Abstract
For treatment of muscle spasms associated pain, combination of nonsteroidal anti-inflammatory drugs like diclofenac (DIC) and muscle relaxants as methocarbamol (MET) is usually utilized. This work represents a novel, rapid, facile, sensitive, and selective first derivative synchronous fluorescence spectroscopy (FDSFS) for the simultaneous determination of DIC and MET in their combined tablets. Factors influencing method’s sensitivity were investigated, and the best findings were accomplished applying Δ λ = 60 nm and using water as a diluting solvent. Through applying the optimized experimental conditions, DIC showed a lower detection limit of 0.15 µg/mL and a quantitation limit of 0.30 µg/mL, while MET corresponding values were 0.03 µg/mL and 0.05 µg/mL. Diclofenac was measured at 288 nm, while methocarbamol was measured at 346 nm, exhibiting linearity over the concentration ranges of 0.3–2.5 and 0.05–5.0 µg/mL, respectively. Through application to several laboratory-prepared mixtures and commercial formulation, the suggested method’s applicability was determined. When comparing the proposed method to the reported HPLC method using the student’s t-test and F-ratio test, no discernible differences were found. Due to simplicity and economical advantage of the method, it can be applied in quality control laboratories for analysis of the studied drugs. The evaluation of the method’s eco-friendliness and greenness was also performed using Analytical GREEnness (AGREE), Green Analytical Procedure Index (GAPI) and Analytical Green Star Area (AGSA) metrics. Complete validation procedures were applied to the suggested approach in compliance with the International Conference on Harmonization’s criteria.
Similar content being viewed by others
Introduction
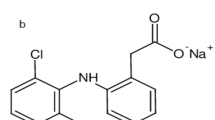
Diclofenac (DIC) is a phenyl acetic acid derivative, chemically designated as [2-(2,6-Dichloroanilino) phenyl] acetic acid1 (Fig. 1A). As a non-steroidal anti-inflammatory medication2, it acts by blocking the cyclooxygenase 1 and 2 enzymes, which prevents prostaglandin synthesis3. It is regarded as a first-line treatment for rheumatoid arthritis, osteoarthritis, and other illnesses that cause both acute and chronic pain and inflammation4,5.
Structural formulae of DIC (A) and MET (B).
Methocarbamol (MET); a guaiacol glyceryl ether (Fig. 1B) [2-hydroxy-3-(2-methoxyphenoxy) propyl] carbamate1. A centrally-acting skeletal muscle relaxant with an unclear mode of action6,7,8. Methocarbamol is used to treat muscle spasms in individuals with pyramidal spine abnormalities, such as ruptured intervertebral discs9.
Several techniques, such as spectrophotometry10,11,12,13,14,15,16, gas chromatography17,18,19,20,21, high performance liquid chromatography22,23,24,25,26,27,28,29, thin layer chromatography30,31,32,33 and ultra performance liquid chromatography34,35 were reported in earlier studies to quantify DIC and MET wheather alone or in conjunction with other medications.
The routine quality control analysis of the drugs in concern still requires an alternative to these techniques, as liquid chromatography needs skilled analysts to operate, constant maintenance, uses costly, high-grade chemicals, and consumes a lot of organic solvents which deprive it from the required sustainability, and spectrophotometry’s sensitivity is low. These facts gave us the motivation to carry out this work and because up to the moment, no analytical tool has been reported for the simultaneous determination of the concerned drugs using a direct fluorescence spectroscopic method which is advantageous by being rapid, sensitive, economic, and nondestructive requiring minimal sample preparation. However, the analysis of drug mixtures by single-wavelength spectrofluorimetry is limited since their spectra overlap36. This limitation is overcomed by synchronous fluorescence spectroscopy (SFS), which reduces overlap between the resulting spectra, improves selectivity, and narrows spectral bands37. Variable angle synchronous luminescence, constant energy, and constant wavelength are some of the several modalities of SFS methods. Constant-energy SFS (CESFS), proposed by Inman et al.38. In CESFS, the excitation and the emission monochromators are scanned synchronized so that a constant energy difference is maintained between two monochromators. This technique significantly increased the selectivity of the analysis of PAHs mixtures39. Another technique is to quantify fluorescence by simultaneously scanning the excitation and the emission monochromators at various rates. This technique, known as variable-angle SFS (VASFS), is known for both its great selectivity and versatility40,41. Because of its special characteristics, constant wavelength synchronous fluorescence spectroscopy (CWSFS) was used in this study. It entails scanning both the excitation and emission wavelengths simultaneously at constant Δλ values37. Synchronous fluorimetry in conjunction with derivatization is beneficial in terms of sensitivity and selectivity; it frequently yields peaks that are well-resolved in multicomponent mixes, simplifies intricate spectra, and produces narrow-width, sharp bands42, as well as high tolerance limits and recovery percentages. For these reasons, it was adopted in this work42.
Experimental
Apparatus
Shimadzu RF-6000 (A40245801833SA) Spectro fluorophotometer equipped with a 150 W Xenon arc lamp, (Shimadzu, Kyoto, Japan). The pH was adjusted using a “Jenway 3503 digital pH meter (Stone, Staffs, UK)”. “Stakpure Pure water system OmniaTap12 UV” used to get deionized water. “Sartorius Entris 224-1S laboratory balance” used to weigh the raw materials of drugs.
Materials and reagents
Diclofenac acid of purity (100.85%), (Batch #678) was generously supplied by Swiss pharma Co., Ciba-Geigy, Switzerland and Methocarbamol of purity (99.98%), (Batch# B/MC-IPA/12/20/010) was kindly gifted by (MUP); Medical Union Pharmaceuticals Co., Cairo, Egypt. Purity of both drugs was determined by applying the methods published by United States Pharmacopeia (USP). Methoquick® tablets: 50 mg DIC and 500 mg MET/ tablet, (Batch #240381), manufactured by (Sigma pharmaceutical & chemical industries, Egypt), were obtained from local pharmacies.
Methanol, acetone and acetonitrile (99.9% purity, a product of Fisher scientific, UK) HPLC grade. Potassium dihydrogen phosphate obtained from PIOCHEM Laboratory Chemicals, 1st industrial zone, 6th of October, Egypt.
Standard stock solutions
Serial dilution was performed with methanol to reach stock solutions of concentrations 100.0 µg/mL. Methanol was used to further dilute the DIC and MET working standard solutions. This solution remained stable in the refrigerator for at least a week.
Construction of calibration curves
Appropriate volumes of 0.3, 0.5,1.0, 1.5, 2.0 and 2.5 mL for DIC and 0.05, 0.25, 0.5, 1.0, 2.0, 3.0, 3.5, 4.0, 5.0 mL for MET of stock solutions of concentration 100 µg/mL were transferred to 10.0 mL volumetric flasks, 1 mL phosphate buffer (pH 7) was added to each flask then the volume was completed to the mark with deionized water as a diluent. These concentrations covering the linearity ranges of 0.3–2.5 and 0.05–5.0 µg/mL for DIC and MET, respectively, were prepared to construct the calibration curve. Monochromators were scanned at (Δλ) 60 nm to record synchronous fluorescence spectra. The excitation and emission monochromators’ bandwidth were adjusted at 5 nm using a scan rate of 6000 nm/min. A blank experiment was conducted concurrently with each measurement. Shimadzu Lab Solution RF® software was then used to derivatize the resulting SFS spectra. DIC was measured selectively at 288 nm (zero crossing point for MET), while MET could be estimated at 346 nm (zero crossing point for DIC) applying FDSFS. By plotting the corrected peak amplitudes versus the final drug concentration (µg/mL), the calibration graphs were established, or the appropriate regression equations were determined.
Analysis of laboratory prepared mixtures
The marketed formulation’s ratio as well as various laboratory- prepared mixtures with varying DIC and MET concentrations and ratios were prepared. Appropriate volumes of the drug stock solutions were put into 10.0 mL volumetric flasks and diluted with deionized water to the appropriate volume. The steps described in " Construction of calibration curves " were then carried out. The percentage recoveries were calculated using the appropriate regression equations.
Analysis of Methoquick® tablets
Methoquick® tablets were precisely weighed. About 30 mL of methanol was added to a conical flask containing an amount of the properly grinded ground tablets equivalent to 50 mg of DIC and 500 mg of MET. Filtration was finished after 20 min of sonicating the contents of the flask, and the filtrate was quantitatively transferred to a volumetric flask (100.0 mL). Two additions of 30 mL of methanol were made to complete the extraction procedures. The volume was then completed to the appropriate level using the same solvent. After that, the steps described under " Construction of calibration curves " were carried out, and the relevant regression equations were used to determine the content of DIC and MET in their tablets.
Results and discussion
Compared to conventional fluorescence spectroscopy, SFS provides a lot of benefits, such as low interference and high selectivity43. SFS is a relatively straightforward and efficient way to obtain data for quantitative determination in a single run because of its resultant sharp, narrowed spectra36. It has been previously employed to simultaneously determine various pharmaceutical compounds binary mixtures in different matrices44,45,46,47. Figure 2 demonstrates that the native fluorescence spectra of DIC and MET significantly overlap, making it unfeasible to determine them simultaneously. SFS has been utilized to overcome this overlap. As shown in Fig. 3, the SFS spectra of DIC and MET still exhibit significant spectral overlap. Hence first derivative synchronous fluorescence spectroscopy (FDSFS) has been applied where DIC can be detected at 288 nm and MET at 346 nm upon using Δλ = 60 nm as Fig. 4 illustrates.
Emission spectra of 1.0 µg/mL DIC and 0.25 µg/mL MET.
Synchronous fluorescence spectroscopy of 1.0 µg/mL DIC and 0.25 µg/mL MET at Δλ 60 nm using 1 mL phosphate buffer pH 7.
First derivative synchronous fluorescence spectra of 1.0 µg/mL DIC and 0.25 µg/mL MET at Δλ 60 nm using 1 mL phosphate buffer pH 7.
Method optimization
The developed method’s selectivity was enhanced by carefully investigating and optimizing several experimental parameters including:
Selection of Δλ
The choice of Δλ in CWSFS should be thoroughly studied because it has significant effect on band width and peak intensity42. Scanning of each of DIC and MET was performed at different values of Δλ, ranging from (20 to 120) nm. Different SFS of DIC (Fig. 5A), and MET (Fig. 5B) at various Δλ values reveal that Δλ = 60 nm is the optimum regarding peaks symmetry and resolution when simultaneously analyzed in alkaline media, providing the highest resolution and enabling DIC quantification at 288 nm following first derivatization. whereas, MET was quantified at 346 nm following first derivatization.
SFS of (A) 1.0 µg/mL DIC at Δƛ: (a) 20 nm, (b) 40 nm, (c) 60 nm, (d) 80 nm, (e) 100 nm and (f) 120 nm. (B) 0.25 µg/mL MET at Δƛ: (a) 20 nm, (b) 40 nm, (c) 60 nm, (d) 80 nm, (e) 100 nm and (f) 120 nm.
Selection of diluting solvent
A variety of solvents, including water, methanol, ethanol, acetone, and acetonitrile, were tested. The highest sensitivity and selectivity between the studied drugs have been obtained when water was used as a solvent, which adds another advantage to the suggested method.
Selection of buffer
Acetate buffer (pH 3-5.5) and phosphate buffer (pH 6–10) were tested. The highest sensitivity for the studied drugs has been attained when phosphate buffer (pH 7) was used.
Selection of buffer volume
Different volumes of phosphate buffer pH 7 were tried (1–3 mL), 1 mL resulted in the maximum fluorescence intensity, hence it was recommended throughout the work (Fig. 6). Different volumes below 1 mL were experimentally tried, but such volumes were not enough to reach the required pH for the prepared solutions.
Effect of buffer volume on the intensity values (1.0 µg/mL DIC and 0.25 µg/mL MET).
Method validation
The proposed method was fully validated according to ICH guidelines48 regarding linearity, range, precision, accuracy, detection limit (LOD), quantitation limit (LOQ) and robustness.
Linearity and range
Upon applying the proposed method DIC and MET responses were linear over the concentration ranges of 0.3–2.5 µg/mL and 0.05–5.0 µg/mL, respectively. Plotting the peak amplitudes versus the final concentration of each drug in µg/mL led to the construction of calibration curves, which in turn permitted the deduction of partial last square regression equations (Table 1). Low scattering of the data points around the calibration line, endorsed by correlation coefficient values that approach unity, confirms the linearity of the measured responses with concentration. Furthermore, the regression data pertaining to standard deviation of the slope (Sb), standard deviation of the intercept (Sa), and standard deviation of residuals (Sy/x) were used to assess the validity of the suggested method (Table 1).
LOQ and LOD
The lowest drug concentration that can be identified under the stated experimental conditions but not necessarily quantified is known as (LOD)48. The lowest analyte concentration that can be identified with acceptable accuracy and precision is known as the (LOQ)48. The calculated values are listed in (Table 1).
Precision
The intra-day and inter-day precision were evaluated using three replicate analyses of three concentrations of pure DIC and MET within their linearity ranges on the same day or on three consecutive days respectively. The obtained results are summarized in (Table 2). The small SD values demonstrate the high precision of the proposed approach.
Accuracy
Results obtained from drug analyses applying the proposed method in pure form and in laboratory prepared mixtures (Table 3) showed acceptable accuracy as revealed from the percent recoveries obtained. By comparing the results obtained upon application of the proposed method to the analysis of the analytes in their tablets with those of comparison method49, it is possible to conclude that the proposed method is accurate as illustrated from the low t and F test values50.
Robustness
Consistency of peak amplitudes with deliberately minor changes in various experimental conditions was used to establish it. These changes related to method and involve the buffer volume (1 mL ± 0.1) as shown in (Fig. 6). The robustness of the method was demonstrated by the fact that peak amplitudes of the drugs under study were not influenced by these little changes that may happen during the experimental process.
Applications
Application of the proposed method to dosage form
Diclofenac and methocarbamol in its tablets were effectively analyzed using the suggested method, the results exhibited were in good agreement with those obtained via the reference HPLC method which used a C18 column with mobile phase composed of phosphate buffer: methanol (30:70), adjusted to pH 4.5 with orthophosphoric acid, adopting a flow rate 1 mL/min., with UV detection at 281 nm49. The calculated t and F values were less than the tabulated ones, which highlighted the correspondence between the suggested and reference method50. Table 4 provides an overview of the obtained data.
Greenness assessment of the proposed method
AGREE51, GAPI52, and AGSA53 were used to assess the proposed method’s greenness. AGREE is a metric system used in the proposed method to determine environmental and occupational hazards. It assesses twelve important environmental criteria and assigns a score between 0 and 1 to represent how environmentally friendly the analytical process is51. On the other hand, GAPI symbolically express the greenness of individual aspects of the analytical procedure via three levels of color scale: green, yellow, or red, indicating high, medium, or low impact respectively52. Eventually, the Analytical Green Star Area (AGSA) presents a thorough, integrated rating system that is easy to use. AGSA ensures objective evaluation while adhering to the 12 GAC Principles by integrating method classification, integrated scoring, and resistance to human prejudice53. Our proposed method is superior in terms of greenness compared to the reported chromatographic method49, as we use spectrofluorimetry, which uses less energy, produces less waste, uses a safer solvent, and takes less time than HPLC. Consequently, the method was observed to be excellent green with a green GAPI pictogram along with higher AGREE and AGSA scores as demonstrated in Fig. 7.
Assessment of the greenness profile of the proposed method by AGREE (a), GAPI (b) and AGSA (c) metrics.
Conclusion
In order to determine DIC and MET in their combined dosage form simultaneously, this work showed how to develop and validate a sensitive, accurate, specific, and precise FDSFS approach accomplishing LOD values 0.15 and 0.03 µg/mL, respectively. Furthermore, the developed method has an economical advantage and do not require complex requirements for sample and data processing. In addition to the reproducibility as well as the simplicity and convenience. So it can be applied in quality control laboratories for analysis of the studied drugs. Applying the procedure to Methoquick® tablets is encouraged by its high sensitivity.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
O’Neil, M. J., Smith, A. & Heckelman, P. E. The Merck Index—An Encyclopedia of Chemicals, Drugs, and Biologicals (Merck and Co. Inc., 2006).
Altman, R., Bosch, B., Brune, K., Patrignani, P. & Young, C. Advances in NSAID development: evolution of diclofenac products using pharmaceutical technology. Drugs 75(8), 859–877. https://doi.org/10.1007/s40265-015-0392-z (2015).
Gan, T. J. Diclofenac: an update on its mechanism of action and safety profile. Curr. Med. Res. Opin. 26(7), 1715–1731. https://doi.org/10.1185/03007995.2010.486301 (2010).
Pirard, D., Vereecken, P., Melot, C. & Heenen, M. 3% diclofenac in 2.5% hyaluronan gel in the treatment of actinic keratoses: a meta-analysis of the recent studies. Arch. Dermatol. Res. 297(5), 185–189. https://doi.org/10.1007/s00403-005-0601-9 (2005).
Tampucci, S. et al. Diclofenac-derived hybrids for treatment of actinic keratosis and squamous cell carcinoma. Molecules 24(9), 1793. https://doi.org/10.3390/molecules24091793 (2019).
See, S. & Ginzburg, R. Skeletal muscle relaxants. Pharmacotherapy. J. Hum. Pharmacol. Drug Therapy. 28(2), 207–213. https://doi.org/10.1592/phco.28.2.207 (2008).
Sibrack, J., Patel, P. & Hammer, R. Methocarbamol. In StatPearls [Internet] 2024 Sep 10. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK565868/
Beebe, F. A., Barkin, R. L. & Barkin, S. A clinical and pharmacologic review of skeletal muscle relaxants for musculoskeletal conditions. Am. J. Ther. 12(2), 151–171 (2005).
Jung, H. & Chae, H. K. The safety and efficacy of methocarbamol as a muscle relaxant with analgesic action: analysis of current data. Innov. Biosyst Bioeng. 3, 201–221 (2019).
Elkady, E. F. Simultaneous spectrophotometric determination of diclofenac potassium and methocarbamol in binary mixture using chemometric techniques and artificial neural networks. Drug. Test. Anal. 3(4), 228–233 (2011).
El-Din, M. S., Eid, M. & Zeid, A. M. Simultaneous determination of methocarbamol and aspirin binary mixture in their combined tablets by derivative and ratio derivative spectrophotometry. Anal. Methods. 7(13), 5674–5681. https://doi.org/10.1039/C3AY42163E (2015).
El-Malla, S. F., Hamza, A. A. & Elagamy, S. H. Effective spectrophotometric methods for resolving the superimposed spectra of Diclofenac Potassium and Methocarbamol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 296, 122662. https://doi.org/10.1016/j.saa.2023.122662 (2023).
Patel, N. P. & Hinge, M. Development and validation of UV spectroscopic method for simultaneous estimation of methocarbamol and diclofenac sodium in injection dosage form. Wjpps 5(4), 1625–1636. https://doi.org/10.20959/wjpps20164-6451 (2016).
Nagamani, M., Ramana, H., Bhadru, B. & Vasanthi, R. Method development and validation of methocarbamol in bulk and its formulation by UV spectroscopy. Int. J. Pharm. Chem. Biol. Sci. 5(1) (2015).
Vijayasree, P., Devika, G. S., Sravani, M., Krishna, G. S. & Vali, K. D. Simple UV spectrophotometric estimation of methocarbamol by co-solubilization technique. Res. J. Pharm. Technol. 7(8), 2 (2014).
Kabra, P., Patel, C., Kimbahune, R., Raj, N. & LVG N. Simultaneous spectrophotometric estimation of Metaxalone and Diclofenac Potassium tablet dosage form. Int. J. Pharm. Sci. Rev. Res. 15(1), 36–40 (2012). Available online at: www.globalresearchonline.net.
Yilmaz, B. & Ciltas, U. Determination of diclofenac in pharmaceutical preparations by voltammetry and gas chromatography methods. J. Pharm. Anal. 5(3), 153–160. https://doi.org/10.1016/j.jpha.2014.10.005 (2015).
Ramana Rao, G., Avadhanulu, A. B., Vatsa, D. K. & Pantulu, A. R. Gas liquid chromatographic determination of paracetamol and methocarbamol in single and combined dosage forms. Indian Drugs. 27(11), 576–580 (1990).
Sioufi, A., Pommier, F. & Godbillon, J. Determination of diclofenac in plasma and urine by capillary gas chromatography—mass spectrometry with possible simultaneous determination of deuterium-labelled diclofenac. J. Chromatogr. B Biomed. Sci. Appl. 571(1–2), 87–100. https://doi.org/10.1016/0378-4347(91)80436-G (1991).
Sane, R. T., Samant, R. S. & Nayak, V. G. Gas chromatographic determination of methocarbamol from pharmaceutical preparations. Indian Drugs. 24(4), 196–198 (1987).
Sane, R. T., Surve, S. R., Gangrade, M. G. & Bapat, V. V. Simultaneous gas chromatographic estimation of combined dosages, I) Amitriptyline hydrochloride with chlordiazepoxide, II) Methocarbamol with Ibuprofen, III) Paracetamol with Diclofenac sodium and IV) Paracetamol with Promethazine hydrochloride. INDIAN DRUGS-BOMBAY-. 30, 66 (1993).
Kalokhe, S., Nalwade, S., Patil, P. & Raskar, P. Development and Validation of a Stability-Indicating High-Performance Liquid Chromatographic Method for the Quantification of Methocarbamol and Its Impurities in Pharmaceutical Dosage Forms. J. Chromatogr. Sci. 59(6), 555–565. https://doi.org/10.1093/chromsci/bmaa125 (2021).
Bassuoni, Y. F., AboulMagd, A. M. & Ibrahim, M. M. Green chromatographic approach to determine methocarbamol, aspirin and their related impurities in their combined pharmaceutical formulation and human plasma: with computational evaluation. BMC Chem. 19(1), 136. https://doi.org/10.1186/s13065-025-01500-7 (2025).
Hafsa, D., Chanda, S. & Prabhu, J. Simultaneous HPLC determination of methocarbamol, paracetamol and diclofenac sodium. J. Chem. 8(4), 1620–1625. https://doi.org/10.1155/2011/614748 (2011).
Qiushi, P. et al. Simultaneous determination of methocarbamol and ibuprofen in the presence of five related impurities by reversed phase liquid chromatography. Anal. Chem. Lett. 7(5), 623–631. https://doi.org/10.1080/22297928.2017.1385419 (2017).
Elkady, E. F., Fouad, M. A. & Mozayad, A. N. Application of Box-Behnken experimental design and response surface methodology for selecting the optimum RP-HPLC conditions for the simultaneous determination of methocarbamol, indomethacin and betamethasone in their pharmaceutical dosage form. BMC Chem. 16(1), 114. https://doi.org/10.1186/s13065-022-00908-9 (2022).
Mohamed, S. A. & Belal, F. F. A micellar HPLC method for simultaneous determination of methocarbamol in three different tablets using single run; application to human plasma and evaluation of the method greenness. Microchem. J. 148, 262–269. https://doi.org/10.1016/j.microc.2019.04.079 (2019).
Walash, M. I., Ibrahim, F. & Abo El Abass, S. Development and validation of HPLC method for simultaneous estimation of famotidine, paracetamol and diclofenac in their raw materials and pharmaceutical formulation. Anal. Chem. Lett. 7(3), 421–437. https://doi.org/10.1080/22297928.2017.1353921 (2017).
AlNahwa, L. H. et al. Development of an RP-HPLC method for quantifying diclofenac diethylamine, methyl salicylate, and capsaicin in pharmaceutical formulation and skin samples. Molecules 29(12), 2732. https://doi.org/10.3390/molecules29122732 (2024).
El-Malla, S. F., Hamza, A. A. & Elagamy, S. H. Thin-layer chromatography–densitometric method for the simultaneous determination of paracetamol and methocarbamol in their dosage form. JPC–Journal Planar Chromatography–Modern TLC. 37(6), 547–556. https://doi.org/10.1007/s00764-024-00326-8 (2024).
El-Yazbi, F. A., Amin, O. A., El-Kimary, E. I., Khamis, E. F. & Younis, S. E. HPTLC and spectrophotometric estimation of febuxostat and diclofenac potassium in their combined tablets. J. Chromatogr. Sci. 54(7), 1146–1152. https://doi.org/10.1093/chromsci/bmw091 (2016).
Kaale, E., Nyamweru, B. C., Manyanga, V., Chambuso, M. & Layloff, T. The development and validation of a Thin Layer Chromatography densitometry method for the analysis of diclofenac sodium tablets. Int. J. Chem. Anal. Sci. 4(2), 73–79. https://doi.org/10.1016/j.ijcas.2013.05.001 (2013).
Thongchai, W., Liawruangrath, B., Thongpoon, C. & Machan, T. High performance thin layer chromatographic method for the determination of diclofenac sodium in pharmaceutical formulations. Chiang Mai J. Sci. 33(1), 123–128 (2006).
Zheng, Y. et al. Simultaneous determination of methocarbamol and paracetamol in the presence of three related substances by ultra performance liquid chromatography. Curr. Pharm. Anal. 15(5), 505–510. https://doi.org/10.2174/1573412914666180702150357 (2019).
Elzayat, E. M. et al. A validated stability-indicating UPLC method for determination of diclofenac sodium in its pure form and matrix formulations. Arab. J. Chem. 10, S3245–S3254. https://doi.org/10.1016/j.arabjc.2013.12.022 (2017).
Patra, D. & Mishra, A. K. Recent developments in multi-component synchronous fluorescence scan analysis. TRAC Trends Anal. Chem. 21(12), 787–798. https://doi.org/10.1016/S0165-9936(02)01201-3 (2002).
Andrade-Eiroa, A., de-Armas, G., Estela, J. M. & Cerda, V. Critical approach to synchronous spectrofluorimetry. II. TRAC Trends Anal. Chem. 29(8), 902–927. https://doi.org/10.1016/j.trac.2010.05.002 (2010).
Inman, E. L., Files, L. A. & Winefordner, J. D. Theoretical optimization of parameter selection in constant energy synchronous luminescence spectrometry. Anal. Chem. 58(11), 2156–2160. https://doi.org/10.1021/ac00124a011 (1986).
Inman, E. L. & Winefordner, J. D. Constant energy synchronous fluorescence for analysis of polynuclear aromatic hydrocarbon mixtures. Anal. Chem. 54(12), 2018–2022. https://doi.org/10.1021/ac00249a025 (1982).
Kubic, T. A., Lasher, C. M. & Dwyer, J. Individualization of Automobile Engine Oils I: The Introduction of Variable Separation Synchronous Excitation Fluorescence to Engine Oil Analysis. J. Forensic Sci. 28(1), 186–199. https://doi.org/10.1520/jfs12251j (1983).
Blanco, M. et al. Spectrofluorimetric identification of polycyclic aromatic hydrocarbons at PPB level. Anal. Lett. 29(9), 1603–1617. https://doi.org/10.1080/00032719608001508 (1996).
Li, Y. Q. et al. Synchronous fluorescence spectroscopy and its applications in clinical analysis and food safety evaluation. InReviews in fluorescence 95–117 (Springer, 2010). https://doi.org/10.1007/978-1-4419-9828-6_5.
Chen, G. Z., Huang, X. Z., Xu, J. G., Zheng, Z. Z. & Wang, Z. B. The Methods of Fluorescence Analysis 112 (Science Press., 1990).
Abuseada, H. H., Abdel Sattar, O. I., Madkour, A. W. & Taha, A. S. Ecofriendly first-derivative synchronous fluorometric method for simultaneous determination of atorvastatin and aspirin in pharmaceutical preparations. Sci. Rep. 15(1), 17787. https://doi.org/10.1038/s41598-025-99718-x (2025).
Abd El-Aziz, H., Fathy, M. E., El-Enany, N., Aly, F. A. & Tolba, M. M. Concurrent estimation of some co-administered antimicrobial drugs applying conventional and first derivative synchronous fluorescence spectroscopy techniques. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 264, 120255. https://doi.org/10.1016/j.saa.2021.120255 (2022).
Ramadan, H. S., Abdel Salam, R. A., Hadad, G. M., Belal, F. & Salim, M. M. First derivative synchronous spectrofluorimetric method for the simultaneous determination of tramadol and celecoxib in their dosage forms and human plasma. Luminescence 39(5), e4774. https://doi.org/10.1002/bio.4774 (2024).
Madbouly, E. A., El-Shanawani, A. A., Sobhy, M. & Abdelkhalek, A. S. Eco-friendly novel deconvoluted synchronous spectrofluorimetric approach for the determination of favipiravir, levodropropizine and moxifloxacin hydrochloride as an effective therapeutic combination for COVID-19; application in laboratory prepared mixtures and spiked human plasma. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 309, 123823. https://doi.org/10.1016/j.saa.2023.123823 (2024).
ICH Q2 (R2). Guidelines, Validation of analytical procedures (2022).
Patel, N. P. & Hinge, M. Development and validation of RP-HPLC method for simultaneous determination of methocarbamol and diclofenac sodium in injection dosage form. World J. Plast. Reconstr. Surg. 5(4), 1833–1842. https://doi.org/10.20959/wjpps20164-6506 (2016).
Miller, J. & Miller, J. C. Statistics and Chemometrics for Analytical Chemistry (Pearson Education, 2018).
Pena-Pereira, F., Wojnowski, W. & Tobiszewski, M. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 92(14), 10076–10082. https://doi.org/10.1021/acs.analchem.0c01887 (2020).
Płotka-Wasylka, J. A new tool for the evaluation of the analytical procedure: Green Analytical Procedure Index. Talanta 181, 204–209. https://doi.org/10.1016/j.talanta.2018.01.013 (2018).
Mansour, F. R., Bedair, A., Belal, F., Magdy, G. & Locatelli, M. Analytical Green Star Area (AGSA) as a new tool to assess greenness of analytical methods. Sustainable Chem. Pharm. 46, 10205. https://doi.org/10.1016/j.scp.2025.102051 (2025).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Maram Attia: Methodology, Investigation, Validation, Writing original draft; Ghada Hadad: Conceptualization, Writing-review & editing, Supervision; Randa Abdel Salam: Validation, Writing-review& editing; Mary Wahba: Conceptualization, Methodology, Resources, Writing-review and editing, Supervision. All authors have approval the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Attia, M., Hadad, G.M., Salam, R.A.A. et al. Comparative greenness assessment for the simultaneous estimation of diclofenac and methocarbamol in their tablets applying synchronous fluorimetry. Sci Rep 16, 9666 (2026). https://doi.org/10.1038/s41598-026-41615-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-41615-y