Abstract
This study was designed to assess the prognostic significance of nutritional risk index (NRI) in nasopharyngeal carcinoma (NPC) patients. In this dual-center study, 1,174 patients were enrolled. Kaplan-Meier analysis assessed the association between NRI and prognosis. Independent prognostic factors for nomogram construction were identified using multivariate Cox regression analysis. The predictive nomogram underwent assessment through decision curve analysis (DCA) and calibration curves. The median NRI value was 112.11. Patients with high NRI (≥ 112.11) demonstrated significantly better survival outcomes than those with low NRI (< 112.11) in both cohorts. Multivariate analysis identified that NRI (≥ 112.11 vs. < 112.11) was an independent prognostic factor for distant metastasis-free survival in the training cohort, and for overall survival (OS) and locoregional relapse-free survival in the validation cohort. The constructed nomogram showed strong discriminatory ability (C-index: 0.715 for training; 0.678 for validation) and good calibration. DCA indicated superior clinical net benefit compared to default strategies. Risk stratification revealed that low-risk patients achieved significantly higher 3-year OS (97.8% vs. 81.1%, P < 0.001) and progression-free survival (PFS, 93.0% vs. 69.3%, P < 0.001) than high-risk patients. Within the high-risk group, the induction chemotherapy (IC) plus radiotherapy (RT)/concurrent chemoradiotherapy (CCRT) regimen significantly improved OS and PFS compared to RT/CCRT alone. Among low-risk patients, the two regimens yielded comparable outcomes. The NRI serves as a reliable prognostic indicator for NPC patients. The proposed nomogram enables personalized risk stratification and offers reference for individual treatment regimen selection.
Similar content being viewed by others
Introduction
Nasopharyngeal carcinoma (NPC), arising from the nasopharyngeal epithelium, predominantly affects young and middle-aged adults1. Its incidence increases with age, peaking between 45 and 60 years. Due to frequent absence of apparent symptoms in early stages, clinical recognition is often delayed, and disease severity is commonly underestimated at initial presentation2. Current management relies primarily on radiotherapy-based comprehensive treatment3,4. Notably, epidemiological data show that approximately 70% of cases are diagnosed as locoregionally advanced NPC (LA-NPC)5.
Induction chemotherapy (IC) followed by concurrent chemoradiotherapy (CCRT) represents the preferred treatment strategy for LA-NPC6. Several prospective phase III trials have established that IC plus CCRT significantly reduces tumor burden, eliminates occult micrometastases, lowers distant metastasis incidence, and enhances long-term survival compared to CCRT alone6,7,8. Conversely, IC offers no significant survival advantage in early-stage NPC and increases the risk of adverse events9,10. For stage I and low-risk stage II disease, radiotherapy alone remains the primary approach, while CCRT is indicated for high-risk stage II9. Treatment decisions regarding IC must be individualized, considering patient-specific factors including age, performance status, and nutritional condition11.
Malnutrition is a significant risk factor associated with adverse prognosis, prolonged hospitalization, and increased healthcare costs in patients with cancer12,13. Accumulating evidence underscores the impact of nutritional status on outcomes across several malignancies14,15. Therefore, accurate nutritional assessment in NPC patients is essential to guide targeted interventions and improve prognosis. In current practice, clinicians often rely on isolated objective measures such as weight loss or serum albumin levels to evaluate nutritional status16,17. This approach provides a limited evaluation, particularly in older NPC patients, and can lead to inconsistent conclusions. Given that nutritional status is inherently multifaceted, it should be evaluated using composite indices. The nutritional risk index (NRI), a composite tool calculated from serum albumin and the ratio of actual to ideal body weight, provides a more holistic assessment. While the NRI has demonstrated prognostic value in gastrointestinal malignancies18,19,20, its significance in NPC remains relatively understudied, and its potential utility in guiding IC selection is unexplored.
This study aimed to evaluate the prognostic significance of the NRI in patients with non-metastatic NPC. We constructed a prognostic nomogram integrating the NRI with other independent prognostic factors to predict survival outcomes. Furthermore, we assessed the potential benefit of IC within the risk strata defined by this nomogram.
Methods
Patient characteristics
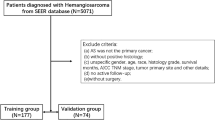
We reviewed data from patients treated at two institutions: The Second Affiliated Hospital of Fujian Medical University (Center 1: training cohort; January 2016 through October 2024) and Fujian Cancer Hospital (Center 2: external validation cohort; July 2018 through December 2019). Inclusion criteria: (1) Newly diagnosed, pathologically confirmed stage I-IVa NPC (AJCC 8th edition staging system); (2) Treatment with intensity-modulated radiotherapy (IMRT); (3) Availability of complete pretreatment laboratory data; (4) Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–1. Exclusion criteria: (1) History of or concurrent malignant tumors; (2) Incomplete follow-up data; (3) Major organ dysfunction or uncontrolled comorbidities. The patient selection process according to the exclusion criteria is illustrated in Supplementary file 1.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committees of The Second Affiliated Hospital of Fujian Medical University (Approval No. 2025 − 544) and Fujian Cancer Hospital (Approval No. K2025-321-01). Given the observational and retrospective nature, the requirement for written informed consent was waived.
Data collection and definition
Demographic and clinical information, including age, gender, height, weight, clinical stage, treatment details, and laboratory parameters from one week prior to treatment, were electronically retrieved from the hospital information system. In this study, the body mass index (BMI), prognostic nutritional index (PNI), and NRI were calculated using the established formulas21,22. Patient height and body weight were measured in a standing position.
BMI = body weight (kg) / height (m2).
PNI = serum albumin (g/L) + 5 × 10− 9 × lymphocyte count (109/L).
NRI = [1.519 × serum albumin (g/L)] + [41.7 × (current body weight / ideal body weight)].
Ideal body weight (kg) = 22.5 × height (m2).
Treatment protocol
All patients were treated with IMRT. In both cohorts, over 80% of patients received IC. Detailed treatment parameters, including chemotherapy regimens, dosing schedules, and radiation field delineation protocols, are provided in Supplementary file 2.
Follow-up and outcomes
The final follow-up was conducted in May 2025 for the training cohort (median follow-up: 35 months) and March 2025 for the validation cohort (median follow-up: 57 months). The primary endpoint was progression-free survival (PFS), defined as the duration from radiotherapy completion to the first occurrence of disease progression (including local recurrence, distant metastasis) or death. Overall survival (OS), distant metastasis-free survival (DMFS), and locoregional relapse-free survival (LRRFS) were defined as the duration from the conclusion of radiotherapy to death from any cause, distant metastasis (or death), and locoregional failure (or death), respectively.
Construction of the prognostic model and statistical analysis
Survival outcomes were estimated using the Kaplan-Meier method and compared with the log-rank test. Independent prognostic factors were identified through univariate and subsequent multivariate Cox regression analyses. The nomogram’s discriminative ability and accuracy were assessed by the concordance index (C-index), calibration plots, and decision curve analysis (DCA). A two-sided p-value < 0.05 was considered statistically significant.
Results
Baseline characteristics and follow-up
A total of 1,174 patients were included, comprising 314 in the training cohort and 860 in the validation cohort. In both cohorts, approximately 84.0% presented with locally advanced disease, and over 80.0% received IC. The male patients accounted for more than 70% of each cohort. The two cohorts were largely well-balanced in terms of key clinical characteristics, including gender distribution, T category, N category, overall stage, and the proportion of patients receiving IC (all P > 0.05). Patients in the validation cohort were younger (median age: 48 vs. 52 years), had lower median NRI (107.60 vs. 112.11) and PNI (51.20 vs. 54.05) values, and were more likely to receive higher radiotherapy doses to both the primary tumor and lymph nodes (all P < 0.05). Detailed baseline characteristics are summarized in Table 1. During follow-up, the training cohort experienced 41 (13.0%) deaths, 26 (8.2%) locoregional recurrences, 25 (7.9%) distant metastases, and 64 (20.3%) disease progressions. Corresponding events in the validation cohort were 114 (13.2%) deaths, 80 (9.3%) locoregional recurrences, 62 (7.2%) distant metastases, and 169 (19.6%) disease progressions. The causes of death from the two cohorts are detailed in Supplementary file 3.
Prognostic impact and clinical correlates of NRI
Patients were stratified into high-NRI (≥ 112.11) and low-NRI (< 112.11) groups based on the median NRI value. Kaplan-Meier analysis demonstrated markedly inferior survival in low-NRI patients compared to high-NRI patients in both cohorts (Figs. 1 and 2).
A comparative analysis of baseline characteristics between NRI groups (Supplementary file 4) revealed that patients in the high-NRI group were significantly younger (median age: 50.0 vs. 54.0 years; P = 0.011). No significant associations were found between NRI category and T category (P = 0.272), N category (P = 0.259), or overall stage (P = 0.429).
Kaplan-Meier survival curves of OS (A), PFS (B), DMFS (C), and LRRFS (D) between low- and high-NRI patients in training cohort.
Kaplan-Meier survival curves of OS (A), PFS (B), DMFS (C), and LRRFS (D) between low- and high-NRI patients in validation cohort.
Univariate and multivariate analyses
Univariate and multivariate analyses were performed on the training cohort. Univariate analysis identified several factors significantly associated with survival outcomes: age with OS and LRRFS; T category and overall stage with multiple endpoints; N category and BMI with PFS and DMFS; NRI with OS, PFS, and DMFS (Supplementary file 5, Table S2).
These variables were included in the subsequent multivariate analysis (Fig. 3). Multivariate analysis confirmed age as an independent prognostic factor for OS, PFS, and LRRFS. Advanced T category (T3/T4) was an independent predictor of poorer survival across multiple endpoints. Advanced N category (N2/N3) was a risk factor for PFS (HR: 2.04, 95%CI: 1.14–3.67, P = 0.016) and DMFS (HR: 2.04, 95%CI: 1.06–3.90, P = 0.032). Additionally, a high NRI (≥ 112.11) was an independent protective factor for superior DMFS (HR: 0.37, 95%CI: 0.14–0.97, P = 0.042).
A similar analytical approach was applied to the validation cohort. Univariate analysis identified age, T category, N category, overall stage, NRI, and PNI as significant predictors (Supplementary file 6, Table S3). In the multivariate model, age, T category, N category, and NRI retained independent prognostic significance (Fig. 4). Notably, a high NRI (≥ 112.11) was independently associated with superior OS (HR: 0.40, 95%CI: 0.22–0.75, P = 0.004) and LRRFS (HR: 0.57, 95%CI: 0.36–0.90, P = 0.015).
Multivariate Cox regression analysis of OS (A), PFS (B), DMFS (C), and LRRFS (D) for the 314 NPC patients from training cohort. NPC = nasopharyngeal carcinoma; NRI = nutritional risk index; BMI = body mass index.
Multivariate Cox regression analysis of OS (A), PFS (B), DMFS (C), and LRRFS (D) for the 860 NPC patients from validation cohort. NPC = nasopharyngeal carcinoma; NRI = nutritional risk index; PNI = prognostic nutritional index.
Development of a nomogram model
A prognostic nomogram was developed by integrating independent predictors (age, T category, N category, and NRI group) to PFS, providing individualized estimates of 1-, 2-, and 3-year PFS probabilities (Fig. 5A).
The model demonstrated good discriminatory power, with a C-index of 0.715 in the training cohort and 0.678 in the validation cohort. Calibration curves for both cohorts showed excellent agreement between nomogram-predicted and observed survival outcomes (Fig. 5B and E). DCA indicated that the nomogram provided a superior net benefit across a wide range of threshold probabilities compared to individual prognostic factors (Fig. 5C and F). Furthermore, the area under the receiver operating characteristic curve of the model was 0.774 and 0.681 for predicting 3-year PFS in the training and validation cohorts, respectively (Fig. 5D and G).
Nomogram model established by relevant prognostic factors (A). Calibration plot of the nomogram at 1-, 2-, and 3-year PFS in the training (B) and validation cohorts (E). Decision curves analysis of the nomogram at 3-year PFS in the training (C) and validation cohorts (F). The area under the receiver operating characteristic curve of the model at 3-year PFS in the training (D) and validation cohorts (G). NRI = nutritional risk index; AUC = area under curve.
Risk stratification
Based on the nomogram, a risk score was calculated for each NPC patient. Using the training cohort data, the median risk score was identified as 168.5, which served as the cutoff to stratify patients into high- and low-risk groups. Risk stratification revealed significantly superior outcomes in the low-risk group, with higher 3-year OS (97.8% vs. 81.1%, P < 0.001, Fig. 6A) and PFS (93.0% vs. 69.3%, P < 0.001, Fig. 6B) compared to the high-risk group. Consistent results were observed in the validation cohorts, where low-risk patients exhibited significantly improved OS and PFS, as shown in the survival curves (Fig. 6C-D, all P < 0.05). These results further validate the strong discriminative ability of the prognostic model.
Kaplan-Meier survival curves of different risk groups. OS (A) and PFS (B) for patients in training cohort; OS (C) and PFS (D) for patients in validation cohort. (low-risk group: risk score < 168.5; high-risk group: risk score ≥ 168.5).
Subgroup analysis
Analysis of treatment efficacy within each risk stratum revealed distinct outcomes, particularly among high-risk patients (Fig. 7). In the training cohort, high-risk patients receiving IC plus RT/CCRT achieved significantly better OS and PFS than those receiving RT/CCRT alone (Fig. 7A-B). This survival benefit was consistently observed in the validation cohort (Fig. 7C-D). In contrast, among low-risk patients in both cohorts, OS and PFS were comparable between the two treatment regimens, with no significant difference (Supplementary file 7, Figure S3).
The Kaplan-Meier survival curves of induction chemotherapy in high-risk group. OS (A) and PFS (B) for patients in training cohort; OS (C) and PFS (D) for patients in validation cohort. IC = induction chemotherapy.
Discussion
This multicenter study establishes the NRI as an independent prognostic marker in NPC. We developed and validated a clinically applicable nomogram that integrates the NRI with other clinicopathological factors to predict survival outcomes from two independent centers. The model effectively stratified patients into distinct risk groups, with high-risk patients showing significantly inferior survival outcomes. Importantly, within the high-risk group, the addition of IC to RT/CCRT was associated with improved OS and PFS compared to RT/CCRT alone. These findings indicate that the proposed nomogram not only provides reliable risk stratification but may also help identify patients who are more likely to benefit from intensive treatment with IC.
The tumor lymph node metastasis (TNM) staging system remains the cornerstone of clinical decision-making and prognosis in oncology23,24. However, it does not account for differences in systemic function among individuals25. Moreover, due to tumor heterogeneity, TNM staging based on anatomy might not adequately reflect the tumor’s biological behavior. Consequently, there is a growing need for reliable biomarkers or composite tools to refine prognostication and guide personalized treatment. Various studies have combined accessible clinical indicators and molecular biomarkers to create predictive models for stratified treatment in NPC patients24,26,27. Previous research has demonstrated that, in addition to clinical stage, factors such as radiomics28, plasma Epstein-Barr virus (EBV) DNA levels29, and nutritional status30 also play significant roles in prognostic evaluation.
In our study, overall stage was significantly associated with survival outcomes in univariate analysis, reaffirming its prognostic relevance. However, it did not retain independent prognostic significance in our final multivariate Cox regression model. This observation indicates that the prognostic information of overall stage may be shared with or captured by other covariates in the model. Here, we propose and validate a nutrition-based prognostic nomogram that integrates the NRI with key clinicopathological variables, offering a readily applicable tool for outcome prediction and therapeutic stratification. Therefore, the prognostic nomogram is not intended to replace the TNM system but to complement it by integrating key host factors, thereby enabling a more personalized assessment of risk and informing therapeutic strategy.
The prognostic importance of age, T category, N category, and NRI differed across survival endpoints. These variations, likely due to differences between cohorts in size, event rates, and follow-up, highlight that a host factor like nutrition has multifaceted effects, which may emerge differently depending on the patient population and endpoint measured. The NRI consistently predicted at least one critical endpoint in each cohort, confirming its overall relevance. Thus, the observed differences reflect biological reality, including separate aspects of the disease (local and nodal spread) and the patient (age and nutritional status) contribute differently to distinct modes of treatment failure. The strength of our final model is its integration of these four dimensions into a single, clinically useful predictor for the composite endpoint of PFS.
Nutritional status is a well-established prognostic factor in NPC. The NRI, calculated from serum albumin levels and body weight, is a straightforward and effective method for evaluating nutritional status and forecasting survival outcomes in diverse cancers31,32,33. For instance, a meta-analysis confirmed that a low NRI is significantly associated with worse OS in patients with head and neck cancer31, and it has also been recognized as a significant predictor of OS in elderly gastric cancer patients32. Tang et al. conducted a study focusing on NPC, identifying the NRI as a prognostic indicator for patients undergoing IMRT33. Our findings strongly corroborate this existing evidence. We consistently identified a low baseline NRI as an independent predictor of inferior survival outcomes across both cohorts. Notably, the median NRI value in our cohort (112.11) aligns with the established prognostic cut-off value reported in prior NPC research30. In our study, we found no statistically significant association between NRI category and T category, N category, or overall stage. It implies that nutritional status at diagnosis may be influenced more strongly by intrinsic host factors (such as age and individual metabolic response) than by the anatomic extent of the disease alone. Consequently, this observation reinforces the independent prognostic value of the NRI in NPC prognosis. Importantly, the nutritional risk can identify vulnerable patients across all disease stages who might be at heightened risk for adverse outcomes, potentially offering a complementary layer of risk stratification beyond the TNM system.
The association between a low NRI and poorer survival in NPC may be explained by several mechanisms. First, malnutrition in these patients can compromise immune function, diminishing anti-tumor immunity and increasing infection risk34. Second, evidence from other centers suggests that poor nutritional status may exacerbate radiation-induced toxicities, such as mucositis and dysphagia, potentially impairing treatment tolerance and adherence35,36. Clinically, incorporating the NRI into prognostic models may help tailor adaptive treatment strategies. For example, patients with a low NRI might benefit from more intensive nutritional interventions and closer monitoring to reduce treatment-related complications and improve outcomes.
While acknowledging the established prognostic value of PNI in other contexts21,22, our statistical findings within this specific cohort did not support its inclusion as an independent predictor in the final model. Instead, our findings demonstrated that the NRI independently predicted survival outcomes and was therefore selected for nomogram construction. Previous study has indicated the prognostic value of the NRI in NPC patients using internal validation37. The major strength of this study lies in confirming its generalizability through external validation in a large, dual-center cohort of non-metastatic NPC patients, thereby providing robust evidence for the NRI as a prognostic marker. We developed a risk-stratification model that integrates both anatomical factors (clinical stage) and nutritional indicator (NRI). Importantly, this integrative approach helps overcome limitations inherent in the conventional TNM system alone. Going beyond prognosis to prediction, the NRI-based prognostic nomogram not only exhibits superior predictive performance compared with individual biomarkers but also effectively stratifies patients for IC benefit. Furthermore, the model ensures clinical practicality through the use of an accessible biomarker, offering a valuable tool for guiding treatment selection.
In clinical practice, the decision to administer IC is multifaceted, incorporating factors such as performance status, comorbidities, age, and nutritional status, rather than being based solely on tumor stage. Our analysis revealed that the addition of IC to RT/CCRT was associated with significantly improved OS and PFS specifically in high-risk patients. Several mechanisms may underlie this benefit. First, high-risk patients often present with a higher tumor burden or extensive lymph node involvement, which correlates with increased risks of recurrence and distant metastasis. For these patients, IC may effectively reduce tumor load, clear plasma EBV DNA, and thereby lower the incidence of relapse, ultimately improving long-term survival36,38. In contrast, for low-risk patients, survival outcomes with RT/CCRT alone were comparable to those achieved with IC plus RT/CCRT. This suggests that more intensive therapy may be unnecessary in this subgroup, as local control can often be achieved with radiotherapy-based treatment alone. Moreover, the addition of IC could introduce unnecessary risks of adverse events such as myelosuppression and gastrointestinal toxicity without conferring additional survival benefits39. Second, IC has been associated with potential delays in initiating definitive radiotherapy, which may compromise local control40,41. Therefore, low-risk patients are unlikely to benefit from IC and may instead be ideal candidates for treatment de-escalation to avoid undue toxicity. Collectively, these findings underscore the clinical utility of our nomogram. It facilitates personalized treatment strategies by identifying patients who are most likely to benefit from intensive therapy (IC) while sparing those at low risk from unnecessary treatment intensification and its associated toxicities.
Although EBV DNA load is a crucial biomarker in NPC pathogenesis and prognosis42, it was not included as a parameter in this study. This decision was based on several practical challenges that limit its standardized application across multi-center studies. First, there is significant inter-institutional variability in EBV DNA detection methodologies due to differences in diagnostic kits, equipment, and laboratory protocols43. Second, there is currently no consensus regarding the optimal sample type for EBV DNA analysis (tissue, serum, or plasma). Additionally, the lack of standardized measurement units and reference values across laboratories severely hampers reliable cross-center comparison and consistent data interpretation.
This study has several limitations. First, as a retrospective analysis, it is subject to inherent selection bias, and the non-randomized allocation of treatments may introduce confounding. Second, while we assessed global nutritional status using the NRI, the specific mode of nutritional support (e.g., enteral tube feeding) was not evaluated as a predictor. Third, data on radiotherapy-related adverse events were incomplete, limiting assessment of their association with nutritional status. Finally, the model incorporated only baseline NRI values and did not account for dynamic changes in nutritional status during or after treatment, which may hold additional prognostic significance. Future research should focus on prospective, multi-center validation of this nomogram. Furthermore, investigating the biological mechanisms linking nutritional status, host immunity, and treatment response, as well as conducting interventional trials to determine whether tailored nutritional support improves outcomes in patients with low NRI, are important next steps.
Conclusions
We constructed and validated a prognostic nomogram incorporating the NRI and key clinical parameters to predict prognosis in non-metastatic NPC. This model provides a practical tool for personalized risk stratification and therapeutic guidance.
Data availability
The data used and/or analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- NPC:
-
Nasopharyngeal carcinoma
- NRI:
-
Nutritional risk index
- PS:
-
Performance status
- PNI:
-
Prognostic nutritional index
- BMI:
-
Body mass index
- RT:
-
Radiotherapy
- CCRT:
-
Concurrent chemoradiotherapy
- IC:
-
Induction chemotherapy
- IMRT:
-
Intensity-modulated radiotherapy
- TNM:
-
Tumor lymph node metastasis
- DCA:
-
Decision curve analysis
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- DMFS:
-
Distant metastasis-free survival
- LRRFS:
-
Locoregional relapse-free survival
- LA-NPC:
-
Locoregionally advanced nasopharyngeal carcinoma
- EBV:
-
Epstein-Barr virus
- AUC:
-
Area under curve
References
Wong, K. C. W. et al. Nasopharyngeal carcinoma: an evolving paradigm. Nat. Rev. Clin. Oncol. 18 (11), 679–695 (2021).
Chen, Y. P. et al. Nasopharyngeal carcinoma. Lancet 394 (10192), 64–80 (2019).
Blanchard, P. et al. Chemotherapy and radiotherapy in nasopharyngeal carcinoma: an update of the MAC-NPC meta-analysis. Lancet Oncol. 16 (6), 645–655 (2015).
Ji, M. F. et al. Incidence and mortality of nasopharyngeal carcinoma: interim analysis of a cluster randomized controlled screening trial (PRO-NPC-001) in southern China. Ann. Oncol. 30 (10), 1630–1637 (2019).
Peng, H. et al. Optimize the cycle of neoadjuvant chemotherapy for locoregionally advanced nasopharyngeal carcinoma treated with intensity-modulated radiotherapy: A propensity score matching analysis. Oral Oncol. 62, 78–84 (2016).
Zhang, Y. et al. Final Overall Survival Analysis of Gemcitabine and Cisplatin Induction Chemotherapy in Nasopharyngeal Carcinoma: A Multicenter, Randomized Phase III Trial. J. Clin. oncology: official J. Am. Soc. Clin. Oncol. 40 (22), 2420–2425 (2022).
Yang, Q. et al. Induction chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: long-term results of a phase III multicentre randomised controlled trial. Eur. J. Cancer. 119, 87–96 (2019).
Li, W. F. et al. Concurrent chemoradiotherapy with/without induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: Long-term results of phase 3 randomized controlled trial. Int. J. Cancer. 145 (1), 295–305 (2019).
Su, S. F. et al. Long-Term Outcomes of Early-Stage Nasopharyngeal Carcinoma Patients Treated With Intensity-Modulated Radiotherapy Alone. Int. J. Radiation Oncology*Biology*Physics. 82 (1), 327–333 (2012).
Wu, P. et al. Management of Chemotherapy for Stage II Nasopharyngeal Carcinoma in the Intensity-Modulated Radiotherapy Era: A Review. Cancer Manage. Res. 12, 957–963 (2020).
Liu, S. L. et al. Comparing three induction chemotherapy regimens for patients with locoregionally advanced nasopharyngeal carcinoma based on TNM stage and plasma Epstein-Barr virus DNA level. BMC Cancer. 20 (1), 89 (2020).
Pingping, J. et al. Nutritional status and its correlation to prognosis of nasopharyngeal carcinoma patients in different ages in China: a multicenter cohort study. Support. Care Cancer. 31 (11), 638 (2023).
Lavalette, C. et al. Cancer-Specific and General Nutritional Scores and Cancer Risk: Results from the Prospective NutriNet-Santé Cohort. Cancer Res. 78 (15), 4427–4435 (2018).
Hamaker, M. E. et al. Nutritional status and interventions for patients with cancer - A systematic review. J. Geriatric Oncol. 12 (1), 6–21 (2021).
Wiseman, M. J. Nutrition and cancer: prevention and survival. Br. J. Nutr. 122 (5), 481–487 (2018).
Ou, Q. et al. Grading and prognosis of weight loss before and after treatment with optimal cutoff values in nasopharyngeal carcinoma. Nutrition 78, 110943 (2020).
Li, G. et al. Influence of pretreatment ideal body weight percentile and albumin on prognosis of nasopharyngeal carcinoma: Long-term outcomes of 512 patients from a single institution. Head Neck. 36 (5), 660–666 (2014).
Sasaki, M. et al. The Geriatric Nutritional Risk Index predicts postoperative complications and prognosis in elderly patients with colorectal cancer after curative surgery. Sci. Rep. 10 (1), 10744 (2020).
Abe, A. et al. Correlation between geriatric nutritional risk index and oral condition in gastric cancer patients. Oral Dis. 29 (2), 836–842 (2021).
Miyoshi, N. et al. Geriatric nutritional risk index as a prognostic factor in patients with recurrent pancreatic cancer. Plos One. 17 (7), e0271073 (2022).
Duan, Y. Y. et al. Construction of a comprehensive nutritional index and comparison of its prognostic performance with the PNI and NRI for survival in older patients with nasopharyngeal carcinoma: a retrospective study. Support. Care Cancer. 29 (9), 5371–5381 (2021).
Song, J. et al. Prognostic value of prognostic nutritional index in patients with nasopharyngeal carcinoma treated with endostar and concurrent chemoradiotherapy. Support. Care Cancer. 33 (3), 225 (2025).
Pan, J. J. et al. Ninth Version of the AJCC and UICC Nasopharyngeal Cancer TNM Staging Classification. JAMA Oncol. 10 (12), 1627–1635 (2024).
Du, X. J. et al. Refining the 8th edition TNM classification for EBV related nasopharyngeal carcinoma. Cancer Cell. 42 (3), 464–473e463 (2024).
Lee, V. H. F. et al. The addition of pretreatment plasma Epstein–Barr virus DNA into the eighth edition of nasopharyngeal cancer TNM stage classification. Int. J. Cancer. 144 (7), 1713–1722 (2018).
Cheng, G. Y. et al. A prognostic nomogram for patients with III–IV nasopharyngeal carcinoma based on dynamic changes in the inflammatory and nutrition index. Clin. Transl. Oncol. 27 (6), 2638–2650 (2024).
Mao, J. R. et al. Can the prognosis of individual patients with nasopharyngeal carcinoma be predicted using a routine blood test at admission? Radiother. Oncol. 179, 109445 (2023).
Wang, S. X. et al. Radiomics-based nomogram guides adaptive de-intensification in locoregionally advanced nasopharyngeal carcinoma following induction chemotherapy. Eur. Radiol. 34 (10), 6831–6842 (2024).
Lin, J. C. et al. Quantification of plasma Epstein-Barr virus DNA in patients with advanced nasopharyngeal carcinoma. N Engl. J. Med. 350 (24), 2461–2470 (2004).
Fang, W. N. et al. A scoring system based on inflammatory and nutritional indicators to predict the long-term survival of patients with non-metastatic nasopharyngeal carcinoma. Sci. Rep. 14 (1), 20229 (2024).
Huang, Y. C., Chen, S. W. & Chiang, Y. S. Prognostic role of geriatric nutritional risk index (GNRI) and controlling nutritional status (CONUT) on outcomes in patients with head and neck cancer: a systematic review and meta-analysis. BMC Cancer. 25 (1), 242 (2025).
Hirahara, N. et al. Prediction of postoperative complications and survival after laparoscopic gastrectomy using preoperative Geriatric Nutritional Risk Index in elderly gastric cancer patients. Surg. Endosc. 35 (3), 1202–1209 (2020).
Tang, Q. N. et al. Geriatric nutritional risk index as an independent prognostic factor in locally advanced nasopharyngeal carcinoma treated using radical concurrent chemoradiotherapy: a retrospective cohort study. Annals Translational Med. 9 (7), 532–532 (2021).
Kubo, N. et al. The impact of geriatric nutritional risk index on surgical outcomes after esophagectomy in patients with esophageal cancer. Esophagus 16 (2), 147–154 (2018).
de Pinho, N. B. et al. Malnutrition associated with nutrition impact symptoms and localization of the disease: Results of a multicentric research on oncological nutrition. Clin. Nutr. 38 (3), 1274–1279 (2019).
Jin, Y. N. et al. Neoadjuvant chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in elderly patients with stage III-IVa nasopharyngeal carcinoma: A real‐world study based on medical comorbidities. Head Neck. 46 (8), 2020–2030 (2024).
Lin, X. et al. Insights into the Prognostic Efficacy of the Geriatric Nutritional Risk Index for Nasopharyngeal Carcinoma in the Era of Volumetric Modulated Arc Therapy: A Nomogram for Predicting Long-Term Survival Outcomes. Curr. Oncol. 32 (7), 372 (2025).
Xie, W. H. et al. Four cycles of docetaxel plus cisplatin as neoadjuvant chemotherapy followed by concurrent chemoradiotherapy in stage N2-3 nasopharyngeal carcinoma: phase 3 multicentre randomised controlled trial. Bmj 389, e081557 (2025).
Qu, W. L. et al. Chemotherapy for locoregionally advanced nasopharyngeal carcinoma: Who really needs it. Cancer Med. 12 (6), 6994–7004 (2022).
Liang, H. et al. Survival impact of waiting time for radical radiotherapy in nasopharyngeal carcinoma: A large institution-based cohort study from an endemic area. Eur. J. Cancer. 73, 48–60 (2017).
Jiang, Y. T. et al. Prognostic significance of wait time for radical radiotherapy in locoregionally advanced nasopharyngeal carcinoma. Head Neck. 44 (5), 1182–1191 (2022).
Li, W. et al. Integrating pre- and post-treatment Plasma Epstein-Barr Virus DNA levels for better prognostic prediction of Nasopharyngeal Carcinoma. J. Cancer. 12 (9), 2715–2722 (2021).
Lee, A. W. M. et al. A systematic review and recommendations on the use of plasma EBV DNA for nasopharyngeal carcinoma. Eur. J. Cancer. 153, 109–122 (2021).
Funding
This work was supported by the National Natural Science Foundation of China (82473376); Major Scientific Research Program for Young and Middle-aged Health Professionals of Fujian Province, China (2021ZQNZD010); Startup Fund for scientific research, Fujian Medical University (2020QH1107).
Author information
Authors and Affiliations
Contributions
Jingjing Su and Sufang Qiu: Conceptualization, Formal analysis, Writing-original draft; Sunqin Cai and Jue Wang: Study design, Writing-review and editing; Ying Li: Structural characterization; Zongwei Huang: Data curation; Sundong Cai: Clinical supervision, Project administration. All authors critically reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committees of The Second Affiliated Hospital of Fujian Medical University (Approval No. 2025 − 544) and Fujian Cancer Hospital (Approval No. K2025-321-01). Given the observational and retrospective nature, the requirement for written informed consent was waived.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cai, S., Wang, J., Li, Y. et al. A nutritional risk index-based nomogram for predicting prognosis and identifying induction chemotherapy beneficiaries in nasopharyngeal carcinoma. Sci Rep 16, 8848 (2026). https://doi.org/10.1038/s41598-026-40939-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40939-z