Abstract
Preventing anastomotic leakage (AL) is crucial in rectal surgery, and several surgical techniques have been proposed to reduce the risk and severity of AL. This study evaluated the burst pressure at the double-stapling anastomosis in a porcine model to provide evidence to support the clinical efficacy of powered circular staplers and anastomotic reinforcing sutures. An experimental study using an ex vivo porcine model was performed to measure burst pressure as an AL test. We compared four groups with and without the placement of reinforcing sutures at the anastomotic site in each model using a powered and manual circular stapler. Four models were created in each of the four groups, for a total of 16 porcine test models. Anastomotic reinforcing sutures with a powered circular stapler improved the burst pressure by approximately 8% (p = 0.996), whereas those performed using a manual circular stapler improved the burst pressure by approximately 45% (p = 0.403). The powered circular stapler with anastomotic reinforcing sutures (n = 4, 28.7 ± 2.3 mmHg) increased burst pressure by about 63% compared to manual circular stapler without anastomotic reinforcing sutures (n = 4, 17.6 ± 1.0 mmHg) (p = 0.048). Our study suggests that the intraluminal pressure at the double-stapling anastomotic site may be improved by using a powered circular stapler or by adding an anastomotic reinforcing suture.
Similar content being viewed by others
Introduction
Anastomotic leakage (AL) is among the most serious complications after rectal cancer surgery, contributing to increased morbidity, mortality, and poorer oncological outcomes. Despite improvements in surgical instruments and the standardization of techniques, its incidence remains around 10% in recent studies1,2.
Gastrointestinal surgeons are particularly concerned with perioperative complications, especially postoperative AL, which is influenced by factors such as anastomotic vascular perfusion, anastomotic tension, and intraluminal pressure at the anastomotic site3. Preventing AL is therefore crucial in rectal surgery, and several surgical techniques have been proposed to reduce the risk and severity.
With the increasing global adoption of circular staplers for rectal surgery, operative time has been reduced and safety has improved compared with hand-sewn anastomosis. However, the manual circular stapler has a relatively high incidence of technical errors, particularly among gastroenterological surgeons with smaller hand sizes4, and these errors have been associated with a significantly higher risk of gastrointestinal bleeding as well as postoperative AL5.
In March of 2019, the first ETHICON CIRCULAR Powered Stapler (ECP, Ethicon Endo-Surgery, Cincinnati, OH, USA) was introduced to the US markets and subsequently launched in Japan approximately six months later. Powered stapling systems are operated by battery packs that deliver consistent firing force, enabling uniform staple formation and more reliable anastomoses regardless of the surgeon’s grip strength. Several studies have reported that the ECP improves stability and safety of stapling formation, helps prevent AL, and reduces healthcare costs compared with the manual ETHICON Circular Stapler (CDH, Ethicon Endo-Surgery, Cincinnati, OH, USA)6,7.
In addition, several reports have suggested that placement of reinforcing sutures at the anastomotic site can reduce anastomotic traction and help prevent AL8. Because double-stapling anastomosis may create weak points where staple lines intersect, reinforcing sutures may improve intraluminal pressure in the presence of high intrarectal pressure.
The aim of this study was to evaluate the burst pressure at the double-stapling anastomosis in a porcine model to support the clinical efficacy of ECP and anastomotic reinforced suturing.
Methods
To determine the relevant burst pressure for testing of AL, a porcine model was used to establish an end-to-end anastomosis with the double-stapling technique (DST). We used commercial porcine colon specimens sourced from Tokyo Shibaura Zouki Co., Ltd., a certified supplier of biological research materials in Japan. Because the specimens were supplied as isolated colon segments on a commercial basis, the exact anatomical level of resection could not be determined. All procedures and steps in our study were performed in accordance with the ethical standards of NHO Beppu Medical Center and were performed in accordance with relevant guidelines and regulations.
The colon was transected with a GST60T linear stapler using the Powered ECHELON FLEX + 60 (Ethicon Endo-Surgery, Cincinnati, OH, USA) in all groups. The colon stump was fitted with a purse-string suture and the anvil head of the circular stapler (ECP or CDH) was fixed at the edge of the colon stump. The ECP or CDH was inserted through the open stump of the colon and positioned in the closed stump, and end-to-end anastomosis using DST was performed. After performing the anastomosis, reinforcing sutures using 3-0 VICRYL (Ethicon Endo-Surgery, Cincinnati, OH, USA) were placed at the anastomotic site. Four reinforcing sutures were placed along the staple line: two at the crossing points of the staple lines, which represent potential weak areas8,9, and two on the anterior wall.
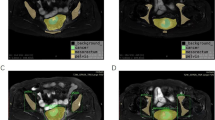
To confirm the burst pressure at the staple line, the intraluminal pressure needed to cause a visually definite bubble leakage was determined by inflating the colon samples underwater, and leakage was measured using a handheld digital manometer (PG-100 N; Nidec Components Corporation, Tokyo, Japan) (Fig. 1). We compared four groups with and without reinforcing sutures at the site of anastomosis in each DST model using ECP and CDH. DST models were created in each of the four specimens, resulting in a total of 16 DST models. All anastomoses were constructed and tested on the same day in the same room under identical ambient conditions to minimize handling bias and atmospheric variation. Furthermore, all procedures were performed by the same experienced surgeon familiar with both manual and powered circular staplers, with burst pressure measurements initiated approximately 10 min after completion of each anastomosis to standardize tissue conditions. The handheld digital manometer measured in kilopascal (kPa), which were converted to millimeters of mercury (mmHg; 1 kPa = 7.501 mmHg).
Measurement of burst pressure at the staple line. The intraluminal pressure required to cause a visually definite bubble leakage was measured using a digital manometer.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics version 28 (Chicago, IL, USA). Testing for a significant difference in means between each burst pressure of the four groups was performed using multiple comparisons according to the Dunnett’s T3 post-hoc test. All tests were two-sided, and statistical significance was set at p < 0.05.
Results
The burst pressure at the double-stapling anastomotic site was measured in all animal models. For the DST models using CDH, the manual circular stapler, the burst pressures of the group with and without anastomotic reinforcing sutures were 25.5 ± 7.45 mmHg (n = 4) and 17.6 ± 1.98 mmHg (n = 4), respectively. Additionally, following anastomosis using ECP, the powered circular stapler, the burst pressures of the group with and without anastomotic reinforcing sutures were 28.7 ± 6.90 mmHg (n = 4) and 26.6 ± 7.97 mmHg (n = 4), respectively.
In both DST models using CDH and ECP, there were no significant differences in burst pressure improvement in the presence or absence of anastomotic reinforcing sutures; however, the anastomotic reinforcing sutures with ECP improved the burst pressure by approximately 8% (p = 0.996), whereas CDH improved the burst pressure by approximately 45% (p = 0.403), respectively. However, ECP with anastomotic reinforcing sutures (n = 4, 28.7 ± 2.3 mmHg) significantly increased the burst pressure by approximately 63% compared to CDH without the need of anastomotic reinforcing sutures (n = 4, 17.6 ± 1.0 mmHg) (p = 0.048) (Fig. 2). In most models, bubble leakage was observed at the crossing points of the staple lines, which have been reported to represent structurally weak areas of the double-stapling line9.
Box plot showing burst pressure differences across the four porcine models used in this study. ECP with anastomotic reinforced suturing increased burst pressure resistance by approximately 63% compared to CDH without anastomotic-reinforced suturing (p = 0.048).
Discussion
Although the introduction of the circular stapler has dramatically reduced postoperative complications related to anastomosis compared with those occurring following hand-sewn anastomoses, the causes of complications cannot be attributed solely to technical errors at the anastomosis. Various studies have reported a reduction in the incidence of AL after rectal cancer surgery, and anastomotic vascular perfusion, anastomotic tension, and intraluminal pressure at the anastomotic site have been suggested to be associated with the risk of developing postoperative AL3.
We previously showed that there was no significant difference in the incidence of AL depending on the level of inferior mesenteric artery (IMA) ligation in rectal cancer surgery10, although the clinical impact with respect to the level of IMA ligation has been controversial. On the other hand, Japanese EssentiAL Trial Group recently showed that indocyanine green fluorescence imaging (ICG-FI), which assesses anastomotic vascular perfusion, significantly reduced the AL rate by 4.2% in rectal cancer surgery11. ICG-FI is a revolutionary technology that can objectively evaluate blood flow at the anastomotic site and has the potential to advance colorectal surgery further by quantifying fluorescent signals.
Rojatkar et al. showed that ECP had lower stapling and compressive forces than CDH and created secure anastomoses with higher intraluminal pressure and less bleeding than CDH, with no significant difference in anastomotic vascular perfusion3. Furthermore, several studies have shown that ECP allows safe stapling by reducing workload on anastomotic tissue, and the creation of an anastomosis with ECP is associated with lower incidence rates of several surgical complications6,12. Although our study found no statistically significant differences, ECP increased the burst pressure by 51% compared with CDH (p = 0.356). The burst pressure of ECP without anastomotic reinforcing sutures was nearly equivalent that of CDH with anastomotic reinforcing sutures (26.6 ± 7.97 mmHg vs. 25.5 ± 7.45 mmHg), and also ECP with or without anastomotic reinforcing sutures maintained a higher intraluminal pressure than the manual type. This finding may be explained by the mechanical stability of the powered device, which standardizes the compression force and minimizes surgeon-dependent variation in staple formation, thereby reducing the incremental effect of additional reinforcing sutures. In addition, the ECP incorporates several mechanical innovations − 3D stapling technology, controlled tissue compression, and gripping surface technology - which distribute compression evenly, minimize mucosal injury at the anastomotic site, and enable more stable full-thickness tissue approximation3. These design elements likely contribute to improved mechanical stability and burst pressure performance at the anastomotic site.
Several studies have recently addressed that the placement of reinforcing sutures may improve anastomotic intraluminal pressure and reduce the incidence of AL after rectal cancer surgery. Maeda et al. demonstrated that intracorporeal reinforcing sutures placed along the staple line significantly reduced the incidence of AL after laparoscopic rectal cancer surgery, particularly in patients with tumors located close to the anal verge or with large tumor size8. In our study, the anastomotic reinforcing sutures improved the intraluminal pressure, although the difference was not statistically significant. This suggests that while the powered circular stapler itself ensures stable compression and staple formation, reinforcing sutures may provide an additional benefit in selected situations, especially when anastomotic tension is high or staple overlap may create potential weak areas. However, their application may be technically limited in cases with a narrow pelvis or lower anastomotic location, where access to the suture site is restricted.
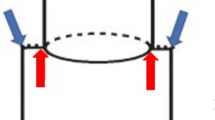
From a physiological standpoint, although no consistent threshold burst pressure predictive of clinical AL has been established, manometric data show that rectal pressures during defecatory maneuvers can vary widely. MacDonald et al. reported intrarectal pressures reaching approximately 70 cmH₂O during balloon evacuation13. Therefore, our burst pressure measurements should be interpreted as comparative mechanical indicators rather than definitive clinical cutoffs. Taken together, these results suggest that both powered circular staplers and reinforcing sutures contribute to strengthening anastomotic integrity through different mechanisms. While the powered stapler provides consistent compression and staple formation, reinforcing sutures may offer additional security in situations with higher anastomotic tension or weak points along the staple line. Thus, selecting the appropriate technique according to intraoperative conditions could help reduce postoperative AL (Fig. 3).
Intracorporeal reinforcing sutures. Reinforcing sutures were placed laparoscopically at the staple line after double-stapling anastomosis.
A major limitation of this study is the small sample size, with only four porcine models per group. This ex vivo study should therefore be regarded as a preliminary proof-of-concept investigation, providing limited data to guide future studies with larger sample sizes. Nevertheless, our findings indicate that both powered stapling and anastomotic reinforcement contribute to improved anastomotic integrity through distinct mechanisms. The powered stapler provides mechanical consistency, whereas reinforcing sutures may offer additional support in technically demanding cases. Future clinical trials with larger cohorts are warranted to validate these findings and to determine when and how powered stapling and reinforcing sutures should be optimally applied to maximize anastomotic safety.
Conclusions
Many factors contribute to postoperative AL, and continuous surgical research together with advances in clinical instruments is essential to elucidate its underlying causes. To the best of our knowledge, this is the first study to investigate the changes in burst pressure associated with powered circular staplers and anastomotic reinforcing sutures in an ex vivo model. Although the sample size was small, our findings suggest that intraluminal pressure at the double-stapling anastomotic site can be improved by using a powered circular stapler and/or adding an anastomotic reinforcing suture. Further studies with larger sample sizes are warranted to validate these results and to determine whether the favorable outcomes observed ex vivo can be translated into clinical practice.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Watanabe, T. et al. Prediction model for complications after low anterior resection based on data from 33,411 Japanese patients included in the National clinical database. Surgery 161 (6), 1597–1608 (2017).
Hida, K. et al. Open versus laparoscopic surgery for advanced low rectal cancer: A large, multicenter, propensity score matched cohort study in Japan. Ann. Surg. 268 (2), 318–324 (2018).
Rojatkar, P. et al. A novel powered circular stapler designed for creating secure anastomoses. Med. Devices Diagn. Eng. 2 (2), 94–100 (2017).
Kono, E. et al. Rating and issues of mechanical anastomotic staplers in surgical practice: A survey of 241 Japanese gastroenterological surgeons. Surg. Today. 42(10), 962–972 (2012).
Offodile II, A. C. et al. High incidence of technical errors involving the EEA circular stapler: a single institution experience. J. Am. Coll. Surg. 210 (3), 331–335 (2010).
Pla-Martí, V. et al. Impact of the novel powered circular stapler on risk of anastomotic leakage in colorectal anastomosis: a propensity score-matched study. Tech. Coloproctol. 25 (3), 279–284 (2021).
Pollack, E. et al. Economic analysis of leak complications in anastomoses performed with powered versus manual circular staplers in left-sided colorectal resections: A US-based cost analysis. Clinicoecon Outcomes Res. 13, 531–540 (2021).
Maeda, K. et al. Efficacy of intracorporeal reinforcing sutures for anastomotic leakage after laparoscopic surgery for rectal cancer. Surg. Endosc. 29(12), 3535–3542 (2015).
Fiorillo, C. et al. Circular staplers and anastomotic leakage in colorectal surgery. BJS Open. 9(2), zrae170 (2025).
Yoshida, D. et al. Oncological impact of the level of inferior mesenteric artery ligation in low rectal cancer surgery. Anticancer Res. 43(7), 3225–3233 (2023).
Watanabe, J. et al. Blood perfusion assessment by indocyanine green fluorescence imaging for minimally invasive rectal cancer surgery (EssentiAL trial): A randomized clinical trial. Ann. Surg. 278(4), e688–e694 (2023).
Sylla, P. et al. Outcomes associated with the use of a new powered circular stapler for left-sided colorectal reconstructions: a propensity score matching-adjusted indirect comparison with manual circular staplers. Surg. Endosc. 36(4), 2541–2553 (2022).
MacDonald, A. et al. Relationship between intra-abdominal and intrarectal pressure in the proctometrogram. Br. J. Surg. 80(8), 1070–1071 (1993).
Acknowledgements
The authors would like to thank Editage (www.editage.com) for the English language editing.
Funding
No funding was received for the conduct of this study.
Author information
Authors and Affiliations
Contributions
D.Y., T.H., and S.N. contributed to the study conception and design, data collection, data analysis, and manuscript writing.W.O., S.I., and K.N. contributed to critical revisions that were important for the intellectual content. K.Y., T.M. and H.K.approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All experiments were conducted on ex vivo porcine large intestine models obtained from animals sacrificed for food purposes and not for the current study. All procedures performed in accordance with the ethical standards of the NHO Beppu Medical Center Institutional Review Board.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yoshida, D., Hasegawa, T., Nakashima, S. et al. Assessment of reinforcing sutures of double-stapling anastomosis with manual and powered circular staplers in Porcine models. Sci Rep 16, 9710 (2026). https://doi.org/10.1038/s41598-026-40474-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40474-x