Abstract
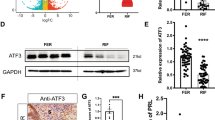
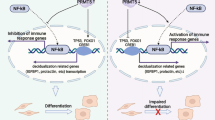
Recurrent implantation failure (RIF), a important contributor to infertility in assisted reproductive technologies, is believed to be primarily to abnormal endometrial decidualization. However, its related molecular mechanisms are still incompletely clear. It has been reported that PANX1 is an ATP-permeable channel to regulate the extracellular ATP, and ATP level could influence the progress of endometrial decidualization. Therefore, we conduct this study to explore the potential role of PANX1 during the development of RIF. In this study, we first compared the the expression of PANX1 in RIF patients and control patients. Then, the human endometrial stromal cells (HESCs) was used to the explore the regulatory function of PANX1 on endometrial decidualization by Immunofluorescence staining, Western blot, RT-PCR, Plasmid transfection and ATP detection. Finally, animal model was established to further confirm the results which were found in vitro. We observed a significant upregulation expression of ATP release channel protein Pannexin1 (PANX1) in endometrial tissue of RIF patients. Moreover, after overexpressing PANX1 in human endometrial stromal cells (HESCs), we found a significantly high concentration of extracellualr ATP (eATP) and the inhibition of HESCs decidualization, which was characterized as the decreasing expression of prolactin (PRL) and insulin-like growth factor binding protein 1 (IGFBP-1), as well as the aberrant morphologic cytoskeletion of HESCs. Surprisingly, knockdown the expression of PANX1 in HESCs also impaired the cellular decidualization, indicating the important role of normal PANX1 expression during the process of endometrial decidualization. The subsequent animal study further confirmed our findings in vitro. The present study clearly showed that abnormal high expression of PANX1 hindered endometrial decidualization through the upregulation of eATP concentrations, ultimately leading to embryo implantation failure. Our findings suggested a novel cause of RIF and identified a potential therapeutic target for RIF.
Similar content being viewed by others
Data availability
The datasets used in this study are available from the corresponding author on reasonable request.
References
Franasiak, J. M. et al. A review of the pathophysiology of recurrent implantation failure. Fertil. Steril. 116, 1436–1448 (2021).
Bashiri, A., Halper, K. I. & Orvieto, R. Recurrent implantation failure-update overview on etiology, diagnosis, treatment and future directions. Reprod. Biol. Endocrinol. 16, 121 (2018).
Ma, J., Gao, W. & Li, D. Recurrent implantation failure: A comprehensive summary from etiology to treatment. Front. Endocrinol. 13, 1061766 (2022).
Cimadomo, D., Craciunas, L., Vermeulen, N., Vomstein, K. & Toth, B. Definition, diagnostic and therapeutic options in recurrent implantation failure: An international survey of clinicians and embryologists. J. Hum. Reprod. 36, 305–317 (2021).
Wang, X. & Yu, Q. An update on the progress of transcriptomic profiles of human endometrial receptivity. J. Biol. Reprod. 98, 440–448 (2018).
Lédée, N. et al. Endometrial immune profiling: A method to design personalized care in assisted reproductive medicine. Front. Immunol. 11, 1032 (2020).
Diedrich, K., Fauser, B. C., Devroey, P. & Griesinger, G. The role of the endometrium and embryo in human implantation. Hum. Reprod. Update 13, 365–377 (2007).
Cha, J., Sun, X. & Dey, S. K. Mechanisms of implantation: Strategies for successful pregnancy. Nat. Med. 18, 1754–1767 (2012).
Jia, Z., Wei, Y., Zhang, Y., Song, K. & Yuan, J. Metabolic reprogramming and heterogeneity during the decidualization process of endometrial stromal cells. J. Cell. Commun. Signal. 22, 385 (2024).
Gellersen, B. & Brosens, J. J. Cyclic decidualization of the human endometrium in reproductive health and failure. J. Endocr. Rev. 35, 851–905 (2014).
Shi, J. W. et al. An IGF1-expressing endometrial stromal cell population is associated with human decidualization. J. BMC Biol. 20, 276 (2022).
Gibori, G. & Keyes, P. L. Luteotropic role of estrogen in early pregnancy in the rat. J. Endocrinol. 106, 1584–1588 (1980).
Evans, J. et al. Fertile ground: Human endometrial programming and lessons in health and disease. J. Nat. Rev. Endocrinol. 12, 654–667 (2016).
Gu, X. W. et al. Blastocyst-induced ATP release from luminal epithelial cells initiates decidualization through the P2Y2 receptor in mice. Sci. Signal. https://doi.org/10.1126/scisignal.aba3396 (2020).
Sang, Y., Li, Y., Xu, L., Li, D. & Du, M. Regulatory mechanisms of endometrial decidualization and pregnancy-related diseases. J. Acta Biochim. Biophys. Sin (Shanghai). 52, 105–115 (2020).
Rusiecka, O. M., Tournier, M., Molica, F. & Kwak, B. R. Pannexin1 channels-a potential therapeutic target in inflammation. J. Front. Cell. Dev. Biol. 10, 1020826 (2022).
Dahl, G. ATP release through pannexon channels. Philos. Trans. R. Soc. Lond. B Biol. Sci. https://doi.org/10.1098/rstb.2014.0191 (2015).
Vultaggio-Poma, V., Sarti, A. C. & Di Virgilio, F. Extracellular ATP: A feasible target for cancer therapy. Cells https://doi.org/10.3390/cells9112496 (2020).
Kobayashi, T. et al. ATP drives eosinophil effector responses through P2 purinergic receptors. J. Allergol. Int. 64 Suppl, S30–36 (2015).
Burnstock, G. Purinergic signalling in the reproductive system in health and disease. Purinergic Signal. 10, 157–187 (2014).
Gu, X. W. et al. ATP mediates the interaction between human blastocyst and endometrium. J. Cell Prolif. 53, e12737 (2020).
Huang, J. Y., Yu, P. H., Li, Y. C. & Kuo, P. L. NLRP7 contributes to in vitro decidualization of endometrial stromal cells. Reprod. Biol. Endocrinol. 15, 66 (2017).
杨倩 顾艳, 石燕, 何亚萍, 王健 米非司酮导致孕激素功能不足的自然流产小鼠模型的建立及其评价. J. 实验动物与比较医学. 36, 327–333 (2016).
Xu, X. B., He, B. & Wang, J. D. Menstrual-like changes in mice are provoked through the pharmacologic withdrawal of progesterone using mifepristone following induction of decidualization. Hum. Reprod. 22, 3184–3191 (2007).
Chang, S. J., Tzeng, C. R., Lee, Y. H. & Tai, C. J. Extracellular ATP activates the PLC/PKC/ERK signaling pathway through the P2Y2 purinergic receptor leading to the induction of Early Growth Response 1 expression and the inhibition of viability in human endometrial stromal cells. Cell. Signal. 20, 1248–1255 (2008).
Zhang, S. et al. Physiological and molecular determinants of embryo implantation. Mol. Aspects Med. 34, 939–980 (2013).
Benkhalifa, M. et al. Endometrium immunomodulation to prevent recurrent implantation failure in assisted reproductive technology. Int. J. Mol. Sci. https://doi.org/10.3390/ijms232112787 (2022).
Smith, M. B. & Paulson, R. J. Endometrial preparation for third-party parenting and cryopreserved embryo transfer. Fertil. Steril. 111, 641–649 (2019).
Deryabin, P. I. & Borodkina, A. V. Stromal cell senescence contributes to impaired endometrial decidualization and defective interaction with trophoblast cells. Hum. Reprod. 37, 1505–1524 (2022).
Bond, S. R. & Naus, C. C. The pannexins: Past and present. J. Front. Physiol. 5, 58 (2014).
Good, M. E. et al. Endothelial cell Pannexin1 modulates severity of ischemic stroke by regulating cerebral inflammation and myogenic tone. JCI Insight https://doi.org/10.1172/jci.insight.96272 (2018).
Scemes, E., Suadicani, S. O., Dahl, G. & Spray, D. C. Connexin and pannexin mediated cell-cell communication. J. Neuron Glia Biol. .3, 199–208 (2007).
Pelegrin, P. & Surprenant, A. Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. Embo J. 25, 5071–5082 (2006).
Acknowledgements
The authors thank all the staff, nurses, and physicians at the Reproductive Medicine Center for their support in generating this manuscript. This work was supported by the National Natural Science Foundation of China (82360318), the Science Project of Science and Technology Department of Jiangxi Province (20232BAB206029), the Outstanding Youth Fund Program of Science and Technology Department of Jiangxi Province (20224ACB216005), the Youth Fund Program of Science and Technology Department of Jiangxi Province (20232BAB216024) and the JXHC Key Laboratory of Fertility Preservation.
Author information
Authors and Affiliations
Contributions
Conception and design of the study: Jun Tan. Cell experiments: Qi Yang. Animal experiments: Xia-Lu Liu. Sample collection: Pei-Pei Liu and Li-Yun Cao. Data analysis and interpretation: Xin Li and Zhi-Qin Zhang. Draft of the manuscript: Xia-Lu Liu and Qi Yang. Supervision and critical revision of the manuscript for important intellectual content: Xian-E Sun, Jun Tan and Yao Cheng. All authors have read and confirm that they meet the authorship criteria.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was conducted in accordance with the guidelines of the Declaration of Helsinki and was approved by the Clinical Ethical Committee of Jiangxi Maternal and Child Health Hospital, and informed consents from patients were obtained before the initiation of the study. All the authors consented to participate in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, XL., Yang, Q., Liu, PP. et al. Abnormal elevated PANX1 expression hampers endometrial decidualization by upregulating extracellular ATP concentration in patients with recurrent implantation failure. Sci Rep (2026). https://doi.org/10.1038/s41598-026-41800-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-41800-z