Abstract
This study aims to investigate the efficacy of area-wide management (AWM) approach on a large scale implemented in a tropical region against subterranean termite infestations. This AWM was carried out using a slow acting Chitin Synthesis Inhibitor (CSI), chlorfluazuron, as bait in the study area. A total of 11,511 in-ground stations were employed to intercept outdoor termite activities and 40 indoor above-ground stations were installed on infested structures in the study area. This study was carried out from April 2021 till March 2024. At the in-ground stations, total elimination of termite activity was recorded within 16 months from the start of the study, and no further interceptions were observed until the end of the study. The average days-to-elimination (DTE) of termite activity was recorded at 39.71 ± 2.47 days for in-ground stations. Above-ground stations had a high number of active stations from April to September 2021, followed by a sharp decrease after September 2021. No re-invasions were reported from March to December 2022. However, two active above-ground stations were recorded between January to March 2023, after which no termite activity was observed inside buildings during monitoring period till the end of the study. The above-ground stations recorded a slightly longer time to elimination than in-ground stations with 64.63 ± 8.30 days. This study shows the effectiveness of baiting in eliminating termite activities and the feasibility of implementing an area-wide baiting program to control termite infestation on a large scale.
Similar content being viewed by others
Introduction
Subterranean termites are found in large parts of the world in both undisturbed and man-made habitats, and as the decomposers of cellulose material, they contribute greatly in recycling nutrients such as carbon and nitrogen back to the ecosystem1. However, termites can also cause massive damage to human structures especially wooden made structures such as door frames and cabinets2. Subterranean termite pest species are estimated to cost more than $32B in annual damages globally3. In Southeast Asia, the damage caused by termites is estimated at around 400 million US dollars per year4. Malaysia suffers from damage caused by termite pests, especially the genus Coptotermes, such as Coptotermes curvignathus, which is the most aggressive species4. The extent of their damage has been reported mainly in urban areas where termites destroy floorboards and other wooden structures5, but also on agricultural land, where along with Coptotermes gestroi, C. curvignathus have been found feeding on trees, such as oil palm6,7, and trees in urban landscapes8.
The two most used methods for controlling termite infestations are soil treatment with a liquid termiticide or installing bait stations9,10. The termiticides commonly used for such applications in Malaysia are fipronil, imidacloprid and chlorpyrifos (which as of 2023 is now banned for agriculture use but allowed for public health and urban pest control114. In the USA, 33 to 42% of pest management providers reported sole reliance on liquid termiticide treatments as a solution to subterranean termite infestations and 4 to 16% reported using chitin synthesis inhibitor (CSI) baits as a standalone solution12.
The active ingredient in CSIs does not readily affect individual survival of termites that consume the bait, rather it interferes with the molting process of termites13. Summarized in Chouvenc12, CSI baits rapidly alter a termite colony demography from a healthy productive one to a very old one rapidly, i.e., CSI baits artificially accelerate a colony senescence process beginning with foragers that initiate feeding on a CSI bait station, to spread of a CSI throughout the colony indirectly through trophallaxis and directly through exposed workers’ movement14 and demographic profile alterations that ultimately lead to the complete collapse of the brood within 20–30 days after initial CSI bait feeding by the foragers12. For installation of CSI bait stations, a tubular plastic container with a lid into which a bait (bait matrix containing alpha-cellulose powder with an active ingredient such as hexaflumuron, chlorfluazuron and bistifluron) is placed at an infested area15. Both laboratory and field studies have demonstrated the elimination of subterranean termite colonies using CSI baits (e.g.,16,17,18,19). Most laboratory experiments involving CSI baits involved relatively small groups of termites originating from a field-collected foraging population, which may not represent bait impact at the colony level20. Laboratory studies involving larger colonies of Coptotermes spp. termites reported that colonies were eliminated with CSI baits within 90 days21,22. A colony of termites in the field can contain hundreds of thousands or even millions of foraging termites15, and colony densities can be as high as 300 colonies per hectare23, although this varies geographically and by habitat type24. With such a high colony density, the control of single or multiple nests in a small area may not be very effective in preventing nearby termite populations from recolonising the cleared area. In such cases, large-scale application of an insecticide is better suited as a long-term solution for pest control.
The aim of the area-wide management (AWM) strategy is to reduce overall pest pressure by concentrating control at the population level rather than focusing on individual infestations25. Several studies have reported mixed results of the efficacy of area-wide management using baiting systems for sustainable termite infestation management in the USA, particularly on major species such as the Formosan subterranean termite, C. formosaus17,26,27,28,29,30. In contrast, a study conducted by Smith et al. (2006)16, Reticulitermes flavipes infestations were successfully eliminated in a low-income community in Santiago, Chile, within a 15-month period. Remarkably, termites were not detected in the study area for up to three years and till the end of the research. However, in tropical countries like Malaysia, similar approaches remain largely unexplored. Therefore, in this study, we aimed to investigate the efficacy of a baiting approach within a large-scale using an area-wide management strategy to control termite infestations in a tropical region, specifically at the Kampus Alam, Universiti Malaysia Perlis (UniMAP), Malaysia.
Materials and methods
Study site and termite infestation
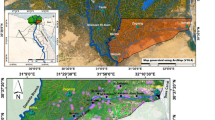
This study was conducted at the main campus of Kampus Alam, Universiti Malaysia Perlis (UniMAP), Malaysia. (6° 27’ 44’’ N, 100° 9’ 52’’ E) (Fig. 1). The campus is about 20 km from the Kangar town of Perlis state, Malaysia. Initially, this study commenced as a contract project awarded by UniMAP to the pest control company, Insepro Malaysia Sdn Bhd with the primary objective to control termite infestations in their campus. A research team was established to make the project a commercial scientific study of termite control. The team installed bait stations, monitored the progress of the study, conducted all the data collection and data analyses to ensure proper documentation of the project. This project was conducted from April 2021 to December 2023 (32 months) and the monitoring of the termite bait stations was extended till March 2024 (total 36 months).
Map of the main campus of Kampus Alam, Universiti Malaysia Perlis (UniMAP), Malaysia (Geographic boundaries adapted from OpenStreetMap).
The study area covered an approximate total area of 1,050 acres. The campus had been infested with subterranean termites Coptotermes sp. (Blattodea: Heterotermitidae) since the development of the campus, mainly because the land was a rubber plantation prior to the construction of the campus and the colonies of subterranean termites, C. curvignathus had remained in the soil. The infestation of termites in the area were reported in buildings on campus where termites were reported to have damaged the structure and wood furniture of buildings. The infestation also involved trees and the landscape of the campus. Discussions with university representatives revealed that previous treatments in the form of spot spraying had not contained the termite infestation in the campus. Thus, area-wide management was proposed to deal with the termite infestations.
Termite bait station and treatment
Two types of bait stations were employed in the study area: in-ground stations for outdoor sites and above-ground stations for indoor application of termite bait. The in-ground stations were installed and placed near buildings, trees, and landscapes and were placed approximately 5 m apart. To ensure the installation of the in-ground stations were carried out systemically within the campus area, the study area was divided into 21 zones. Zones A-H and J, K and P, S covered the building complex of the campus and these zone areas that were mostly made up of buildings, with tree and landscape interspersed between buildings (Table 1). A total of 7,672 of in-ground stations were installed in these zones; with 1,743 in-ground stations installed surrounding the buildings, 2,559 to protect trees and 3,370 in-ground stations installed in open areas without trees. The other zones L, N, T, Q, R, V, W, X and Y were areas covering tree and landscape areas. The total number of installed in-ground stations in these zones were 3,839 with 2,597 to protect tress and 1,242 in-ground stations installed in the landscape area (Table 1). Overall, 11, 511 in-ground stations were installed in the study area.
Upon installation, each in-ground station was equipped with six wooden ‘interceptors’ and powdered attractant FOCUS®, a non-toxic formulation composed of natural materials31. After two weeks, all the in-ground stations were inspected and observed for termite activities. The collection of termite samples (20–50 termites) was carried out in all active in-ground stations using soft forceps and samples were placed in universal bottles filled with 75% alcohol and brought back to the laboratory. The termites were identified to genus level following Lee (2003)32 and Lee (2014)33. After the collection of termite samples, termite baits were placed in all active in-ground stations.
In this study, Requiem Exterra® bait (0.1% chlorfluazuron) was used as termite baits and to prepare the baits, 250 g of bait powder was mixed with 1500 ml of water to obtain bait dough as specified by the manufacturer. 250 g of the resulting bait dough (0.1% chlorfluazuron) was placed and secured at each bait station. All the termite bait preparation was handled by well-trained technicians from the pest control company, Insepro Sdn. Bhd., with supervision from the research team. All the in-ground stations were monitored every three months from April 2021 to March 2024. The feeding of the bait was monitored until the termite activity ceased, i.e., the in-ground station was inactive. Inactive in-ground stations then cleaned and refilled with wooden ‘interceptors’ and powdered attractant FOCUS®. This was to ensure the station would detect reinfestation of termite activity in the area. The number of active in-ground stations and the days required to eliminate termite activity in all active in-ground stations in the study area were recorded.
Above-ground stations were exclusively deployed in buildings in the entire study area upon visual confirmation of termite infestations during monthly monitoring surveys. All above-ground stations were baited and monitored until elimination of termite activity. However, in contrast to in-ground stations, above-ground stations did not employ interceptors or attractants and the above-ground stations were removed after successful elimination of termite activity in the area. The collection of termite samples was done similar to that of in-ground stations. After the first bait application, the above-ground stations were checked monthly and refilled if necessary. The acceptance of the bait was monitored until no more termite activity was detected at the stations. Above-ground stations were monitored every month from April 2021 to April 2023, after which no termite activity was observed within buildings hence no above-ground stations were installed. Monitoring of termite activity within buildings continued till end of the study in March 2024.
Data analysis
The data of days-to-elimination (DTE), i.e., the number of days recorded for elimination of termite activity at active bait stations, were recorded according to visits to all the installed in-ground and above-ground stations. All the data was subjected to the Shapiro-Wilk test to determine their normality. As the data was not parametric, the Kruskal-Wallis test was performed to compare the DTE of the termite colony in three different habitats. In addition, a generalised linear mixed model (GLMM), which can handle non-normal data, was used to investigate the relationship between DTE and the type of termite habitat. The GLMM was performed in R Studio (R Core Team, 2024)34 using the package lme4 (Bates et al., 2015)35 and the function glmer. Visual representations of the density and prevalence of termite activity at the study site, i.e., Figs. 1, 2, 3, 4 and 5 were manually generated using Adobe Photoshop 2023 (v24.5.0, Adobe Inc., San Jose, CA, USA), with maps adapted from OpenStreetMap (OpenStreetMap contributors), licensed under the Open Database License (ODbL, https://www.openstreetmap.org/copyright).
Density and prevalence of active termite activity at IGS in 1st quarter of the study (April - June 2021) Red represents active stations and circle area size signifies the number of active IGS. (Figure generated using Adobe Photoshop based on OpenStreetMap data)
Density and prevalence of termite activity at IGS in the study area. (A) 2nd period (July to September 2021) (B): 3rd period (October to December 2021), (C): 4th period (January to March 2022), (D) 5th period (April-June 2022). Red represents active IGS and green elimination of termite activities. The circle area signifies the number of active IGS or elimination of termite activity. (Figure generated using Adobe Photoshop based on OpenStreetMap data)
Density and prevalence of termite activity at IGS in the study area. (A) 6th period (July to September 2022) (B): 7th period (October to December 2022), (C): 8th period (January to March 2023). Red represents active IGS and green indicates inactive IGS. The circle area signifies the number of active IGS or elimination of termite activities. (Figure generated using Adobe Photoshop based on OpenStreetMap data)
Density of AGS in the study. Circle area size signifies the number of AGS where termite feeding activity were no longer recorded after stations were deployed. (Figure generated using Adobe Photoshop based on OpenStreetMap data)
Results
Termite activity at in-ground stations
Coptotermes sp. termites were the sole species recorded at in-ground stations throughout the whole study period. Raw data of the results of the monitoring of in-ground stations is available as Supplementary Material 1. Figure 2 illustrates the termite infestation recorded during the first period of the study (April - June 2021). At this early stage, a total of five in-ground stations were active with termites. Two in-ground stations were located near Pusat Latihan Kejuruteraan (C266 and C110), and three active in-ground stations were located near FTK Elektronik (E26, E67, and E157).
A total of eight in-ground stations were active in the second period of the study (July to September 2021), including the five previously active ones (E26, E67, E157, C266, and C110) and an additional one, E266. Two new active in-ground stations (A596 and A598) were detected near the ‘FTK Mekanikal’ building (Fig. 3A). No termite activity eliminations were recorded during this period. In the third period (October to December 2021) (Fig. 3B), there was a total of 29 active in-ground stations, with elimination of termite feeding activity at nine in-ground stations. The highest number of active in-ground stations were detected near the ‘Perpustakaan’ building (N3, N8, N9, N15, N25, N26, N28, N189, and N191). The ‘Kompleks Sukan’ area had five active in-ground stations, and three new areas (‘FTK Electric,’ ‘Asrama Pelajar,’ and ‘Surau’) showed termite activity. The study recorded elimination of termite activities near ‘Pusat Latihan Kejuruteraan,’ FTK Electronic, and ‘FTK Mekanikal. However, termite activities were detected at in-ground stations E238, E865, and E866. Furthermore, the ‘Pusat Latihan Kejuruteraan’ location had two eliminations of termite activity (C266 and C110) and one new active station (C324), while ‘FTK Mekanikal’ had one elimination (A596) and four active stations (A598, A421, and A87).
In the fourth period (January to March 2022) and fifth period (April-June 2022), the elimination of termite colonies in the in-ground stations around the campus was evident (Fig. 3C and D). An increasing number of active in-ground stations transitioned to recording no feeding activities of termites. In the fourth period, a total of 30 in-ground stations recorded the elimination of termite activity, leaving only 12 in-ground stations around the study site, namely those close to Komplex Sukan (S67, S74 and S670), Dewan Ilmu (J165 and J278), Perpustakaan (N15 and N191) FTK Mekanikal (A598, A421 and A97) and FTK elektronik (F526 and F54). In the fifth period, a total of 42 in-ground stations in the study area recorded no termite activity, with one additional active in-ground station detected.
In the sixth (July to September 2022) and seventh period (October to December 2022) (Fig. 4A and B), the elimination of termite activities continued in in-ground stations. In the sixth period, a cumulative total of 44 in-ground stations recorded no feeding activities of termite, and an additional five new active stations were recorded. These active stations primarily originated from new termite activities at in-ground stations Q85 and Q87 near HEPA, as well as in-ground stations A1 and A2 near FTK Mekanikal, and J30 near the Dewan Ilmu building. In the seventh period, a total of 49 cumulative in-ground stations with termite feeding ceased and two new active in-ground stations were recorded during this period: A437 near FTK Mekanikal and P153 near Asrama Pelajar (PF1-1). In the eight period (January to March 2023), a total of 51 in-ground stations with termite feeding ceased (Fig. 4C). Termite activities were no longer detected after this period. Overall, the records of active stations of in-ground stations were limited to the period from April 2021 until March 2023. No termite activity was detected in in-ground stations within the campus till the end of the study in March 2024.
The overall average DTE of termite activity of in-ground stations was 39.71 ± 2.47 days. Taking the median as a measure of central tendency and comparing the interquartile range (IQR) for days-to-elimination (DTE) in in-ground stations across habitat types, there were no significant differences (Kruskal-Wallis chi-squared = 0.492, df = 2, p-value = 0.7821). The mean DTE for all in-ground stations employed near buildings was recorded at 43.80 ± 5.27 days, followed by landscape habitat at 39.40 ± 2.08 days, and in-ground stations that were installed to protect trees at 37.49 ± 6.99 days. The total mean of DTE for all the in-ground stations employed in the study area was 39.71 ± 2.47 days. When analysing the relationship between the termite habitat and the DTE at in-ground stations, a significant correlation (p = 0.00336) was found between the building habitat and the DTE of the termite colony with a positive coefficient (Table 2), which means that the higher the number of building habitats, the longer the days to elimination of termite activity.
Termite activity at above-ground stations
A total of 40 above-ground stations were installed throughout the study period. Similar as in in-ground stations, Coptotermes sp. was the only species found in above-ground stations. Raw data of the results of the monitoring of above-ground stations is available as Supplementary Material 2. Termite activity at above-ground stations exhibited a pattern resembling that of in-ground stations, displaying high numbers of active stations from early stage of the study from April to September 2021, followed by a steep reduction after September 2021 (Fig. 6). No new termite activity was reported from March 2022 to December 2022. However, two active stations were installed between January 2023 and March 2023, after which no new termite activity were recorded till end of the study in December 2023.
Number of active AGS throughout the study period from April 2021 till last detected termite activity in April 2023.
The density and prevalence of termite activity at above-ground stations on UNIMAP campus, along with the number of active stations throughout the study period are shown in Fig. 5. The highest infestation was recorded at the FTK Mekanical where 10 above-ground stations were employed in the building namely AGS1, AGS2, AGS5, AGS6, AGS14, AGS15a, AGS15b, AGS21, AGS23 and AGS25. Similarly, the FTK Elektrik recorded 10 active above-ground stations (AGS3, AGS4, AGS9, AGS10, AGS11, AGS12, AGS35, AGS36, AGS37 and AGS38). The Asrama Pelajar (AGS17, AGS18, AGS19 and AGS20) and Kompleks Sukan (AGS22, AGS24, AGS29a and AGS29b) both recorded 4 active above-ground stations. Dewan Kuliah and Surau recorded 3 above-ground stations and the rest of buildings were employed only with single above-ground stations (Fig. 5). The average DTE for all 40 above-ground stations employed in the campus of UniMAP was 64.63 \(\:\pm\:\) 8.30 days.
Discussion
This study to implement an area-wide management (AWM) strategy on 4.25 km2 using 11,551 bait stations (11, 511 in-ground stations and 40 above-ground stations) required approximately 16 months (in-ground stations) and 24 months (above-ground stations) to achieve complete elimination of Coptotermes termite activities in the study area. Our study utilized in-ground and above-ground CSI bait stations instead of liquid termiticides which usage has decreased over the years despite once being the most used method for managing subterranean termites36. Termite baiting uses a minimal amount of active ingredients, unlike soil termiticide treatment such as fipronil, to eliminate a termite colony, hence a significantly reduced impact on non-target soil organisms10,37. Additionally, no reemergence of termite activity was recorded at least 15 months after the last termite activity was observed.
Our study extended the demonstration by Mullins et al. (2011)29, who conducted an AWM at Louis Armstrong Park in New Orleans, Louisiana USA, which was 32 acres in size and took 23 months for termite colonies elimination, and further reported that six to 12 months elapsed before new termite activity was detected at the site38. The timeframe for elimination of the detected termite activity in our study was also consistent with the study by Shults et al. (2021)30, who reported efficacy of AWM using novaluron bait against existing colony subterranean termites Reticulitermes flavipes in Texas, USA took 17 months of baiting for complete elimination of the species and new invading colonies were unable to establish within the site.
The use of hexaflumuron bait in an AWM strategy successfully eliminated almost all R. flavipes infestations in a low-income community in Santiago, Chile within a 15-month period and the R. flavipes was not detected in the study area up to three years16. Similarly, Osbrink et al., (2011)17 reported AWM using hexaflumuron and chlorfluazuron active ingredients significantly reduced termite populations C. formosanus and R. flavipes in approximately 3 years of baiting in the campus of the Southern Regional Research Centre, New Orleans, USA. The reports of several previous studies using AWM with termite CSI baits against invasive termite species are also encouraging26,28. Baiting for termite control in the French Quarter of New Orleans, Louisiana, USA showed efficacy in reducing populations and preventing rapid reinvasion. However, challenges such as absentee property owners and difficulty treating structures with common walls persist. Improved detection methods and comprehensive treatments are enhancing effectiveness. A study conducted in Chatsworth, California, demonstrated the efficacy of AWM in controlling subterranean termite colonies in a condominium complex residential area. Over the course of the study, significant reduction in termite activity was observed, leading to minimal presence by the end of the monitoring period27. A study by Messenger et al. (2005)39 focused on eliminating only three colonies of C. formosanus in New Orleans, Louisiana, USA. The elimination method employed hexaflumuron baits, which successfully eliminated all selected colonies within three months. Monitoring was conducted to observe the reinvasion of the vacated territories by neighbouring C. formosanus colony and R. flavipes. Overall, the research highlighted the potential for reinvasion of termite colonies following elimination, underscoring the challenges in long-term termite control efforts. Wan & Ab Majid (2020)40 demonstrated that placing the same CSI bait, i.e. chlorfluazuron baits, in one active station effectively eliminated termite infestations at the study site was a bit longer at 4–8 weeks. Bait stations can be intercepted by re-invading colonies of subterranean termites and such events may occur multiple times in a single station, though such re-invading colonies were reported to be eliminated with continued baiting programs38.
The use of above-ground bait stations along with in-ground stations are reported to have the potential for faster colony elimination12. Our study showed that the days-to-elimination of termite activity for all in-ground stations employed in the study area was 39.71 ± 2.47 days, while above-ground stations recorded a slightly longer elimination at 64.63 ± 8.30 days. There could be several factors causing the longer elimination times in above-ground stations; Lee et al. (1999)41, Sajap et al. (2000)42 and Chouvenc (2024)12 suggest that efficacy differences in baits are influenced by how different termite species respond to them. Colony size and health could also influence time needed for final colony elimination, as larger, healthy colonies may succumb to treatment slower compared to relatively small or unhealthy colonies15. Additionally, Coptotermes sp. exhibit increased feeding rates under tropical conditions. In warmer climates, termite feeding rate, activity, and abundance will also increase43,44. In Southeast Asia, where the temperature is constant, the foraging activity of C. gestroi is triggered by high precipitation during the rainy season45. In Australia, colonies of C. acinaciformis and C. frenchi were terminated within 16 weeks (112 days) using chlorfluazuron bait. In Malaysia, C. curvignathus colonies were eliminated within 56 days with hexaflumuron bait46. Similarly, in The Philippines, C. vastator colonies required the same DTE to eliminate the species using chlorfluazuron bait46. Furthermore, Peters et al. (2003)47 reported the mound-building termite C. acinaciformis took 16 weeks to be eliminated when exposed to chlorfluazuron bait. In a laboratory condition, Rasib and Wright (2018)19 demonstrated that hexaflumuron at 1000 ppm effectively eliminated all R. santonensis termites within a 3-week period.
Our study recorded mean days-to-elimination (DTE) for in-ground stations was longest for stations employed near buildings at 43.80 ± 5.27 days, followed by landscape habitat at 39.40 ± 2.08 days and in-ground stations installed to protect trees at 37.49 ± 6.99 days. The result shows that building habitat is significantly associated with time to elimination. This could be because building habitat provides more food resources, including various wooden structures such as door frames and cabinets, and hiding places compared to trees and landscapes. This was documented by Lee (2002)2, who found that Coptotermes sp. frequently consumed wooden structures and eventually developed larger and more numerous termite nests near buildings. In addition, the result of the current research is consistent with those of Haagsma and Rust (1995)48, who found that populations of another subterranean termite, R. Hesperus, travelled greater distances when foraging in urban habitats and took longer to eliminate due to higher food availability. Besides, another supporting rationale of significant association of buildings with longer termite activity termination time could be due to the microclimate, such as temperature, humidity, and light intensity in the form of shade, which are highly suitable for termite foraging. Notably, from the observation of certain habitats in our study, termites displayed a higher rate of station attacks in shaded areas, which supported the observation of previous studies such as Fei and Henderson (2004)49 where the termites were characterized by reduced fluctuations in these environmental variables. Furthermore, Wan and Ab Majid (2022)50 reported that termite worker abundance increases with temperature within the species’ tolerance threshold and Woon et al. (2019)51 reported termites exhibit susceptibility to extreme humidity levels, avoiding areas with excessive moisture and display low tolerance for desiccation.
Conclusion
Our large-scale study in a tropical region, specifically the UniMAP campus in Perlis, demonstrated the effectiveness of area-wide management (AWM) strategy in eliminating Coptotermes sp. activity within the campus though termite baiting. By employing in-ground and above-ground baiting stations using chlorfluazuron-based bait, we recorded complete termite activity elimination in 16–24 months, with no termites reinvading the study area at least 15 months after the last recorded termite activity. Our study contributes novel field data on the effectiveness of chlorfluazuron-based bait in eliminating Coptotermes sp. activity in the study area, supporting the feasibility of implementing area-wide termite baiting programs in tropical regions.
Data availability
All data generated or analysed during this study are included as Supplementary Material.
References
Khan, M. A. & Ahmad, W. Termites: an overview. Termites and Sustainable Management: Volume 1-Biology, Social Behaviour and Economic Importance 1–25 (Springer, 2018).
Lee, C. Y. Subterranean termite pests and their control in the urban environment in Malaysia. Sociobiology 40 (1), 3–10 (2002).
Rust, M. K. & Su, N. Y. Managing social insects of urban importance. Annu. Rev. Entomol. 57 (1), 355–375. https://doi.org/10.1146/annurev-ento-120710-100634 (2012).
Lee, C. Y., Vongkaluang, C. & Lenz, M. Challenges to subterranean termite management of multi-genera faunas in Southeast Asia and Australia. Sociobiology 50 (1), 213–222 (2007).
Hassim, A. D. M., Appalasamy, S. & Arumugam, N. Termite species and structural pest identification in selected rural areas of Kelantan, Malaysia. IOP Conf. Ser. Earth Environ. Sci. 549(1), 012053 (2020).
Keng, W. M. & Rahman, H. Logistic regression to predict termite occurrences with environmental variables in primary forest and oil palm ecosystem: the case study in Sabah, Malaysia. Apcbee Procedia. 4, 53–57 (2012).
Kon, T. W., Bong, C. F., King, J. H. & Leong, C. T. Biodiversity of termite (Insecta: Isoptera) in tropical peat land cultivated with oil palms. Pakistan J. Biology. 15 (3), 108–120 (2012).
Arinana, A. et al. Termite diversity in urban landscape, South Jakarta, Indonesia. Insects 7 (2), 20 (2016).
Eger, J. E. Jr et al. Elimination of subterranean termite (Isoptera: Rhinotermitidae) colonies using a refined cellulose bait matrix containing noviflumuron when monitored and replenished quarterly. J. Econ. Entomol. 105 (2), 533–539 (2012).
Neoh, K. B. & Lee, C. Y. Asian subterranean termite, Coptotermes gestroi. In Biology and Management of the Formosan Subterranean Termite and Related Species (eds. Su, N. Y. & Lee, C. Y.) 323–352 (CAB International, 2023). https://doi.org/10.1079/9781800621596.0016.
Department of Agriculture. Status Pendaftaran Racun Makhluk Perosak Chlorpyrifos. (2021). https://www.doa.gov.my/doa/resources/aktiviti_sumber/sumber_awam/maklumat_racun_perosak/pekeliling/surat_pekeliling_lrmp_04_2021.pdf.
Chouvenc, T. How do termite baits work? implication of subterranean termite colony demography on the successful implementation of baits. J. Econ. Entomol. 2024, 243. https://doi.org/10.1093/jee/toae243 (2024).
Su, N. Y. & Scheffrahn, R. H. Laboratory evaluation of two chitin synthesis inhibitors, hexaflumuron and diflubenzuron, as bait toxicants against Formosan and eastern subterranean termites (Isoptera: Rhinotermitidae). J. Econ. Entomol. 86 (5), 1453–1457. https://doi.org/10.1093/jee/86.5.1453 (1993).
Kakkar, G., Osbrink, W. L. A. & Su, N. Y. Molting site fidelity accounts for colony elimination of the Formosan subterranean termites (Isoptera: Rhinotermitidae) by chitin synthesis inhibitor baits. Sci. Rep. 8 (1), 1259. https://doi.org/10.1038/s41598-018-19603-8 (2018).
Su, N. Y. & Lee, C. Y. Biology and Management of the Formosan Subterranean Termite and Related Species 403 (CAB International, 2023).
Smith, J., Su, N. Y. & Escobar, R. N. An areawide population management project for the invasive eastern subterranean termite (Isoptera: Rhinotermitidae) in a low-income community in Santiago, Chile. Am. Entomol. 52 (4), 253–260 (2006).
Osbrink, W. L., Cornelius, M. L. & Lax, A. R. Areawide field study on effect of three chitin synthesis inhibitor baits on populations of Coptotermes formosanus and Reticulitermes flavipes (Isoptera: Rhinotermitidae). J. Econ. Entomol. 104 (3), 1009–1017 (2011).
Chouvenc, T. Comparative impact of chitin synthesis inhibitor baits and non-repellent liquid termiticides on subterranean termite colonies over foraging distances: colony elimination versus localized termite exclusion. J. Econ. Entomol. 111 (5), 2317–2328. https://doi.org/10.1093/jee/toy210 (2018).
Rasib, K. Z. & Wright, D. J. Comparative efficacy of three bait toxicants against the subterranean termite Reticulitermes santonensis (Isoptera/Blattoidea: Rhinotermitidae). Biomed. J. Sci. Tech. Res. 11, 3 (2018).
Su, N. Y. Response of the Formosan subterranean termites (Isoptera: Rhinotermitidae) to baits or nonrepellent termiticides in extended foraging arenas. J. Econ. Entomol. 98, 2143–2152 (2005).
Chouvenc, T. & Su, N. Y. Subterranean termites feeding on CSI baits for a short duration still results in colony elimination. J. Econ. Entomol. 110 (6), 2534–2538. https://doi.org/10.1093/jee/tox282 (2017).
Chouvenc, T. Subterranean termite (Coptotermes gestroi (Blattodea: Rhinotermitidae)) colony elimination through exposure to a novaluron CSI bait formulation in laboratory. J. Econ. Entomol. 114 (3), 1249–1255. https://doi.org/10.1093/jee/toab061 (2021).
Deheer, C. J. & Vargo, E. L. Colony genetic organization and colony fusion in the termite Reticulitermes flavipes as revealed by foraging patterns over time and space. Mol. Ecol. 13 (2), 431–441 (2004).
Howard, R. W., Jones, S. C., Mauldin, J. K. & Beal, R. H. Abundance, distribution, and colony size estimates for Reticulitermes spp.(Isoptera: Rhinotermitidae) in southern Mississippi. Environ. Entomol. 1 (6), 1290–1293 (1982).
Vreysen, M. J. B., Robinson, A. S., Hendrichs, J. & Kenmore, P. Area-wide integrated pest management (AW-IPM): principles, practice, and prospects. In Area-Wide Control of Insect Pests (eds. Vreysen, M. J. B., Robinson, A. S. & Hendrichs, J.) 3–33. (Springer, 2007). https://doi.org/10.1007/978-1-4020-6059-5_1 .
Lax, A. R., Guillot, F. S. & Ring, D. R. Area-wide management of the Formosan subterranean termite in New Orleans’ French Quarter (IAEA-CN–131) (International Atomic Energy Agency (IAEA), 2005).
Getty, G. M., Solek, C. W., Sbragia, R. J., Haverty, M. I. & Lewis, V. R. Large-scale suppression of a subterranean termite community using the Sentricon® termite colony elimination system: a case study in Chatsworth, California, USA. Sociobiology 50 (3), 1041–1050 (2007).
Guillot, F. S. et al. Area-wide management of the Formosan subterranean termite, Coptotermes formosanus Shiraki (Isoptera: Rhinotermitidae), in the New Orleans French Quarter. Sociobiology 55 (2), 311–338 (2010).
Mullins, A. J., Su, N. Y. & Owens, C. Reinvasion and colony expansion of Coptotermes formosanus (Isoptera: Rhinotermitidae) after areawide elimination. J. Econ. Entomol. 104 (5), 1687–1697 (2011).
Shults, P. et al. Area-wide elimination of subterranean termite colonies using a novaluron bait. Insects 12 (3), 192 (2021).
Broadbent, S., Farr, M., Bernklau, E. J., Siderhurst, M. S. & James, D. M. Field attraction of termites to a carbon dioxide-generating bait in Australia (Isoptera). Sociobiology 48 (3), 1–9 (2006).
Lee, C. Y. Identification Key to Common Subterranean Termites Found in and Around Buildings and Structure in Malaysia 34 (Dow AgroSciences (Malaysia), 2003).
Lee, C. Y. Termites of Singapore -- A Scientific Guide for Pest Management Professionals 176 (Singapore Pest Management Association, 2014).
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria (2024). https://www.R-project.org/.
Bates, D., Maechler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67 (1), 1–48. https://doi.org/10.18637/jss.v067.i01 (2015).
Lee, C. Y. & Neoh, K. B. Management of subterranean termites using liquid termiticides. In Biology and Management of the Formosan Subterranean Termite and Related Species (eds. Su, N. Y. & Lee, C. Y.) 238–272 (CAB International, 2023). https://doi.org/10.1079/9781800621596.0016.
Su, N. Y. Termite IPM in historic sites. J. Integr. Pest Manag. 15 (1), 26. https://doi.org/10.1093/jipm/pmae013 (2024).
Su, N. Y., Guidry, E. & Cottone, C. Sustainable management of subterranean termite populations (Isoptera: Rhinotermitidae) in Armstrong Park, New Orleans, with durable baits. J. Econ. Entomol. 109 (3), 1326–1332. https://doi.org/10.1093/jee/tow051 (2016).
Messenger, M. T., Su, N. Y., Husseneder, C. & Grace, J. K. Elimination and reinvasion studies with Coptotermes formosanus (Isoptera:Rhinotermitidae) in Louisiana. J. Econ. Entomol. 98, 916–929 (2005).
Wan Umar, W. A. S. & Ab Majid, A. H. Efficacy of minimum application of chlorfluazuron baiting to control urban subterranean termite populations of Coptotermes gestroi (Wasmann) (Blattodea: Rhinotermitidae). Insects 11 (9), 569. https://doi.org/10.3390/insects11090569 (2020).
Lee, C. Y., Yap, H. H., Chong, N. L. & Jaal, Z. Urban Pest Control–A Malaysian Perspective (Vector Control Research Unit, Universiti Sains Malaysia, 1999).
Sajap, A. S., Amit, S. & Welker, J. Evaluation of hexaflumuron for controlling the subterranean termite Coptotermes curvignathus (Isoptera: Rhinotermitidae) in Malaysia. J. Econ. Entomol. 93, 429–433. https://doi.org/10.1603/0022-0493-93.2.429 (2000).
Cao, R. X. & Su, N. Y. Temperature preferences of four subterranean termite species (Isoptera: Rhinotermitidae) and temperature-dependent survivorship and wood-consumption rate. Ann. Entomol. Soc. Am. 109, 64–71 (2016).
Zanne, A. E. et al. Termite sensitivity to temperature affects global wood decay rates. Science 377, 1440–1444 (2022).
Sornnuwat, Y., Tsunoda, K., Yoshimura, T., Takahashi, M. & Vongkaluang, C. Foraging populations of Coptotermes gestroi (Isoptera: Rhinotermitidae) in an urban area. J. Econ. Entomol. 89, 1485–1490 (1996).
Peters, B. C., Broadbent, S. & Dhang, P. Evaluating a baiting system for management of termites in landscape and orchard trees in Australia, Hong Kong, Malaysia, and the Philippines. In Proceedings of the 6th International Conference on Urban Pests; Budapest, Hungary 379–383 (2008).
Peters, B. C. & Fitzgerald, C. J. Field evaluation of the bait toxicant chlorfluazuron in eliminating Coptotermes acinaciformis (Froggatt) (Isoptera: Rhinotermitidae). J. Econ. Entomol. 96 (6), 1828–1831 (2003).
Haagsma, K. A. & Rust, M. K. Colony size estimates, foraging trends, and physiological characteristics of the western subterranean termite (Isoptera: Rhinotermitidae). Environ. Entomol. 24 (6), 1520–1528 (1995).
Fei, H. & Henderson, G. Effects of temperature, directional aspects, light conditions, and termite species on subterranean termite activity (Isoptera: Rhinotermitidae). Environ. Entomol. 33 (2), 242–248 (2004).
Wan Umar, W. A. S. & Ab Majid, A. H. Ecological factors affecting the abundance of worker–soldier proportions, and wood consumption of Coptotermes gestroi (Wassman) (Blattodea: Rhinotermitidae) in a tropical urban environment. Int. J. Trop. Insect Sci. 42 (3), 2593–2602 (2022).
Woon, J. S., Boyle, M. J. W., Ewers, R. M., Chung, A. & Eggleton, P. Termite environmental tolerances are more linked to desiccation than temperature in modified tropical forests. Insectes Soc. 66, 57–64 (2019).
Acknowledgements
We thank the Development Department of Universiti Malaysia Perlis (UniMAP) for awarding the contract project of termite control to Insepro Sdn Bhd. We also thank both UniMAP and Insepro for allowing us to carry out this research. We thank Ensystex (Malaysia) Sdn. Bhd. who provided funding to the research team and provided the logistic support needed. We extend our thanks to all personnel involved in the project especially from Insepro Malaysia Sdn Bhd who provided well trained technicians who were involved in installation of both in-ground and above-ground bait stations in the study site.
Funding
No funding was obtained for this study.
Author information
Authors and Affiliations
Contributions
HS, AAA, SQO and AHA: Conceptualization; Formal analysis; Investigation; Methodology; Validation; Visualization; Writing. SR: Writing. KAI, KRA, NS and FMY: Funding acquisition; Writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Salim, H., Alymann, A.A., Ong, SQ. et al. A study on area-wide management of subterranean termites through baiting strategies in a tropical region. Sci Rep 16, 11073 (2026). https://doi.org/10.1038/s41598-026-40987-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40987-5