Abstract
This study aimed to evaluate the diagnostic efficacy of 68Ga-labeled DOTA-ibandronic acid positron emission tomography/computed tomography (68Ga-DOTA-IBA PET/CT) and 99mTc-labelled methylene diphosphonate (99mTc-MDP) bone scintigraphy (BS) in detecting bone metastases. Patients who underwent both 99mTc-MDP bone scintigraphy and ⁶⁸Ga-DOTA-IBA PET/CT within a one-week interval between March 2022 and April 2024 were retrospectively enrolled in this study. A total of 133 patients (67 males and 66 females; age: 59.98 ± 12.58) were included. The resulting images were subsequently analyzed by seasoned nuclear medicine specialists. Diagnostic efficacy was assessed based on the detection rates of bone metastatic lesions by the two imaging modalities. The maximum standardized uptake values (SUVmax) of metastatic and benign bone lesions identified on ⁶⁸Ga-DOTA-IBA PET/CT were recorded to evaluate the diagnostic performance of ⁶⁸Ga-DOTA-IBA in differentiating malignant from benign bone lesions. A total of 1453 lesions were identified as bone metastases, with 1423 (97.9%) detected using 68Ga-DOTA-IBA PET/CT, and with 1208 lesions (83.1%) identified through optional SPECT-assisted 99mTc-MDP bone scintigraphy (p < 0.001). At the individual patient level, the detection rates of bone metastases using ⁶⁸Ga-DOTA-IBA PET/CT and SPECT-assisted 99mTc-MDP bone scintigraphy were both 132 out of 133 (99.2%), with no statistically significant difference observed. The SUVmax for malignant lesions was markedly higher than that observed in benign lesions (7.18(5.18 ~ 10.59) vs. 2.6(2.0 ~ 3.3), p < 0.001). The area under the curve (AUC) for SUVmax when diagnosing bone metastases with 68Ga-DOTA-IBA PET/CT was 0.958, utilizing a threshold of 4.0 to differentiate between benign and malignant lesions. 68Ga-DOTA-IBA PET/CT has a higher detection rate for skeletal metastases than SPECT-assisted 99mTc-MDP bone scintigraphy. Furthermore, ⁶⁸Ga-DOTA-IBA PET/CT has demonstrated a strong ability to differentiate between benign and malignant bone lesions.
Similar content being viewed by others
Introduction
Bone is the most frequent site for distant metastasis of malignant tumors, following the lung and liver. Prostate, breast, lung, liver, and thyroid carcinomas are particularly prone to metastasizing to bone1,2,3. Complications associated with bone metastases encompass bone pain, pathological fractures (with an incidence ranging from 16% to 60%), hypercalcemia, and spinal cord compression, all of which significantly diminish patients’ quality of life4,5,6. Early diagnosis and treatment of bone metastases are crucial for enhancing the quality of life and extending patient survival7,8.
Routine 99mTc-MDP bone scintigraphy uses technetium-99m-labeled bisphosphonates and is widely recommended for detecting bone metastases. However, its sensitivity and specificity remain suboptimal9,10,11. This is due to the fact that 99mTc-MDP bone scintigraphy depends on osteoblastic activity. Early bone metastases may be overlooked as metastatic cancer cells tend to be preferentially localized within the bone marrow microenvironment12. In addition to tumor formation, local trauma, infection, and degeneration can result in an increased accumulation of skeletal radionuclides. False-negative results may occur when reactive changes are absent or when lesions grow slowly, grow rapidly as purely osteolytic metastases, or lack sufficient vascularity. Such lesions can appear as photon-deficient areas, commonly referred to as “cold spots” 13,14. Although computed tomography (CT) is a routine method for detecting bone metastases, it often has limitations15,16. To enhance the diagnostic sensitivity and specificity of bone metastases, positron-emission tomography (PET) emerges as one of the most promising advanced imaging modalities.
Ibandronic acid (IBA), a third-generation bisphosphonate, possesses anti-resorptive and anti-hypercalcemic properties. The successful synthesis of DOTA-IBA, a DOTA-conjugated ligand, offers a novel strategy for the diagnosis and treatment of bone metastases. In preclinical studies, 68Ga/177Lu-DOTA-IBA exhibits a high affinity for hydroxyapatite and a favorable target-to-background ratio and demonstrates rapid renal clearance, which are key characteristics for effective theranostic agents in bone metastases17. Across multiple solid tumors, 68Ga-DOTA-IBA PET/CT provides a higher detection rate of bone metastases than 99mTc-MDP bone scintigraphy. In addition, it offers a useful imaging platform to guide 177Lu-DOTA-IBA–based diagnosis and therapeutic response assessment in skeletal metastatic disease18. Nevertheless, the scope of tumor types and cases included in existing studies assessing the diagnostic efficacy of 68Ga-DOTA-IBA PET/CT is limited. Consequently, the objective of this study was to assess the diagnostic efficacy of 68Ga-DOTA-IBA PET/CT, BS, and CT scanning for bone metastatic lesions across a range of tumors.
Patients and methods
Patients
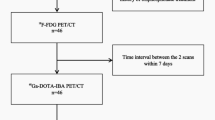
This retrospective study investigates the use of 68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy for the evaluation of bone metastatic lesions. The study was carried out between March 2022 and April 2024 at the Affiliated Hospital of Southwest Medical University (Clinical Trial Registration Number: ChiCTR2200064487). The study was approved by the Research Ethics Committee (Institutional Review Board) of The Affiliated Hospital of Southwest Medical University (approval No. KY2022114), and all patients provided written informed consent before undergoing 68Ga-DOTA-IBA PET/CT. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Patient inclusion criteria included: (1) imaging findings or clinical symptoms indicative of suspected bone metastases; (2) undergoing both 99mTc-MDP bone scintigraphy and ⁶⁸Ga-DOTA-IBA PET/CT; and (3) an interval of no more than one week between the two examinations. Exclusion criteria were (1) pregnant or lactating women, (2) incomplete clinical or imaging data to determine the nature of the bone lesion, (3) diffuse systemic bone metastatic lesions that did not allow for easy documentation of the number of bone metastases, as well as for easy comparison of 99mTc-MDP bone scintigraphy and 68Ga-DOTA-IBA PET/CT bone metastatic lesions. Finally, a total of 133 patients were ultimately enrolled in the study.
Imaging
Weighing is the sole requirement before the 68Ga-DOTA-IBA scan, with no additional preparatory steps required. The labeling of 68Ga-DOTA-IBA is conducted as detailed in the preceding methodology, with an intravenous dose of 68Ga-DOTA-IBA being 1.85MBq/kg (corresponding to 0.05mCi/kg)17. PET/CT imaging (uMI780, United Imaging Healthcare) was conducted approximately 45 to 60 min after the intravenous administration of the tracer. An initial unenhanced high-resolution CT was carried out from the vertex to toes, which was subsequently used for attenuation correction of the PET data. The CT scan parameters were as follows: tube voltage of 120 kV, current of 120 mA, slice thickness of 3.00 mm, slice interval of 5 mm, pitch of 0.813, and reconstructed with bone-specific algorithms. Subsequent PET scans were conducted using 3D acquisition mode, utilizing the same scanner bed as the CT. The resulting images underwent attenuation correction and iterative reconstruction, resulting in transverse, coronal, and sagittal views of the PET/CT scans.
99mTc-MDP Bone scintigraphy was performed 3 to 4 h after intravenous administration of 740–925 MBq (20–25 mCi) of 99mTc-MDP. Whole-body planar bone scintigrams, both anterior and posterior, were obtained from the skull vertex to the toes. Lesion identification on bone scintigraphy was based on visual detection of foci of increased tracer uptake on these planar images. Localized SPECT imaging is performed when the area of increased radiotracer uptake on planar imaging cannot be precisely localized due to anatomical overlap or when the characterization of the lesion remains unclear. SPECT imaging was performed within 25 min following the completion of planar scanning, using a 128 × 128 matrix and a 50 × 40 cm field of view. SPECT/CT was not routinely acquired in all patients, and SPECT/CT was not used as an independent whole-body screening tool to search for additional lesions beyond those detected on planar scintigraphy.
Image analysis
68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy were interpreted by two experienced nuclear medicine physicians. Each reader independently reviewed all examinations in random order, blinded to clinical information and to the findings of the other modality. Visual assessment was used without predefined SUV cut-offs. Any focus of visually increased tracer uptake was considered positive on that modality, whereas CT abnormalities without increased uptake were not regarded as positive findings for the corresponding tracer.
For tracer-avid foci, the readers used the CT component, additional cross-sectional imaging when available (e.g., MRI), and imaging follow-up to classify lesions as metastatic or benign. Owing to technical and ethical limitations, systematic histopathological confirmation of all suspected bone lesions was not feasible in this cohort. A lesion was considered metastatic if at least one of the following criteria was met: (1) progression of a specific skeletal lesion on imaging over ≥ 2 months; (2) a typical multifocal metastatic pattern on bone scintigraphy or ⁶⁸Ga-DOTA-IBA PET/CT, or an increase in lesion number over time; or (3) post-treatment CT demonstrating osteosclerotic or other characteristic malignant changes19. Lesions with imaging appearances clearly compatible with benign entities (e.g., bone islands, hemangiomas, bone cysts, vascular structures) were classified as non-metastatic and excluded from lesion-based analyses. Any discrepancies in lesion detection or classification between the two readers were resolved in a joint consensus review; formal inter-observer or intra-observer agreement statistics (e.g., κ values) were not calculated.
Lesions that remained indeterminate despite follow-up were excluded from lesion-based analyses. Because the present study focused on comparing the diagnostic performance of the two imaging modalities, CT/MRI-suspicious lesions without increased uptake on either tracer were not included in the lesion-level analysis. In cases of diffuse involvement of a single vertebral body, the entire vertebra was counted as one lesion; patients with diffuse skeletal disease in whom individual lesions could not be reliably counted were excluded from lesion-based analysis. For each patient, SUVmax on 68Ga-DOTA-IBA PET/CT was recorded for all metastatic lesions and for up to five benign lesions (bone capillaries or intraosseous vascular channels, bone cysts, bone islands, hemangiomas). Based on baseline or follow-up CT, lesions in the composite reference dataset were categorized as osteoblastic, osteolytic, mixed, or without a clear CT abnormality at baseline. Given that many lesions exhibited mixed or evolving patterns after treatment, this morphology-based stratification should be regarded as exploratory.
Statistics
Statistical analyses were conducted utilizing SPSS version 27.0 (IBM Corporation, Armonk, New York, USA). This analysis aimed to compare the detection rates between the two imaging modalities using the McNemar test. Comparisons of SUVmax between lesions identified on 68Ga-DOTA-IBA scans were performed using the Wilcoxon test, and the differentiation between benign and malignant lesions based on cut-off values was assessed using the receiver operating characteristic (ROC) curve. P-values less than 0.05 were considered statistically significant.
Results
Patient characteristics
The clinical characteristics of the enrolled patients are shown in Table 1. A total of 133 patients (66 females and 67 males; age: 59.98 ± 12.58) were enrolled in the trial. All patients underwent 68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy within one week. Follow-up imaging was assessed for a period of up to 1 year post-study imaging, with a minimum interval of 2 months.
Comparative analysis in 68Ga-DOTA-IBA and 99mTc-MDP scintigraphy
A total of 1,470 skeletal foci of increased tracer uptake were initially identified on 68Ga-DOTA-IBA PET/CT and/or 99mTc-MDP bone scintigraphy. Among these, 14 lesions showed increased uptake only on 68Ga-DOTA-IBA (10 vertebral, 4 iliac), 2 lesions showed increased uptake on both 68Ga-DOTA-IBA and 99mTc-MDP (both vertebral), and 1 lesion showed increased uptake only on 99mTc-MDP (vertebral). Despite CT/MRI correlation and imaging follow-up, these lesions remained indeterminate and were excluded from the lesion-level analysis. Consequently, 1,453 tracer-avid foci were finally confirmed as metastatic and constituted the lesion-based reference dataset for subsequent analyses. Of these, 1,423/1,453 (97.9%) were detected by ⁶⁸Ga-DOTA-IBA PET/CT and 1,208/1,453 (83.1%) by 99mTc-MDP bone scintigraphy with SPECT assistance (P < 0.001). Overall, 1,178 lesions were positive on both modalities, 245 lesions were detected only by 68Ga-DOTA-IBA PET/CT (PET+/MDP−), and 30 lesions were detected only by 99mTc-MDP bone scintigraphy (MDP+/PET−). The detection rates of the two imaging tests in the tumor subgroups are detailed in Table 2.
In the 66 patients in whom 68Ga-DOTA-IBA PET/CT identified more metastatic bone lesions than 99mTc-MDP bone scintigraphy, the primary tumours included breast cancer (n = 20), lung cancer (n = 19), prostate cancer (n = 16), hepatocellular carcinoma (n = 4), colon cancer (n = 3), rectal cancer (n = 3) and nasopharyngeal carcinoma (n = 1). Across these patients, 68Ga-DOTA-IBA PET/CT detected 245 additional metastatic lesions that were not identified as metastatic on 99mTc-MDP scintigraphy. These lesions were located in the skull (n = 8), clavicle and scapula (n = 11), spine (n = 119), ribs and sternum (n = 51), pelvis (n = 44), and extremities (n = 12). The median SUVₘₐₓ of these 245 lesions was 4.9 (interquartile range 4.1–6.4), with a range of 2.9–15.3. On CT, 95 lesions showed a mixed pattern, 86 were osteoblastic, and 60 were osteolytic. Four lesions appeared morphologically normal on baseline CT but developed new osteoblastic changes at follow-up, confirming metastatic involvement. Conversely, in the 13 patients in whom ⁹⁹ᵐTc-MDP bone scintigraphy detected more metastatic lesions than 68Ga-DOTA-IBA PET/CT, the primary tumours were lung cancer (n = 5), breast cancer (n = 5), prostate cancer (n = 2), and hepatocellular carcinoma (n = 1). Altogether, ⁹⁹ᵐTc-MDP scintigraphy identified 30 additional metastatic lesions that were not detected as metastatic on 68Ga-DOTA-IBA PET/CT. These lesions were located in the vertebrae (n = 5), ribs (n = 19), scapula (n = 3), ilium (n = 2), and ischium (n = 1). On CT, 7 of these lesions showed osteoblastic changes, 9 were osteolytic, and 14 exhibited a mixed pattern.
On an individual patient basis, the detection rate of bone metastases was 132/133(99.2%) for 68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy with optional SPECT-assisted. The detection rates of the two imaging tests in the tumor subgroups are detailed in Table 3. No statistically significant differences were detected between 68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy with optional SPECT-assisted. One patient with hepatocellular carcinoma demonstrated no ⁶⁸Ga-DOTA-IBA uptake in bone metastatic lesions on ⁶⁸Ga-DOTA-IBA PET/CT, while exhibiting increased radiotracer uptake on 99mTc-MDP bone scintigraphy. This patient with hepatocellular carcinoma had two lesions in the left scapula, both of which demonstrated osteolytic changes on CT. One patient with colorectal cancer demonstrated increased uptake of ⁶⁸Ga-DOTA-IBA in a bone metastatic lesion on ⁶⁸Ga-DOTA-IBA PET/CT, but showed no radiotracer uptake on 99mTc-MDP bone scintigraphy. This patient with colorectal cancer had two bone metastatic lesions in the spine, which demonstrated osteolytic changes on CT.
When stratified according to CT-based morphology, 517 lesions were classified as osteoblastic, 336 as osteolytic, and 595 as mixed. The lesion-based detection rates of 68Ga-DOTA-IBA PET/CT versus 99mTc-MDP bone scintigraphy were 98.6% (510/517) vs. 83.4% (431/517) for osteoblastic metastases, 97.3% (327/336) vs. 82.1% (276/336) for osteolytic metastases, and 97.6% (581/595) vs. 84.0% (500/595) for mixed lesions. Thus, 68Ga-DOTA-IBA PET/CT showed uniformly high lesion-level detection across all three morphologic subtypes, whereas 99mTc-MDP bone scintigraphy demonstrated consistently lower detection rates. All 5 lesions without baseline CT changes demonstrated increased 68Ga-DOTA-IBA uptake, whereas only 1 of these 5 lesions was detected by 99mTc-MDP bone scintigraphy.
Comparative analysis of SUVmax of benign and malignant bone lesions in 68Ga-DOTA-IBA PET/CT
A total of 1,423 malignant and 547 benign bone lesions were recorded in the ⁶⁸Ga-DOTA-IBA PET/CT scans, along with their corresponding SUVmax values. The SUVmax values of malignant lesions were significantly higher than those of benign lesions (p < 0.001): the median SUVmax of malignant lesions was 7.18(5.18 ~ 10.59), while the median SUVmax of benign lesions was 2.6 (2.0 ~ 3.3). To assess the diagnostic performance, an ROC curve analysis was conducted based on SUVmax values from 68Ga-DOTA-IBA PET/CT scans for benign and malignant lesions (as shown in Fig. 1). The findings revealed that the SUVmax AUC for diagnosing bone metastases in the total cancer on 68Ga-DOTA-IBA PET/CT was 0.958, indicating that SUVmax values from 68Ga-DOTA-IBA PET/CT demonstrated high accuracy in distinguishing between benign and malignant lesions. The maximum Yoden index calculated was 0.79, with the corresponding SUVmax threshold of 4.0.
ROC curves comparing the SUVmax of bone metastatic lesions to benign lesions in 68Ga-DOTA-IBA PET/CT.
Discussion
Previous studies compared the diagnostic efficacy of 68Ga-DOTA-IBA PET/CT and bone scintigraphy for bone metastatic lesions, finding that the detection rate of 68Ga-DOTA-IBA PET/CT was superior to that of bone scintigraphy for lesions17,18,19. Our study evaluated the diagnostic performance of ⁶⁸Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy with optional SPECT-assisted imaging in comparison to previous studies that included patients with various tumor types. In addition, a comparative evaluation of the diagnostic performance of the two modalities within a single tumor type was performed.
68Ga-DOTA-IBA PET/CT has the ability to identify occult bone metastases that are not discernible through conventional imaging techniques. The accurate detection of osseous metastases significantly impacts staging, treatment planning, and the monitoring of therapeutic responses20,21,22. It has been demonstrated that 68Ga-DOTA-IBA PET/CT exhibits a higher detection rate for bone metastases compared to 99mTc-MDP bone scintigraphy17,19. Xiang et al.18 encompassed 45 patients with breast cancer and identified 546 bone metastatic lesions. The detection rate was significantly higher with 68Ga-DOTA-IBA PET/CT at 100% (546/546) compared to 99mTc-MDP WBBS at 67.8% (370/546). In a study conducted by Deng J23 et al., 24 patients were enrolled to compare the diagnostic performance of 68Ga-DOTA-IBA PET/CT and18F-NaF PET/CT for bone metastatic lesions. 68Ga-DOTA-IBA PET/CT exhibited an 81% detection rate, and no significant difference was observed in the diagnostic performance when compared with18F-NaF PET/CT. The detection rate of 68Ga-DOTA-IBA PET/CT in our study modestly varied from the results obtained in the aforementioned studies, potentially attributable to the number of cases included and the type of tumors encompassed. In the analysis of ⁶⁸Ga-DOTA-IBA PET/CT images, only PET data were documented in this study, and areas with osteolytic or osteoblastic changes on CT that lacked increased radiotracer uptake on PET were not considered bone metastases. This may also explain why the lesion detection rate observed in this study slightly differed from that reported in the other two studies.
Although the addition of SPECT to planar acquisitions enhances the diagnostic accuracy of 99mTc-MDP bone scintigraphy in detecting malignant bone involvement9,24, our findings indicate that 68Ga-DOTA-IBA PET/CT possesses a superior detection rate. In comparison to conventional bone scintigraphy, 68Ga-DOTA-IBA PET/CT exhibits superior spatial resolution. In clinical practice, PET/CT routinely acquires tomographic images of the entire skeleton, whereas additional SPECT acquisitions are needed to obtain tomographic data with 99mTc-MDP. This difference likely contributes to the higher detection rates of 68Ga-DOTA-IBA PET/CT for bone metastases. Furthermore, Ibandronate, a third-generation amino bisphosphonate, demonstrates a higher bone affinity compared to MDP and EDTMP. 68Ga-DOTA-IBA PET/CT, utilizing the 68Ga-labeled ligand DOTA-ibandronate, demonstrated a strong affinity for hydroxyapatite and favorable target-to-background ratios (as shown in Fig. 2). This is consistent with the findings from various studies that highlight the high bone targeting and target-to-non-target ratio for bone metastases with 68Ga-DOTA-IBA19,25. This improved bone targeting is probably an important reason for the superior diagnostic performance of 68Ga-DOTA-IBA PET/CT, as it allows more precise localization of bone metastases26. In comparison with 99mTc-MDP bone scintigraphy, certain lesions were identified as positive on 68Ga-DOTA-IBA PET/CT but showed no corresponding osteolytic or osteoblastic changes on CT. This lesion was subsequently confirmed as a bone metastasis through follow-up imaging. These findings may indicate early reactive changes of osteoblasts in response to metastatic deposits, highlighting the potential of 68Ga-DOTA-IBA PET/CT to detect bone metastases at an earlier stage11.
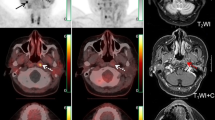
A direct comparison of 99mTc-MDP bone scintigraphy and 68Ga-DOTA-IBA PET/CT images in a prostate cancer patient was conducted over a one-week interval. An 80-year-old man with metastatic prostate cancer. 68Ga-DOTA-IBA PET/CT (B) demonstrated a higher number of bone metastatic lesions when compared with a 99mTc-MDP bone scan (A) and presented them with greater clarity.
As both IBA and MDP are bisphosphonates, the mechanism of radiotracer accumulation in malignant bone lesions is considered similar between ⁶⁸Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy. The radiotracer is incorporated into the bone surface, and its uptake is influenced by local blood flow and osteoblastic activity27,28. A dynamic balance between osteoclastic and osteoblastic activity characterizes the process of bone remodeling. The simultaneous activity of both cell types accounts for the appearance of bone metastases as osteolytic (bone resorptive), osteoblastic (bone forming), or mixed lesions on imaging29. Previous work has suggested that bisphosphonate-based tracers may be relatively less sensitive for purely osteolytic metastases, such as those frequently observed in breast cancer and hepatocellular carcinoma30,31, and more sensitive for predominantly osteoblastic metastases, such as those typical of prostate cancer32,33. However, in this cohort, the lesion-level detection rates of ⁶⁸Ga-DOTA-IBA PET/CT differed only minimally between tumour types. ⁶⁸Ga-DOTA-IBA PET/CT maintained high lesion-level detection rates (> 97%) for osteoblastic, osteolytic, and mixed metastases, with only small numerical differences between these subgroups. Because this morphology-stratified analysis was derived from a tracer-avid reference dataset, these findings should be regarded as exploratory and hypothesis-generating rather than as definitive evidence of performance differences according to CT morphology.
In some lesions, CT demonstrated osteoblastic/sclerotic changes without corresponding increased ⁶⁸Ga-DOTA-IBA uptake, which may reflect low residual osteoblastic activity or treatment-induced healing. The pattern of “CT sclerosis with absent PET uptake” should therefore be interpreted with caution, particularly in treated patients. This discordance may represent a known pitfall in post-treatment imaging, where low or absent uptake in sclerotic lesions could indicate successful therapy-induced healing rather than a low osteoblastic phenotype per se10,34,35. Accordingly, the differential diagnosis should take into account both treatment effects and variability in osteoblastic metabolic activity.
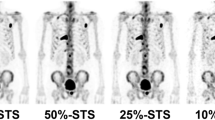
In our lesion-based analysis, 68Ga-DOTA-IBA PET/CT detected more metastatic lesions overall than 99mTc-MDP bone scintigraphy; however, a small subset of lesions (30 lesions in 13 patients) were visualized only on 99mTc-MDP bone scintigraphy and not on 68Ga-DOTA-IBA PET/CT (as shown in Fig. 3). These discordant foci were mainly located in the vertebrae, ribs, scapula, and pelvis, and most showed osteosclerotic changes on CT. Several mechanisms may account for this pattern. Although both 99mTc-MDP and 68Ga-DOTA-IBA are bone-seeking bisphosphonate-based tracers, they differ in pharmacokinetics (e.g., 68Ga-DOTA-IBA exhibits rapid renal clearance and high bone retention, while 99mTc-MDP has slower soft tissue clearance), binding characteristics (e.g., ibandronic acid in 68Ga-DOTA-IBA has stronger antiresorptive affinity but may be influenced by mineralization regulation), and imaging time-points (e.g., 68Ga-DOTA-IBA at 40–60 min post-injection versus 99mTc-MDP at 3–4 h)26,27,36,37. In addition, some of the MDP-positive but IBA-negative lesions were small sclerotic foci in anatomically complex regions of the axial skeleton, where partial-volume effects, local noise, and background activity may have further reduced lesion conspicuity on PET38,39. These factors likely contribute to the small number of 99mTc-MDP–positive but 68Ga-DOTA-IBA–negative lesions and help explain why the number of lesions detected by 68Ga-DOTA-IBA PET/CT (1,423) was slightly lower than the total number of metastatic lesions defined by the combined tracer-based reference dataset (1,453).
A 54-year-old woman with treated breast cancer showing lesions visible on 99mTc-MDP bone scintigraphy but not on 68Ga-DOTA-IBA PET/CT. 99mTc-MDP bone scintigraphy (A, B) demonstrates increased tracer uptake in parts of the spine, the left fifth rib, the right tenth rib, and the right scapula (arrows). 68Ga-DOTA-IBA PET MIP image (C) shows increased uptake in only one vertebral body and the left fifth rib(arrows).
99mTc-MDP bone scintigraphy, a commonly employed method for evaluating bone metastases in oncology patients, suffers from low specificity, resulting in a higher incidence of inconclusive findings40. Numerous pathological conditions, such as trauma, postoperative alterations, degenerative disorders, and infections, may result in false-positive scans, thereby complicating the differentiation between benign and malignant diseases13,14. Within the skull, distinguishing metastases from both normal anatomical variants, including arachnoid granulomas and venous lakes, and benign lesions presents a significant diagnostic challenge41. Our study included a quantitative analysis of lesions detected by 68Ga-DOTA-IBA PET/CT. We established an SUVmax threshold to help distinguish benign from malignant lesions. This threshold is particularly useful in cases with diagnostic uncertainty.
In this study, 68Ga-DOTA-IBA PET/CT and 99mTc-MDP bone scintigraphy identified the same number of patients with bone metastases at the patient level. However, at the lesion level, 68Ga-DOTA-IBA PET/CT detected a greater number of metastatic sites. For lesions that appear indeterminate on 99mTc-MDP bone scintigrams, 68Ga-DOTA-IBA PET/CT may offer additional diagnostic value by providing quantitative SUVmax information. This more detailed depiction of skeletal involvement can help clinicians better assess overall disease burden and refine treatment planning. Furthermore, the theranostic pair 68Ga/177Lu-DOTA-IBA provides a foundation for precision treatment in patients with cancer.
While our study demonstrated that ⁶⁸Ga-DOTA-IBA PET/CT has excellent diagnostic performance for detecting bone metastases, several limitations should be acknowledged. First, because of the high metastatic burden, diffuse skeletal involvement, and the procedural complexity of PET-guided bone biopsy, a uniform histopathologic gold standard could not be obtained. Bone metastases were therefore defined using an imaging-based composite reference standard that combined multimodality imaging and radiologic follow-up. By design, the lesion-level analysis only included lesions showing increased uptake on at least one of the two tracers (⁶⁸Ga-DOTA-IBA PET/CT or ⁹⁹ᵐTc-MDP bone scintigraphy); CT- or MRI-suspicious lesions without radiotracer uptake were not counted in the total number of metastatic lesions. Consequently, the lesion-based detection rates reported here reflect the performance of both tracers within this tracer-avid subset, and the overall metastatic burden—particularly very early or minimally osteoblastic lesions without detectable uptake—may be underestimated. At the lesion level, 17 tracer-avid foci lesions that remained indeterminate despite follow-up were excluded from the lesion-based analysis; this conservative approach may slightly overestimate lesion-level detection rates and limit the generalizability of our findings to patients with highly equivocal or very early lesions, although the number of excluded lesions was small relative to the overall lesion burden. In addition, many patients (72.9%) had received systemic or local therapy before imaging, which may influence tracer uptake patterns; however, this reflects real-world practice and increases the clinical relevance of our results. Third, this was a retrospective study, and data collection focused on the number of bone metastases detected by each modality and on the ability of ⁶⁸Ga-DOTA-IBA to differentiate benign from malignant bone lesions; formal sensitivity and specificity were not calculated and should be further validated in larger, prospectively designed cohorts. Finally, ⁹⁹99mTc-MDP SPECT/CT was performed only in selected regions with increased or equivocal planar uptake, and was primarily used for localisation and morphological characterisation rather than as a systematic whole-body tomographic acquisition. This restricted use of SPECT/CT may have led to some underestimation of the maximum achievable lesion detection performance of bone scintigraphy and should be regarded as a methodological limitation when interpreting the comparison with ⁶⁸Ga-DOTA-IBA PET/CT.
Conclusion
Our retrospective study demonstrated that 68Ga-DOTA-IBA PET/CT exhibits a higher detection rate compared to conventional 99mTc-MDP bone scintigraphy for the detection of bone metastases. In addition, high accuracy in differentiating between benign and malignant lesions is demonstrated by 68Ga-DOTA-IBA PET/CT.
Data availability
The datasets we used during the current study are not publicly available due to privacy issues, but are available from the corresponding author on reasonable request.
References
Chow, E. et al. Dexamethasone for the prophylaxis of radiation-induced pain flare after palliative radiotherapy for bone metastases—a pilot study. Support Care Cancer. 15, 643–647. https://doi.org/10.1007/s00520-007-0217-z (2007).
Gdowski, A. S., Ranjan, A. & Vishwanatha, J. K. Current concepts in bone metastasis, contemporary therapeutic strategies and ongoing clinical trials. J. Exp. Clin. Cancer Res. 36, 108. https://doi.org/10.1186/s13046-017-0578-1 (2017).
Siegel, R. L., Giaquinto, A. N., Jemal, A. & Cancer statistics CA. Cancer J. Clin. 74, 12–49. (2024). https://doi.org/10.3322/caac.21820 (2024).
Hortobagyi, G. N. et al. Long-term prevention of skeletal complications of metastatic breast cancer with pamidronate. Protocol 19 Aredia breast cancer study group. J. Clin. Oncol. 16, 2038–2044. https://doi.org/10.1200/JCO.1998.16.6.2038 (1998).
Roodman, G. D. Biology of osteoclast activation in cancer. J. Clin. Oncol. 19, 3562–3571. https://doi.org/10.1200/JCO.2001.19.15.3562 (2001).
Rubens, R. D. Bone metastases–the clinical problem. Eur. J. Cancer. 34, 210–213. https://doi.org/10.1016/s0959-8049(97)10128-9 (1998).
Eshghi, A., Covington, M. F., Eshghi, N. & Kuo, P. H. Utility of PET to appropriately select patients for PSMA-Targeted theranostics. Clin. Nucl. Med. 47, 488–495. https://doi.org/10.1097/rlu.0000000000004196 (2022).
Kabunda, J. et al. Comparing 99mTc-PSMA to 99mTc-MDP in prostate cancer staging of the skeletal system. Clin. Nucl. Med. 46, 562–568. https://doi.org/10.1097/rlu.0000000000003702 (2021).
Cook, G. J. & Fogelman, I. The role of positron emission tomography in skeletal disease. Semin Nucl. Med. 31, 50–61. https://doi.org/10.1053/snuc.2001.18746 (2001).
Mohseninia, N. et al. Bone metastasis in prostate cancer: bone scan versus PET Imaging. Semin. Nucl. Med. 54, 97–118. https://doi.org/10.1053/j.semnuclmed.2023.07.004 (2024).
Cook, G. J. R. & Goh, V. Molecular imaging of bone metastases and their response to therapy. J. Nucl. Med. 61, 799–806. https://doi.org/10.2967/jnumed.119.234260 (2020).
Algra, P. R. et al. Detection of vertebral metastases: comparison between MR imaging and bone scintigraphy. Radiographics 11, 219–232. https://doi.org/10.1148/radiographics.11.2.2028061 (1991).
Galasko, C. S. & Doyle, F. H. The detection of skeletal metastases from mammary cancer. A regional comparison between radiology and scintigraphy. Clin. Radiol. 23, 295–297. https://doi.org/10.1016/s0009-9260(72)80051-5 (1972).
Horiuchi-Suzuki, K. et al. Skeletal affinity of Tc(V)-DMS is bone cell mediated and pH dependent. Eur. J. Nucl. Med. Mol. Imaging. 31, 388–398. https://doi.org/10.1007/s00259-003-1364-1 (2004).
Noguchi, S. et al. Deep learning-based algorithm improved radiologists’ performance in bone metastases detection on CT. Eur. Radiol. 32, 7976–7987. https://doi.org/10.1007/s00330-022-08741-3 (2022).
Luo, H. et al. Spectral CT assists differentiation of osteoblastic bone metastasis from bone Island in newly diagnosed cancer patients. Eur. Radiol. 34, 60–68. https://doi.org/10.1007/s00330-023-10036-0 (2024).
Qiu, L. et al. Safety and efficacy of 68Ga- or 177Lu-Labeled DOTA-IBA as a novel theranostic radiopharmaceutical for bone metastases. Clin. Nucl. Med. 48, 489–496. https://doi.org/10.1097/rlu.0000000000004634 (2023).
Xiang, F. et al. Prospective comparison of 68Ga-DOTA-ibandronate and bone scans for detecting bone metastases in breast cancer. Front. Oncol. 14, 1428498. https://doi.org/10.3389/fonc.2024.1428498 (2024).
Bénard, F. et al. Intra-individual comparison of 18F-sodium fluoride PET–CT and 99mTc bone scintigraphy with SPECT in patients with prostate cancer or breast cancer at high risk for skeletal metastases (MITNEC-A1): a multicentre, phase 3 trial. Lancet Oncol. 23, 1499–1507. https://doi.org/10.1016/s1470-2045(22)00642-8 (2022).
Wang, Y. et al. Preparation, biological characterization and preliminary human imaging studies of 68Ga-DOTA-IBA. Front. Oncol. 12, 1027792. https://doi.org/10.3389/fonc.2022.1027792 (2022).
von Moos, R. et al. Management of bone health in solid tumours: from bisphosphonates to a monoclonal antibody. Cancer Treat. Rev. 76, 57–67. https://doi.org/10.1016/j.ctrv.2019.05.003 (2019).
Thurairaja, R., McFarlane, J., Traill, Z. & Persad, R. State-of-the-art approaches to detecting early bone metastasis in prostate cancer. BJU Int. 94, 268–271. https://doi.org/10.1111/j.1464-410X.2003.04960.x (2004).
Bortot, D. C. et al. 18F-Fluoride PET/CT is highly effective for excluding bone metastases even in patients with equivocal bone scintigraphy. Eur. J. Nucl. Med. Mol. Imaging. 39, 1730–1736. https://doi.org/10.1007/s00259-012-2195-8 (2012).
Deng, J., Yang, J., Wang, Y., Liu, G. & Chen, Y. Comparison of the relative diagnostic performance of 68Ga-DOTA-IBA and 18F-NaF for the detection of bone metastasis. Front. Oncol. 14, 1364311. https://doi.org/10.3389/fonc.2024.1364311 (2024).
Ghanem, N. et al. Diagnostic value of MRI in comparison to scintigraphy, PET, MS-CT and PET/CT for the detection of metastases of bone. Eur. J. Radiol. 55, 41–55. https://doi.org/10.1016/j.ejrad.2005.01.016 (2005).
Yang, J. et al. Biodistribution and internal dosimetry of 68 Ga-DOTA-IBA PET imaging for patients with bone metastases. Clin. Nucl. Med. 48, 847–852. https://doi.org/10.1097/RLU.0000000000004757 (2023).
Wang, Q. et al. Lutetium177-Labeled DOTA-Ibandronate: A novel radiopharmaceutical for targeted treatment of bone metastases. Mol. Pharm. 20, 1788–1795. https://doi.org/10.1021/acs.molpharmaceut.2c00978 (2023).
Blake, G. M., Park-Holohan, S. J., Cook, G. J. & Fogelman, I. Quantitative studies of bone with the use of 18F-fluoride and 99mTc-methylene diphosphonate. Semin Nucl. Med. 31, 28–49. https://doi.org/10.1053/snuc.2001.18742 (2001).
Malhotra, P. & Berman, C. G. Evaluation of bone metastases in lung cancer. Improved sensitivity and specificity of PET over bone scanning. Cancer Control. 9, 254–260. https://doi.org/10.1177/107327480200900311 (2002).
Theriault, R. L. & Hortobagyi, G. N. Bone metastasis in breast cancer. Anticancer Drugs. 3, 455–462. https://doi.org/10.1097/00001813-199210000-00002 (1992).
Becker, A. K., Tso, D. K., Harris, A. C., Malfair, D. & Chang, S. D. Extrahepatic metastases of hepatocellular carcinoma: A spectrum of imaging findings. Can. Assoc. Radiol. J. 65, 60–66. https://doi.org/10.1016/j.carj.2013.05.004 (2014).
Mandal, C. C. Osteolytic metastasis in breast cancer: effective prevention strategies. Expert Rev. Anticancer Ther. 20, 797–811. https://doi.org/10.1080/14737140.2020.1807950 (2020).
Guise, T. A. et al. Basic mechanisms responsible for osteolytic and osteoblastic bone metastases. Clin. Cancer Res. 12, 6213s–6216. https://doi.org/10.1158/1078-0432.CCR-06-1007 (2006). s.
O’Sullivan, G. J., Carty, F. L. & Cronin, C. G. Imaging of bone metastasis: an update. World J. Radiol. 7, 202–211. https://doi.org/10.4329/wjr.v7.i8.202 (2015).
Ulmert, D., Solnes, L. & Thorek, D. L. J. Contemporary approaches for imaging skeletal metastasis. Bone Res. https://doi.org/10.1038/boneres.2015.24 (2015). 3.
Wong, K. K. & Piert, M. Dynamic bone imaging with 99mTc-labeled diphosphonates and 18F-NaF: mechanisms and applications. J. Nucl. Med. 54, 590–599. https://doi.org/10.2967/jnumed.112.114298 (2013).
Zuckier, L. S. & Martineau, P. Altered biodistribution of radiopharmaceuticals used in bone scintigraphy. Semin Nucl. Med. 45, 81–96. https://doi.org/10.1053/j.semnuclmed.2014.07.007 (2015).
Grecchi, E. et al. Multimodal Partial-Volume correction: application to 18F-Fluoride PET/CT bone metastases studies. J. Nucl. Med. 56, 1408–1414. https://doi.org/10.2967/jnumed.115.160598 (2015).
Marquis, H., Willowson, K. P. & Bailey, D. L. Partial volume effect in SPECT & PET imaging and impact on radionuclide dosimetry estimates. Asia Ocean. J. Nucl. Med. Biol. 11, 44–54. https://doi.org/10.22038/AOJNMB.2022.63827.1448 (2023).
Nicolini, A., Ferrari, P., Sagripanti, A. & Carpi, A. The role of tumour markers in predicting skeletal metastases in breast cancer patients with equivocal bone scintigraphy. Br. J. Cancer. 79, 1443–1447. https://doi.org/10.1038/sj.bjc.6690230 (1999).
Gerety, E. L. et al. Prospective study evaluating the relative sensitivity of 18F-NaF PET/CT for detecting skeletal metastases from renal cell carcinoma in comparison to multidetector CT and 99mTc-MDP bone scintigraphy, using an adaptive trial design. Ann. Oncol. 26, 2113–2118. https://doi.org/10.1093/annonc/mdv289 (2015).
Acknowledgements
We are grateful to the members of the Department of Nuclear Medicine, The Affiliated Hospital, Southwest Medical University, and Nuclear Medicine and Molecular Imaging Key Laboratory of Sichuan Province for their technical guidance, cooperation, and assistance in completing this study. We are also grateful for the financial support given by the major science and technology project in Gansu Province (23ZDFA014) and the school-level scientific research project of Southwest Medical University (Grant No. 2024ZKY082).
Funding
This work was supported by the major science and technology project in Gansu Province (23ZDFA014) and the school-level scientific research project of Southwest Medical University (Grant No. 2024ZKY082).
Author information
Authors and Affiliations
Contributions
Xinyi Lin, Na Zhang, Tingting Xu, and Yue Chen contributed to the study design, and Xinyi Lin wrote the manuscript. Xinyi Lin, Na Zhang, Rongliang Wang, Huajun Liu, and Wei Wang collected the clinical data of patients. Xinyi Lin and Na Zhang analyzed the clinical data of patients. Yue Chen and Tingting Xu were responsible for revising important intellectual content. Yue Chen and Tingting Xu contributed equally to this paper and shared joint corresponding authorship. All authors read and approved the final manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lin, X., Zhang, N., Wang, R. et al. The performance of 68Ga-DOTA-IBA PET/CT for detecting bone metastases compared with 99mTc-MDP bone scintigraphy. Sci Rep 16, 10174 (2026). https://doi.org/10.1038/s41598-026-40411-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40411-y