in the second part of Cupressales pollen season (April–May).
Whether the observed variation in pollen contribution is due to differences in pollen production or species abundance and distribution requires further investigation. In Central and Western Poland, only two native Cupressales species occur: Taxus baccata and Juniperus communis55. Both are relatively scarce, particularly T. baccata, which persists mainly at a few natural sites and is considered endangered. In forests, yew occurs singly or in small groups in the understory, limiting its pollen emission and dispersal56. J. communis is more widespread, typically growing as an understory species on poor, sandy podzolic and brown soils dominated by pine and pine–spruce stands57. However, its greater abundance does not translate into higher airborne pollen concentrations. In Poznań, pollen levels during the second part of the Cupressales season (flowering time of Juniperus) are much lower than in February–March, when yew releases pollen. Therefore, the high Cupressales pollen concentrations observed in the city are likely driven mainly by ornamental and planted taxa in the urban environment (parks and private gardens), rather than by native populations.
In our study, botanical gardens served as reference sites for phenological observations of well-identified Cupressales taxa, complemented by monitoring at several additional urban locations. The onset and peak of pollen release across the city were broadly consistent with the botanical garden records. Although some spatial variation between sites can be expected due to microclimatic differences—for example, as observed for birch in Poznań58—this variation is unlikely to exceed 1–2 weeks. Given that the Cupressales pollen season in Poznań lasts almost four months (February–May) and is characterized by several distinct peaks, we assume that such microclimatic shifts in flowering timing are too small to significantly affect the overall seasonal pattern.
Studies on the genus Cupressus have shown that pollen production can vary substantially between species. For example, C. macrocarpa produces significantly more pollen—nine times more than C. arizonica and 18 times more than C. sempervirens59. Comparable interspecific variation in pollen production has also been reported in other genera, such as Artemisia. A. campestris, for instance, produces considerably fewer pollen grains per plant compared to A. vulgaris and A. absinthium. Nevertheless, due to its widespread presence in rural environments and relatively high allergenicity43, A. campestris contributes substantially to overall airborne pollen loads and poses an increased risk to sensitized individuals in certain areas60.
Without such fundamental data, it is not possible to accurately assess the allergenic potential of specific plant species. While simplified pollen calendars fulfill general public needs, they are often insufficient for urban-scale assessments, where pollen exposure is highly localized. Greater efforts are needed within the scientific community to rigorously evaluate the allergenic characteristics of key species. Remarkably, even after decades of research, essential data—such as pollen production rates and allergen content per pollen grain—remain unknown for many highly allergenic taxa.
Species selection for urban planting
The results of this study suggest that yews (Taxus spp.) and thujas (Thuja spp.) could be favorable choices for urban greening projects in terms of reducing exposure to group 1 allergens. However, further research is needed before these taxa can be confidently recommended as low-allergenic species for urban plantings. It should be borne in mind that other allergenic proteins not investigated in this study may be present in their pollen. In addition, published studies indicate that yew and thuja pollen may indeed cause allergic reactions61,62. Nevertheless, in comparison, to other investigated species, yew and thujas exhibited very low levels of the investigated pectin lyase allergens. An additional advantage of yews is their dioecious nature1, allowing for the selective planting of female individuals, which do not produce pollen, thereby further reducing airborne pollen exposure in urban environments Similarly, when selecting junipers (Juniperus spp.), it would be beneficial to prefer dioecious species, wherever possible. Although not all analyzed juniper species are dioecious, preference should be given to those that are (J. sabina and J. communis), as this also enables the strategic planting of female plants to minimize pollen emissions.
Beyond species selection, other considerations are critical for allergen-sensitive urban greening. Monocultures and dense hedgerows composed of highly allergenic species should be avoided, as they can lead to locally elevated pollen concentrations63,64. A diverse plant palette should be promoted, incorporating species and cultivars known for low pollen production and minimal allergenic potential. Trees with broad leaves and lower pollen dispersal, such as Acer (mapple) or Tilia (linden) can serve as effective buffer species, especially when placed strategically to intercept airborne allergens from more potent sources65. Additionally, the introduction of highly allergenic exotic species, such as Cryptomeria japonica22, which recently has been increasingly available in commercial garden shops (own observations), should be avoided to prevent the introduction of new allergenic sources into urban areas.
Overall, the findings highlight the importance of incorporating allergenicity into urban planting guidelines. Strategic selection of plant species based on their reproductive biology, flowering phenology, and allergenic cross-reactivity, combined with diversification of urban greenery, can significantly contribute to the creation of healthier urban environments and the improvement of life quality for allergy-prone individuals.
Conclusion
Many factors influence how healthy and resident-friendly an urban space is, and one of them—often overlooked—is the allergenic potential of plants. As our research demonstrates, even closely related plant species can differ significantly in their allergenicity, pollination periods, and contribution to the total airborne pollen load. These differences can directly impact the severity and frequency of allergic symptoms among the population.
Our findings highlight the need for continued research, particularly to assess additional factors such as pollen production, spatial distribution, and plant abundance within urban environments. Furthermore, given the wide range of cultivars among ornamental plants, their allergenic properties should be evaluated prior to commercial distribution. Introducing standardized labelling of a species’ allergenic potential could offer valuable guidance to both landscape planners and individual buyers, helping to mitigate unintended health risks associated with urban greening.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Senata, W. & Dolatowski, J. Dendrologia (PWN, 2021).
Willson, C. J., Manos, P. S. & Jackson, R. B. Hydraulic traits are influenced by phylogenetic history in the drought-resistant, invasive genus Juniperus (Cupressaceae). Am. J. Bot. 95(3), 299–314. https://doi.org/10.3732/ajb.95.3.299 (2008).
Nagalingam, S. et al. Unexpectedly strong heat stress induction of monoterpene, methylbutenol, and other volatile emissions for conifers in the cypress family (Cupressaceae). Sci. Total Environ. 956, 177336. https://doi.org/10.1016/j.scitotenv.2024.177336 (2024).
Caballero, T. et al. Cupressaceae pollinosis in the Madrid area. Clin. Exp. Allergy 26, 197–201. https://doi.org/10.1111/j.1365-2222.1996.tb00080.x (1996).
Burbach, G. J. et al. GA2LEN skin test study II: Clinical relevance of inhalant allergen sensitizations in Europe. Allergy 64(10), 1507–1515. https://doi.org/10.1111/j.1398-9995.2009.02068.x (2009).
Bousquet, J., Cour, P., Guerin, B. & Michel, F. B. Allergy in the Mediterranean area. I. Pollen counts and pollinosis of Montpellier. Clin. Allergy 14, 249–258. https://doi.org/10.1111/j.1365-2222.1984.tb02204.x (1984).
Charpin, D., Sénéchal, H. & Poncet, P. Respiratory allergy to conifers. Conifers Recent Adv. https://doi.org/10.5772/intechopen.101217 (2022).
de Weger, L. A. et al. Method to develop a regional guide for the allergenic potential of tree pollen. Sci. Total Environ. 926, 171575. https://doi.org/10.1016/j.scitotenv.2024.171575 (2024).
D’Amato, G. & Liccardi, G. Pollen-related allergy in the European Mediterranean area. Clin. Exp. Allergy 24, 210–219. https://doi.org/10.1111/j.1365-2222.1994.tb00222.x (1994).
Velasco-Jiménez, M. J., Candau, P., De Linares, C. & Alba-Sánchez, F. Pollen season trends in winter flowering trees in South Spain. Aerobiologia 36, 213–224. https://doi.org/10.1007/s10453-019-09622-x (2020).
Charpin, D., Hughes, B. & Mellea, M. Seasonal allergenic symptoms and their relation to pollen exposure in south-east France. Clin. Exp. Allergy 23, 435–439. https://doi.org/10.1111/j.1365-2222.1993.tb00350.x (1993).
Charpin, D., Calleja, M., Lahoz, C., Pichot, C. & Waisel, Y. Allergy to cypress pollen. Allergy 60, 293–301. https://doi.org/10.1111/j.1398-9995.2005.00731.x (2005).
Caimmi, D. et al. Epidemiology of cypress pollen allergy in Montpellier. J. Investig. Allergol. Clin. Immunol. 22(4), 280 (2012).
Guerra, F. et al. Sensitivity to Cupressus. Allergenic significance in Cordoba (Spain). J. Investig. Allergol. Clin. Immunol. 6, 117–120 (1996).
De Linares, C. et al. Airborne Cupressaceae pollen and its major allergen, Cup a 1, in urban green areas of southern Iberian Peninsula. Forests 12(2), 254. https://doi.org/10.3390/f12020254 (2021).
Gomes, C., Ribeiro, H. & Abreu, I. Aerobiology of Cupressaceae in Porto city, Portugal. Aerobiologia 35, 97–103. https://doi.org/10.1007/s10453-018-9543-1 (2019).
Caiaffa, M. F. et al. Airborne Cupressaceae pollen in Southern Italy. Ann. Allergy 71, 45–50 (1993).
Asero, R., Ceriotti, V. & Bonini, M. Cypress pollen allergy in Milan: The story of an ongoing growth. Eur. Ann. Allergy Clin. Immunol. 53(5), 209–213. https://doi.org/10.23822/EurAnnACI.1764-1489.155 (2020).
Gonianakis, M. I. et al. A 10-year aerobiological study (1994–2003) in the Mediterranean island of Crete, Greece: Trees, aerobiologic data, and botanical and clinical correlations. Allergy Asthma Proc. https://doi.org/10.2500/aap.2006.27.2935 (2006).
Geller-Bernstein, C., Waisel, Y. & Lahoz, C. Environment and sensitization to cypress in Israel. Allerg. Immunol. 32, 92–93 (2000).
Sin, A. Z. et al. Prevalence of cypress pollen sensitization and its clinical importance in Izmir, Turkey, with cypress allergy assessed by nasal provocation. J. Investig. Allergol. Clin. Immunol. 18(1), 46 (2008).
Hashimoto, M. et al. Sensitivity to two major allergens (Cry j I and Cry j II) in patients with Japanese cedar (Cryptomeria japonica) pollinosis. Clin. Exp. Allergy 25(9), 848–852. https://doi.org/10.1111/j.1365-2222.1995.tb00027.x (1995).
Ito, H., Nishima, J. & Suzuki, M. Specific IgE to Japanese cypress (Chamaecyparis obtusa) in patients with nasal allergy. Ann. Allergy Asthma Immunol. 74, 299–303 (1995).
Iacovacci, P. et al. Juniperus oxycedrus: A new allergenic pollen from the Cupressaceae family. J. Allergy Clin. Immunol. 101(6), 755–761. https://doi.org/10.1016/S0091-6749(98)70304-5 (1998).
White, J. F. & Bernstein, D. I. Key pollen allergens in North America. Annals of Allergy, Asthma Immunol. 91(5), 425–435. https://doi.org/10.1016/S1081-1206(10)61509-8 (2003).
Midoro-Horiuti, T., Tiwari, R., Schein, C. H. & Goldblum, R. M. Molecular cloning of the mountain cedar (Juniperus ashei) pollen major allergen, Jun a 1. J. Allergy Clin. Immunol. 104(3), 613–617. https://doi.org/10.1016/S0091-6749(99)70332-5 (1999).
Arilla, M. C. et al. Quantification of the major allergen from cypress (Cupressus arizonica) pollen, Cup a 1, by monoclonal antibody-based ELISA. Int. Arch. Allergy Immunol. 134(1), 10–16. https://doi.org/10.1159/000077528 (2004).
Marín-Rodríguez, M. C., Orchard, J. & Seymour, G. B. Pectate lyases, cell wall degradation and fruit softening. J. Exp. Bot. 53(377), 2115–2119. https://doi.org/10.1093/jxb/erf089 (2002).
Schenk, M. F. et al. Proteomic analysis of the major birch allergen Bet v 1 predicts allergenicity for 15 birch species. J. Proteomics 74(8), 1290–1300. https://doi.org/10.1016/j.jprot.2011.03.021 (2011).
Jung, S. et al. Grass pollen production and group V allergen content of agriculturally relevant species and cultivars. PLoS ONE 13(3), e0193958. https://doi.org/10.1371/journal.pone.0193958 (2018).
Bortenschlager, S. Aspects of pollen morphology in the Cupressaceae. Grana 29(2), 129–138. https://doi.org/10.1080/00173139009427743 (1990).
Flonard, M., Lo, E. & Levetin, E. Increasing Juniperus virginiana L. pollen in the Tulsa atmosphere: Long-term trends, variability, and influence of meteorological conditions. Int. J. Biometeorol. 62, 229–324. https://doi.org/10.1007/s00484-017-1444-8 (2018).
Galán, C., Fuillerat, M. J., Comtois, P. & Domínguez-Vilches, E. Bioclimatic factors affecting daily Cupressaceae flowering in southwest Spain. Int. J. Biometeorol. 41, 95–100. https://doi.org/10.1007/s004840050059 (1998).
Monroy-Colín, A., Valencia-Barrera, R. M., Damián-Rodríguez, E. & Belmonte, J. Phenology of Cupressaceae urban infrastructure related to its pollen content and meteorological variables. Aerobiologia 36, 459–479. https://doi.org/10.1007/s10453-020-09645-9 (2020).
Woś, A. Klimat Polski w drugiej połowie XX wieku. (Wydawnictwo Naukowe Uniwersytetu im. Adama Mickiewicza, 2010).
Hess, M. et al. Use of the extended BBCH scale—general for the descriptions of the growth stages of mono; and dicotyledonous weed species. Weed Res. 37(6), 433–444. https://doi.org/10.1046/j.1365-3180.1997.d01-70.x (1997).
Galán, C. et al. Airborne olive pollen counts are not representative of exposure to the major olive allergen Ole e 1. Allergy 68(6), 809–812. https://doi.org/10.1111/all.12144 (2013).
Stach, A. & Kasprzyk, I. Metodyka badań zawartości pyłku roślin i zarodników grzybów w powietrzu z zastosowaniem aparatu Hirsta. (Bogucki Wydawnictwo Naukowe, 2005).
Goldberg, C., Buch, H., Moseholm, L. & Weeke, E. V. Airborne pollen records in Denmark, 1997–1986. Grana 27, 209–217. https://doi.org/10.1080/00173138809428928 (1988).
Buters, J. et al. Variation of the group 5 grass pollen allergen content of airborne pollen in relation to geographic location and time in season. J. Allergy Clin. Immunol. 136, 87–95. https://doi.org/10.1016/j.jaci.2015.01.049 (2015).
Darnhofer, B. et al. Comparative proteomics of common allergenic tree pollens of birch, alder, and hazel. Allergy 76(6), 1743–1753. https://doi.org/10.1111/all.14694 (2021).
Asero, R. et al. Giant ragweed specific immunotherapy is not effective in a proportion of patients sensitized to short ragweed: Analysis of the allergenic differences between short and giant ragweed. J. Allergy and Clin. Immunol. 11(5), P1036-1041. https://doi.org/10.1016/j.jaci.2005.08.019 (2005).
Grewling, Ł et al. Atmospheric exposure to the major Artemisia pollen allergen (Art v 1): Seasonality, impact of weather, and clinical implications. Sci. Total Environ. 713, 136611. https://doi.org/10.1016/j.scitotenv.2020.136611 (2020).
Acar Şahin, A., Aslım, B., Tan, S., Alan, Ş & Pınar, N. M. Differences in structure, allergenic protein content and pectate lyase enzyme activity of some Cupressaceae pollen. Turk. J. Biochem. 43(4), 435–446. https://doi.org/10.1515/tjb-2017-0260 (2018).
Barberini, S. et al. Different allergenicity of pollen extracts of three Mediterranean cypress species accounted for cytological observations. Eur. Ann. Allergy Clin. Immunol. 47(5), 149–155 (2015).
Liu, X. Q., Xia, X. M., Chen, L. & Wang, X. Q. Phylogeny and evolution of Cupressaceae: Updates on intergeneric relationships and new insights on ancient intergeneric hybridization. Mol. Phylogenet. Evol. 177, 107606. https://doi.org/10.1016/j.ympev.2022.107606 (2022).
Barré, A. et al. Identification of potential IgE-binding epitopes contributing to the cross-reactivity of the major Cupressaceae pectate-lyase pollen allergens (Group 1). Allergies 2(3), 106–118. https://doi.org/10.3390/allergies2030010 (2022).
Charpin, D. et al. Cypress pollinosis: From tree to clinic. Clin. Rev. Allergy Immunol. 56, 174–195. https://doi.org/10.1007/s12016-017-8602-y (2019).
Griffith, I. J. The cDNA cloning of Cry j 1, the major allergen of Cryptomeria japonica (Japanese cedar). J. Allergy Clin. Immunol. 91, 339 (1993).
Di Felice, G., Barletta, B., Tinghino, R. & Pini, C. Cupressaceae pollinosis: Identification, purification and cloning of relevant allergens. I. Arch. Allergy Immunol. 125(4), 280–289. https://doi.org/10.1159/000053828 (2001).
Midoro-Horiuti, T., Goldblum, R. M. & Brooks, E. G. Identification of mutations in the genes for the pollen allergens of Juniperus virginiana (Eastern red cedar). Clin. Exp. Allergy 31(5), 771–778. https://doi.org/10.1046/j.1365-2222.2001.01079.x (2001).
Radauer, C., Bublin, M., Wagner, S., Mari, A. & Breiteneder, H. Allergens are distributed into few protein families and possess a restricted number of biochemical functions. J. Allergy Clin. Immunol. 121(4), 847–52.e7. https://doi.org/10.1016/j.jaci.2008.01.025 (2008).
Chothia, C. & Lesk, A. M. The relation between the divergence of sequence and structure in proteins. EMBO J. 5(4), 823–826. https://doi.org/10.1002/j.1460-2075.1986.tb04288.x (1986).
Lesk, A. M. & Chothia, C. How different amino acid sequences determine similar protein structures: The structure and evolutionary dynamics of the globins. J. Mol. Biol. 136(3), 225–270. https://doi.org/10.1016/0022-2836(80)90373-3 (1980).
Zając, A. & Zając, M. Atlas rozmieszczenia roślin naczyniowych w Polsce. Distribution atlas of vascular plants in Poland. (Instytut Botaniki Uniwersytetu Jagiellońskiego, 2001).
Bugała, W. Cis (Taxus L.). Systematyka i zmienność. In: Cis pospolity Taxus baccata L. Nasze Drzew Leśne. Monografie Popularnonaukowe, Tom III (ed. Białobok, S.) (PAN, Inst. Dendrol.; PWN, 1975).
Stypiński, P. & Michalczyk, J. Występowanie jałowca pospolitego (Juniperus communis L.) w lasach państwowych na Równinie Mazurskiej. The occurrence of common juniper (Juniperus communis L.) in the state forests in Masuria Plain. Sylwan 128, 25–33 (1984).
Bogawski, P., Grewling, Ł & Jackowiak, B. Predicting the onset of Betula pendula flowering in Poznań (Poland) using remote sensing thermal data. Sci. Total Environ. 658, 1485–1499. https://doi.org/10.1016/j.scitotenv.2018.12.295 (2019).
Hidalgo, P. J., Galán, C. & Domínguez, E. Pollen production of the genus Cupressus. Grana 38(5), 296–300. https://doi.org/10.1080/001731300750044519 (1999).
Bogawski, P., Grewling, Ł & Frątczak, A. Flowering phenology and potential pollen emission of three Artemisia species in relation to airborne pollen data in Poznań (Western Poland). Aerobiologia 32, 265–276. https://doi.org/10.1007/s10453-015-9397-z (2016).
Guerin, B., Kanny, G., Terrasse, G., Guyot, J. L. & Moneret-Vautrin, D. A. Allergic rhinitis to Thuja pollen. Int. Arch. Allergy Immunol. 110(1), 91–94. https://doi.org/10.1159/000237317 (1996).
Maguchi, S. & Fukuda, S. Taxus cuspidata (Japanese yew) pollen nasal allergy. Auris Nasus Larynx 28(Suppl), S43–S47. https://doi.org/10.1016/S0385-8146(01)00062-1 (2001).
D’Amato, G., Cecchi, L., D’amato, M. & Liccardi, G. Urban air pollution and climate change as environmental risk factors of respiratory allergy: An update. J. Investig. Allergol. Clin. Immunol. 20(2), 95–102 (2010).
Cariñanos, P. & Casares-Porcel, M. Urban green zones and related pollen allergy: A review. Some guidelines for designing spaces with low allergy impact. Landsc. Urban Plan. 101(3), 205–214. https://doi.org/10.1016/j.landurbplan.2011.03.006 (2011).
Cariñanos, P. et al. Estimation of the allergenic potential of urban trees and urban parks: Towards the healthy design of urban green spaces of the future. Int. J. Environ. Res. Public Health 16(8), 1357. https://doi.org/10.3390/ijerph16081357 (2019).
Acknowledgements
We would like to express our sincere gratitude to the Botanical Garden of Adam Mickiewicz University in Poznań and the Dendrological Garden of the Poznań University of Life Sciences for providing pollen material for our study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
OW, AF, ŁG conceptualization. OW methodology, investigations, visualization and writing—orginal draft. AF investigations, data analysis, writing—review and editing. ŁG methodology, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wieczorek, O., Frątczak, A. & Grewling, Ł. Allergenic potential of ornamental Cupressales species and its consequences for urban planting. Sci Rep 16, 8887 (2026). https://doi.org/10.1038/s41598-026-40332-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40332-w